Abstract
Inorganic arsenic (iAs) is an important pollutant associated with various chronic-degenerative diseases. The cytoprotective protein nuclear factor erythroid 2-related factor (NRF2) has been proposed as an important responsive mechanism against iAs exposure. The aim of this study was to determine whether the risk of skin lesions in people exposed to iAs-contaminated water could be modified by the presence of single nucleotide polymorphisms in the NRF2 coding gene. We studied 117 individuals with long-term iAs exposure and 120 nonexposed individuals. Total As was determined in water, meanwhile iAs and its metabolites were measured in urine. The iAs-induced skin lesion status was evaluated by expert dermatologists. We sequenced the promoter region of NRF2 in a sample of 120 healthy donors. We found four polymorphisms previously reported and one novel polymorphism in the 5′ regulatory region of the NRF2. In this study, we did not find allelic and genotype association of NRF2 polymorphisms with iAs-related skin lesion. However, the analysis of haplotypes composed by –653GA, and –617CA NRF2 single nucleotide polymorphisms showed a significant association with protection against skin lesions in the low-As exposure group. This is the first report studying the association between NRF2 polymorphisms and susceptibility of As-related skin lesions. Increasing the sample size will allow us to confirm this data.
Keywords
Introduction
Inorganic arsenic (iAs) is a naturally occurring contaminant present in food, soil, and water. Ground water contaminated with iAs is the main source of human exposure worldwide, and it represents an important health issue. 1 Longtime iAs exposure via drinking water has been associated with hyperkeratosis, cutaneous pigmentations changes, and skin cancer. 2 In fact, hyperkeratosis and hyperpigmentation are early hallmarks of chronic As exposure, which has been also associated to various human diseases such as cancer, atherosclerosis, and diabetes among others. 3 –6
Although the exact molecular mechanisms explaining the biological effects of iAs remain unclear, the toxic effects of iAs are highly associated with its accumulation in cells. Besides, chemical interaction with protein thiol groups, alteration of DNA methylation pattern and generation of reactive oxygen species (ROS) have been implicated in the actions of this metalloid. 7 –9
Numerous epidemiological studies highlight existence of interindividual variability in the susceptibility to the cytotoxic effects and disease outcome in populations chronically exposed to iAs. 10 Several evidences suggest that the occurrence of polymorphisms in genes associated with iAs metabolism and detoxification may be the cause for the individual variability. Accordingly, variants in arsenic methyltransferase and methylenetetrahydrofolate reductase genes have been found associated with bladder cancer in As exposure individuals. 11 Similarly, a dinucleotide repeat polymorphism in the heme oxygenase-1 gene (HMOX-1) has been associated with cardiovascular mortality risk in a population of chronically exposed to iAs. 12
The expression levels of the HMOX-1 gene are regulated by the transcription factor nuclear factor erythroid 2-related factor gene (NRF2), which is involved in one of the major cellular mechanisms against xenobiotics and oxidative stress. 13 In unstressed cells, NRF2 is degraded in the cytoplasm, by its protein inhibitor Kelch-like erythroid cell-derived protein with cap‘n’collar homology-associated protein 1 (KEAP1). 14,15 In the presence of different electrophilic chemicals or arsenical compounds, NRF2 is released from KEAP1 inhibition and promotes the expression of several antioxidant and detoxificant genes, such as glutathione S-transferase (GST), nicotinamide adenine dinucleotide phosphate quinone oxidoreductase (NQO1), and HMOX-1. 16 –18 Interestingly, downregulation of NRF2 by small interfering RNA-sensitized cells to iAs and its metabolite monomethylated As (MMAs)-induced toxicity; meanwhile, its activation by tert-butyl-hydroquinone or sulforaphane reduced not only As accumulation but also cellular toxicity. 19,20 Hence, NRF2-KEAP1 pathway activation has been proposed as an important cellular response mechanism against iAs exposure in different cellular types. 21 –23
Recently, the presence of several single nucleotide polymorphisms (SNPs) has been determined in the genomic sequence of the gene NRF2. At least two of them (–653GA, and –617CA) are located at the promoter region of the gene. 24,25 The presence of the minor allele from both SNPs was associated with a reduced promoter activity in luciferase assays and with several human diseases including gastric mucosal inflammation, acute lung injury, and lupus-related nephritis. 26 –28
Thus, the aim of this study was to evaluate the possible association between skin lesions in people exposed to As-contaminated water and the presence of regulator SNPs in the NRF2 gene.
Methods
Study population
A cross-sectional case-control study was carried out in a total of 117 Mestizos residents of Zimapan, Hidalgo, Mexico, where long-term iAs exposure has been recorded since 1992. Participants were recruited for a baseline visit.
Interviews and sample collection
Subjects were recruited through door-to door contact between January and March 2005. All individuals were older than 15 years and have been living in Zimapan for the past 2 years. Parents and all grandparents of the participants were from Mexican origin. The iAs-induced skin lesion status was evaluated, quantified, and validated by our study physicians and expert dermatologists. The iAs-related skin lesions are known to be hallmark of chronic iAs poisoning. These lesions include alterations in pigmentation and discoloration of skin, and in many cases, these symptoms are accompanied by keratosis, the thickening of the skin of the palms, soles, and trunk. 29 Clinical examination protocols used for assessing skin lesions were previously described. 30 Water, urine, and buccal cells were collected. Water samples were collected from the wells used by the study participants in 15-mL acid-washed tubes and stored at −20°C until they were analyzed. Spot urine samples were collected in 100-mL acid-washed bottles. A detailed description of the urine collection procedure has been reported elsewhere. 31
As determination
Total As (TAs) concentrations were measured as previously described using hydride generation-atomic absorption spectrometry (HG-AAS; model 3100, Perkin Elmer, Norwall, Connecticut, USA) equipped with a FIAS-200 flow injection atomic spectroscopy system. 32 Standard Reference Material (SRM) 1613e was used for quality control during the analyses of TAs in water. The certified TAs concentration in SRM 1613e is 60.15 ± 0.72 µg/L. The value determined in our laboratory was 61.3 ± 0.68 µg/L (n = 3).
Urinary iAs and its metabolites, MMAs, and dimethylated As (DMAs) were measured by HG-AAS, using cryotrapping for preconcentration and separation of arsines. 33,34 The TAs value was calculated as the sum of iAs, MAs, and DMAs. We used the SMR 2670 with the certified As content of 480 ± 100 µg/L to validate the calculated TAs in urines samples with high As concentrations. The low As urine SRM 2570 with a reference value at 60 µg/L was used to validate analyses of urines with low levels of As. Triplicates measurements at the high and low SMR standards provided the values 507 and 51 µg/L, respectively.
DNA samples from individuals exposed to As
Buccal cells were collected with a cytobrush. To avoid contamination, all participants were instructed to brush their teeth and refrain from eating and drinking for at least 1 h before sample collection. Subjects were also instructed to brush and twirl the cytobrush in each cheek for at least 30 s. Each cytobrush was then placed in a 15-mL sterile plastic tube. Genomic DNA was isolated from the buccal samples using a commercial DNA purification kit according to the manufacturer’s protocol (Amersham GFX Genomic blood. Amersham Pharmacia Biotech Inc., Uppsala, Sweden).
Polymorphisms identification in healthy individuals
A total of 120 healthy individuals were enrolled from a blood bank in Mexico City. All were greater than 18 years of age and were diagnosed as clinically healthy after general examination and questionnaire. To participate in this study, parents and all grandparents were required to be from Mexican origin. Local ethics and research committees approved this protocol, and an informed written consent was obtained from all participants. All the procedures were in accordance with the Declaration of Helsinki 1975, revised Hong Kong 1989. In order to identify the presence of new SNPs in the Mexican population, we screened the regulatory region of NRF2 gene in all healthy individuals. Genomic DNA was isolated from whole blood samples using the QIAamp DNA Blood Maxi kit (Qiagen, Valencia, California, USA), according to manufacturer’s instructions. Overlapping primers were designed to span the proximal 1 kb of the NRF2 promoter and its noncoding exon 1. In all cases, polymerase chain reaction (PCR) amplification was started with 100 ng of genomic DNA using Taq Gold polimerase (Applied Biosystem, Foster City, California, USA). Thermal protocol was composed of a denaturing step at 95°C for 10 min, 30 cycles of 95, 60, and 72°C of 30 s each, and a final step of 72°C for 10 min. PCR products were purified with the QiAquick PCR Purification Kit (Quiagen, Valencia, California, USA) and sequencing reactions were performed with deoxyguanosine triphosphate dye terminator kits (Applied Biosystems, Foster City, California, USA). Purified products were analyzed in a 96-capillary 3730xl DNA analyzer (Applied Biosystems, Foster City, California, USA). Electropherograms were aligned with the reference sequence (Genebank accession number: NM_006164) using the SeqMan software (Lasergene, DNASTAR, Inc., Madison, Wisconsin, USA).
Genotyping for –617CA, and –653GA SNPs
The polymorphisms –617CA (rs6721961) and –653GA (rs35652124) were genotyped by the 5′exonuclease assay (TaqMan) in the iAs-exposed population. Allelic discrimination was performed with the ABI Prism 7900HT Sequence Detection System (Applied Biosystems, Foster City, California, USA). The results were analyzed with the SDS 3.2 software (Applied Biosystems, Foster City, California, USA). Call rate was over 98% for both SNPs. A random 5% of the samples were sequenced to validate genotyping procedures.
Statistical analysis
We calculated the population characteristics and tested the significant differences between cases and controls using the χ 2 and t test. To evaluate the significance of differences in genotype and allele frequencies between subjects exposed to As with skin lesions (cases) and As exposed-without skin lesions (controls), we used χ 2 analysis. Also, allele frequencies for cases and controls were used to calculate the odds ratio (OR) and the 95% confidence interval (95% CI). For all statistical calculations and departures from the Hardy–Weinberg equilibrium (HWE), StatCalc software (Epi Info 2005 v3.3.2; Centers of Disease Control and Prevention, Atlanta, Georgia, USA) and the FINETTI program with the exact test (http://ihg.gsf.de/cgi-bin/hw/hwa1.pl) were used.
In the haplotype analysis, Haploview v3.2 (http://www.broad.mit.edu/mpg/haploview) was used to estimate haplotype frequencies; this software uses an implementation of the expectation–maximization (EM) algorithm. The values of p < 0.05 were considered statistically significant; they were determined using one degree of freedom. Pairwise linkage disequilibrium (LD) measures (r2) between SNPs and maximum-likelihood haplotype frequencies were estimated with the EM algorithm.
Results
Study population
Study sample was composed of 104 females and 13 males (n = 117) and their age was in a range from 18 to 50 years, with a median of 35 years. The number of male participants in our study was very low due to the high migration of males to work. All participants were exposed to different levels of iAs through drinking water. The concentrations of iAs in samples of drinking water ranged from 1 to 203 ppb (nanograms of As per mmilliliter; Table 1). The concentrations of TAs in urine ranged from 9 to 342 ng As/mg creatinine (Table 1). The TAs concentration in urine was used as a measure of exposure and allowed to classify our population in low and high iAs exposure with a cutoff point of 50 μg/g creatinine of As in urine. From these values, of the total subjects 41 (35%) and 76 (65%) were exposed to low and high levels of iAs, respectively. In our population, of the total individuals 67 (57%) showed at least one type of lesion independently of the level of iAs exposure. As expected, the occurrence of skin lesions significantly increased in high-exposure individuals (67%) compared to subjects with low iAs exposure (39%). It is important to mention that even under high levels of iAs exposure, a considerable number of subjects did not develop skin lesions (33%; Table 2).
General characteristics of the study population.a

TAs= Total arsenic.
aValues are expressed in mean and range.
Nonmalignant skin lesions in individuals exposed to different levels of iAs.
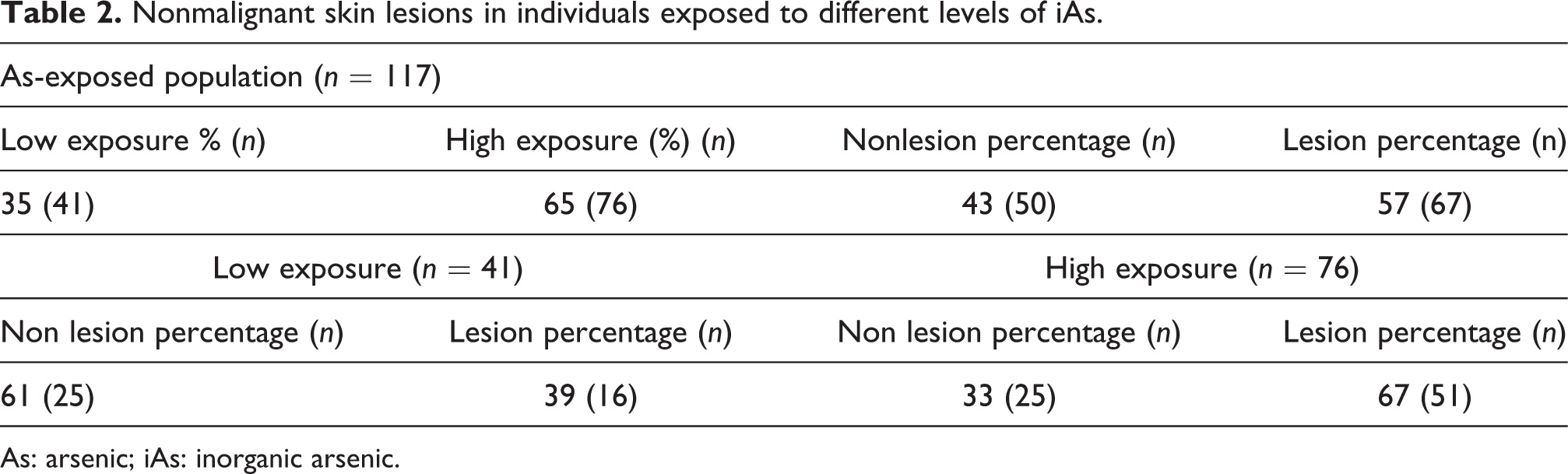
As: arsenic; iAs: inorganic arsenic.
Determination of polymorphisms at the regulatory region of NRF2 gene
We searched for new polymorphisms in 120 healthy donors by analyzing approximately 1 kb of the NRF2 promoter and its noncoding exon 1. We found five different polymorphisms, four were located at the promoter region of NRF2 and one at the noncoding exon (Table 3, Figure 1). Four of these polymorphisms have been described previously in Caucasian and Japanese individuals (–653GA, –651GA, –617CA, and 12delCCG). In the case of the 12delCCG polymorphism, we found in two independent subjects a new variant composed of only two-time CCG triplet repeat, instead of the previously reported four- or five-time triplet repeat. 24 The gene variants –653GA, and 12delCCG were found in high LD (r 2 > 0.95). The minor alleles from –653GA, –617CA, and 12delCCG polymorphisms were relatively common in our population (46, 26, and 45%, respectively); meanwhile, those from –651CA, and –513delC were quite rare compared to the ancestral allele (3 and 1%, respectively).
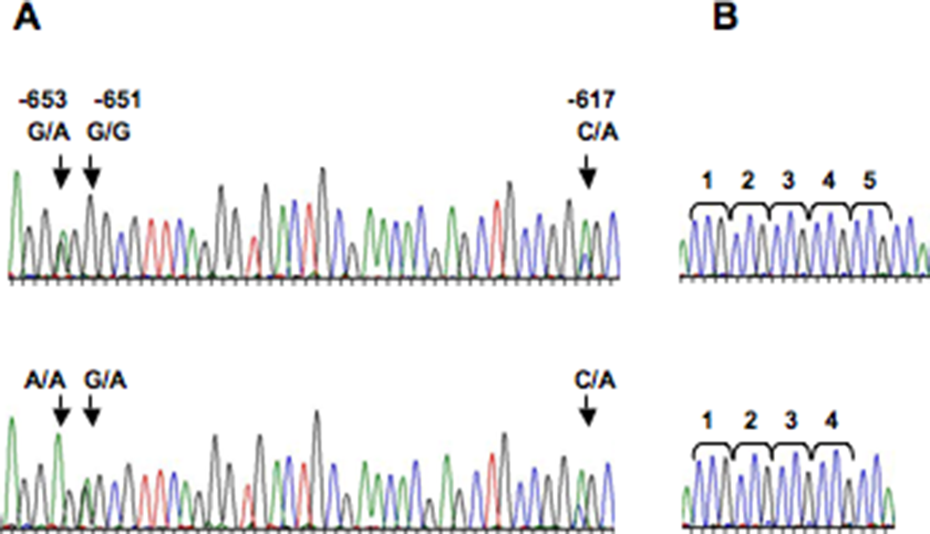
Polymorphisms located at regulatory region of NRF2 gene. (a) Representative electropherograms of the NRF2 promoter region. Arrows show position of the –653 G/A, –651 G/A, and –617 C/A SNPs. (b) Representative electropherograms of homozygote for four (bottom) and five (top) times triplet repeat polymorphisms. NRF2: nuclear factor erythroid 2-related factor gene; SNPs: single nucleotide polymorphisms.
Allelic frequencies of polymorphisms located in the 5′ proximal region of the NRF2 gene in Mexican population.
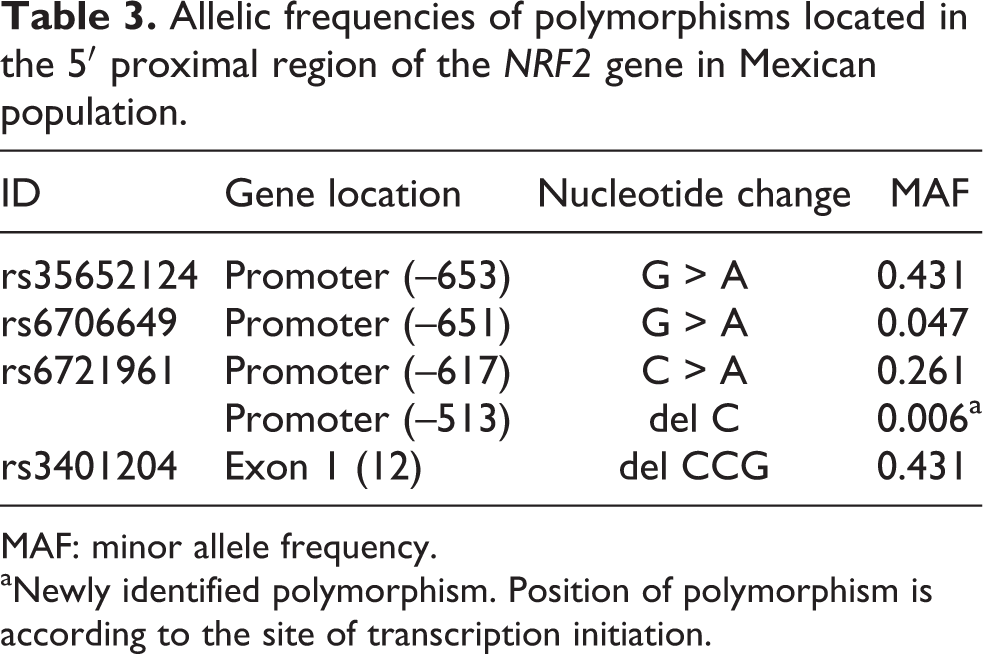
MAF: minor allele frequency.
aNewly identified polymorphism. Position of polymorphism is according to the site of transcription initiation.
NRF2 polymorphisms and As-related skin lesions
We evaluated whether NRF2 SNPs were associated with individual susceptibility to develop nonmalignant skin lesion after chronic exposure to iAs. Thus, we compared polymorphisms frequencies in subjects exposed to iAs with (cases) and without skin lesions (controls). Since the minor allele frequency of almost all polymorphisms was very low (<3%) and 12delCCG polymorphism was in almost complete LD with the –653GA SNP, we only included in the case-control analysis the –653GA and –617CA SNPs. The two SNPs, –653GA and –617CA, were in HWE, in both cases and controls. Allelic and genotype distribution of –653GA and –617CA SNPs showed no significant difference among cases and controls (Table 4).
Allelic and genotypic frequencies of –653G/A and –617C/A NRF2 SNPs in iAs-associated skin lesion.

NRF2: nuclear factor erythroid 2-related factor gene; SNPs: single nucleotide polymorphisms; iAs: inorganic arsenic; C: cytosine; A: adenine; G: guanine; OR: odds ratio; CI: confidence interval.
aThe genotypes and allele distribution of NRF-2 polymorphism in individuals exposed to iAs are shown for groups with and without lesions. Two-sided χ2 test is followed.
NRF2 haplotypes and As-related skin lesions
We also analyzed the possible association of NRF2 haplotypes with the occurrence of As-related skin lesions. We found three different haplotypes: the CG composed of the two ancestral alleles; the AA, made up of the risk alleles, and CA comprising the minor allele and ancestral allele from the –617 and –653 SNPs (63, 21, and 16%, respectively). There was not a significant difference in haplotypes frequencies between the presence and absence of skin lesions in people exposed to high levels of iAs (Table 5).
NRF2 haplotypes and skin lesions in iAs individuals.

NRF2: nuclear factor erythroid 2-related factor gene; iAs: inorganic arsenic; C: cytosine; A: adenine; G: guanine; OR: odds ratio; CI: confidence interval.
aThe haplotypes in individuals exposed to iAs are shown for groups with and without lesions, stratified by low and high exposure considering a cutoff point of 50 μG/g creatinine. Two-sided χ2 test is followed.
However, at low As exposure the CA haplotype was significantly associated with the absence of skin lesion (0.24 vs. 0.06; OR = 0.43, p = 0.037). This result suggested that the combination of the major allele of the –653 SNP and the minor allele of the –651 SNP have a protective effect against low levels of As exposure.
Discussion
Contaminated water with iAs is a global health issue, particularly in developing countries. Chronic iAs exposure is associated with skin lesions, such as hyperkeratosis and hyperpigmentation. Also, drinking iAs-contaminated water increases risk for skin, bladder, and lung carcinomas. Although the molecular mechanisms of iAs toxicity are not completely understood, oxidative stress has been proposed as one of the main iAs effects in the cell. 35 The existence of individual variations in susceptibility to iAs-induced diseases has been extensively reported. 11,36,37
The NRF2 transcription factor regulates the expression of several genes involved in iAs detoxification, such as GST and several multidrug-resistant proteins (MRPs). 38,39 Accordingly, reduced expression of NRF2-sensitized cells to iAs-induced toxicity; meanwhile, induction of NFR2 activity increase cellular resistance to As. 19,20 Polymorphisms located at the promoter region of NRF2 may influence ROS-related diseases by altering the expression of its mRNA. In fact, case-control studies have highlighted the association between the presence of SNPs at the promoter region of NRF2 and gastric mucosal inflammation, acute lung injury, and lupus-related nephritis. 26 –28
Polymorphisms in the promoter region of NRF2 have been previously reported in Japanese and Caucasian population but have been poorly studied in samples of different ethnical origin. Screening of only 10 samples from four ethnically diverse populations (European, African, Native American, and Asian) showed very different frequencies for the –653GA, –651GA, and –617CA SNPs. 27 Since Mexican population is characterized by a high level of admixture, we decided to resequencing promoter region of NRF2 gene in 120 Mexican individuals. The sample size allows detecting at least 95% of polymorphic sites with a minimal allele frequency of 1%. 40 From the previously reported nine polymorphisms, we only found four in our population (–653GA, –651GA, –617CA, and 12delCCG), a new allele for one of them (12delCCG) and a new polymorphism. The frequencies of the –653GA, –651GA, and 12delCCG polymorphisms in our population were very similar to those reported in Native American or Asian population; meanwhile, the –617C/A SNP showed significant differences in the three populations. Taken together, these data highlighted the great diversity in the frequency of each polymorphism in accordance with the ancestry of the population and the importance of these data in case-control study.
Neither allelic nor genotypic frequencies from –653GA or –617C/A SNPs showed significant differences between any of the comparison groups (lesion vs. no lesion; at low- or high-exposure groups). However, after we constructed the haplotypes, we found a significantly higher frequency of the CA haplotype among the individuals without lesions than in those affected with skin lesions in the low As exposure group. This result indicates a protective role of the NRF2 CA haplotype against skin lesions, only under low levels of iAs exposure. A recent study has reported the association of the CA haplotype of NRF2 with a delay in the progression of Parkinson’s disease. 41 The CA haplotype (composed of the ancestral and minor alleles from the –617 and –653 SNPs) is also associated with a higher expression of NRF2 than the other haplotypes, at least in vitro assays. 27 These data suggest that individuals chronically exposed to iAs and carrying haplotypes other than the CA would be in an increased risk to develop iAs-related diseases. The major limitations of this study are the small sample of population and the fact that most of the participants are females. Results obtained in this study may not generalize to males or broad population.
To our knowledge, this is the first report to highlight the possible association between polymorphisms of NRF2 and As susceptibility. Increasing the sample size will allow us to confirm this data. Besides, further studies should analyze the role played by genetic variants in components of the NRF2-KEAP1 signaling pathway in As susceptibility.
Footnotes
Acknowledgments
We acknowledge Ms C Salvador Hernandez for helping in sample collection and DNA sequencing.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This work was financially supported by the CONACyT-FOSSIS [grant number 71053].
