Abstract
Accumulating evidence has shown that ethanol-induced iron overload plays a crucial role in the development and progression of alcoholic liver disease. We designed the present study to investigate the potential protective effect of quercetin, a naturally occurring iron-chelating antioxidant on alcoholic iron overload and oxidative stress. Ethanol-incubated (100 mmol/L) rat primary hepatocytes were co-treated by quercetin (100 µmol/L) and different dose of ferric nitrilotriacetate (Fe-NTA) for 24 h. When the hepatic enzyme releases in the culture medium, redox status of hepatocytes and the intercellular labile iron pool (LIP) level were assayed. Our data showed that Fe-NTA dose dependently induced cellular leakage of aspartate transaminase and lactate dehydrogenase, glutathione depletion, superoxide dismutase inactivation, and overproduction of malondialdehyde) and reactive oxygen species (ROS) of intact and especially ethanol-incubated hepatocytes. The oxidative damage resulted from ethanol, Fe-NTA, and especially their combined treatment was substantially alleviated by quercetin, accompanying the corresponding normalization of intercellular LIP level. Iron in excess, thus, may aggravate ethanol hepatotoxicity through Fenton-active LIP, and quercetin attenuated ethanol-induced iron and oxidative stress. To maintain intercellular LIP contributes to the hepatoprotective effect of quercetin besides its direct ROS-quenching activity.
Introduction
It is now well accepted that the fundamental pathogenesis of alcoholic liver diseases (ALD) is implicated in reactive oxygen species (ROS)-mediated oxidative stress. 1,2 Such harmful by-products are overproduced during ethanol metabolism mainly through microsomal ethanol oxidizing cytochrome P4502E1 (CYP2E1), mitochondrial respiratory chain of hepatocytes, and nicotinamide adenine dinucleotide phosphate reduced oxidase of Kupffer cells and infiltrating granulocytes. 3,4 Moreover, ALD has been demonstrated to be frequently associated with iron overload. 5 Even mild to moderate alcohol consumption could increase the prevalence of iron overload. 6 What is worse is that superoxide (O2•−) and hydrogen peroxide can be transformed into hydroxyl radical (•OH), the most toxic fraction among various ROS, in the presence of the catalysis of “free” iron via Fenton/Haber–Weiss reaction. 7 Whatever may be the cause, iron overload gives rise to high labile iron pool (LIP), which is defined as a low-molecular weight pool of weakly chelated and redox-active iron and a cellular source available for Fenton reaction. 8 –10 Hence, it is presumed that iron overload may synergistically amplify ethanol-induced oxidative stress. 11 What should be focused on the prevention of ALD, therefore, is to avoid the synergistic prooxidative hepatotoxicity of iron with ethanol.
Quercetin (3,5,7,3′,4′-pentahydroxy flavon), one of the most widely distributed flavonoids in plants including various vegetables, fruits, herb medicine, and red wine, is generally regarded as a prominent ROS scavenger. 12 Quercetin has been shown to protect hepatocytes from ethanol-induced oxidative stress in vitro 13,14 and exhibit favorable hepatoprotective effect against ethanol intoxication in vivo. 10,15 In addition, quercetin has also demonstrated to significantly decrease hepatic iron and prevent hepatic lipid peroxidation and protein oxidation as a result of iron overload. 16,17 Up to now, however, little is known about the effects of quercetin on the synergistic oxidative stress of ethanol and iron overload. Therefore, we designed the present study to explore the potential protective effects of quercetin on alcoholic iron overload and subsequent oxidative stress.
Methods
Chemicals
Quercetin (purity: ≥98%), insulin, collagenase (type IV), 2-thiobarbituric acid (TBA), bilirubin, Percoll, 5,5′-dithio-bis (2-nitrobenzoic acid) (DTNB), 1,1,3,3-tetraethoxypropane, glutathione (GSH), ferric ammonium citrate, and hydrocortisone were obtained from Sigma-Aldrich (St Louis, Missouri, USA). Nitrilotriacetic acid disodium salt (Na2-NTA) was provided by Sigma-Fluka (St Louis, Missouri, USA). Bovine serum albumin (BSA) and 4-hydroxyethyl piperazine ethanesulfonic acid (HEPES) were supplied from Amersco Inc. (Framingham, Massachusetts, USA). Calcein–acetomethoxy (AM) was from Molecular Probes (Eugene, Oregon, USA). The kits of superoxide dismutase (SOD), aspartate transaminase (AST), and lactate dehydrogenase (LDH) were provided by the Nanjing Jiancheng Corporation (Nianjing, China). ROS assay kit and trypan blue were obtained from Beyotime Corporation (Jiangsu, China). All other reagents were of the purest grade available and purchased from Sigma unless indicated otherwise.
Preparation of Fe-NTA solution
Ferric-nitrilotriacetate (Fe-NTA) solution was freshly prepared immediately before its use by the method described by Awai et al. 18 with slight modification. Briefly, ferric ammonium citrate and Na2-NTA were dissolved in double distilled water. The pH was adjusted to 7.4 with sodium bicarbonate by constant stirring.
Isolation and culturing of rat primary hepatocytes
Hepatocytes were isolated from adult male Sprague Dawley rats, weighing 200–250 g, using a two-step collagenase perfusion procedure described by Figliomeni and Abdel-Raham. 19 Hepatocytes were purified by Percoll (25%) centrifuge and the viability was assessed by trypan blue exclusion test. The freshly harvested rat hepatocytes (with over 90% viability) were suspended in Dulbecco’s modified Eagle’s medium (DMEM) containing 100 U/ml penicillin, 100 U/ml streptomycin, 10% fetal bovine serum, 32 IE/L insulin, 0.1 µmol/L hydrocortisone, and 15 mol/L HEPES and then inoculated in rat tail collagen-coated six-well plates for culture in a humidified incubator with 95% air and 5% carbon dioxide at 37°C. On the following day, the attached hepatocytes were incubated with the fresh medium containing absolute ethanol (100 mmol/L), quercetin (100 μmol/L), and/or Fe-NTA (different doses). After 24 h, the supernatant and hepatocytes were collected for various bioassays according to the corresponding experimental protocol.
LDH and AST leak assays
Following each treatment, the supernatant from each well was collected to measure the LDH and AST activity leaked from cultured rat hepatocytes with enzyme kinetic method in the light of the instructions of reagent kits. Results are expressed in units per liter.
Determination of cellular GSH, SOD, and MDA
Cellular reduced GSH, the most important nonenzymatic tripeptide antioxidant present in cells, was measured according to its reaction with DTNB into 2-nitro-5-thiobenzolic acid, following deproteinization by 5% trichloroacetic acid. 20 SOD was determined on the basis of the enzymatic disproportionating rate of superoxide radicals originated from xanthine/xanthine oxidase system, and one unit is defined as the amount of enzyme with 50% dismutation of the superoxide radical. 21 As one main end product of lipid peroxidation, cellular malondialdehyde (MDA) level was measured using TBA colorimetry slightly modified by Ohkawa et al. 22 All results were standardized by cellular protein content measured by Lowry’s method, 23 using bovine serum albumin (BSA) as standard.
Assay of cellular ROS and LIP level
ROS was estimated according to its oxidizing capacity on dichlorodihydrofluorescein (DCFH) into dichlorofluorescein with fluorescent emission (excited fluorescence (Ex) = 488 nm and emission fluorescence (Em) = 525 nm). Briefly, DCFH diacetate (DCFH-DA) was incubated with rat hepatocytes at 37°C for 30 min to degrade into DCFH. Following washing with phosphate-buffered saline (PBS) for three times, cellular fluorescence intensity was detected by fluorescence microscope with Image-pro plus 6.0 densitometric analysis program (IPP 6.0 software package; Media Cybernetics, Inc., Rockville, Maryland, USA). Cellular content of LIP-Fe was measured by fluorospectrophotometry using calcein-AM ester, a cell-permeable nonfluorescent probe when hydrolyzed into fluorescent calcein by intracellular esterases. 24,25 Briefly, rat hepatocytes in 96-well plates were washed twice with PBS and incubated with calcein-AM (0.25 μmol/L) for 30 min at 37°C and the fluorescence intensity was measured by fluorospectrophotometer (Ex = 495 nm, Em = 530 nm) using IPP 6.0 software package. Following the incubation in the medium-containing desferrioxamine (DFO, 100 μmol/L) for 30 min at 37°C, the fluorescence intensity was measured again. The LIP was calculated as the difference in calcein-AM cells treated with or without DFO. The increased ROS and LIP level of hepatocytes following various treatments were expressed as fold change over control.
Statistical analysis
Data were expressed as the mean ± standard deviation from replicate experiments. All statistic analyses were performed using Statistical Package for the Social Sciences software package 16.0 (SPSS 16.0 for Windows, SPSS, Inc., Chicago, IL, USA), and the differences in the mean among groups were assessed for significance by one-way analysis of variance followed by Students–Newman–Keuls multiple range test. Statistical significance was defined as
Results
Dose response of Fe-NTA on oxidative damage of ethanol-incubated hepatocytes
Rat primary heptocytes were treated with different doses of Fe-NTA (approximately 5–100 µmol/L) alone and/or with ethanol (100 mmol/L) for 24 h and the cellular enzymatic leakage and redox parameters were determined. Compared with normal intact hepatocytes, Fe-NTA dose dependently induced the leakage of cellular AST and LDH into the culture medium, accompanying GSH depletion, SOD inactivation, and the elevation of ROS and MDA. As observed previously, ethanol (100 mmol/L) also stimulated redox disequilibrium and enzymatic leakage in comparison with normal hepatocytes. Of note, ethanol-derived oxidative damage was further aggravated in the presence of Fe-NTA especially when the concentration of Fe-NTA was above 50 µmol/L (Figure 1).
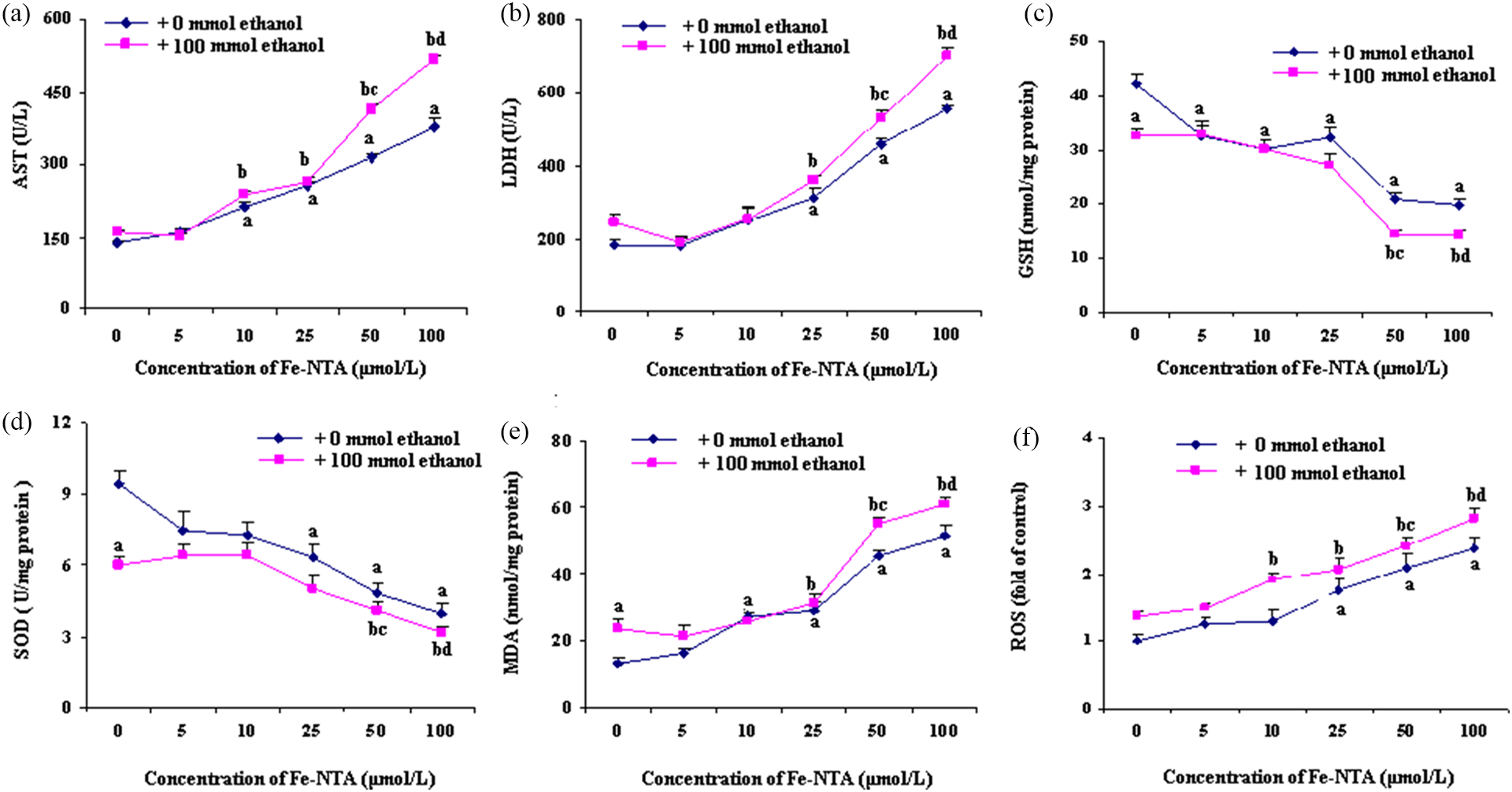
Effects of ethanol and/or different doses of Fe-NTA on oxidative damage induced by ethanol in rat primary hepatocytes (
Quercetin treatment against Fe-NTA induced oxidative damage on ethanol-incubated hepatocytes
Fe-NTA (50 μmol/L)- and/or ethanol (100 mmol/L)-incubated rat hepatocytes were co-treated by quercetin (100 μmol/L) to further explore the potential beneficial effect on ethanol-derived iron overload. As reported previously, quercetin evidently suppressed the enzymatic leakage of cellular AST and LDH, normalized GSH level and SOD activity, and decreased ROS and MDA content of ethanol-incubated hepatocytes in comparison with normal control. The same protective effect of quercetin was also observed on Fe-NTA-treated rat hepatocytes. Fe-NTA further exacerbated ethanol hepatotoxicity reflected by more severe oxidative damage than hepatocytes treated by Fe-NTA or ethanol alone. Notably, such combined hepatotoxicity of ethanol along with Fe-NTA was still partially reversed by quercetin, suggesting a substantial hepatoprotective role of quercetin in counteracting oxidative damage during alcoholic iron overload. However, quercetin had no effect on basal redox indices of normal hepatocytes (Figures 2 –4).

Quercetin against ethanol and Fe-NTA-derived AST and LDH leakage (

Quercetin against ethanol and Fe-NTA derived GSH depletion, lipid peroxidation marked by MDA level and SOD activity (
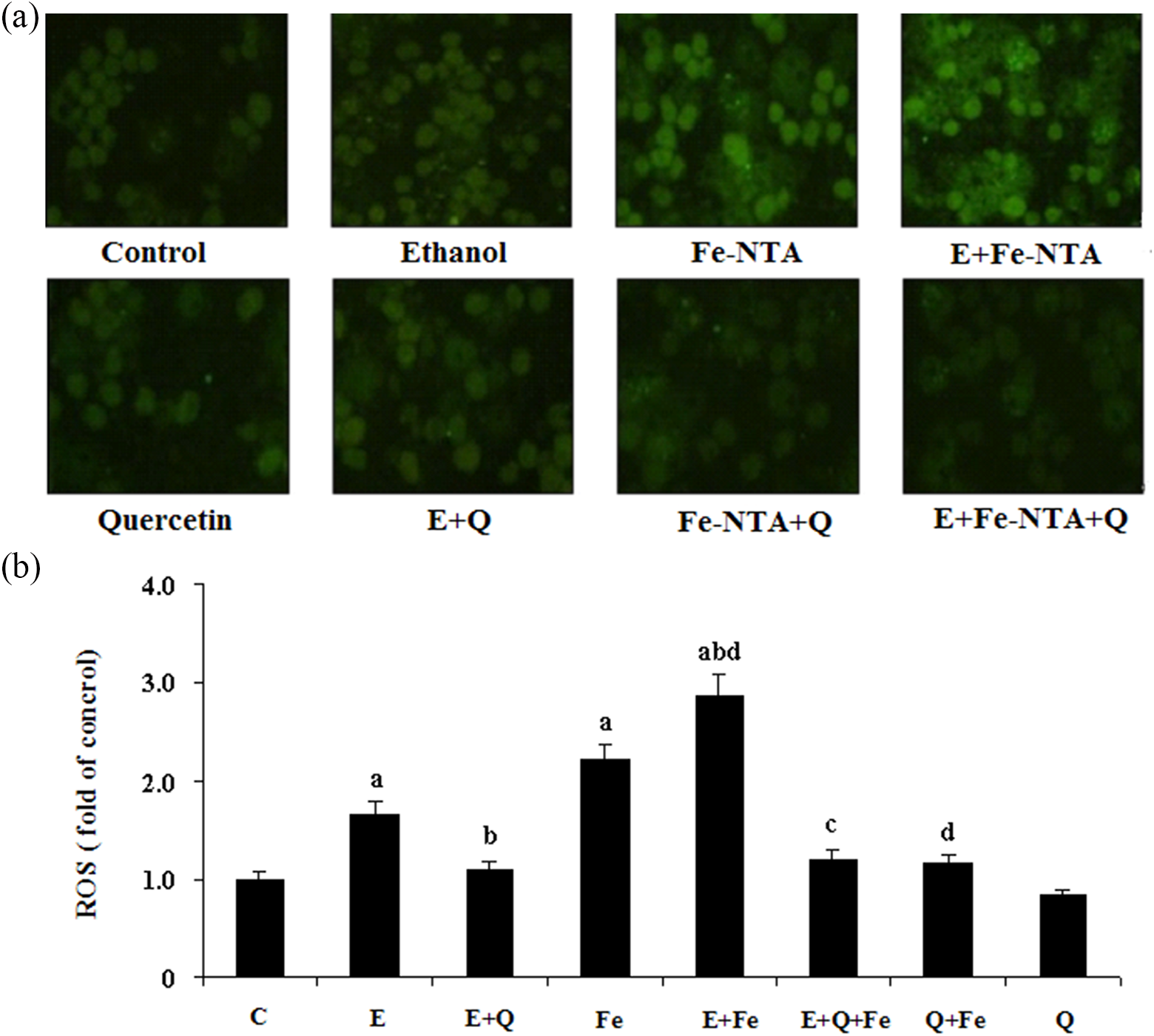
Effect of quercetin on ROS level in rat primary hepatocytes incubated with ethanol and Fe-NTA (
Influence of quercetin on intracellular LIP treated by Fe-NTA and/or ethanol
To investigate the potential mechanism by which ethanol triggered oxidative stress during alcoholic iron overload and quercetin exerted hepatoprotective effect, intracellular LIP level was measured following various treatments. As expected, Fe-NTA incubation dramatically increased LIP content compared with normal control. Meanwhile, ethanol exposure also resulted in the elevation of intercellular LIP concentration in spite of relatively lower value than Fe-NTA-treated hepatocytes. The LIP level distinctly peaked when rat hepatocytes were co-treated with ethanol and Fe-NTA, which was well in line with the observation on the extent of oxidative damage induced by ethanol and/or Fe-NTA. Of importance, the increased LIP level as a result of ethanol/Fe-NTA incubation was effectively reduced by quercetin treatment, which paralleled with quercetin-derived amelioration on oxidative damage provoked by ethanol and Fe-NTA. The basal LIP level, vital for normal iron metabolism and cell functions, was not affected by quercetin for intact hepatocytes (Figure 5).
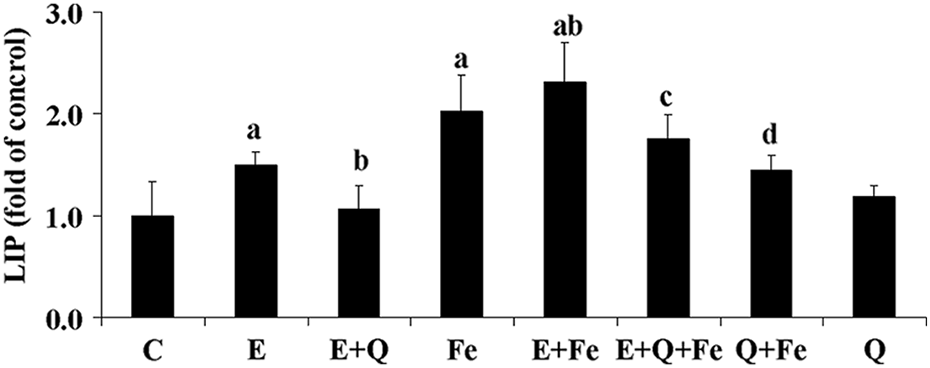
Influence of ethanol and Fe-NTA co-treatment on intracellular labile iron pool (
Discussion
Chronic alcohol consumption, even in moderate quantities, has been demonstrated to increase serum iron and ferritin concentration and transferrin–iron saturation (TS), which promote hepatic iron stores. Alcoholic iron overload as a consequence of heavy consumption has once been falsely diagnosed as hereditary hemochromatosis. Moreover, such iron mismanagement and overload for ALD patients are frequently associated with advanced and sustained deterioration of pathological condition. 26 On the other hand, the cessation of alcohol intake usually leads to fairly prompt reduction in serum TS and ferritin. 27 –29 Recently, iron chelation has been presumed as one of the new approaches to treat and prevent ALD. 30 Manganese SOD overexpression prevents liver mitochondrial DNA depletion after an alcohol binge but worsens this effect after prolonged alcohol consumption in mice. 31 In the present study, both ethanol and iron treatment induced evident damage on rat hepatocytes. Furthermore, the cell damage and redox imbalance were dramatically exacerbated when co-treated by ethanol + iron. Sakurai et al. 32 found that Fe-NTA incubation to a HepG2 cell line expressing CYP2E1, a microsomal cytochrome response for excessive ethanol metabolism and ROS overproduction, resulted in lipid peroxidation and mitochondrial damage. These results suggest that iron may potentiate and participate in the hepatotoxicity of ethanol during alcoholic iron overload.
Oxidative stress has been well documented as a central mechanism implicated in the development and progression of ALD.
1,2
Serving as a catalyst for Fenton/Haber–Weiss reaction, iron in excess is cytotoxic by markedly aggravating oxidative stress in spite of an essential micronutrient required for a variety of biochemical processes. Under normal conditions, such redox-active LIP is tightly regulated and homeostatically maintained to meet the metabolic demands for iron while minimizing its potential engagement in ROS formation. However, in extreme cases of iron overload or deprivation, the changes might exceed the cell homeostatic capacity, thus compromising its integrity.
33
Our data in the current study showed that ethanol or iron incubation, especially their combined treatment, increased intercellular LIP level with corresponding cell oxidative damage. However, Sergent et al.
34
reported that iron overload alone significantly increased the hepatic iron content but failed to affect either LIP or lipid peroxidation in vivo. The disparate results might be due to the difference in iron source and experimental model. Xiong et al.
35
found that iron accumulation in hepatic macrophages/Kupffer cells and the level of LIP increased remarkably during the alcohol liver injury, which in turn aggravated alcoholic steatohepatitis. The increased release of iron from endosomes containing transferrin-receptor complexes may contribute to the abnormal LIP level during ethanol exposure.
34
Several clinical trials showed that iron-chelating antioxidants for LIP might be useful for the treatment of a wide variety of disease with high level of LIP, including heart disease, neuropathy, and free-radical-mediated injury.
25,36,37
Antioxidant supplementation is one plausible strategy to counteract oxidative stress and maintain redox homeostasis. Plant-derived quercetin possesses miscellaneous bioactivity based on or implicated in its prominent antioxidative properties.
38
Besides direct free radicals-quenching effect at physiological concentrations,
39
quercetin has been shown to facilitate electron export and thereby reduces intracellular ROS accumulation by functioning as an electron donor and actual substrate of duodenal transmembrane oxidoreductase cytochrome
Conclusion
Quercetin reduced intracellular LIP level and ameliorated oxidative damage of rat primary hepatocytes induced by ethanol and/or iron. Thus, the heptaprotective effects of quercetin against ALD is involved in the chelation of iron during secondary alcoholic iron overload, which shed light on the treatment and prevention of ALD by naturally occurring iron-chelating flavonoid antioxidants from a new prospective.
Footnotes
Funding
This work was supported by the National Natural Science Foundation of China (No. 30872117), Program for New Century Excellent Talents in University of China (NCET 08-0222), and Wuhan Planning Project of Science and Technology (No. 201260523192).
