Abstract
Cigarette smoking is one of the most important risk factors for kidney cancer, but the molecular mechanism is poorly understood. To examine the expression change of histone H3 on lysine 27 trimethylase (H3K27me3) demethylases ubiquitously transcribed TPR gene on the X chromosome (UTX) in kidney cancer cell line 786-O after nicotine treatment, quantitative real-time-polymerase chain reaction and western blotting analysis were carried out. These results showed that nicotine can increase UTX messenger RNA and protein levels and also decrease the content of H3K27me3. The decreased content of H3K27me3 may activate specific gene expression and lead to kidney cancer. Future investigation on nicotine induced UTX expression and its epigenetic effect would deepen our understanding on nicotine toxicity and carcinogenicity.
Introduction
Renal cell carcinoma (RCC) is the predominant form of kidney cancer and is responsible for an estimated 120,000 deaths per year worldwide. 1 Epidemiological data have showed that cigarette smoking, obesity and hypertension are the most important risk factors for RCC. 2 Cigarette smoking is important in accelerating RCC and accounts for approximately 20% of all RCC cases. 3,4 Nicotine is one of the component of cigarette smoking and can promote the progression of kidney disease. 5 However, the molecular mechanisms by which nicotine may promote kidney cancer progression remain incompletely elucidated. 6
Many studies have shown that an altered pattern of epigenetic modifications is central to many common human diseases, including cancer. 7,8 Epigenetic modifications of histones include acetylation, methylation, phosphorylation and ubiquitination, which affect chromatin structure and function resulting in altered gene expression and changes in cell behavior. 9,10 Alterations in modifications of histones occur frequently in cancer and have been linked to deregulated expression of many genes with important roles in cancer development and progression. 11,12 Several studies also suggested that cigarette smoking can induce cancer-associated epigenetic alterations, especially histone modifications. 13,14 These results indicated histone modifications may play a critical role in nicotine-induced cancer development.
Histone H3 on lysine 27 (H3K27) methylation is a reversible process that is catalyzed by histone methyltransferase enhancer of zeste homolog 2 (EZH2), demethylases ubiquitously transcribed TPR gene on the X chromosome (UTX) and jumonji domain-containing protein 3 (JMJD3). 15 H3K27 trimethylation (H3K27me3) is a transcription-suppressive histone mark and correlated with tumour development and/or progression. 15 –19 H3K27me3 mark is a unique epigenetic state of specific cell and the rapid decrease in the H3K27me3 is important for HOX genes expression. UTX gene is located on the X chromosome, and its encoded protein contains a JmjC-domain and catalyzes the demethylation of H3K27me3. UTX plays a central role in stem cell development, cell senescence and carcinogenesis. Up to date, however, the dynamics of H3K27me3 and UTX in nicotine-induced gene expressions have not been investigated.
In this study, we investigated the changes in H3K27me3 and three of the H3K27me3 modifying enzymes EZH2, UTX and JMJD3 in kidney cancer cell line after nicotine treatment. By utilizing a combination of qualitative polymerase chain reaction (qPCR) and Western blot analyses, we found that demethylases UTX is induced in 786-O by nicotine and upregulated expression of UTX is correlated with decreased levels of H3K27me3. The result is beneficial to deepening the understanding of cancer development.
Material and methods
Cell culture and treatment
The renal carcinoma cell line 786-O was obtained from cell resource center of Shanghai Institutes for Biological Sciences, Chinese Academy of Science. 786-O cells were cultured in Dulbecco’s modified Eagle’s medium (GIBCO, Grand Island, Nebraska, USA) supplemented with 10% fetal bovine serum (Hyclone, Logan, Utah, USA). Cells were grown in a humidified atmosphere with 5% CO2 at 37°C. 786-O cells were treated with given concentrations of nicotine (Cerilliant Co., Round Rock, Texas, USA) for 24 h, and untreated cells were used as control. After removal of culture medium, 786-O cells were rinsed with cold phosphate-buffered saline (pH 7.4) and then collected for total RNA and protein extraction.
Quantitative real time-PCR
Total RNA was extracted with Trizol reagent (Invitrogen, Carlsbad, California, USA) according to the manufacturer’s protocol. The total RNA concentration was determined using a NanoDrop ND-1000 spectrophotometer (Thermo Scientific, Wilmington, Delaware, USA). Complementary DNA was synthesized from 2 μg of total RNA using a RT system, according to the manufacturer’s instructions (Invitrogen). The messenger RNA (mRNA) expression levels of UTX, JMJD3 and EZH2 were analyzed using SYBR green PCR Mix (Tiangen, Beijing, China), with 18S ribosomal RNA (rRNA) as an internal reference. Quantitative real time-PCR (qRT-PCR) was performed using a 7500 RealTime PCR System (Applied Biosystems, Foster City, California, USA). Primer sequences were synthesized by Sangon (Shanghai, China) and included: UTX forward 5′-TTTGTCAATTAGGTCACTTCAACCTC-3′ and UTX reverse 5′-AAAAAGGCAGCATTCT TCCAGTAGTC-3′, JMJD3 forward 5′-GGAGGCCACACGCTGCTAC-3′ and JMJD3 reverse 5′-GCCAGTATGAAAGTTCCAGAGCTG-3′, EZH2 forward 5′-GGGACAGTAAA AATGTGTCCTGC-3′ and EZH2 reverse 5′-TGCCAGCAATAGATGCTTTTTG-3′, 18S rRNA forward 5′-CGGCGGCTTT GGTGACTCTAG-3′ and 18S rRNA reverse 5′-CCGTTTCTCA GGCTCCCTCTCC-3′. Relative levels of the three genes were normalized to the internal reference 18S rRNA. Data were analyzed using the comparative threshold cycle (2−ΔCT) method.
Western blot analysis
786-O cells were lysed in radioimmunoprecipitation assay buffer (Beyotime, Haimen, Jiangsu Province, China) and then sonicated. After centrifugation, supernatants were collected and solubilized in loading buffer. A total of 60 μg of protein was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred onto nitrocellulose (NC) membrane. The NC membrane was blocked with 5% non-fat milk dissolved in Tris-buffered saline with Tween 20 (TBST) buffer (50 mM Tris–hydrochloric acid, 150 mM sodium chloride, 0.1% Tween-20) for 2 h and then washed three times with TBST buffer. Blots were incubated with primary antibodies against UTX (1:1000, Abcam, Hong Kong, China), JMJD3 (1:1500, Abcam), H3K27me3 (1:1500, Epigentek, Brooklyn, New York, USA), H3 (1:2000, Sigma-Aldrich, St Louis, Missouri, USA) and β-actin (1:2500, Sigma, St Louis, Missouri, USA) overnight at 4°C. After washing with TBST buffer three times, blots were incubated with secondary anti-rabbit immunoglobulin G conjugated to horseradish peroxidase (1:5000 dilution, Sigma) for 1 h at room temperature. The NC membranes were exposed to enhanced chemiluminescence CL solution (Pierce, Rockford, Illinois, USA) and detection was performed using a Fujifilm LAS-4000 imaging system (GE Healthcare, Bucks, UK). The proteins were quantified by densitometric scanning FUJIFILM Multigauge V3.1 (Fujifilm, Tokyo, Japan).
Statistical analysis
All experiments were repeated three times. All data were expressed as means ± SD. Statistical significance was determined with Student’s t test (two-tailed). A p value of ≤0.05 was considered to indicate a statistically significant difference between treated group and control group.
Results
Nicotine upregulates mRNA expression of UTX
To understand the role of nicotine on H3K27 methylation, the mRNA expression levels of the two H3K27 demethylase genes, UTX and JMJD3, and the H3K27 methyltransferase EZH2 were determined after nicotine treatment by qRT-PCR. The results indicated UTX transcript was significantly increased when treated with 100 or 500 nM of nicotine for 24 h (p < 0.05, Figure 1), while the mRNA levels of JMJD3 and EZH2 were not significantly changed.

qRT-PCR analysis of three H3K27 methylation-modifying enzymes. Relative mRNA level of UTX is obviously increased by nicotine treatment (100 and 500 nM) than control (Con) for 24 h in 786-O cell line (*p < 0.05). The experiment was repeated three times. qRT-PCR: Quantitative real time- polymerase chain reaction; H3K27: histone H3 on lysine 27; mRNA: messenger RNA; UTX: ubiquitously transcribed TPR gene on the X chromosome.
Nicotine increases the protein content of UTX and decreases the level of H3K27me3
In order to further observe the effect of nicotine on H3K27 methylation, the protein contents of UTX, JMJD3 and H3K27me3 were measured by Western blot analysis. Level of UTX protein was obviously increased when treated with 100 or 500 nM nicotine for 24 h (p < 0.05, Figure 2), while the JMJD3 was not obviously affected. The level of H3K27me3 was reduced in accordance with UTX upregulation (p < 0.05, Figure 2).
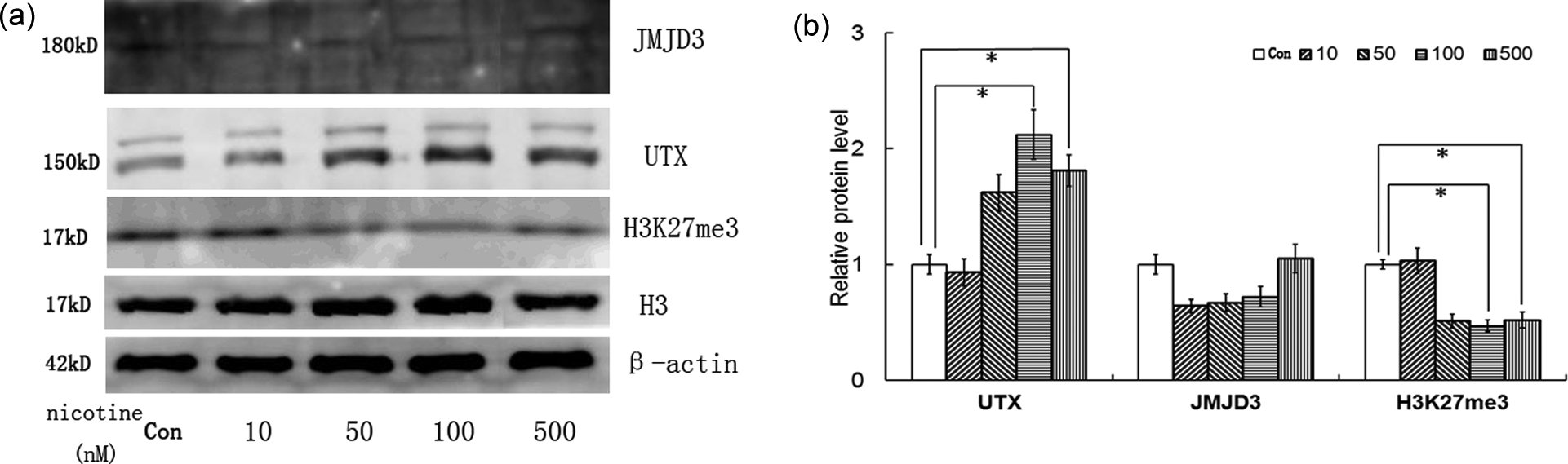
Western blotting analysis of UTX, JMJD3 and H3K27me3. (a) The Western blotting results of three proteins; (b) quantification of the data presented in panel (a). The content of UTX is significantly increased in 786-O cell line after nicotine treatment (100 and 500 nM) than control (Con) for 24 h (*p < 0.05). Similarly, the level of H3K27me3 is lower accompanying with increased UTX (*p < 0.05). The experiment was repeated three times. H3K27: histone H3 on lysine 27; UTX: ubiquitously transcribed TPR gene on the X chromosome; JMJD3: jumonji domain-containing protein 3; H3K27me3: H3K27 trimethylase.
Discussion
Histone methylation has emerged as a critical player in the regulation of gene expression, cell cycle and genome stability, 20 and abnormal histone methylation has been be closely related to tumour formation. 21 Epigenetic alterations including histone methylation are a hallmark in renal tumorigenesis. 22 Till date, several demethylases such as JARIDIC and UTX have been indentified inactivating mutations in RCC. 23 Our previous study has also indicated an association between the H3K9 demethylase JMJD1A and RCC. 24 These facts imply the investigation on histone methylation, which is essential for understanding the RCC development and improve cancer therapy.
In 2007, UTX was identified as one of the histone demethylases, which specifically demethylates H3K27me3. 25 –27 Frequently, mutations of UTX have been identified in many kinds of cancers including kidney, bladder, prostate, leukaemia, and medulloblastoma. 28 –33 Alterations in the expression of UTX are commonly seen in human cancers including prostate and bladder cancer, and our previous research also indicated UTX was upregulated in kidney cancer tissues compared with normal tissues. 34 Because H3K27me3 is highly correlated with gene silencing and repression of transcription, UTX catalyzed demethylation of H3K27me3 is important for gene activation. There are many roles of UTX, including animal posterior development, embryonic development, somatic and germ cell epigenetic reprogramming. 27,35,36 The possible mechanism by which UTX contributes to cancer development is UTX-regulated cell cycle progression through the RB pathway. 37 In vitro and in vivo, UTX regulates the expressions of Retinoblastoma (RB) and RB-binding proteins, which affect cell proliferation as well as cell fate decision. 38 –40
Although a great deal is known about cigarette smoke carcinogens and the mechanisms by which they cause cancer, 41 most of these research are focused on lung cancer. The molecular link between cigarette smoking and RCC is poorly understood. In this study, nicotine induces the expression of UTX and reduced the content of H3K27me3 in kidney cancer cell. These results imply that UTX mediated demethylation of H3K27me3 is one cause of nicotine induced cancer development though activation of specific genes. But whether nicotine-induced UTX upregulation affect the RB pathway in kidney cancer is required to be determined.
Several studies have demonstrated nicotinic acetylcholine receptors (nAChRs) are important in cancer promotion and progression. 42,43 It was determined that nicotine can cause the epigenetic and gene-expression effects. 44,45 Nicotine can change gene expression, thereby affecting cellular replication and differentiation, through the activation of nAChR. 46 After activation of nAChR, nicotine signal transduction was mediated by Ca2+-dependent activation of protein kinase A, protein kinase C and mitogen-activated protein kinase through the induction of the transcription factors nuclear factor-κB and mammalian target of rapamycin (mTOR). 47 It is known that mTOR pathway is essential in kidney cancer development. 48 So we suppose that mTOR may be an important regulator in nicotine induced UTX expression. Our primary study also support the speculation (Guo et al.).
In summary, our results reveal that carcinogenicity of nicotine may be caused by histone methylation, especially H3K27. The increased UTX can change specific gene expression that is dependent or independent of its enzymatic activity, which may be important for cancer development. This study also provides new insight on nicotine-induced carcinogenesis. Further investigation of molecular mechanism underlying UTX involved in cancer development is required.
Footnotes
Authors’ Note
The authors X.G. and X.L. contributed equally to this work.
Acknowledgements
The authors declared no conflicts of interest.
Funding
This research was supported by Emerging Scientist Project of Shenzhen Second People’s Hospital (No 2012001) and National Natural Science Foundation of China (No 30900817).
