Abstract
Objectives:
This study aimed to evaluate the antidotal effect of a newly developed supramolecular complex, ferric porphyrins and a cyclodextrin dimer (FeIIIPIm3CD), that possess a higher binding constant and quicker binding rate to cyanide ions than those of hydroxocobalamin (OHCbl) in the presence of serum protein.
Methods:
First, in vitro cytochrome activity and cell viability were evaluated in murine fibroblast cells cultured with various doses of FeIIIPIm3CD and potassium cyanide (KCN). Next, BALB/c mice were pretreated with intravenous OHCbl (0.23 mmol/kg), FeIIIPIm3CD (0.23 mmol/kg), or saline and then received KCN (lethal dose 100% (LD100): 0.23 mmol/kg) through a stomach tube. Finally, as a resuscitation model, KCN-induced apnea was treated with a bolus injection of an equimolar dose of antidotes followed by a slow infusion of the same reagent.
Results:
FeIIIPIm3CD showed dose-dependent antidotal effects in vitro. Pretreatment with FeIII PIm3CD prevented KCN-induced apnea significantly better than OHCbl. Resuscitation with FeIIIPIm3CD resulted in an earlier resumption of respiration than that seen with OHCbl. However, 24-h survival was similar among the treatments (FeIIIPIm3CD, nine of nine mice; OHCbl, eight of nine mice).
Conclusion:
FeIIIPIm3CD exerted significant antidotal effects on cyanide toxicity in vitro and in vivo, with a potency equal in the mortality of cyanide-poisoned mice or superior in the respiratory status during an acute phase to those of OHCbl.
Introduction
Exposure to cyanide ions (CN−) rapidly results in the loss of consciousness and respiratory arrest,1,2 thus requiring immediate and accurate treatment. 3 Previous antidotes for cyanide poisoning have had several safety and efficacy issues; sodium nitrite may reduce oxygen transport because of methemoglobin overproduction, whereas sodium thiosulfate takes a relatively long time to convert cyanide into thiocyanate.4,5 Hydroxocobalamin (OHCbl) has been reported to be a safer antidote because it binds to CN−, forming cyanocobalamin, which is then excreted through the urine. 6 Although its binding constant to CN− is high, this is reduced to approximately 1/600 in the presence of serum proteins (Table 1).7,8 In addition, OHCbl has a slow rate of binding to CN− (Table 1), which may cause a delay in action and indirectly result in inadequate effects. 7 Kano and colleagues7,8 synthesized a supramolecular complex, ferric porphyrins and a cyclodextrin dimer (FeIIIPIm3CD; Figure 1(a)), which is composed of a per-O-methylated β-cyclodextrin dimer with an imidazole linker (Im3CD) and 5,10,15,20-tetrakis-(4-sulfonatophenyl) porphinatoiron (FeIIITPPS). Although the binding constant of FeIIIPIm3CD to CN− is lower than that of OHCbl in vitro without serum proteins, only a 50% reduction was observed in the presence of serum proteins, 7 resulting in a much higher binding constant and quicker binding rate in vivo (Table 1).7,8 Based on these findings, we evaluated the antidotal effects of FeIIIPIm3CD and compared these effects to those of OHCbl in vitro and in vivo.
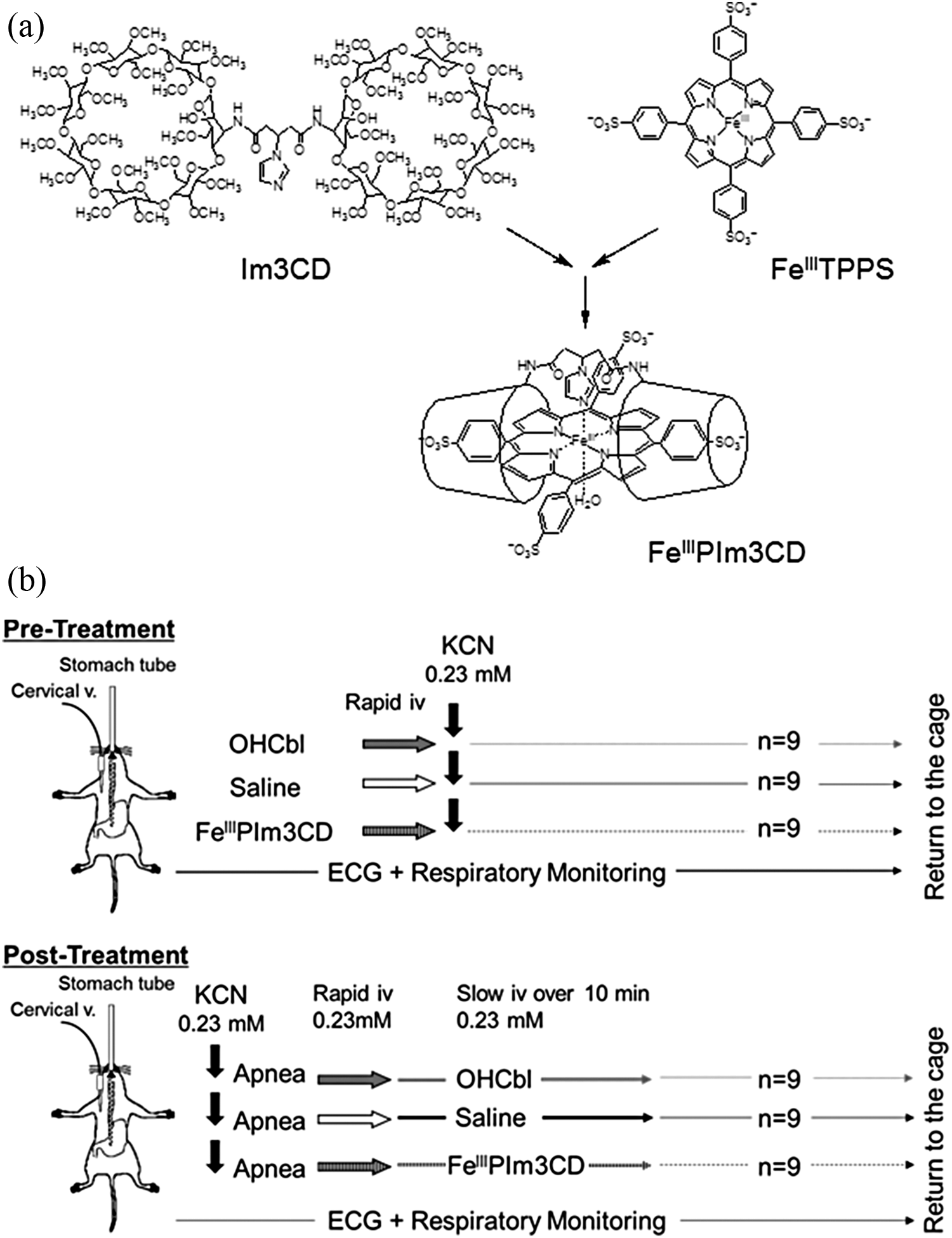
(a) Structure of FeIIIPIm3CD. Im3CD and FeIIITPPS were mixed to prepare FeIIIPIm3CD as reported by Kano and collegues.7,8 (b) Pretreatment procedures (upper panel). Mice were pretreated with intravenous FeIIIPIm3CD, OHCbl, or saline and were then administered with intragastric KCN. (b) Posttreatment procedures (lower panel). Intragastric KCN-induced apnea was immediately treated with intravenous FeIIIPIm3CD, OHCbl, or saline, followed by a slow infusion of another equimolar dose of the same reagent for 10 min. FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; Im3CD: per-O-methylated β-cyclodextrin dimer with an imidazole linker; FeIIITPPS: 5,10,15,20-tetrakis-(4-sulfonatophenyl) porphinatoiron; OHCbl: hydroxocobalamin; KCN: potassium cyanide.
Physical characteristics of FeIIIPIm3CD and OHCbl.

FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; OHCbl: hydroxocobalamin.
a T 1/2 indicates the half-life (h) in the blood after intravenous administration in human volunteers (OHCbl) or in Wistar rats (FeIIIPIm3CD).
Methods
Ethical statement
The present study was approved by the Institutional Animal Care and Use Committee of Tokai University (Kanagawa, Japan) and was conducted in accordance with the Guidelines for the Care and Use of Animals for Scientific Purposes of Tokai University.
Chemicals
OHCbl was purchased as Cyanokit (Merck Serono, Japan). Im3CD and FeIIITPPS (Figure 1(a)) were mixed to prepare FeIIIPIm3CD before use in every experiment.7,8
In vitro studies
In vitro studies were conducted as described previously (Yamagiwa et al., submitted for publication). Briefly, NIH-3T3 murine fibroblast cells (Cell Bank, RIKEN, Japan) were seeded in a 96-well microplates (Iwaki, Japan) at a density of 1 × 104 per well and exposed to various concentrations of potassium cyanide (KCN; Wako, Japan) and FeIIIPIm3CD in 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES)-buffered media (pH 9.2) for 1 h at 37°C in 100% air. After incubation, mitochondrial integrity was measured in terms of reduction in mitochondrial enzymatic activity using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, 10 and cell viability was determined using the trypan blue dye exclusion (TBDE) assay (Yamagiwa et al., submitted for publication).
In vivo studies
Male BALB/c mice (8–10 weeks old) were purchased from CLEA (Japan). The mice were reared in polypropylene cages in a room maintained at a constant temperature of 21–22°C and a 12-h light/12-h dark cycle and were fed with rodent diet CE-2 (CLEA, Japan) and provided water ad libitum. The mice underwent 12-h fasting before experiment.
Pretreatment study
Mice were anesthetized through inhalation of 2% sevoflurane and maintained under spontaneous respiration with 1.0 L/min of indoor air and 1–2% sevoflurane using a facemask (Figure 1(b); upper panel). A 24-G indwelling catheter was inserted into the jugular vein. Saline (Otsuka, Japan; 0.9% sodium chloride), FeIIIPIm3CD (181.7 mg/mL in saline), or OHCbl solution (64.7 mg/mL in saline) was injected into the mice through the jugular vein using an aliquot with a concentration of 5 mL/kg (0.23 mmol/kg for FeIIIPIm3CD or OHCbl, n = 9 in each group). A stomach tube (FUCHIGAMI, Japan; length: 38 mm, φ0.92 × 0.62 mm) was then immediately inserted to administer KCN (1.5 mg/mL in saline), using an aliquot with a concentration of 10 mL/kg (0.23 mmol/kg, the oral lethal dose 100% (LD100) in mice). 11
Posttreatment study
KCN solution, administered through the stomach tube at a concentration of 10 mL/kg (0.23 mmol/kg), rapidly resulted in respiratory arrest. Immediately after that, FeIIIPIm3CD, OHCbl, or saline were administered by rapid intravenous injection using an aliquot with a concentration of 5 mL/kg (0.23 mmol/kg for both FeIIIPIm3CD and OHCbl), followed by continuous injection of another equimolar dose for the next 10 min (n = 9 in each group) (Figure 1(b), lower panel). Each mouse eventually received an antidote dose double (0.46 mmol/kg) that of KCN by the end of the experiment.
Assessments
The antidotal effect was evaluated in terms of the respiratory pattern and heart rates during the first 20 min after KCN administration and by the 24-h survival rate. Respiratory rate was monitored by Moor FLPI Laser Doppler flowmetry (Moor, UK); four regions of interest were placed on both sides of the precordial region and the abdomen of the mice to monitor the thoracic and abdominal wall motions, and amplitudes over 1000 perfusion units were recorded as respiratory motion. Respiratory arrest was defined as the time point after 3 s of continuous apnea. Time of death was defined as the time point 5 min after the termination of spontaneous respiration. Heart rate was measured by a lead II inductive electrocardiography connected to PowerLab® (AD Instruments, Bella Vista NSW, Australia). After the animals were monitored for 20 min, the surviving animals were returned to their cages.
Statistical analysis
Statistical analyses conducted using Prism 5.0. Values obtained by colorimetric analysis, respiratory, and heart rates were compared using the Mann-Whitney U test. The incidence of apnea and the 24-h survival rate were compared using Fisher’s exact test. Survival rate was calculated using the Kaplan-Meier method. Differences were considered significant if the p value was <0.05.
Results
In vitro studies
Exposure to FeIIIPIm3CD alone did not significantly suppress cytochrome activity (Figure 2(a)) or cell viability (Figure 2(b)). FeIIIPIm3CD showed dose-dependent antidotal effects on cytochrome activity as monitored using the MTT assay (Figure 3(a)), and cell viability, as measured using the TBDE assay (Figure 3(b)). An equimolar amount of FeIIIPIm3CD completely reversed KCN toxicity; no significant differences in cytochrome activity and cell viability were observed between this treatment group and the control without KCN.

(a) The effects of FeIIIPIm3CD on cytochrome activity evaluated by MTT assay. (b) The effects of FeIII PIm3CD on cell viability evaluated by TBDE assay. Various concentrations of FeIIIPIm3CD in HEPES-buffered media (pH 9.2) were added to the cells (n = 6 in each group), which were then incubated for 1 h at 37°C in 100% air. The effect of FeIIIPIm3CD was evaluated in terms of cytochrome activity by MTT assay (a) and cell viability by TBDE assay (b). FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; TBDE: trypan blue dye exclusion; HEPES: 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid.

(a) The antidotal effect of FeIIIPIm3CD evaluated by MTT assay. (b) The antidotal effect of FeIIIPIm3CD evaluated by TBDE assay. Various doses of FeIIIPIm3CD and KCN in HEPES-buffered media (pH 9.2) were added simultaneously to the cells (n = 6 in each group), which were then incubated for 1 h at 37°C in 100% air. The antidotal effects of FeIIIPIm3CD were evaluated by the MTT assay (a) and by TBDE assay (b). *Significant at p < 0.05 compared to the control with no KCN or FeIIIPIm3CD. FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; TBDE: trypan blue dye exclusion; KCN: potassium cyanide; HEPES: 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid.
In vivo pretreatment study
Although there was no response to the pretreatments (Figure 4(a)), all animals developed tachypnea immediately after KCN administration. Tachypnea gradually resolved among the FeIIIPIm3CD-pretreated mice, whereas reduction in the respiratory rate to apnea was observed in five of nine (55.6%; p < 0.05) of the OHCbl-pretreated mice and in nine of nine (100%) of the saline-pretreated mice (Table 2). Although the OHCbl-pretreated mice resumed its respiratory rate to its baseline level within 4 min after KCN administration, the saline control group showed termination of respiration approximately 10 min after KCN administration. Bradycardia was observed 1.5 min after KCN administration only in the saline group, whereas the OHCbl and FeIIIPIm3CD groups showed stable heart rates (Figure 4(b)). The 24-h survival rates (Figure 4(c)) of the FeIIIPIm3CD (eight of nine mice, 88.9%) and OHCbl groups (eight of nine mice, 88.9%) were similar, whereas no saline-pretreated mice survived 24-h (zero of nine mice, 0.0%).

Changes in the respiratory pattern (a), heart rate (b), and survival (c) in pretreated mice. Mice were pretreated with intravenous FeIIIPIm3CD (circles), OHCbl (rectangles), or saline (asterisks) and received intragastric KCN. FeIII PIm3CD: ferric porphyrins and a cyclodextrin dimmer; OHCbl: hydroxocobalamin; KCN: potassium cyanide.
Animals, cyanide treatments, and outcomes of the pretreatment study.

FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; OHCbl: hydroxocobalamin.
Posttreatment study
Tachypnea and apnea occurred in all mice within 1 min of KCN administration (Figure 5(a)) and no significant differences were observed in the time of resumption of respiration (Table 3). The respiratory frequency at 2–3.5 min after KCN administration was significantly higher in the FeIIIPIm3CD group than in the OHCbl group, although these differences were not significant at the 4-min time point and thereafter (Figure 5(a)). In contrast, saline-treated control mice did not resume regular respiration and ceased gasping approximately 10 min after KCN administration. All mice showed temporary tachycardia (Figure 5(b)) for approximately 30 s after KCN administration and the heart rates returned to the baseline levels in FeIII PIm3CD and OHCbl groups, whereas the saline-treated mice showed remarkable and continuous bradycardia similar to that observed after pretreatment. The 24-h survival rates (Figure 4(c)) of the FeIIIPIm3CD (nine of nine mice, 100%) and OHCbl groups (eight of nine mice, 88.9%) were similar and significantly better than that of the saline-treated control mice (zero of nine mice, 0%).

Changes in the respiratory pattern (a), heart rate (b), and survival (c) in posttreated mice. Mice had KCN-induced apnea and were immediately treated with intravenous FeIIIPIm3CD (circles), OHCbl (rectangles), or saline (asterisks). *Significant at p < 0.05 compared to OHCbl. FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; KCN: potassium cyanide; OHCbl: hydroxocobalamin.
Animals, cyanide treatments, and outcomes of the posttreatment study.

FeIIIPIm3CD: ferric porphyrins and a cyclodextrin dimmer; OHCbl: hydroxocobalamin.
Discussion
Administration of FeIIIPIm3CD resulted in a dose-dependent antidotal effect on KCN in terms of cytochrome activity and cell viability in vitro. An equimolar amount of FeIIIPIm3CD totally reversed KCN toxicity, in contrast to the significant difference observed with OHCbl in our previous study (Yamagiwa et al., submitted for publication). The difference may be attributable to the binding constant of OHCbl, which was drastically reduced in the presence of serum proteins 7 as well as its slower binding rate compared to that of FeIIIPIm3CD (Table 1). The difference may be attributable to the presence of serum proteins in the media, which holds true during in vivo resuscitation.
Although intravenous administration of FeIII PIm3CD exerted antagonistic effects similar to those of OHCbl in terms of heart rate and 24-h survival, respiratory patterns showed significantly better and/or faster recovery; less frequent apnea in the pretreatment study and faster recovery of respiratory rate from apnea were observed in the resuscitation model. These differences during the acute phase of exposure to KCN may be attributable to the higher binding rate of FeIIIPIm3CD to CN−, which is 16 times faster than that of OHCbl (Table 1), and the higher binding constant in the presence of serum proteins in the blood. 7
OHCbl is currently used as a prehospital treatment for hydrogen cyanide (HCN) poisoning, which is commonly associated with carbon monoxide (CO) poisoning during house fires. 12 Interference of CO hemoglobin (COHb) levels in patients treated with OHCbl poses a serious problem because it may result in a delay in diagnosis and/or treatment for CO poisoning. 13 Although this has been attributable to the 532-nm peak wavelength of OHCbl, which is close to the peak wavelengths of 525 and 575 nm of CO-oximetry and COHb, respectively, 14 this interference in the measurement of serum COHb levels by FeIII PIm3CD was not observed (Yamagiwa et al., submitted for publication) because its peak wavelength at pH 7.0 and 25°C is 422 nm. 8
The half-life of the injected FeIIIPIm3CD in the blood was much shorter than that of OHCbl (Table 1); approximately 69% of the infused FeIIIPIm3CD was excreted in the urine within 1 h compared to 72 h required for the excretion of the infused OHCbl in the urine. 9 While a prefixed dose of OHCbl (75 mg/kg) is used in clinical resuscitation, monitoring the urinary FeIIIPIm3CD level, conjugated or unconjugated with cyanide, 7 may serve as a surrogate to determine the existing amount of cyanide in vivo and enable to decide the dose and timing of addition or cessation of FeIIIPIm3CD administration. Although such a short intravascular half-life may be disadvantageous for prophylaxis, the half-life may be prolonged by chemical modification. 15 Therefore, further studies are required to determine the pharmacokinetics of FeIIIPIm3CD in the extracellular fluid, similar to the observations in in vitro study (Yamagiwa et al., submitted for publication), as well as in the intracellular environment and to determine the putative domain of CN− toxicity.1–3
We focused on respiratory patterns after cyanide administration and used spontaneous respiration as a primary outcome measure to observe KCN toxicity and efficacy of antidotes. Therefore, our study does not clarify the exact efficacy of FeIIIPIm3CD against cyanide in smoke inhalation during house fires. Because orally administered KCN reacts with gastric juices and is converted to and absorbed as HCN, it simulates the self-poisoning, which is another clinical scenario of cyanide intoxication. Dose–response evaluation, delayed treatment, and alternative routes of administration such as the intramuscular route or bone-marrow injection in the case of mass casualties are currently being investigated to simulate clinical resuscitation. Thus, not only acute cyanide toxicity and reversal but also the long-term toxicity of FeIII PIm3CD on the functional and morphological features of organs and the entire body should be addressed in future studies.
In conclusions, FeIIIPIm3CD exerts significant antidotal effects on cyanide toxicity in vitro and in vivo, with a potency equal in the mortality of cyanide-poisoned mice or superior in the respiratory status during an acute phase to those of OHCbl.
Footnotes
Acknowledgments
We thank Masako Kidokoro for her technical advice and Education and Research Support Center, Tokai University for technical assistance.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was supported by a Grant-in-Aid for Young Scientists (B) (23792089), a Grant-in-Aid for Challenging Exploratory Research (23659749), and a Grant-in-Aid for Scientific Research (A) (24249086).
