Abstract
The effect of exposure to inhaled lead acetate in guinea pigs was evaluated. The present study comprised of five groups of guinea pigs including control (C), sensitized to ovalbumin (OA; S) and three groups exposed to 0.1, 0.2, and 0.4 M inhaled lead (Pb; n = 6 for each group). Tracheal responsiveness to methacholine and OA, total and differential white blood cells (WBCs) count in lung lavage, serum levels of cytokines (interferon γ (IFN-γ) and interleukin 4 (IL-4)), histamines, and immunoglobulin E (IgE), and Pb concentration in lung were measured. Tracheal responsiveness to methacholine, OA, total and differential WBC types as well as IL-4, IFN-γ, histamine, and IgE were significantly increased but IFN-γ/IL-4 were significantly decreased in sensitized animals as well as those exposed to high Pb concentrations when compared with the control group (from p < 0.05 to p < 0.001). In addition, there was not a significant difference in most measured values between animals exposed to high Pb concentration and group S. The Pb concentration in lung tissues of animals exposed to all three Pb concentrations was significantly higher than that of group C (p < 0.001 for all cases).These results showed that inhaled lead acetate exposure can induce lung inflammatory changes similar to sensitized animals. Therefore, exposure to environmental Pb pollution may cause asthma-like changes.
Introduction
Exposure to pollutants, such as ozone and various components of particulate matter including transition metals, diesel exhaust, and biological substances such as endotoxins are the causes of asthma exacerbation. Among many air toxic elements, lead (Pb) is one that is highly toxic and has no known beneficial effects in the body. 1 –3 Although the respiratory system is the major target organ of Pb-induced toxicity, the effects of environmental Pb on respiratory system are not yet well known. 4,5 The contribution of Pb pollution to pathogenesis of pulmonary cancers, asthma, and chronic obstructive pulmonary disease is suggested, but the results in this field are still controversial. 6 –8 There are reports regarding increased incidence of asthma in workers exposed to Pb and in children living in places with high level of Pb pollution. 9,10
There are also reports regarding increase of immunoglobulin E (IgE) levels and some inflammatory cytokines in serum of laboratory models and children exposed to Pb as well as release of inflammatory mediators from T helper (Th) cells and macrophages exposed to Pb in a cell culture model. However, some studies indicated a decrease of serum Igs in animals exposed to Pb. 11 –16 In experimental studies on animal models exposed to Pb, morphological changes in the respiratory system as well as increased tracheal responsiveness were observed. 17 These observations can demonstrate the role of Pb in pathogenesis of asthma disease but definite conclusion regarding the role of Pb in the pathogenesis of asthma or other respiratory diseases is not yet available. 17 Therefore, more studies are needed for the evaluation of the immunotoxic effects of Pb exposure and the role of this metal in developing severity of asthma and other respiratory diseases.
Airway hyperresponsiveness (AHR) to different stimuli 18 is the main characteristic feature of asthma, which is due to airway inflammation. 19 Airway inflammation correlates closely with asthma severity and AHR. 19 Various inflammatory cells with overproduction of inflammatory mediators are involved in pathogenesis of airway inflammation in asthma. 20 The immunologic feature of asthma is shifting from Th1 to Th2 cells, which leads to the airway inflammation and AHR. 21
In asthmatic patients and animal model of asthma, increased white blood cells (WBC), especially eosinophil counts, 22,23 and serum histamine levels 24 were documented. There is also a link between total IgE and asthma that is reported to be independent of allergen sensitization. 25,26
In the present study, tracheal responsiveness, total and differential blood cell count in lung lavage, and serum cytokine levels in guinea pigs exposed to inhaled Pb were examined.
Methods
Exposure of animals to Pb, animals sensitization, and groups
A total of 30 adult Dunkin Hartley guinea pigs (400–700 g, both sexes) were used throughout the study. They were allowed to get acclimatized to the laboratory conditions for 10 days. The animals were group housed in individual cages in climate-controlled animal quarters and were given water and food ad libitum, while a 12-h on/12-h off light cycle was maintained. Animals were randomly divided to five groups as follows (n = 6 for each group)
Control group (nonexposed animals to Pb, group C) Animals exposed to 0.1 M Pb concentration (group 0.1 M Pb) Animals exposed to 0.2 M Pb concentration (group 0.2 M Pb) Animals exposed to 0.4 M Pb concentration (group 0.4 M Pb) Sensitized to ovalbumin (OA) and nonexposed to Pb (group S)
Animals exposure to lead acetate (groups 0.1 M Pb, 0.2 M Pb, and 0.4 M Pb) was performed according to the method described by Fortoul et al. 27 Briefly, different animal groups were exposed to aerosol of three lead acetate concentrations of 0.1, 0.2, and 0.4 M (Sigma Chemical Co., St Louis, Missouri, USA) in a closed chamber (30 × 20 × 20 cm) connected to an ultranebulizer (Ultra-Neb 99, DeVilbiss Healthcare GmbH, Kamenzer, Mannheim, Germany) with an air flow of 10 L min−1 for 1 h, thrice a week for 2 weeks.
Sensitization of animals (group S) to OA was performed using the method described previously.
28
–30
Briefly, guinea pigs were sensitized to 10 mg OA (Sigma Chemical Ltd, UK) and 100 mg aluminum hydroxide (Al(OH)3) intraperitoneal
Tissue preparation
Sensitized, control, and Pb-exposed animals were killed the next day after the end of inhalation period (for sensitized control groups, 33 days and for Pb-exposed animals, 15 days after the beginning of protocol) and trachea was removed. In each animal, one tracheal chain was prepared as follows: trachea was cut into eight rings (each containing —two to three cartilaginous rings). All the rings were sutured together by cotton thread to form a tracheal chain, and their cartilage were cut open opposite the trachealis muscle. 31,32 Tissue was then suspended in a 20-mL organ bath (Schuler organ bath type 809, Hugstetten, March, Germany) containing Krebs–Henseleit solution with the following composition: sodium chloride, 120 mM; sodium bicarbonate, 25 mM; magnesium sulphate, 0.5 mM, monopotassium phosphate, 1.2 mM, potassium chloride, 4.72 mM, calcium chloride, 2.5 mM, and dextrose, 11 mM. The Krebs solution was maintained at 37°C and gassed with 95% oxygen and 5% carbon dioxide. Tissue was suspended under isometric tension of 1 g and allowed to equilibrate for at least 1 h while it was washed with Krebs solution every 15 min.
Responses were measured using Vernier control type 850 N sensor with the sensitivity range of 0–20 g and resolution of 0.2 mm turn−1 (Hugo-Sachs Elektronik, March, Germany) and amplified using an ML/118 quadribridge amplifier (Hugstetten, March, Germany) and recorded on Powerlab (ML-750, four channel recorder, Hugstetten, March, Germany).
Assessment of tracheal response to methacholine
In each experiment, a cumulative log concentration–response curve of methacholine hydrochloride (Sigma Chemical Ltd, UK)-induced contraction of tracheal chain was obtained. Consecutive concentrations (including 10−7–10−1 mM dissolved in saline) were added every 3 min, and the contraction due to each concentration was recorded at the end of 3 min with the effect reaching a plateau in all the experiments. To obtain the curve, the percentage of contraction of the tracheal smooth muscle due to each concentration of methacholine in proportion to the maximum contraction obtained by its final concentration was plotted against log concentration of methacholine. A concentration–response curve of methacholine was obtained for tracheal chain of each studied animal.
The effective concentration of methacholine, causing 50% of maximum response (EC50) was measured from methacholine response curve in each experiment using EC50 response in Y axis and measuring the dose of methacholine causing this response in X axis. Contractility response to 10 μM methacholine as the magnitude of contraction was also measured.
Measurement of tracheal response to OA
The tracheal response to 0.1% solution of OA was measured in each studied animal as follows: 0.5 mL of 4% OA solution (dissolved in saline) was added to 20 mL organ bath and the degree of tracheal chain contraction was recorded after 15 min and was expressed in proportion (in percentage) to the contraction obtained by 10 µM methacholine. The measurement of tracheal response to methacholine and OA was performed in random order.
Lung lavage and its WBC count
When preparing the tracheal chain, the cannula was also located in the remaining trachea and the lungs were lavaged with 5 mL of saline for 4 times (total: 20 mL). To this, 1 mL of lung lavage fluid (LLF) was stained with Turk’s solution and counted in duplicate in a hemocytometer (in a Burker chamber). The Turk solution consisted of 1 mL of glacial acetic acid, 1 mL of 1% gentian violet solution, and 100 mL distilled water.
The remaining LLF was centrifuged at 2500g at 4°C for 10 min and the supernatant was removed. The smear was prepared from the cells and stained with Wright–Giemsa stain. According to staining and morphological criteria, differential cell analysis was carried out under a light microscope by counting 400 cells and the percentage of each cell type was calculated.
Measurement of serum IL-4, IFN-γ, IgE, and histamine levels
For the measurement of serum levels, 5 mL of peripheral blood was obtained and placed at room temperature for 1 h. The samples were then centrifuged at 3500g at 4°C for 10 min. The supernatant was collected and immediately stored at −70°C until analyzed. Finally, the serum levels of interleukin 4 (IL-4) and interferon γ (IFN-γ) were measured using enzyme-linked immunosorbent assay sandwich method according to the manufacturer’s instructions (IL-4 assay kit-628 and IFN-γ assay kit-628, Bender MedSystems Co., South San Francisco, Burlingame, USA; IgE Assay Kit, PT-IgE-96 Co., Pishtaz, Iran; histamine assay kit-1100, BAE Systems, London, England). The ratio of IFN-γ/IL4 as an index of Th1/Th2 was also calculated.
Measurement of Pb concentration in lung tissue
Pb concentration in lung tissue was measured according to the method described by Fortoul et al. using a graphite furnace atomic absorption spectrometer (model 2380, Perkin-Elmer, Boston, MA, USA). 27
Statistical analysis
All experiments were carried out at least in duplicate. Every group consisted of six guinea pigs. One-way analysis of variance was performed and Tukey’s post hoc test was used for multiple comparisons. Statistical analyses were performed using the InStat 3.0 program (GraphPad Software, Inc., La Jolla, USA). The results are expressed as mean ± SEM. Linear correlation tests were also performed. Differences of p < 0.05 were considered significant.
Results
Concentration response curves to methacholine showed leftward shift in group S and those exposed to two higher Pb concentrations as compared to group C (Figure 1).

Cumulative log concentration–response curves of methacholine induced contraction of isolated trachea in control (C), sensitized (S), Pb-exposed guinea pigs, exposed to 0.1, 0.2, and 0.4 M Pb concentrations (for each group, n = 6). Pb: lead.
Tracheal responsiveness and contractility to methacholine as well as tracheal responsiveness and maximum response to OA in group S, tracheal responsiveness to methacholine in guinea pigs exposed to two higher (0.2 and 0.4 M) Pb concentrations, contractility to methacholine and maximum response to OA in guinea pigs exposed to high Pb concentration (0.4 M), and tracheal responsiveness to OA and Pb concentration in lung tissue of animals exposed to all Pb concentrations were significantly higher than those of group C (p < 0.05 – p < 0.001). There was no statistically significant difference in tracheal responsiveness to methacholine, tracheal responsiveness, and maximum response to OA between the animals exposed to high Pb concentration (0.4 M) and group S. Tracheal responsiveness to methacholine and OA in guinea pigs exposed to high Pb concentration (0.4 M) and tracheal responsiveness to methacholine in those exposed to medium concentration (0.2 M) were significantly higher than those exposed to low Pb concentration (0.1 M; p < 0.01 – p < 0.001; Figures 2 and 3). There were also significant differences in Pb concentration of lung tissue between animals exposed to three levels of Pb concentrations (p < 0.01 – p < 0.001; Figure 4).
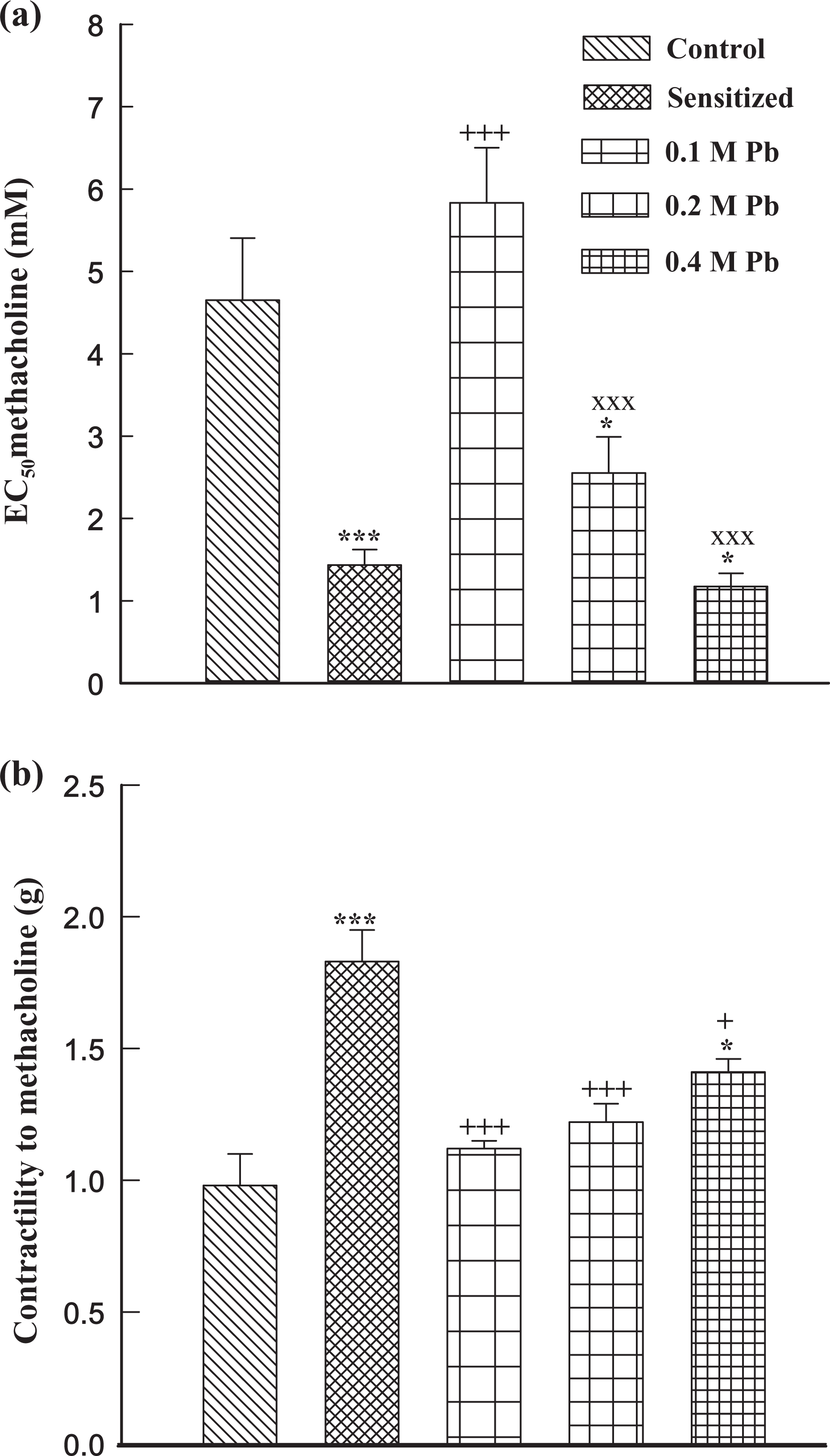
Mean ± SEM values of tracheal response to methacholine, EC50 (a) and tracheal contractility response to 10 μM methacholine (b) in control (C), sensitized (S), Pb-exposed guinea pigs, exposed to 0.1, 0.2, and 0.4 M Pb concentrations (for each group, n = 6). *p < 0.05; ***p < 0.001: statistical significance for the difference between the data of control and other groups. +p < 0.05; +++p < 0.001: statistical significance for the difference between the data of sensitized and Pb-exposed groups. ×××p < 0.001: statistical significance for the difference between the data of 0.4 and 0.2 M vs 0.1 M Pb groups. EC50: 50% maximal effective concentration; Pb: lead.
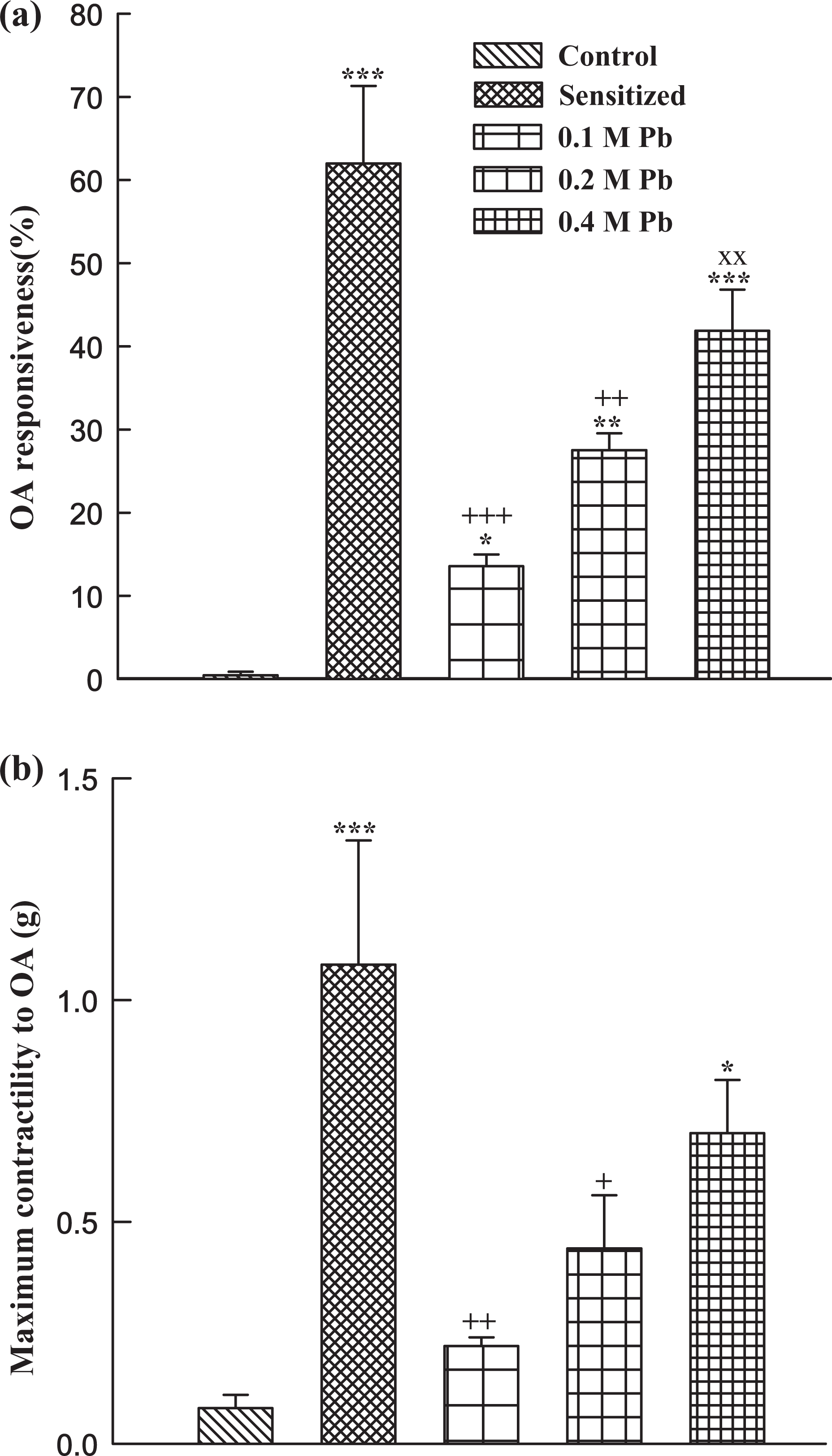
Mean ± SEM values of tracheal response to OA (percentage concentration in proportion to contraction obtained by 10 µM methacholine; (a)) and maximum tracheal contractility response to OA (b) in control (C), sensitized (S), Pb-exposed guinea pigs, exposed to 0.1, 0.2, and 0.4 M Pb concentrations (for each group, n = 6). *p < 0.05; **p < 0.01; ***p < 0.001: Statistical significance for the difference between the data of control and other groups. +p < 0.05; ++p < 0.01; +++p < 0.001: Statistical significance for the difference between the data of sensitized and Pb-exposed groups. ××p < 0.01: Statistical significance for the difference between the data of 0.4 and 0.2 M vs 0.1 M Pb groups. OA: ovalbumin; Pb: lead.

Mean ± SEM values of Pb concentration in the lung of control (C) and Pb-exposed guinea pigs, exposed to 0.1, 0.2, and 0.4 M Pb concentrations (for each group, n = 6). Pb concentration in the lung of group C was zero (0. ± 0.00). *** p < 0.001: Statistical significance for the difference between the data of control and other groups. ×××p < 0.001: Statistical significance for the difference between the data of 0.4 and 0.2 M vs 0.1 M Pb groups. ###p < 0.001: Statistical significance for the difference between the data of 0.4 M Pb and 0.2 M Pb group. Pb: lead.
Total and all differential WBC counts in lung lavage of group S and animals exposed to all Pb concentrations were significantly higher than those of group C (p < 0.001). In addition, total WBC counts in lung lavage of animals exposed to all Pb concentrations were significantly higher than those of group S (p < 0.05 for total WBC counts of 0.1 M Pb group and p < 0.001 for other cases). All differential WBC counts in lung lavage of animals exposed to high Pb concentration (0.4 M) and lymphocyte count in animal exposed to medium Pb concentration (0.2 M) were also significantly higher than those exposed to low Pb concentration (0.1 M; p < 0.05 – p < 0.01; Figure 5).

Mean ± SEM values of total WBC (a) and percentage of different types of WBC (b) in LLF in control (C), sensitized (S), Pb-exposed guinea pigs, exposed to 0.1, 0.2, and 0.4 M Pb concentrations (for each group, n = 6). The data of WBC means that their count in 1 mL of LLF and those of each type means the percentage of total WBC. *p < 0.05; **p < 0.01; ***p < 0.001: Statistical significance for the difference between the data of control and other groups. +p < 0.05; ++p < 0.01; +++p < 0.001: Statistical significance for the difference between the data of sensitized and Pb-exposed groups. ×p < 0.05; ××p < 0.01: Statistical significance for the difference between the data of 0.4 M and 0.2 M vs 0.1 M Pb groups. WBC: white blood cells; LLF: lung lavage fluid; Pb: lead.
Serum levels of IFN-γ, IL-4, histamine, and IgE in group S and in animals exposed to low and high (0.1 and 0.4

Serum levels of IL-4 and IFN-γ (picogram per milli liter) in control (C), sensitized (S) Pb-exposed guinea pigs, exposed to 0.1 and 0.4 M Pb concentrations (for each group, n = 6). Values are presented as mean ± SEM. *p < 0.05; **p < 0.01; ***p < 0.001: Statistical significance for the difference between the data of control and other groups. +p < 0.01; +++p < 0.001: Statistical significance for the difference between the data of sensitized and Pb-exposed groups. ×p < 0.05: Statistical significance for the difference between the data of 0.4 and 0.1 M Pb groups. IFN-γ: interferon γ; IL-4: interleukin 4; Pb: lead.

Serum levels of histamine and IgE (nanogram per milli liter) in control (C), sensitized (S) and Pb-exposed guinea pigs, exposed to 0.1 and 0.4 M lead concentrations (for each group, n = 6). Values are presented as mean ± SEM. *p < 0.05; ***p < 0.001: Statistical significance for the difference between the data of control and other groups. ++p < 0.01; +++p < 0.001: Statistical significance for the difference between the data of sensitized and Pb-exposed groups. ××p < 0.01; ×××p < 0.001: Statistical significance for the difference between the data of 0.4 and 0.1 M Pb groups. IgE: immunoglobulin E; Pb: lead.
Discussion
In order to examine the effects of environmental Pb pollution (inhaled Pb) on respiratory system, in the present study, the effects of Pb on specific and nonspecific tracheal responsiveness (responsiveness to methacholine and OA, respectively), total and differential WBC count, serum inflammatory cytokines, and mediators were evaluated. The results of the present study showed significant increase in tracheal responsiveness to methacholine and OA, total and differential WBC number, serum IL-4, IgE, and histamine but decrease in serum IFN-γ level and IFN-γ/IL-4 ratio in Pb-exposed guinea pigs compared with the control animals. The same findings were also observed in sensitized animals. The increased Pb concentration in lung tissue of guinea pigs exposed to inhaled Pb was also observed.
The increased total WBC number and eosinophil count are well-known changes in asthma disease. 22,23 Increased total WBC and eosinophil in the lung lavage of sensitized guinea pigs were seen in our previous studies. 33,34 The results of the present study also showed increased total WBC number and eosinophil count in sensitized and Pb-exposed animals. In addition, the present study showed increased serum IgE and histamine levels in sensitized animals and those exposed to Pb. Increased serum histamine and IgE levels in asthmatic patients and sensitized animals were also observed. 24 –26 Increased total WBC, eosinophil, serum histamine, and IgE levels in sensitized animals and those exposed to inhaled Pb indicate that Pb exposure could induce lung inflammation, the important change of lungs in asthma. The reason for decreased lymphocyte count in sensitized animals and those exposed to two higher Pb concentrations is their expression in percentage due to significant increase in total WBC. However, the absolute lymphocyte count show their significant increase in sensitized group and those exposed to all Pb concentrations (1123.76 ± 6.59, 1454.05 ± 6.47, 2963.02 ± 3.05, 2981.99 ± 6.92, and 2551.11 ± 4.43 mL−1 for groups C, S, and those exposed to three Pb concentrations (0.1, 0.2, and 0.4 mM), respectively, p < 0.001 for all cases compared with group C). In fact, increased lymphocyte count was observed in Pb-exposed rats, which supports the findings of the present study. 35
Increased nonspecific and specific tracheal responsiveness were observed in the present study in sensitized as well as Pb-exposed guinea pigs. Airway inflammation is the main characteristic feature of asthma, 19 which leads to AHR to many stimuli. 18 Airway inflammation correlates closely with AHR and severity of asthma. 19 Our previous studies also showed increased tracheal responsiveness to methacholine and OA in sensitized guinea pigs using the same method of sensitization. 29,30 Contraction of airway smooth muscle due to subacute exposure to Pb was reported previously. 36 Morphologic changes in the respiratory system as well as increased tracheal responsiveness were also observed in animal models exposed to Pb. 17 These studies support the increased tracheal responsiveness seen in the present study. Therefore, the results showed that inhaled Pb exposure can induce airway inflammation (increased total and differential WBC counts in LLF and serum levels of histamine and IgE), which caused increased specific and nonspecific AHR.
Significant increase in serum IL-4, but decreased serum IFN-γ level and IFN-γ/IL-4 ratio (indicating reduction in Th1/Th2 balance) in Pb-exposed guinea pigs were observed in the present study. An imbalance in the Th1/Th2 cytokine immune response is the immunologic feature of asthma. Th2 is activated in asthma disease and cause airway inflammation and AHR. 21 However, Th1 cells can inhibit Th2 response and thus regulate the anti-inflammatory response. 37 Th1 lymphocytes release IL-2 and IFN-γ, and Th2 cells produce IL-4 and IL-10. 38 Increased serum IL-4 and IFN-γ were also shown in sensitized animals. 33,34 A previous study also showed the possibility that Pb exposure may alter Th subset development and/or function, 11 which confirmed the results of our study. However, the results of this study showed a reduction in IFN-γ in Pb-exposed groups, while the level of this cytokine was increased in sensitized animals. According to Th1/Th2 balance toward Th2 in asthma, the level of IFN-γ in S group should also be increased and the observed reduction could be due to the lower dose of OA with which the guinea pigs were sensitized. In fact, it was also shown that a hallmark of Pb-induced immunotoxicity is a pronounced shift in the balance of T helper cell function toward Th2 responses at the expense of Th1functions, resulting in elevated IL-4 with the reduction in IFN-γ levels, which support the results of the present study. 9
The results of the present study confirmed the process of sensitization (inducing animal model of asthma) in sensitized group. Similar findings observed in Pb-exposed animals suggest that exposure to inhaled Pb may cause asthma-like disease in subjects exposed to environmental Pb pollution. This conclusion is supported by the results of a previous study showing higher prevalence of respiratory symptoms for sputum production, shortness of breath, and diagnosed asthma in industrial workers exposed to Pb. 8
The results of the present study also showed increased Pb concentration in lung tissue of animals exposed to inhaled Pb. The Pb concentration of lung tissue was higher in animals exposed to higher inhaled Pb concentration. In fact, it was shown that severe smoke inhalation of closed-space fires, presenting an urban tertiary burn center, was associated with increased blood Pb levels, 33 which confirms the findings of the present study. In addition, the results showed that tracheal responsiveness, total WBC number, eosinophil count, and decreased lymphocyte count as well as increased serum levels of histamine, IgE, and IL-4, and decreased IFN-γ level in animals exposed to high inhaled Pb concentration were more prominent compared with the animals exposed to low Pb concentration. The important findings of the present study support this claim that exposure to inhaled Pb may result in an asthma-like disorder. The association between blood Pb concentration and increased bronchial hyperresponsiveness was also shown in animal models, 39 which supports the results of our study.
The absence of significant changes in most of the measured parameters in animals exposed to low Pb concentration (0.1 M) may show its safety margin in humans. This inhaled Pb concentration (60 mg/m3, 0.38 g nebulized with 6000 L air) was much higher than Pb concentration in Pb industrial environment of 0.089–0.092 mg/m3. 40 However, the studied animals were exposed to Pb for a short period of time compared to workers exposed to inhaled Pb in industrial environment (14 days daily for 60 min in the present study, compared to several years, each day about 8 h in the industrial environment). Therefore, being exposed to Pb concentration lower than 0.1 M (60 mg/m3) for a long period of time in an industrial environment may cause respiratory disorders. The marginal safety for inhaled Pb concentration in industrial environment remains to be clarified in further studies.
In conclusion, these results showed that exposure to inhaled lead acetate can cause increased total and differential WBC count, tracheal responsiveness to methacholine and OA, and serum levels of IL-4, IgE, and histamine with a reduction in IFN-γ and IFN-γ/IL-4 ratio. All changes observed in animals exposed to inhaled Pb were similar to those of sensitized animals. Therefore, exposure to environmental Pb pollution may cause respiratory disorders similar to asthma disease.
Footnotes
Acknowledgments
This paper is a part of a PhD thesis. The authors would like to thank Dr Ramin Sadeghi for revising the manuscript.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was financially supported by the Research Council of Mashhad University of Medical Sciences, Mashhad, Islamic Republic of Iran.
