Abstract
The aim of this study was to document the effect of hesperidin on the key enzyme activities of carbohydrate metabolism, lipid profile, and membrane-bound adenosine triphosphatases (ATPases) during 7,12-dimethylbenz(a)anthracene (DMBA)-induced breast carcinogenesis. Hesperidin has been reported to have multiple biological properties. Breast cancer was induced by single dose of DMBA (20 mg/kg body weight (bw)). The results revealed that there was a significant increase in the activities of hexokinase, phosphoglucoisomerase, and aldolase and a concomitant decrease in the activities of glucose-6-phosphatase and fructose-1,6-diphosphatase in cancer-induced animals. The activities of ATPases were found to be decreased both in erythrocyte membrane and in the liver of mammary cancer-bearing animals. The lipid profiles such as total cholesterol, free cholesterol, phospholipids, triglycerides, and free fatty acids significantly increased and in contrast the ester cholesterol in plasma was found to be decreased, whereas it was found to be elevated in the liver of cancer-bearing groups. The altered levels of the above-mentioned biochemical parameters in cancer-bearing animals were significantly ameliorated by the administration of hesperidin at the dosage of 30 mg/kg bw for 45 days. The histopathological analysis of breast and liver tissues were well supported the modulatory property of hesperidin, and this might be associated with normalizing the gluconeogenesis process, stabilization of cell membranes, and modulation of lipid biosynthesis.
Introduction
Breast cancer is the most frequently diagnosed cancer in women around the world. It remains the second most frequent cause of cancer-related deaths in women. 1 The onset of breast cancer is often earlier than other types of cancers. 2 Metastasis is often seen in breast cancer patients and accounts for over 90% of their deaths. 3 Several mechanisms have been considered as possible mediators of the effect of stress on neoplastic process, which includes alteration in the antioxidant defense status. The production of reactive oxygen species, such as peroxides, hydroxyl, and superoxide anion radicals, results in cellular oxidative damage, through the DNA strand breaks and lipid peroxidation. 4 It is speculated that oxidative stress causes or aggravates many diseases such as cancer, diabetes, and coronary heart disease. 5 Free radical reactions, especially with participation of oxidative radicals, have been shown to be involved in many biological processes that cause damage to lipids, proteins, membranes, and nucleic acids, thus giving rise to a variety of diseases. 6 –8 In lipid-rich environment, the radical chain reactions result in the production of hydroperoxides. These hydroperoxide residues change the hydrophobic interaction between adjacent chains of phospholipids, allowing water molecules to penetrate the biological bilayer easily. The altered water gradient across the membrane consequently changes the substructure of the membrane and leads to the degradation of lipids and proteins. The search for synthetic or natural chemical agents that inhibit and/or delay neoplastic events has received an attention in cancer therapy. 9 Vijayavel et al. have suggested that various molecules such as flavonoids and polyphenols from medicinal plants can inhibit the formation of free radicals associated with carcinogenesis. 10 Bioactive compounds from plant origin have the potential to subside the biochemical imbalance induced by various toxins associated with free radicals. External supply of antioxidants is essential to countervail the deleterious consequences of oxidative damage. 11 Flavonoids have been reported as a double antioxidant and free radical scavenger and also an excited species quencher. 12 Flavonoids are polyphenolic compounds occurring naturally in the plant kingdom, displaying a wide range of pharmacological properties, including anticarcinogenic, anti-inflammatory, and antioxidants activities. 13 Polyphenols may exert their inhibitory effect by acting as pro-oxidants on cancer cells; and in addition, they were found to suppress malignant cell migration, invasion, and metastasis. 14 Hesperidin (Figure 1) is one of the naturally available flavonoids found predominantly in citrus fruits. 15 Several investigators have examined the antioxidant activity and radical scavenging properties of hesperidin using a variety of assay systems. 16 –19 Recently, we also reported various modulating properties of hesperidin against 7,12-dimethylbenz(a)anthracene (DMBA)-induced breast cancer. 20 –23 Although it is reported to have some prominent biological property, there is still a paucity of information on the role of hesperidin especially on the enzymes of carbohydrate metabolism, lipid profile, and adenosine triphosphatases (ATPases). Hence, the present investigation was designed to evaluate the modulatory impact of hesperidin with reference to the above-mentioned biochemical parameters especially against DMBA-induced breast cancer.

Structure of hesperidin.
Materials and methods
Experimental animals
Adult female Sprague Dawley rats aged 45–48 days were procured from central animal house facility, Dr A.L.M. Post Graduate Institute of Basic Medical Sciences, University of Madras, Taramani, Chennai, India. They were maintained in the controlled environmental conditions of temperature and humidity on alternative 12-h light–dark cycles and were fed with standardized rat pellet and water ad libitum. The experiment was sanctioned and approved by the institutional animal ethical committee (IAEC No. 07/019/08).
Chemicals
Hesperidin and DMBA were purchased from Sigma Chemical Company, St Louis, Missouri, USA. 2,4-Dinitrophenylhydrazine and ethylenediaminetetraacetic acid were procured from Sisco Research Laboratories, Mumbai, India. All other chemicals used in the study were of high purity and analytical grade.
Cancer induction
DMBA was used as a carcinogen for the present investigation. Mammary cancer was induced according to the method of Barros et al. 24 A single dose of 20 mg/kg body weight (bw) of DMBA diluted in soya oil (1 ml) was given intragastrically by gavage to induce breast cancer. After 13 weeks of experimental period, all the animals were subjected to ether anesthesia and were sacrificed by cervical decapitation.
Experimental design
The female Sprague Dawley rats were divided into four groups, comprising six animals in each groups as follows: group I animals served as control and the group II animals were administered with 20 mg/kg bw of DMBA in 1 ml of soya oil to induce breast cancer. The group III breast cancer-bearing rats were treated with hesperidin at the dosage of 30 mg/kg bw for 45 days by intragastric administration and the group IV control rats were treated with hesperidin alone with the same concentrations as mentioned in group III animals.
Collection of blood and tissues
After the experimental period, the animals were subjected to ether anesthesia and were sacrificed by cervical decapitation. Blood was collected and plasma was separated for the assays. The liver tissues were collected and homogenized by a Teflon homogenizer in phosphate buffered saline of pH 7.4 and the samples were stored at −80°C for further assays.
Biochemical analysis
The carbohydrate-metabolizing enzymes such as hexokinase was assayed by the method of Brandstrub et al. 25 Phosphoglucoisomerase was assayed by the method of Horrocks et al. 26 and aldolase was estimated by the method of King. 27 Glucose-6-phosphatase was assayed according to the method of King. 28 Fructose-1,6-diphosphatase was assayed by the method of Gancedo and Gancedo. 29 Total lipid was extracted and quantified according to the method of Floch et al., 30 and the total cholesterol was estimated by the method of Parekh and Jung. 31 Free cholesterol was determined by the method of Horn and Menahan. 32 Phospholipids were estimated by the method of Rouser et al. 33 after digesting the lipid extract with perchloric acid. Triglycerides were estimated by the method of Rice 34 based on the method of Van Handel. 35 Free fatty acid content was determined by the method of Horn and Menahan. 32 The inorganic phosphate was estimated according to the method of Fiske and Subbarow, 36 and the erythrocyte membrane was isolated by the method of Dodge et al., 37 with a change in buffer, according to Quist. 38 Na+, K+-ATPase was estimated by the method of Bonting. 39 The activity of Ca2+-ATPase was assayed, according to the method of Hjerten and Pan. 40 The activity of Mg2+-ATPase was assayed by the method of Ohnishi et al. 41
Histopathological analysis
For histological analysis, the breast and liver tissues were sliced and immersed at once in 10% buffered formalin solution for fixation, dehydrated with graded ethanol solutions from 50 to 100%, and then embedded in paraffin. Sections of 5 μm in thickness were cut and stained with hematoxylin and eosin and the slides were observed under a microscope.
Statistical analysis
Values are expressed as mean ± SD. The results were statistically evaluated using one-way analysis of variance by SPSS 10.0 student version followed by Tukey’s multiple comparison method to compare means of different groups. The mean difference is significant at the 0.05 levels.
Results
Effect of hesperidin on carbohydrate-metabolizing enzymes
The effect of hesperidin on carbohydrate-metabolizing enzymes in the liver of control and experimental animals are shown in Figure 2. The activities of the enzymes such as hexokinase, phosphoglucoisomerase, and aldolase were found to be significantly increased ( p < 0.001); and in contrast, the glucose-6-phosphatase and fructose-1,6-diphosphatase ( p < 0.001) were significantly decreased in group II cancer-bearing animals, when compared with group I animals. On the other hand, all the key enzymes of carbohydrate metabolism were significantly altered in group III hesperidin-treated animals ( p < 0.001), when compared with group II cancer-bearing animals. However, no significant changes were observed in group IV hesperidin-alone-treated animals, when compared with group I control animals.
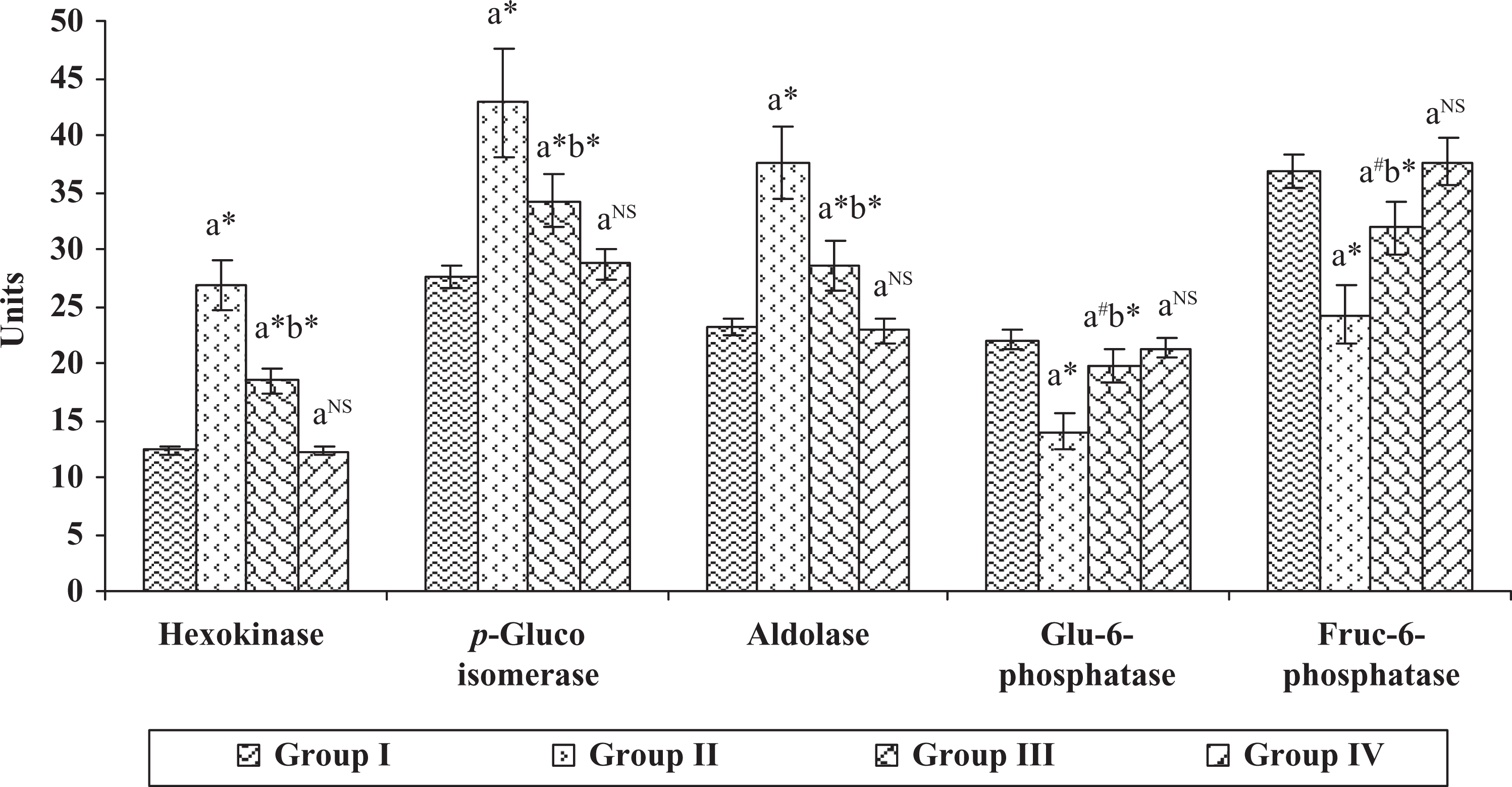
Effect of hesperidin on the levels of carbohydrate-metabolizing enzymes in the liver of control and experimental animals. Units are expressed as hexokinase—n moles of glucose-6-phosphate liberated/mg protein/min; phosphoglucoisomerase—n moles of fructose liberated/mg protein/min; aldolase—n moles of glyceraldehydes liberated/mg protein/min; glu-6-phosphatase and fructose-1,6-diphosphatase—n moles of inorganic phosphate liberated/mg protein/min. Each value represents mean
Effect of hesperidin on lipid profile
The effect of hesperidin on the levels of lipid profile in the plasma of control and experimental animals are presented in Table 1. The levels of total cholesterol, free cholesterol, phospholipids, free fatty acids and triglycerides were found to be significantly increased, whereas the ester cholesterol was found to be decreased in group II cancer-bearing animals, when compared with group I control animals. On the contrary, remarkable alterations in the levels of lipid profile such as phospholipids, free fatty acids ( p < 0.001), total cholesterol ( p < 0.01), free cholesterol, ester cholesterol, and triglycerides ( p < 0.001) were observed in group III hesperidin-treated animals and were comparable with group II cancer-bearing animals. However, no noticeable changes were observed in group IV hesperidin-alone-treated animals when compared with group I control.
Effect of hesperidin on the levels of lipid profile in the plasma of control and experimental animals.

DMBA: 7,12-dimethylbenz(a)anthracene.
Each Value represents mean + SD of Six animals.
Comparisons are made between: aGroup II, III, IV Compared with group I, bGroup III Compared with group II.
Statistical significance: *p < 0.001, **p < 0.01, ***p < 0.05, NS Not Significant.
In addition, Figure 3(a) and (b) shows a similar pattern of lipid profile in the liver of control and experimental animals. The status of lipid profile in group II animals significantly increased ( p < 0.001), when compared with group I control animals. Conversely, all the increased lipid profile status observed in group II cancer-bearing animals were significantly altered in group III hesperidin-treated animals with the significant values of p < 0.01 and p < 0.05, respectively. However, no changes were observed in group IV hesperidin-alone-treated animals when compared with group I control animals.
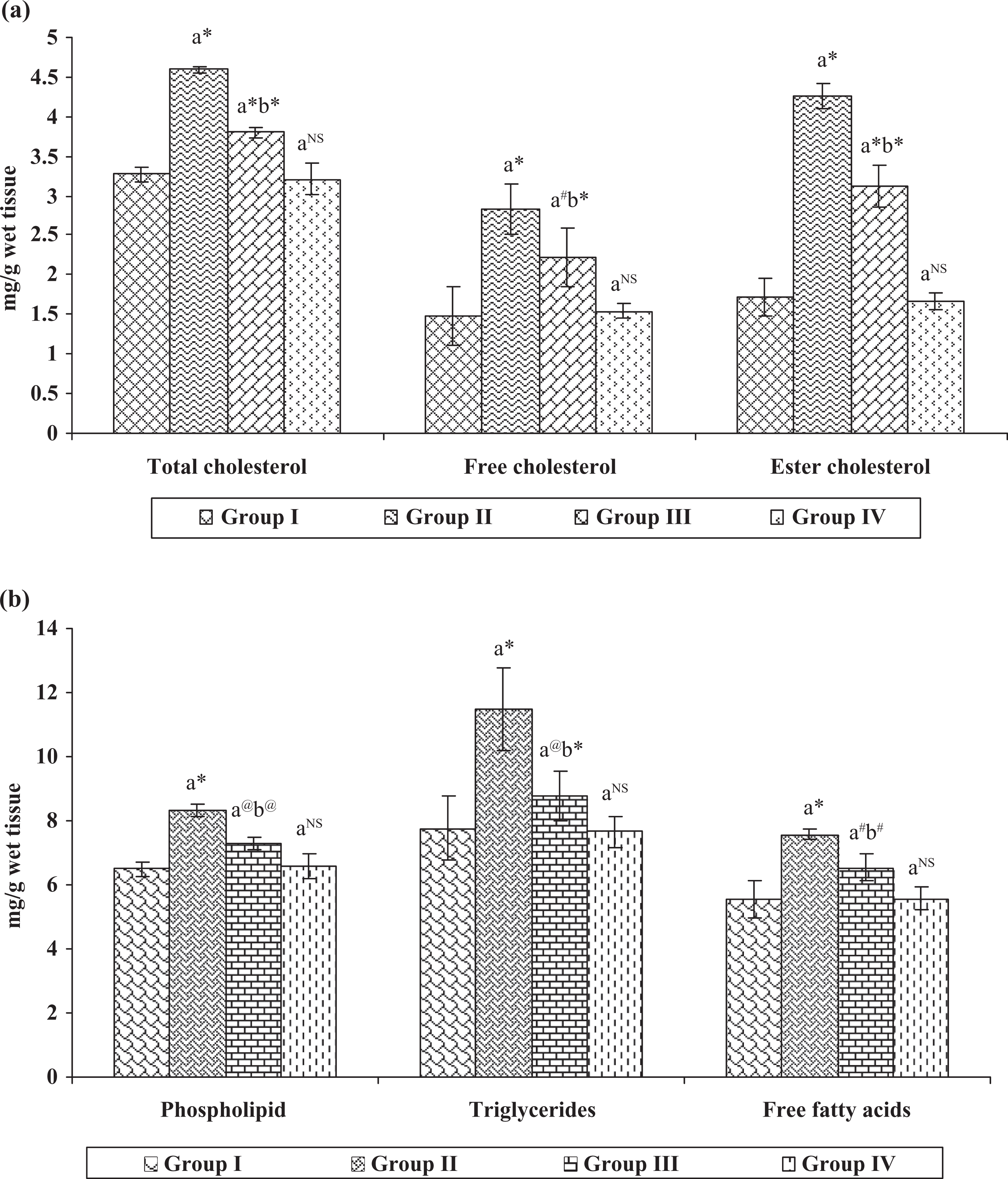
(a and b) Effect of hesperidin on the levels of lipid profile in the liver of control and experimental animals. Each value represents mean
Effect of hesperidin on membrane-bound ATPases
The activities of Na+/K+-, Ca2+- and Mg2+-ATPases in erythrocyte membrane and liver of control and experimental animals are shown in Figures 4 and 5, respectively. The levels of Na+/K+-, Ca2+-, and Mg2+-ATPases ( p < 0.001) were found to be decreased in group II cancer-bearing animals, when compared with group I control animals. On the contrary, the ATPase levels were found to be increased in group III hesperidin-administered animals ( p < 0.05), when compared with group II DMBA-induced breast cancer animals. However, no significant variations were observed in group IV hesperidin-alone-treated animals when compared with group I control animals.

Effect of hesperidin on ATPases in erythrocyte membrane of control and experimental animals. Each value represents mean
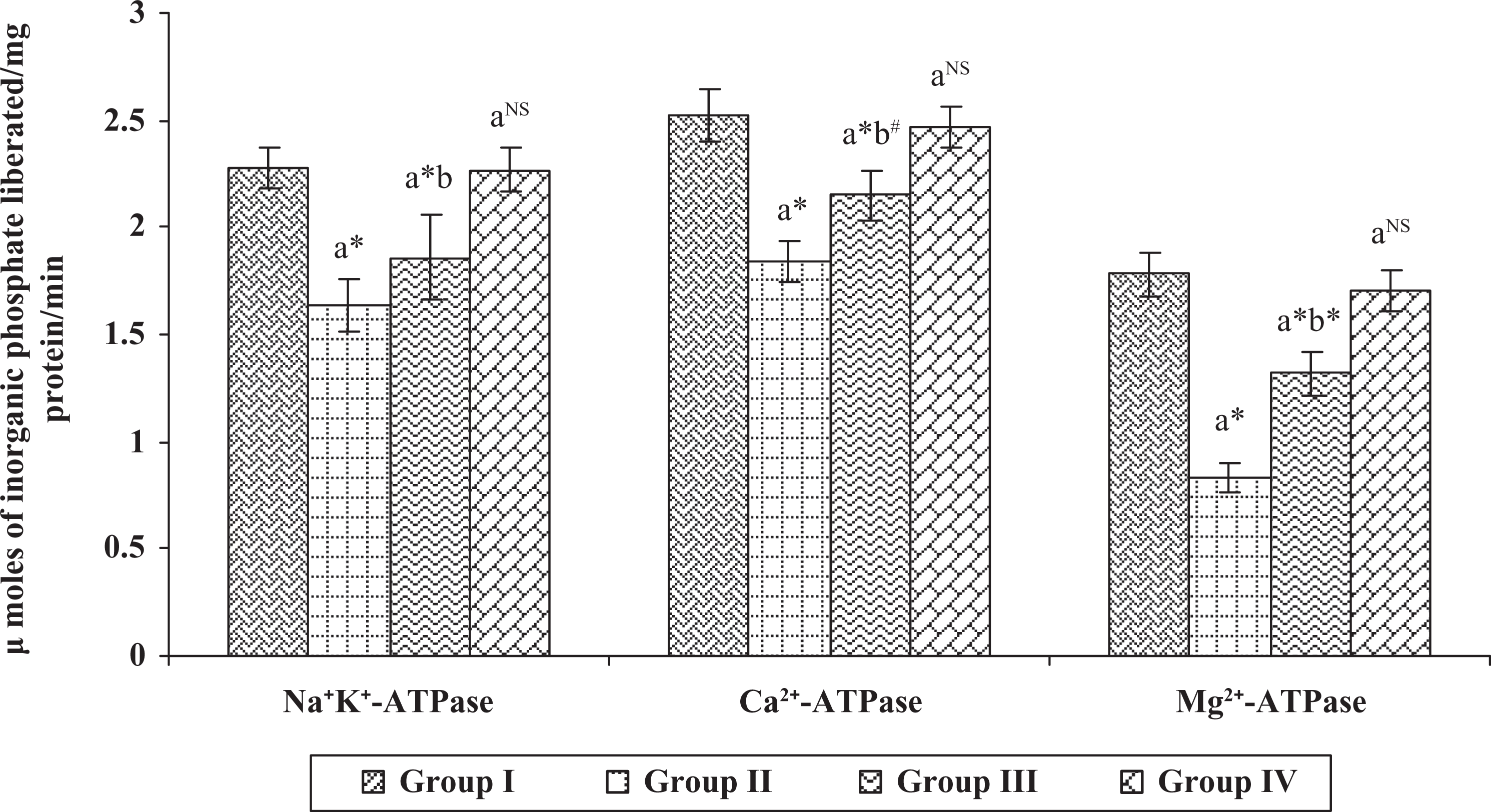
Effect of hesperidin on ATPases in the liver of control and experimental animals. Each value represents mean
Similarly, all the aforementioned ATPase levels were significantly decreased ( p < 0.001) in the liver of group II cancer-bearing animals, when compared with group I control animals. Conversely, the decreased levels were tremendously increased in group III hesperidin-treated animals with the significant values of p < 0.001 and p < 0.05, when compared with group I control animals. Whereas no differences were noticed in group IV hesperidin-alone-administered animals when compared with the group I control animals.
Discussion
Cancer cells have an abnormal energy metabolism. Glucose is the preferred metabolic substrate of most mammalian cells. 42 Cancer-bearing animals have shown a progressive energy loss from the host (i.e. noncancerous) tissues due to the establishment of a systemic energy-depriving cycle in the interaction of tumor glycolysis and host gluconeogenesis. 43 The proliferating cells undergo a shift from oxidative to glycolytic metabolism, where the energy requirements of the rapidly dividing cell are provided by adenosine triphosphate (ATP) from glycolysis. 44 High rates of glucose utilization with production of lactic acid are characteristic features of the neoplastic cell. Gluconeogenesis from lactate is an energy-requiring process that may play an important role in excessive energy expenditure of the host, thus contributing to mechanisms that promote weight loss. 45 Malignant cancer cells exhibit an increased rate of glucose metabolism compared with healthy cells from the same tissue of origin. Transformation of many cell types is accompanied by increased glucose catabolism and increased activities of key glycolytic enzymes. 46 The degree of elevation of these enzymes is directly related to the extent of morphological undifferentiation and growth rate of cancer. 47 The early changes in the carbohydrate metabolism are of particular interest, since anomalies of glycolytic and gluconeogenic pathways are well known from biochemical investigations of cancer conditions. 48 Hexokinase levels occupy an important place in determining the glycolytic capacity of cancer cells. 49 It is a rate-limiting enzyme that catalyses the conversion of glucose to glucose-6-phosphate in the first step of the glycolytic pathway. The glycolytic capacity of cancer cells depends totally on the hexokinase activity for its metabolic fuel. 50 The functional significance of hexokinase is that it commits glucose metabolism. 49 An elevated level of hexokinase of the present study might be due to an increased metabolic need of proliferating cancer cells for energy. This is well in accordance with the previous findings of Hennipman and colleagues. 51 In contrast, administration of hesperidin normalizes the hexokinase level, and this could be due to the intervention of hesperidin in glucose metabolism by blocking the commitments of the hexokinase activity.
Phosphoglucoisomerase serves as a good index of cancer condition. It acts as a catalyst in the conversion of glucose-6-phosphate to fructose-6-phosphate. The increased levels of phosphoglucoisomerase in breast cancer-induced animals might be due to the higher glycolytic rate in cancer condition. This is well in accordance with the earlier findings of Ebrahim et al. 52 Treatment with hesperidin significantly restored this enzyme activity, which might be due to the intervention of hesperidin in the catalytic activity of phosphoglucoisomerase of the glycolytic pathway. Aldolase is another key enzyme in the glycolytic pathway, and it is reported to be increased in cancer-bearing animals. 53 The increased level of aldolase in breast cancer-bearing animals of the present study might be due to the energy requirements of the preneoplastic and neoplastic cells, and it is consistent with the earlier reports of Hennipman and coworkers. 54 The progressive failure of gluconeogenesis has been reported in rapidly growing cancer and is explained partly by a marked decrease or the complete absence of glucose-6-phosphatase activities in cancer condition. 46 Glucose-6-phosphatase is a marker enzyme for liver microsomal activity, and it is greatly inhibited in cancer-bearing animals. In the present study, decreased levels of glucose-6-phosphatase and fructose-1-6-diphosphatase were observed in animals with breast cancer, and it is well in accordance with the earlier findings of Schamhart et al. 55 and Baggetto, 46 according to which the specific activities of a number of enzymes involved in glycolysis, gluconeogenesis, pentose phosphate pathway, and glycogen formation were determined in four rat hepatoma cell lines in culture. It is reported that during the progression of cancer, the production of lactate from glucose has raised and concomitantly glucose production from pyruvate has decreased. It has been proved that cancerous cell utilizes a large proportion of lactate for glycolysis and protein synthesis. Ortega and colleagues have suggested that the drugs that could selectively target the energy metabolism of cancer cells are of great remuneration in the treatment of the cancer. 42 Treatment with hesperidin significantly restored these enzymes, which might be due to the inhibition of glycolytic pathway and activation of gluconeogenesis, thus hesperidin may interrupt the energy requirement of neoplastic tissues and lead to the suppression of cancer progression.
Breast tumor cells require lipids as metabolic fuel and lipids can exert their effect on breast cancer through specific mechanisms by stimulating or influencing the stages of carcinogenesis, modification of structure and function of cell membranes, and alteration of cell signal transduction pathways. 56 –60 Cancer is highly related with higher lipid metabolizing activity, and the cholesterol metabolism is regulated differently during the progression of cancer. 61 The deregulation of cholesterogenesis in cancer implicated an overproduction that could result in the enrichment of cancer cell membrane with cholesterol. 62 The elevated levels of lipid profile of the present investigation are well in accordance with the previous findings of Tang and Eisenbrand 63 according to which the lipid profile was markedly elevated in breast cancer condition. This might be due to the fact that cancer cells utilize the lipids for their fuel supply for progression. Phospholipids play a significant role in the molecular organization and membrane-bound enzymes and also play an important role in maintaining the structural integrity of the cellular membrane. The biological function of membrane-bound proteins depends on phospholipids because they influence the stability and fluidity of the plasma membrane and any alteration in their structure leads to serious consequences. 64 The lipid peroxidation leads to degradation of phospholipids in free radical-mediated tissue injury. The progression of proliferation of the cancer cells depends on the elevation of cholesterol and phospholipid levels, which modifies the lipid fluidity of the cancer cell membranes, thereby increasing the rate of malignancy. 65 In this connection, a report revealed that changes in the distribution of total cholesterol were observed in animals during the progression of cancer. 62 In the present study, elevated levels of lipid profile were observed in animals with breast cancer. This is in line with the earlier findings of Kumar et al. 66 and Veena et al., 67 according to which there was a significant increase in total plasma lipids, total cholesterol, low-density lipoprotein cholesterol, very-low-density lipoprotein cholesterol, phospholipids, triacylglycerols, and free fatty acids in malignant breast cancer patients. The accumulation of triglycerides indicates an imbalance between the rate of synthesis and the rate of release of triglycerides by the parenchyma cells into the systemic circulation. 68 Furthermore, the level of triglycerides in liver reflects a balance between the rates of exogenous supply and endogenous fatty acid synthesis, and it also reflects the rate of free fatty acid oxidation and triglyceride secretion from the tissue. Treatment with hesperidin altered the lipid profiles, which might be due to its inhibitory effect through its free radical quenching activity, since lipids are the primary target of free radicals. In this context, Kaur and colleagues have suggested that hesperidin has significant antioxidant activity and renders protection to the liver against lipopolysaccharide-induced toxicity. 69
ATPase has been described as prominent energy-linked enzymes found in all organisms providing metabolic energy for energy-dependent processes. It is responsible for the transport of ions through the membrane and thus regulates cellular volume, osmotic pressure, and membrane permeability. ATPase is an integral part of the membrane structure. The membrane-bound enzymes such as Na+/K+-, Ca2+-, and Mg2+-ATPases are responsible for transport of sodium/potassium and calcium ions across the cell membrane at the expenses of ATP, and they are lipid-dependent membrane-bound enzymes susceptible to lipid peroxidation. 70,71 In this context, Geetha and colleagues have suggested that lipid peroxidation in cells decreases the activity of membrane-bound ATPase because cell membrane is extremely susceptible to free radical attack, and any alteration in membrane lipid leads to change in membrane fluidity, which in turn alters the ATPase activities and cellular functions. 72 Additionally, Ohta et al. have suggested that the inhibition of membrane ATPases is mediated by oxygen radicals. 73 The oxidation of sulfhydryl groups in ATPase is considered to play a key role in the inhibition of ATPases. The decreased activities of ATPases in breast cancer animals of the present investigation might be due to the free radicals induced by the DMBA administration, and this is well in accordance with the previous reports of several investigators. 74 –77 In contrast, treatment with hesperidin increased the activities of the ATPases. This might be due to the protective role of hesperidin in membrane permeability and stabilizing potential. In addition, it can maintain the structural integrity of cell membrane probably by protecting the membrane ATPases from the deleterious effect of lipid peroxidation caused by the DMBA, as evidenced in the histopathology of breast and liver tissues, respectively (Figures 6 and 7).

(a-d) Shows the histopathological analysis of breast tissues of control and experimental animals. (a) Group I control (20x, HE) and (b) group IV hesperidin alone treated animals were showed normal architecture of fibro fatty tissue structure (20x, HE). (b) Group II DMBA induced cancer bearing animals were showed loss of architecture with neoplastic structure and disturbed fat bodies in the fibro fatty tissue structure (20x, HE). (c) Group III hesperidin treated animals were showed almost normal breast tissue architecture and the fibro fatty tissue structure was maintained normal, which inevitably suggests its protective role in breast (20x, HE).

(a-d) Shows the histopathological analysis of liver tissues of control and experimental animals. (a) Group I control (20x, HE) and (b) group IV hesperidin alone treated animals were showed normal architecture of hepatocytes surround with granulated cytoplasm and small uniform nuclei (20x, HE). (b) Group II DMBA induced cancer bearing animals were showed loss of architecture with disturbed central vein and enlarged sinusoids (20x, HE). (c) Group III hesperidin treated animals were showed almost normal hepatocytes along with congested sinusoids and loosely arranged normal hepatocytes (20x, HE).
Therefore, it can be concluded that the hesperidin significantly ameliorated the changes in carbohydrate metabolism, lipid profile, and ATPases during the treatment through quenching free radicals and thereby suppress the key enzymes of gluconeogenesis, lipids, and ATPases to attenuate the progression of malignant cells as shown in the schematic representation of the overall summary (Figure 8).

Schematic representations of overall summary of hesperidin’s activity during DMBA-induced breast cancer. DMBA: 7,12-dimethylbenz(a)anthracene.
Footnotes
Acknowledgment
The authors are extremely grateful to Dr. R. Venkatakrishna Murali, M.D., Ph.D., Professor and Head, Department of Pharmacology and Environmental Toxicology, Dr A.L.M. Post Graduate Institute of Basic Medical Sciences, University of Madras, Taramani, Chennai, for providing the laboratory facilities.
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
