Abstract
Artemisinins combination therapy (ACT) is the first choice therapy for falciparum malaria. Data on the safety of ACTs in pregnancy are limited and controversial and the use is not recommended on the first trimester. To evaluate the effects of isolated and combined artesunate (AS)/mefloquine (MQ) on embryo rats, pregnant rats were treated orally with AS (15 and 40 mg/kg body weight (bwt)/day), MQ (30 and 80 mg/kg bwt/day) and AS/MQ (15/30 and 40/80 mg/kg bwt/day) on days 9–11 post coitum (pc). The dams were euthanized on day 12 pc and gestational and embryos histological parameters were evaluated. Embryolethality and histopathological anomalies were significant when AS was given alone or combined with MQ. Combination of AS and MQ did not enhance their toxicity compared to their separate administrations; on the other side, there was a reduction in the toxic effects of the AS when combined with MQ. Isolated MQ did not induce developmental toxicity.
Introduction
Malaria remains one of the most important infectious diseases in the world. Plasmodium falciparum, which is responsible for causing severe forms of the disease, is the cause of nearly all of the 1–3 million malaria-related deaths each year. 1 P. falciparum is also adept in developing resistance to drugs, thereby decreasing their efficacy in treatment over a period of time. 2 The simultaneous administration of multiple drugs (combination chemotherapy) combats the development of resistance by preventing the survival of mutated parasites with resistance to a single drug. 3
Pregnant women are in particular risk of adverse malarial outcomes, including death. Malaria during pregnancy is also associated with adverse consequences for the conceptus including intrauterine death, stillbirth, preterm labor and low birth weight, as well as vertical transmission of the disease. 4
Artesunate (AS), dihydroartemisinin (DHA), artemether and arteether are semisynthetic derivatives of artemisinin, which, in nature, is found in leaves and flowers of Artemisia annua L. 5 DHA is the major active metabolite common to several artemisinins including AS, which is the succinate ester of DHA. 6 Since artemisinins are potent and rapidly acting drugs to which there have been few cases of resistance, 7 –9 they have become one of the most important groups of drugs available to treat malaria. The World Health Organization (WHO) has recommended that artemisinin-containing combination therapies should be used to treat P. falciparum in areas of drug resistance. 10
Developmental toxicity of AS and mefloquine (MQ) isolated has been studied. The artemisinins common effect observed is a dose-related increase in postimplantation loss, with total resorptions at higher dose levels. Some studies show morphological abnormalities at doses that cause fetal resorptions, without maternal toxicity. 11 –14 All studies using AS show a steep dose–response curve and some have demonstrated marked sensitivity on the day of dosing between days 10 and 14 post coitum (pc) of gestation in the rat. 15 Within this window, day 10 pc is the most sensitive day for teratogenic effects and day 11 pc the most sensitive day for embryolethality. 12,16
Different laboratories investigated the mechanism of developmental toxicity of artemisinins in animals. These studies showed that embryonic erythroblasts were the primary target of artemisinin toxicity in rat embryos exposed in vivo. 17,18 The hypothesis is that the reduction of red blood cells and associated anemia led to the observed embryolethality or cell damage, causing heart abnormalities (swollen or collapsed chambers), cardiac myopathy and delays in limb and tail development. 14,16
In recent years, the use of the MQ has increased because of the spread of P. falciparum strains that are resistant to other available drugs. MQ is prescribed widely in prophylaxis for malaria in travelers and for the treatment of uncomplicated P. falciparum infections in association with AS. 10 The toxicity of MQ has been studied extensively and the drug is relatively well tolerated. 19,20 Severe central nervous system events requiring hospitalization (e.g. seizures and hallucinations) occur in 1:10,000 patients taking MQ in chemoprophylaxis. 2 During pregnancy, MQ was demonstrated to be teratogenic in animal studies 5–20 times the recommended dosage for humans. 19 Studies on the use of MQ in prophylaxis during the second half of pregnancy have concluded that the drug is safe, but there are few data on the effects of fetal exposure to MQ given for the treatment of malaria during early gestation. 21
Studies on the safety of the association of the AS and MQ are performed, mostly, in nonpregnant subjects; there are few data to support a proper comparison of developmental toxicity of this association. WHO, in 2006, published an assessment of the safety of artemisinin compounds in pregnancy and recommended more detailed studies on the possible interactions of artemisinins and the associated antimalarials, such as MQ. 22 According to the WHO, all artemisinins will be used in combination with other antimalarials in the treatment of uncomplicated malaria, however, the possibilities of interactions must be considered.
A major concern about the safety of the AS/MQ association is the possibility of interactions between the components. Pregnancy is associated with changes in several drugs pharmacokinetics, and, in the case of the artesunate, the target of the developmental toxicity is, apparently, the embryonic erythroblasts, so interactions may not be observed in nonpregnant patients. The objective of this study was to investigate the developmental effects of the antimalarials AS and MQ and their association in the embryo exposed in utero, during the critic period of artemisinins developmental toxicity.
Materials and methods
Animals
All Wistar rats used in this study were from the Federal University of Paraná animal house breeding stock. Animals were maintained under controlled environmental conditions at 22 ± 2°C and a constant 12-h light/12-h dark cycle. Standard pellet food (Nuvital, Curitiba, Paraná, Brazil) and tap water were available ad libitum. The study was approved by the Ethics Committee on Animal Experimentation of Federal University of Paraná (Protocol number: 308/08). Female rats were placed in the male cage—at a ratio of 3:1—for 1 h at the end of the dark period. Mating was confirmed by the presence of sperm in vaginal smears and the day on which the sperm was detected was considered day 0 pc. Pregnant females were randomly assigned to control and treatment groups. Maternal weight was monitored daily during gestation and all animals were observed for clinical signs of toxicity.
Drugs and treatment
AS (artesunic acid; Knoll/Abbott (Germany), lot no. 2.05) and MQ (mefloquine chlorohydrate; Sifavitor (Milano, Italy), lot no. 8700/13/09) were supplied by Fiocruz/PDTIS (Rio de Janeiro, Brazil). Suspension of AS in 1% carboxymethyl cellulose and suspension of MQ in distilled water were prepared daily.
The following are the experimental groups, with 15 females each: (I) vehicle control, (II) AS 15 mg/kg body weight (bwt)/day (AS15), (III) AS 40 mg/kg bwt/day (AS40), (IV) MQ 30 mg/kg bwt/day (MQ30), (V) MQ 80 mg/kg bwt/day (MQ80), (VI) AS 15 mg/kg bwt/day plus MQ 30 mg/kg bwt/day (AS15/MQ30) and (VII) AS 40 mg/kg bwt/day plus MQ 80 mg/kg bwt/day (AS40/MQ80). The doses of AS and MQ recommended by WHO for treatment during three consecutive days were AS 4 mg/kg and MQ 8 mg/kg (1:2). 17 In the present study, the doses of AS were chosen taking into consideration the human therapeutic dose (4 mg/kg). The lower dose of AS (AS15) is the extrapolation of the human therapeutic dose to rat dose estimated by allometry (4 × 3.94 = 15.76 mg/kg). 23 The higher dose (AS40) was 10-fold the human therapeutic dose used for eventual interspecies differences. The MQ dose was based on the proportion of AS and MQ recommended by WHO. 17
All drugs were administered by gavage once a day from days 9 to 11 pc. The 3-day treatment regimen is consistent with a WHO recommendation for the treatment of human malaria with artemisinin-containing combinations of drugs. 17 The volume administered was 5 mL/kg, based on the animal body weight on the day of dosing.
Cesarean section and embryo examination
Pregnant females were euthanized by decapitation and were submitted to cesarean section on day 12 pc. Blood was collected in heparinized tubes; plasma was removed after centrifugation and immediately frozen at −20°C for hormonal analysis. Gravid uteri were removed and weighed. Implantation site, viable embryos and resorptions were counted. The criteria used to determine embryonic death was the presence of embryo severely deformed when examined under a stereomicroscope. Each uterus was cut along the antimesometrial borders and the individual implantation sites removed with their decidual material. Each embryo was divested of the remaining decidual under a stereomicroscope and observed for yolk sac vascularization and embryo viability and morphology under the stereomicroscope. Two viable embryos from each litter were collected and fixed in methacan solution (methanol 60%, chloroform 30% and acetic acid 10%) for 24 h at 4°C for histological examination. The percentage of postimplantation losses was calculated. Selected organs were removed and weighed, including ovaries, liver, kidneys and adrenal glands. Organ weights were evaluated as absolute and relative weights ((organ weight/bwt) × 100)).
Histological analysis
Fixed embryos were washed and dehydrated three times in a bath of absolute ethanol for 30 min followed by overnight bath of absolute ethanol/xylene (16 h) at 4°C and three xylene diafanization baths for 30 min. The embryos were embedded in paraffin for 2 h at 56°C and placed in paraffin blocks. Five to six embryos from each group were selected for histological analysis and completely sectioned, producing about 150–200 serial sagittal sections of 5 μm per embryo. All slides were stained with hematoxylin and eosin and each section was examined under a light microscope. The remaining embryos were preserved but not analyzed. The histological photomicrographs were taken with a Leica EC3 Digital Color Camera (Leica Microsystems) attached to a Leica DM 2500 light microscope and the software Leica Application Suite—LAS EZ 1.4. Results were expressed as a prevalence of histopathological alterations in each embryo, by an adaptation of Bernet and coworkers. 24 This histopathology index is based on the sum of score rankings and importance factors for each lesion found in the tissue. For each embryo investigated, the respective pathological changes were classified into three patterns, which include several alterations. These patterns are (1) red blood cell alterations, including empty vessels or general reduced concentration of primitive erythroblasts and anomalies of erythroblast shape or color; (2) cell death, with general cell death areas and/or cell death in specific locations; (3) architectural and structural alterations of fetal selected organs, changes in tissue structure as well as in shape and arrangement of cells.
Hormone analysis
Plasma progesterone and testosterone levels were determined by enzyme immunoassay. Antibodies of progesterone (polyclonal antiprogesterone R4861; 1:10,000 dilution) and testosterone (polyclonal antitestosterone R156/7; 1:7500 dilution) were obtained from Coralie Munro at the University of California (Davis, California, USA). The progesterone antibody cross-reacted (at 50% binding) 47.3% with 11α-hydroxyprogesterone, 0.4% with 17α-hydroxyprogesterone, 0.2% with 20α-hydroxyprogesterone, 2.4% with 20β-hydroxyprogesterone, 39.0% with 5α-pregnane-3,20-dione, 17.0% with 5β-pregnane-3,20-dione, 0.003% with 17β-estradiol, 0.004% with androstenedione, 0.8% with testosterone and 0.002% with cortisol. Cross-reactivities for antitestosterone are testosterone 100.0%, 5α-dihydrotestosterone 57.4%, androstenedione 0.27%, and androsterone, dehydroepiandrosterone, cholesterol, estradiol, progesterone and pregnenolone <0.05%. Serial dilutions of pooled plasma samples (n = 35; five per group) produced displacement curves parallel to those of the appropriate standard. Inter- and intra-assay coefficients of variation were <10%. Assay sensitivities were 0.78 and 2.3 pg/well for the progesterone and testosterone enzyme immunoassay, respectively.
Statistical analysis
Data from cesarean section and embryo parameters were tested for normal distribution by the D’Agostino and Person normality test. Parametric data were analyzed by analyses of variance, and the differences between groups were assessed by Tukey’s multiple comparison test. Nonparametric data were analyzed by the Kruskal–Wallis test followed by Dunn’s multiple comparison test. The data regarding the histopathological injury indexes were analyzed using the Kruskal–Wallis test followed by Dunn’s multiple comparison test. Differences were considered statistically significant at a probability level of 5% (p < 0.05). The statistical analyses were performed using Prism version 5.0 (GraphPad, San Diego, California, USA).
Results
No signs of maternal toxicity were observed in control and treated animals. Expected weight gain occurred in all groups during pregnancy (Table 1). There was a reduction in the relative maternal weight gain on day 12 pc in the groups treated with AS40 and the association AS40/MQ80, but this reduction was not statistically significant. Maternal organ weight (ovaries, liver, kidneys and adrenal gland) did not differ among groups (data not shown). There was no significant difference in the uterus weight with embryos among groups, and there was also no significant difference between the implantations number in control and treated female (Table 1). Furthermore, no clinical or behavioral changes were observed in the treated animals.
Maternal and pregnancy data of dams treated orally with artesunate, mefloquine and artesunate/mefloquine association on gestational day 9–10 and results of cesarean sectioning on day 12 pc.a

pc: post coitum.
aValues represent mean ± SEM.
bPercentage of maternal weight gain compared to day 0 pc.
c p ≤ 0.01.
d p ≤ 0.001.
eOn day 12 pc.
Maternal progesterone and testosterone levels determined 24 h after the last day of the treatment were not significantly different among the groups (Table 1).
Control embryos
At the time of the cesarean section, all control animals showed well-developed embryos, there was rare occurrence of postimplantation losses (Table 1). The yolk sacs were fully vascularized and not anemic when observed with the naked eye (Figure 1(a)) and under a stereomicroscope. Histological examination reveals normal tissue organization (Table 2 and Figure 4). The red blood cells visible in the sections of embryos examined were nucleated, indicating that they were primitive erythroblast (Figure 1).
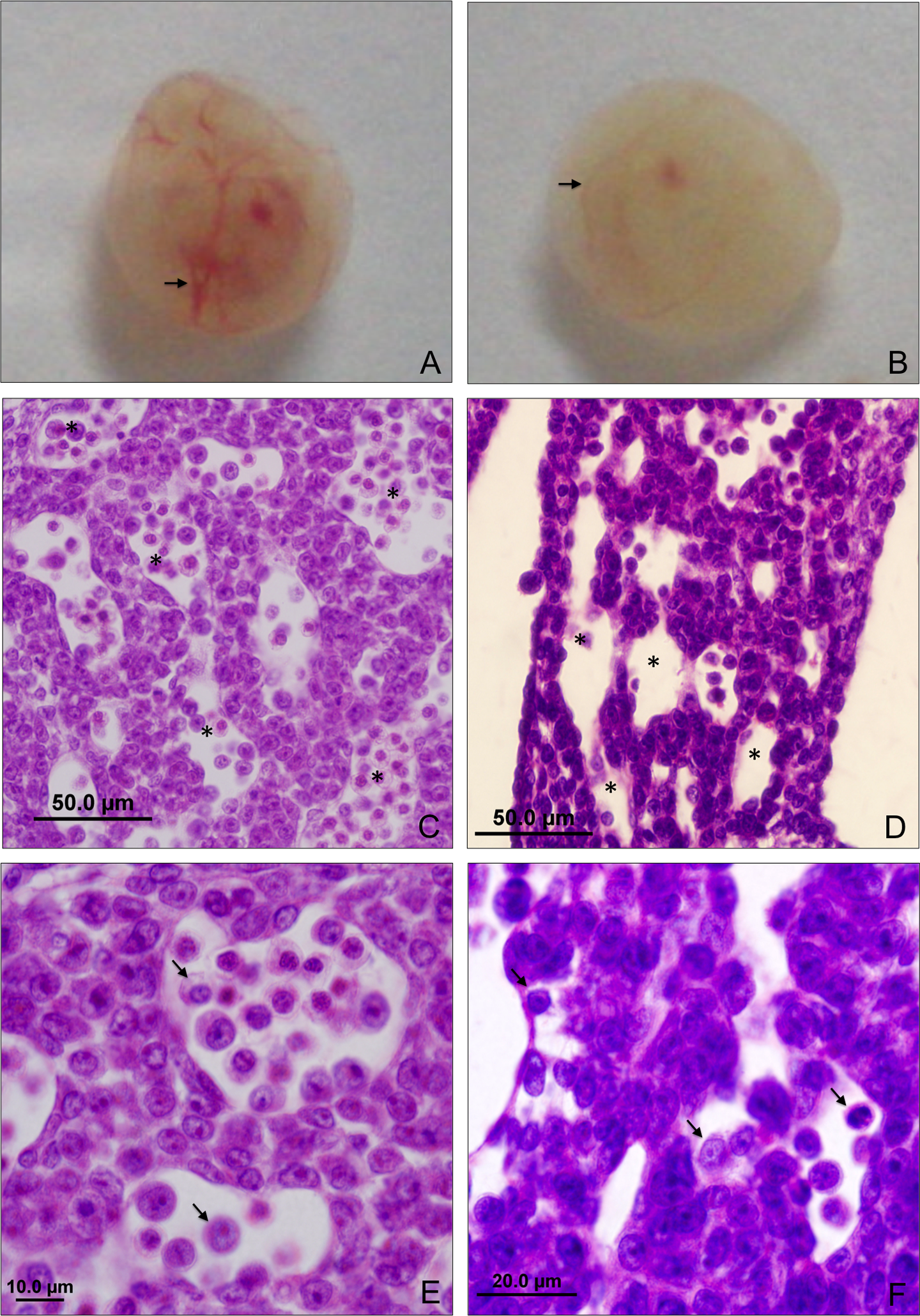
Red blood cell patterns from control (a, c and e) and AS40 (b, d and f) embryos on day 12 pc. (a) Control embryo with well-vascularized yolk sac (arrow), (b) embryo exposed in utero to AS40 with anemic yolk sac (arrow), (c and e) sagittal sections of well-developed livers from the control group, red blood cells are spherical and nucleated (e—arrows) and the vasculature are full of cells (c—*) and (d and f) sagittal sections of AS40 irregular architecture livers from embryos on day 12 pc, red blood cells are fewer in number (d—*) and some with abnormal shape (f—arrow). AS: artesunate; pc: post coitum.

Histological sagittal sections of hearts from control (a and c) and AS40 (b and d) embryos on day 12 pc. (a) Well-developed heart, (b) heart with irregular architecture, (c) control heart with red blood cells (arrow) and (d) heart with almost no red blood cells (arrow). a = atrium; v = ventricle. AS: artesunate; pc: post coitum.

Histological sagittal sections from control (a and c) and AS40 (b and d) embryos on day 12 pc. Control embryo showed well-organized tissues in pharyngeal arches (a) and somites (c). Embryo exposed in utero to AS has areas of cell death in the pharyngeal arches (b) and somites (d; arrows). AS: artesunate; pc: post coitum.

Histopathology index. (a) General histopathology index, (b) indexRBC (index of red blood cells anomalies), indexCD (index of cell death) and index IA (index of organs irregular architecture). AS15 = AS 15 mg/kg bwt/day, AS40 = AS 40 mg/kg bwt/day, MQ30 = MQ 30 mg/kg bwt/day, MQ80 = MQ 80 mg/kg bwt/day, AS15 + MQ30 = AS 15 mg/kg bwt/day plus MQ 30 mg/kg bwt/day and AS40 + MQ80 = AS 40 mg/kg bwt/day plus MQ 80 mg/kg bwt/day. AS: artesunate; MQ: mefloquine; pc: post coitum.
Histological anomalies in the day 12 pc embryos following 3 days of oral doses of artesunate, mefloquine and artesunate/mefloquine association on days 9–11 pc.
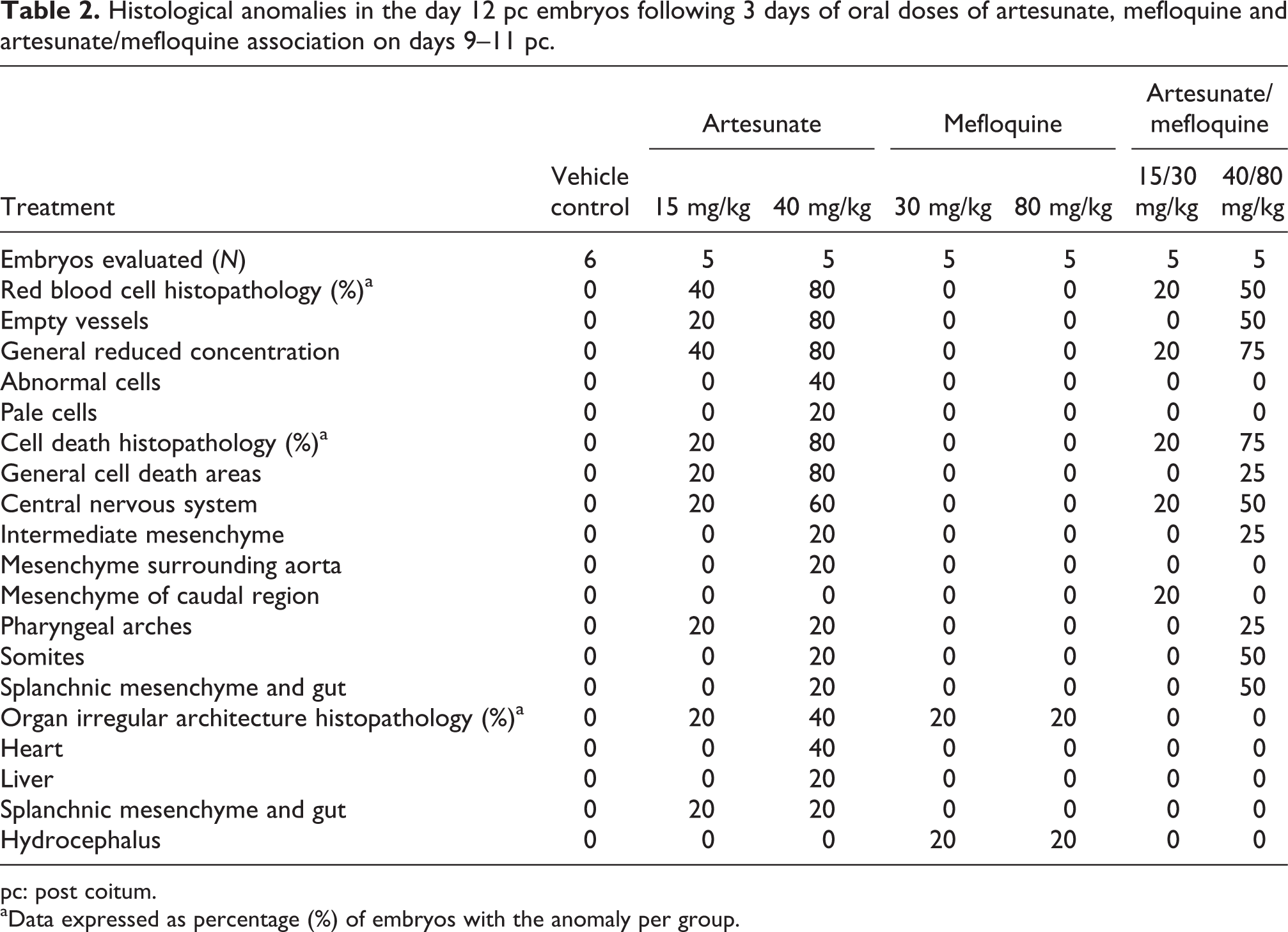
pc: post coitum.
aData expressed as percentage (%) of embryos with the anomaly per group.
AS-exposed embryos
At both the doses of AS, the yolk sac was pale in part of the implants (Table 1). Yolk sac vessels formation and circulation, observed with the naked eye and under a stereomicroscope, appeared normal but was visibly paler than the control group in approximately 40% of embryos exposed to AS15 and 80% in those exposed to AS40 (Table 1 and Figure 1(b)).
In the animals treated with AS15 and AS40, respectively, 30 and 40% of the embryos were dead at macroscopic observation (Table 1), it was significantly different from the control group. The remaining embryos were viable, however, some of them were pale when compared with the control group.
The surviving embryos that were histologically analyzed showed anomalies in the liver (Figure 1) and heart (Figure 2) architecture and some areas of cell death (Figure 3). There was an overall relative reduction in erythroblasts compared with control embryo. The red blood cells were also pale and occasionally abnormally shaped (Figure 1). In the same embryos in which reductions in the number of the red blood cells were observed, the cell death and the irregular architecture of the organs were also observed, showing a direct relationship among these parameters.
In the group exposed to AS15, three of the five analyzed embryos showed histological anomalies (Table 2). These embryos presented anemia, with general reduced concentration of red blood cells and some empty vessels. Cell death was found at the splanchnic mesenchyme, spinal cord mesenchyme, central nervous system and pharyngeal arches. General cell death areas were found in one embryo. Irregular architecture in the histopathology was observed in the splanchnic mesenchyme and gut.
In the group exposed to AS40, four of the five analyzed embryos showed histological anomalies (Table 2), and the histopathology index of these anomalies (10.5) was higher than that observed in the group treated with AS15 (2.8; Table 2 and Figure 4). General reduced concentration of red blood cells was found, with almost no blood in some embryos and misshapen and pale red blood cells. Some embryos presented dilated and abnormally shaped heart, and liver and splanchnic mesenchyme abnormally shaped. Cell death was observed at the central nervous system, intermediate mesenchyme, mesenchyme surrounding aorta, pharyngeal arches, somites and splanchnic and gut. The system most affected by areas of cell death was the central nervous system. General cell death was found in one embryo.
MQ-exposed embryos
The animals treated with MQ, in both the doses, showed well-developed embryos, similar to the control group, with fully vascularized and not anemic yolk sac (Table 1). Histological examination did not reveal alteration in the erythroblast and areas with cell death (Table 2). There were two embryos affected with hydrocephalus, one in each MQ experimental group (Table 2).
AS plus MQ-exposed embryos
There was no significant difference in the number of postimplantation losses and viable embryos among the groups treated with isolated AS or AS plus MQ and both the doses of these groups differ from the control group. In the same way, in both the doses of AS associated with MQ, in part of the implants, the yolk sac was pale. However, the group treated with the association of AS40/MQ80 seems to be less affected than the group treated with AS40 isolated, but it was not statistically significant (Table 1).
Some surviving embryos that were histologically analyzed showed areas of cell death, and there were fewer red blood cells than normal. The groups treated with the association of AS plus MQ had a lower histopathology index when compared with the same doses of isolated AS (Table 2). In the same way, as the group treated with isolated AS, the embryos that presented red blood cells reduction were the same embryos that were observed in cell death and organs with irregular architecture.
In the group exposed to AS15/MQ30, three of the five analyzed embryos showed anomalies (Table 2). These embryos presented anemia, with general reduced concentration of red blood cells. Cell death was found at the central nervous system, mesenchyme surrounding aorta and mesenchyme of the caudal region.
In the group exposed to AS40/MQ80, three of the four analyzed embryos showed anomalies, and the histopathology index of these anomalies (3.1) was higher than that observed in the group treated with AS15 (0.5), but lower when compared with the group treated with isolated AS40 (10.5; Table 2 and Figure 4). Reduced concentration of red blood cells and some empty vessels were found. Focal necrosis was observed in the central nervous system, intermediate mesenchyme, splanchnic mesenchyme, somites, splanchnic mesenchyme and pharyngeal arches. General cell death areas were found in two embryos.
Discussion
The present study investigated the effect of the antimalarials agents, AS and MQ, and their association on pregnancy and embryonic development in rats. The results are consistent with literature data showing that AS impairs rat development. AS15 and AS40 administered on days 9–11 pc were markedly embryotoxic, with significant postimplantation losses and increased incidence of histological alterations.
The results of treatment with AS and MQ, isolated or in association, did not indicate alteration in the body weight gain of dams, maternal progesterone and testosterone dosages and other signs of clinical toxicity, excluding a possible maternal toxicity. The levels of postimplantation losses observed in this study with the treatment with AS40 (40%) were lower than the losses cited in the literature, when in the same dose, some studies reported 100% of postimplantation losses. 12 This probably occurred because the euthanasia was performed just 1 day after the treatment, in the beginning of the alterations. Previous studies showed that embryolethality is the hallmark effect of AS developmental toxicity in rats and that the most critical period of AS treatment in rats is between days 9 and 11 pc, 12,15 these developmental effects were observed largely in the absence of any apparent maternal toxicity. The induction of embryolethality by AS has been reported in, at least, three species: rats, rabbits and monkeys. 12,25
The administration of MQ without AS did not cause embryo death. The embryos death observed with the association AS-MQ could be attributed to AS. The embryolethality was significant when AS was given alone or in combination with MQ, and the association did not enhance the embryolethality, although some embryos exposed to isolated MQ presented hydrocephalus. Studies carried out by Palmer and coworkers and Phillips-Howard demonstrated that MQ was teratogenic in animal 5–20 times the recommended dosage for humans. 19,26 The observed anomalies were domed cranium, hydrocephalus in rats, and cleft palate and skeletal anomalies in mice. In the present study, the litters were exposed to isolated MQ30 and MQ80, respectively, 4 and 10 times the humans dosage. 17 Recently, women of childbearing potential have been advised to take contraceptive precautions up to 3 months after the last dose. It is recommended to use in pregnancy only if the expected benefit justifies the potential risk for the fetus. 21 Studies on the use of MQ in prophylaxis during the second half of pregnancy have concluded that the drug is safe, but there are few data on the effects of fetal exposure to MQ given for the treatment of malaria during early gestation. 20,21,27,28 In the present study, the hydrocephalus observed in a few embryos exposed to isolated MQ was not observed in the embryos exposed to the combination AS plus MQ.
The histological examination reveals normal tissue organization in all the control group embryos. The changes observed in the groups treated with AS are similar to the patterns of anomalies seen in other studies where AS was administered orally in rats and also in monkeys. 14,25,29 This pattern includes reduction, lack of deformed red blood cells in the vasculature, liver and heart architecture anomalies and areas with cell death. The histological effects of AS were significant when AS was given alone or in combination with MQ, however, the association led to a decrease in the histopathology index when compared with isolated AS. The way by which MQ could attenuate the developmental toxic effects on AS is unclear. In principle, observed drugs interactions may involve toxicokinetics, toxicodynamic or both factors.
After being absorbed, the AS ester bond is hydrolyzed within minutes yielding DHA that is believed to be its main active metabolite against malaria parasites and also the proximate developmental toxicant compound. 30 A hypothesis has been advanced that the initial event in the developmental toxicity of AS and DHA is a depletion of primitive embryonic erythroblasts, which led to a prolonged, severe anemia. 16 The initial observation of this anemia was the loss of red color from the blood vessels in the embryo, which was apparent by 6 h after a single administration of 17 mg/kg AS on day 11 pc. After 24 h, there was a marked reduction in primitive erythroblasts, and after 2 days of dosing, there was almost complete elimination of the primitive erythroblasts. 16
In mammalian embryos, an active erythropoietic activity is first detected in the yolk sac. 6 It is generally accepted that hematopoietic stem cells are generated during embryonic development in the yolk sac and sequentially colonize the fetal liver, the spleen and finally the bone marrow. 31 The red blood cells produced by the yolk sac are nucleated and metabolically active, able to synthesize protein and prosthetic components (heme) of hemoglobin. 29 In the same way, the parasitized erythrocytes with plasmodium and primitive erythroblasts have high concentrations of heme and iron, and both have been proposed as activators and as targets of artemisinin derivatives. 32,33 The primitive erythroblasts enter the embryonic circulation about day 10 of gestation in the rat, at the same time the heart begins to beat. 14 The primitive erythrocytes never reach the final enucleated stage in situ, and for this reason they have been called primitive. 31
The marked depletion of embryonic erythroblasts after a single AS dose to rats on days 10 or 11 of gestation is due to the death of the primitive erythroblast population. Since, after day 10 of gestation, the primitive erythroblast population is self-sustaining, once extensively depleted, they cannot be replaced until definitive erythroblasts are introduced into the blood from the liver. 6 Embryonic responses to acute anemia varied from complete recovery to malformation and death, depending on the extension of cell death.
Nonclinical and clinical studies on the safety of antimalarial drug combinations during pregnancy have seldom been performed. An exception was a study by Clark et al. 12 on the developmental toxicity of a three drug combination consisting of chlorproguanil–dapsone and AS (1:1.25:2) in rats and rabbits. Data from the foregoing study showed no evidence of a potentiating effect of this combination compared to AS given separately, on the contrary, results presented in this article suggested that the developmental toxicity of AS may be attenuated when coadministrated with MQ. There were no antagonistic interactions between AS and MQ in studies published on the efficacy and pharmacokinetics of these drugs when performed in nonpregnant subjects. However, during pregnancy, there might be changes in the pharmacokinetics of drugs, with modifications, mainly in the distribution and metabolism, even the artemisinin derivatives are enzymatic inducers of various CYP450 families. Moreover, the target of the toxic action of artemisinins derivatives is the fetal erythroblast, 14,16,29 that is, much more sensitive to artemisinins than the adult erythroblast. 6 The protective action of MQ on the reproductive toxicity of AS may be taking place at the cellular level, however, to prove this hypothesis, further studies on the mechanism of reproductive toxicity and toxicokinetics of this interaction are necessary.
In conclusion, the association AS plus MQ is effective and indispensable as antimalarial, but the exposure during gestation may induce postimplantation losses and malformations, mainly in early gestation. The embryolethality and the histological alterations were significant when AS was given alone or in combination with MQ. However, the results indicate a reduction in some developmental anomalies of AS associated with MQ at both evaluated doses. Due to the dependence of malaria therapy on the use of drug combinations containing artemisinins, and also on the decreased morbidity and mortality of malaria in pregnant women, mechanisms by which MQ and AS interact during gestation deserve further investigation.
Footnotes
Funding
This work was partially supported by CAPES, CNPq and FUNPAR.
Acknowledgment
The authors are grateful to FIOCRUZ/PDTIS for supplying the drugs.
