Abstract
The aim of this study was to investigate the role of serum cholinesterase (SChE) activity and S100B protein in the evaluation of patients with acute organophosphate (OP) poisoning. Patients with acute OP poisoning admitted to the emergency department were included in this cross-sectional study. Twenty healthy volunteers served as controls. The SChE activity and serum S100B were determined on admission. Patients were divided into two groups (low severity and high severity). Thirty-six patients diagnosed with acute OP poisoning were enrolled. Serum S100B concentrations were higher in patients than in the control group (p < 0.05). In the high-severity group, the SChE levels were lower and the S100Bs levels were higher than in the low-severity group. The SChE level was not different between survivors and nonsurvivors. S100B levels were higher in nonsurvivors than in survivors. According to receiver–operating characteristic curve analysis, the optimal cutoff value of serum S100B level to predict mortality was 236.5 pg/mL, with 71.4% sensitivity and 89.7% specificity. Our data suggest that initial SChE level is related to the clinical severity but not with mortality. S100B may be a useful marker in the assessment of clinical severity and prediction of mortality in acute OP poisoning.
Introduction
Organophosphate (OP) poisoning is an important clinical problem in the rural regions of the developing world, and severe acute OP poisoning is a medical emergency. 1 The mortality rate for OP poisoning is high, with fatality often related to a delay in diagnosis or improper management. 2 OPs inhibit the enzymes acetylcholinesterase (AChE), at cholinergic synapses and in red blood cells, and butyrylcholinesterase (BuChE) in plasma. 3 The inhibition of AChE by OPs causes accumulation of acetylcholine at cholinergic synapses, with overstimulation of muscarinic and nicotinic receptors. 4,5 The subsequent central nervous system (CNS) and neuromuscular features of OP poisoning are fairly understood. 1 Serum cholinesterase (SChE) activity is often checked in OP-poisoned patients. Previous studies suggested that although initial low SChE activity supports the diagnosis, decreased SChE activity does not appear to have prognostic value in acute OP poisoning. 6 –8
In addition to the inhibition of AChE activity, glia and neurons may be alternative targets for OP compounds. 9 The various OPs have been shown to inhibit astroglial cell proliferation and to cause neuronal apoptotic death. 10 –12 S100B is a calcium-binding protein that has various effects on glia and neurons. S100B is expressed mainly in astrocytes in the CNS. 9,13 S100B release was shown to increase after brain damage, such as traumatic cortical injury and focal cerebral ischemia as well as in some psychiatric disorders. 14 S100B may have the potential to be a new marker in clinical toxicology; it is associated with levels of consciousness in carbon monoxide poisoning and benzodiazepine overdose. 13,15,16 Recently, it was demonstrated that increased serum levels of S100B are associated with acute chlorpyrifos poisoning in rats. 9 However, no data are available on the serum levels of S100B in patients with OP poisoning. The aim of this study was to investigate the role of SChE activity and S100B protein in the assessment of patients with acute OP poisoning.
Methods
Study population
Prior to the study, the proposed protocol was approved by the Medical Research Ethics Committee of Ondokuz Mayis University (OMU) in Samsun, Turkey, and written informed consent was obtained from patients or, in the case of unconscious patients, from relatives. The authors prospectively studied all patients with acute OP poisoning who were admitted to the emergency department (ED) of OMU Teaching Hospital between June 2008 and May 2010. OMU is a tertiary medical center with 1200 beds and an annual ED registration of approximately 25,000 patients. Adult patients (age ≥18 years) with OP poisoning were included in the study. Exclusion criteria included carbamate poisoning, an uncertain history of exposure or an uncertain time of poisoning, severe preexisting chronic health status, coingestion of other drugs, and patients without SChE activity measurements within 24 h of poisoning. Patients were also excluded if the ingested compound was not identifiable. The diagnosis of OP poisoning was based on the history of exposure to OP insecticides, characteristic clinical features, and decreased SChE activity. On admission, information about the OP agent involved in the exposure was taken from either the patient or the patient’s family. The physical and neurological examinations were carried out and the Glasgow Coma Scale (GCS) score was determined.
The clinical severity of OP poisoning was determined by a form of the grading system developed by Bardin et al. 17,18 A revised form of this grading system categorizes poisoning into three groups, namely mild, severe, and life-threatening poisoning. Mild poisoning is defined by a history of exposure/intake with mild signs of poisoning, including normal consciousness, mild secretions, and little fasciculation. Severe poisoning is defined by a history of exposure/intake and severe signs of poisoning characterized by altered consciousness, copious secretions, and generalized fasciculation. Life-threatening poisoning is defined by a history of exposure/intake resulting from a suicide attempt or characterized by stupor, a partial pressure of oxygen (PaO2) level of <75 mmHg, and an abnormal chest roentgenogram. In that grading system, at least two criteria are required to be satisfied to allocate a patient to that particular grade. If fewer criteria are satisfied, the next lower grade is used. In the current study, severe and life-threatening grades were combined as high-severity poisoning. Based on that criterion, the patients were further divided into two groups as (a) high severity and (b) low severity. A control group of 20 age- and sex-matched healthy volunteers served as a reference for biochemical parameters. They did not have any history of disease or current medical problem and were not on any medication.
After the diagnosis, all of the patients were treated in the emergency intensive care unit (ICU). In addition to general supportive measures (washing of the entire skin surface, gastric lavage, administration of cathartics, and activated charcoal), a standard therapy involving atropine and pralidoxime was administered to the patients. Either continuous infusion or an intermittent dose of atropine was administered until bronchial secretions were controlled. Pralidoxime was administered from 2 g daily (divided into four doses) up to 200–500 mg/h (continuous infusion), according to the clinical severity of the condition. The indications for endotracheal intubation and mechanical ventilation were accepted as cardiorespiratory arrest, depressed level of consciousness causing an inability to maintain the airway, excessive secretions, and severe metabolic acidosis with hemodynamic instability. Data regarding the age, sex, cause of contact, type of OP, clinical features, admission time to the ED after exposure, emergency tests such as chest x-ray, blood gases, need for mechanical ventilation, duration of ICU stay, and clinical outcome of patients were recorded. Outcome was assessed as inpatient mortality. Regarding outcomes, patients were divided into two groups as survivors and nonsurvivors.
Blood samples and measurement of SChE and S100B
Venous blood samples from patients on admission and controls were collected by venipuncture without anticoagulant. After sampling, the blood was centrifuged for 15 min at 1500g, and the serum was separated. The SChE activity was determined by the method using S-butyrylthiocholine iodide as the substrate in the automatic analyzer Cobas Integra 800 (Roche Diagnostics, Rotkreuz, Switzerland) on admission. SChE activity was accepted as normal in the range of 5400–13,200 U/L. 19 A portion of serum was stored at −80°C until analysis of S100B level. The serum S100B levels of patients and controls were measured by enzyme-linked immunosorbent assay with commercially available kits (BioVendor Laboratory Medicine, Inc., Modrice, Czech Republic) in a TECAN Sunrise absorbance reader.
Statistical analysis
All statistical calculations were made using the Statistical Package for the Social Sciences for Windows version 15.0 (SPSS Inc. Headquarters, Chicago, Illinois, USA) software program. Data were analyzed with the Kolmogorov–Smirnov test for normality. Values were reported as median (minimum–maximum) for data that were not distributed normally. A value of p < 0.05 was accepted as statistically significant. To compare the groups, Mann–Whitney U, Kruskal–Wallis analysis of variance, and Bonferroni-corrected Mann–Whitney U (p < 0.017) tests were used. Analysis of variables within categories was done with the chi-square and Fisher’s exact tests, and correlation analysis was performed with the Spearman test. Receiver–operating characteristic curves for predicting mortality were generated from the data. Sensitivity and specificity were also calculated for S100B levels.
Results
A total of 36 patients poisoned by OP (median age 43 years; range, 18–88; 23 women, 13 men) and 20 controls (median age 38 years; range, 22–57; 13 women, 7 men) were included in the study. There were no significant differences in age or sex distribution between the groups.
The clinical characteristics of the patients are summarized in Table 1. Mean time interval between intoxication and ED admission was 3.9 ± 4.2 h (range, 1–22 h). The most common cause of poisoning was attempted suicide (72.2%). The most common route of exposure was oral ingestion (86.1%). Six kinds of OP compounds were involved in the poisonings, the most common of which were dichlorvos (36.1%) and parathion (27.8%). There were 25 patients (69.4%) in the high-severity group and 11 patients (30.6%) in the low-severity group.
Baseline characteristics of patients with organophosphate poisoning on admission.a
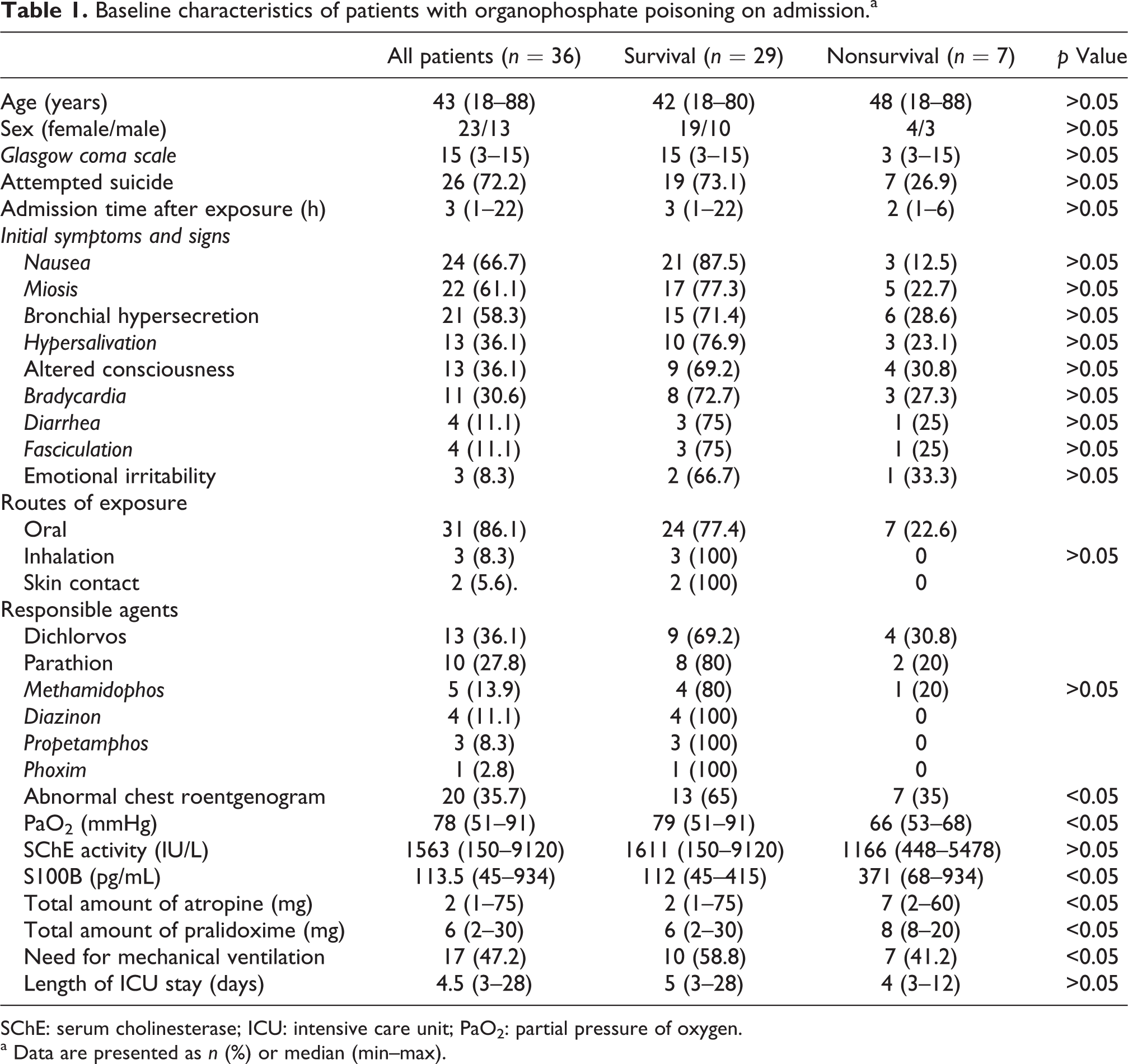
SChE: serum cholinesterase; ICU: intensive care unit; PaO2: partial pressure of oxygen.
a Data are presented as n (%) or median (min–max).
All patients were treated with atropine and pralidoxime. The median total dose of atropine was 2 mg (range, 1–75 mg). The median total dose of pralidoxime was 6 g (range, 2–30 g). Endotracheal intubation and mechanical ventilation were required in 17 patients (47.2%). A total of 7 patients (19.4%) died, with 3, 1, 2, and 1 patients dying on days 3, 4, 9, and 12 after poisoning, respectively. Of these seven patients, four died from intermediate syndrome, and the remaining three died from aspiration pneumonia and multiple organ failure. The remaining 29 patients (80.6%) recovered completely and were discharged from the ICU. The median length of ICU stay was 4.5 days (range, 3–28 days).
The median SChE level in patients was 1563 U/L, with a range of 150–9120 U/L. There was a positive correlation between SChE activity and PaO2 (p < 0.05), and between SChE activity and GCS score (p < 0.05). However, no correlation was found between SChE activity and total amount of pralidoxime, between SChE activity and total amount of atropine, or between SChE activity and length of ICU stay (Table 2). The serum SChE activity in the high-severity group (median = 1453 U/L; range, 150–8011 U/L) was lower than that in the low-severity group (median = 4711 U/L; range, 434–9120 U/L; p < 0.05). A comparison of serum SChE activity in mechanically ventilated patients (median = 1514 U/L; range, 150–5478) and nonmechanically ventilated patients (median = 2138 U/L; range, 325–9120) did not reveal a significant difference (p > 0.05). In addition, the comparison of SChE activity in survivors and nonsurvivors did not reveal a significant difference (p > 0.05; Table 1).
Correlation of SChE and serum S100B levels with six variables in patients with organophosphate poisoning.

SChE: serum cholinesterase; ICU: intensive care unit; PaO2: partial pressure of oxygen.
Serum S100B concentrations were significantly increased in the patients (median = 113.5; range, 45–934 pg/mL) compared with the controls (median = 62.5; range, 24–127 pg/mL; p < 0.05). There was a negative correlation between S100B and PaO2 (p < 0.05), between S100B and SChE activity (p < 0.05), and between S100B and GCS score (p < 0.05), and a positive correlation between S100B and total amount of pralidoxime (p < 0.05). No correlation was found between S100B and total amount of atropine administered or length of ICU stay (p > 0.05; Table 2). Serum S100B levels in the high-severity group (median = 146 pg/mL; range, 52–934 pg/mL) were significantly higher than in the low-severity group (median = 86 pg/mL; range, 45–125 pg/mL; p < 0.017) and in controls (median = 62.5; range, 24–127 pg/mL; p < 0.017). There was no significant difference between the low-severity group and controls with respect to serum S100B levels (p > 0.017). Serum S100B levels were higher in mechanically ventilated patients (median = 186 pg/mL; range, 52–934 pg/mL) than in nonmechanically ventilated patients (median = 108 pg/mL; range, 45–244 pg/mL; p < 0.05). Serum S100B levels were significantly higher in nonsurvivors than in survivors (p < 0.05; Table 1). When the serum S100B level of ≥236.5 pg/mL was used as a cutoff value to predict mortality, the sensitivity was 71.4%, specificity 89.7%, positive predictive value 62.5%, negative predictive value 92.8%, and accuracy 86.1% (Figure 1 and Table 3). A S100B cutoff concentration of 236.5 pg/mL conferred a 21.7-fold increased odds of mortality (odds ratio 21.7 [2.8–164.8]).

Receiver–operator characteristics curve for serum S100B predicting mortality rates. The most appropriate cutoff for sensitivity and specificity is indicated and equals 236.5 pg/mL S100B.
Best cutoff, sensitivity, specificity, and area under the curve (AUC) values of serum S100B for predicting the mortality (95% confidence interval (CI)).

Discussion
OP poisoning is a major problem worldwide, especially in developing countries, with millions of cases and hundreds of thousands of deaths occurring each year. 20 The commonly accepted toxicological effects of OP are primarily, if not entirely, due to the inhibition of AChE and to the subsequent accumulation of acetylcholine at the cholinergic synapses, causing overstimulation and disruption of neurotransmission in both the central and the peripheral nervous systems. 7,21 There are two cholinesterases commonly measured in the diagnosis of OP poisoning. 22 The cholinesterase present in neural tissues and also in erythrocytes is known as true AChE. The cholinesterase found in blood serum and synthesized by liver has been called pseudo or BuChE. 23 The SChE activity is also known as BuChE activity. The BuChE activity usually recovers before red cell AChE activity, and it is less specific for exposure to OP than red cell AChE activity. Additionally, day-to-day variation in the activity of this enzyme in healthy individuals may be as high as 20%. 22 In clinical practice, it may be difficult to evaluate the severity or predict the outcome in acute OP poisoning. The highly variable history of patients and difficulty in determining the ingested dose make predicting the outcome for an individual difficult and potentially hazardous because people admitted in good condition can deteriorate rapidly and require intubation and mechanical ventilation. 21 Therefore, an early biochemical marker to help in the evaluation of a patient with OP poisoning and to predict outcome is needed.
The present study demonstrated that in acute OP poisoning, SChE activity measured at the time of ED admission was lower in the high-severity group (severe and life-threatening poisoning) than that in the low-severity group (mild poisoning). There was a positive correlation between SChE activity and PaO2 and between SChE activity and GCS on admission. Respiratory complications of OP poisoning include the direct pulmonary effects of bronchorrhea and bronchoconstriction, muscular junction failure of the diaphragm and intercostal muscles, and loss of central respiratory drive. These effects can lead to hypoxemia and respiratory arrest. 22 Decreased PaO2 levels can be detected in severe OP poisoning. It has been reported that GCS is an effective and easily used tool for the determination of the severity of OP poisoning. 3,24 GCS is also useful in the determination of prognosis and prevention of complications in patients with OP poisoning. 25 In the literature, there is no consensus on the role of SChE in the assessment of the severity of OP poisoning. It was reported that low SChE and plasma cholinesterase levels support the diagnosis of OP poisoning but are not significantly related to the severity of poisoning. 8,26 In contrast, Rehiman et al. 27 used the peradenya organophosphorus poisoning (POP) scale in the grading of severity of OP poisoning and reported that the severity of poisoning was correlated with the SChE level. In a retrospective study that used the grading system developed by Bardin et al., 17,18 it was demonstrated that SChE activity was depressed significantly in all grades, and that it was related to the severity of OP poisoning. 19 These different results may be associated with the different grading systems used in those studies. However, our results indicate that SChE activity measured on admission may be useful in the assessment of clinical severity in cases of acute OP poisoning.
It was reported that the amount of atropine required to neutralize the effects of OP poisoning reflects the severity of poisoning. 7 In the present study, SChE activity did not correlate with total atropine or pralidoxime doses administered. In contrast, Sungurtekin et al. 21 reported a negative correlation between SChE level and atropine requirement. However, similar to our results, Nouira et al. 7 reported that SChE concentration was not correlated with the required dose of atropine. They asserted that one explanation of that finding is that atropine is only a muscarinic receptor antagonist, whereas cholinergic symptoms are associated with overstimulation, to varying degree, of both muscarinic and nicotinic receptors. The need for assisted ventilation is usually considered in serious patients with acute OP poisoning. In our study, there was no significant difference in SChE activity between mechanically ventilated patients and those who did not require mechanical ventilation. The current study revealed that SChE activity measured on admission may not be a reliable indicator for the selection of therapy or follow up in patients with acute OP poisoning.
In the present study, the prognostic value of SChE activity was also evaluated. No significant difference in SChE activity between survivors and nonsurvivors was found (p > 0.05). Similarly, it was reported that although it is a useful diagnostic parameter, SChE activity does not appear to have prognostic value in acute OP poisoning. 6 Similarly, Nouira et al. 7 also reported that the SChE level at admission has no prognostic value in acute OP poisoning. The SChE level is subject to a high degree of variability induced by a hereditary deficiency of this enzyme, liver dysfunction, malnutrition, iron deficiency anemia, and drugs such as cocaine, morphine, codeine, and succinyl choline, making this enzyme a less-than-perfect biomarker for OP poisoning, if the baseline levels in an individual are unknown. 20 It is not surprising that SChE activity measured only on admission has no prognostic value. The SChE activity may better predict patient mortality if it is determined at different times after poisoning. Serial SChE measurements can provide a guide to physicians in the reevaluation and management of patients with OP poisoning. 6,28
In addition to the inhibition of cholinesterase activity, the toxic effects of OP compounds on glia and neurons are another mechanism of OP poisoning. 9 Glia are the most common cells in the brain, providing critical nutritional, structural, and homeostatic support that is essential to the architectural modeling of the brain and to the establishment and maintenance of synaptic function. 29 It was suggested that glial cells may be affected by the neurotoxicity of OPs through different mechanisms. It was also suggested that glial cells provide neuroprotection against OP toxicity. 30
S100B is a calcium-binding protein primarily produced and released by astrocytes in the CNS. It has neurotrophic and gliotrophic activity, possibly having important roles in normal CNS development and recovery after injury. 13 S100B is also involved in the regulation of energy metabolism and in many immunological functions of the brain. 14 Increased S100B levels are associated with a variety of poisonings affecting the CNS, such as carbon monoxide poisoning, 13,15 aluminum neurotoxicity, 31 and benzodiazepine overdose. 16 S100B is a biomarker for the severity of brain damage and has been shown to assist the prognosis after acute ischemic, hemorrhagic, or traumatic CNS pathologies. 32 –34 Based on these reports, S100B has been proposed as a candidate molecule for guiding the management of patients with OP poisoning.
In a recently published study, it was demonstrated that serum levels of S100B are elevated in the acute phase of chlorpyrifos poisoning (2 h after exposure) in adult rats given a single dose, which indicated its usefulness in assessing OP toxicity. 9 To the best of the authors’ knowledge, the current study is the first to investigate the serum levels of S100B in OP-poisoned patients. This study demonstrated elevated serum levels of S100B in patients with acute OP poisoning. Serum S100B levels in the high-severity group were significantly higher than in the low-severity group. There was a negative correlation between S100B and PaO2, between S100B and SChE levels, and between S100B and GCS scores. In addition, there was a positive correlation between S100B level and total dose of pralidoxime administered. However, there was no correlation between S100B level and the total dose of atropine administered. The present study also found that serum S100B levels were higher in mechanically ventilated patients than in nonmechanically ventilated patients. These results support that increased S100B levels are associated with the clinical severity of OP poisoning. Moreover, high S100B levels may also predict the need for more aggressive therapy in acute OP poisoning. In the present study, the S100B level was significantly higher in nonsurvivors than in survivors. It was also determined that an S100B value over the cutoff value of 236.5 pg/mL was sensitive and specific in the prediction of mortality, at the levels of 71.4 and 89.7%, respectively. These data suggest that S100B may be helpful in predicting the mortality of patients with OP poisoning.
Limitations
This study has several limitations. First, the study is monocentric and limited by the small sample size to the extent that it is not possible to make any strong conclusions. Prospective and controlled studies involving larger numbers of OP-poisoned patients are needed. Second, there was no standard sampling time after the poisoning event for the SChE and S100B sampling. Although this interval was not significantly different between the survival and nonsurvival groups, it may have influenced this study’s results. It is impossible to control the time interval between poisoning and both SChE and S100B sampling in the ED. That interval is mostly dependent on the time between poisoning and the arrival of patients to the ED. Third, SChE activity and S100B were measured on admission. A series of measurements over time may provide more insight into the role of these markers in acute OP poisoning.
Conclusions
The SChE activity is related to the clinical severity of acute OP poisoning; however, it appears that SChE has no prognostic value in acute OP poisoning. The S100B level increases in patients with acute OP poisoning, and it may be a useful marker in the assessment of clinical severity and prediction of mortality. It may also suggest that more aggressive treatment as mechanical ventilation is required in patients with high S100B levels in this poisoning. Considering the preliminary nature of this study, the clinical utility of S100B protein in the management of OP poisoning should be further investigated with more comprehensive studies.
Footnotes
Authors’ Note
This paper was presented at the 2nd EurAsian Congress on Emergency Medicine, October 28–31, 2010, Antalya, Turkey.
Acknowledgments
The authors of this study are grateful for the financial support of the Scientific Research Foundation of Ondokuz Mayis University (OMU) in Samsun, Turkey. The authors also thank Dr Ahmet Tevfik Sunter of OMU for providing advice on the statistical analysis.
Funding
This work was financially supported by the Scientific Research Foundation of Ondokuz Mayis University (OMU) in Samsun, Turkey.
