Abstract
Ocimum sanctum L. (Lamiaceae) is found throughout India and in many parts of world. O. sanctum is used for the treatment of various health indications. In this lieu, it is of prime importance to investigate the safety aspects of the plant. Hence, the present study was conducted to investigate the possible genotoxic potential and acute oral toxicity of the extract of O. sanctum (OciBest™). The standard battery of in vitro genotoxicity tests, namely bacterial reverse mutation, chromosome aberration and micronucleus (MN) tests were employed to assess the possible mutagenic activity. The results showed that OciBest™ (7.9–2500.0 µg/mL) did not increase the number of histidine revertant colonies in Salmonella typhimurium strains (TA98 and TAMix) with and without exogenous metabolic activation (S9). OciBest™ (10.0–100.0 µg/mL) did not show structural chromosomal aberrations or increase in MN induction, with and without S9, at the tested dose range in both 4-h and 18-h exposure cell cultures. Thus, OciBest™ is not genotoxic in bacterial reverse mutation, chromosomal aberration and MN tests. In an acute oral toxicity test, rats were treated with 5 g/kg of OciBest™ and observed for signs of toxicity for 14 days and the results did not show any treatment-related toxic effects to Wistar rats.
Keywords
Introduction
The use of natural products as medicinal agents presumably predates the earliest recorded history. Ocimum is a genus of about 35 species of aromatic annual and perennial herbs and shrubs in the family Lamiaceae. One among them is Ocimum sanctum (synonym – Ocimum tenuiflorum), commonly called as Holy Basil or Tulsi, is a small herb found throughout India from Andaman and Nicobar islands to the Himalayas up to 1800 m above the sea level. 1 It is renowned for its religious and spiritual sanctity as well as for its important role in the traditional ayurveda and unani systems of holistic health and herbal medicine of the East. 2 Different parts of O. sanctum (leaves, flowers, stem, root and seeds) are known to possess therapeutic potentials 3 and have been used in indigenous ayurveda medicine 4 to cure wide array of diseases since time immemorial. One among the popular, well-supported traditional remedy for alleviating stress symptoms is O. sanctum. Charaka samhita, an ayurvedic text, mentioned that O. sanctum is considered to be an adaptogen, balancing different processes in the body and helpful for adapting to stress. 5 O. sanctum is known to possess anticarcinogenic, 6 antistress, 7 wound healing, 8 antioxidant, 9 anti-inflammatory, 10 antibacterial, 11 antidiabetic, 12 neuroprotective, 13 antifertility 14 and radioprotective 15 properties.
A wide range of chemical compounds including eugenol, ursolic acid (UA), linalool, limatrol, caryophyllene and sitosterol have been reported from O. sanctum. 16 Among all, the phytoconstituents eugenol (an essential oil) and UA of O. sanctum are the major active phytochemicals. 17 Many scientific studies have established the pharmacological effects of UA and eugenol on various systems such as immune system, reproductive system, central nervous system, cardiovascular system, gastric system, urinary system and blood circulatory system. 4,18 Besides the widespread therapeutic application, it is integral to investigate the mutagenicity aspects of this important plant. According to our knowledge, very limited scientific data are available on possible mutagenic effect of O. sanctum. Hence, the present study was conducted to investigate the genotoxic potential in a battery of in vitro assay systems (bacterial reverse mutation test (Ames II™ test); chromosomal aberration (CA) test; micronucleus (MN) test) and acute oral toxicity of O. sanctum extract (OciBest™). This study was carried out in compliance with the Test Guidelines of the Organization for Economic Cooperation and Development. 19 –22
Materials and methods
Chemicals
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, benzo (a) pyrene (B[a]P), 2-aminoanthracene (2-AA), 4-Nitroquinoline N-oxide (4-NQO), 2-nitrofluorene (2-NF), mitomycin C (MMC), glucose-6-phosphate, cytochalasin B, nicotinamide adenine dinucleotide phosphate and colchicine were purchased from Sigma–Aldrich (St Louis, Missouri, USA). The metabolic activation system, Aroclor™ 1254-induced S9 fraction, was procured from Moltox (Boone, North Carolina, USA). Fetal bovine serum (FBS) was supplied by Hyclone (Logan, Utah, USA). The Ames II™ Automated System for High Throughput Screening kit containing bacterial strains (TA98, TAMix), growth medium, exposure medium, ampicillin, indicator medium and incubation bag was purchased from Xenometrix (Allschwil, Switzerland). Trypsin–ethylenediaminetetraacetic acid and Ham’s F12K medium were purchased from Gibco Life Technologies (Grand Island, New York, USA). Dulbecco’s phosphate-buffered saline (DPBS) was procured from HiMedia (Mumbai, Maharashtra, India). All other chemicals and solvents used in this study were of the highest analytical grade available.
Bacterial strains and cell line
The Ames II™ test was performed using Salmonella typhimurium histidine auxotrophs, TA98 and TAMix. 10 µL of freshly thawed frozen Salmonella strains were inoculated in 10 mL of growth medium and the cultures were grown overnight at 37°C in an environmental shaker set at 180 r/min in the presence of ampicillin (50 µg/mL). Chinese Hamster Ovary K1 (CHO-K1) cell line (CCL-61TM) was procured from American Type Culture Collection (Rockville, Maryland, USA). The cells were cultured in Ham’s F12K media supplemented with 10% FBS, 10 mM sodium pyruvate in 37°C incubator maintained at 5% carbon dioxide.
Collection and identification of O. sanctum
Whole plant of O. sanctum Linn. was collected from regions of Tamil Nadu, India. Botanical identity was confirmed at National Institute of Science Communication and Information Resources. A voucher specimen (no. 106) was deposited in our herbarium. Extract of whole plant of O. sanctum Linn. was developed and named as OciBest™ by M/s Natural Remedies Pvt. Ltd (Bangalore, Karnataka, India).
Preparation of OciBest™
Coarse ground whole plants of O. sanctum (300 kg) were charged into a stainless steel jacketed extractor fitted with a reflux condenser. Methanol (1200 L) was added to the extractor and the contents were refluxed for 3 h by providing steam in the jacket. The liquid extract was drained from the extractor into a separate vessel and fresh methanol (1000 L) was added to the extractor containing the marc. The extraction procedure as above was carried out two times and the liquid extracts from each extraction step were separately subjected to distillation under vacuum (at <55°C) until a thick paste with a total solid content of 40–50% (w/w) was obtained. Thick paste obtained from the three extraction steps was mixed and dried under vacuum (<65°C) to get lumps of the extract. The extract lumps were then milled and sieved (# 40) to get a uniform-powdered extract of O. sanctum (around 27 kg). Methanol was stripped off from the marc by passing the steam and heating at 80°C. After removal of methanol, demineralized water (1200 L) was added in the extractor containing marc and the contents were refluxed for 3 h by providing steam in the jacket. The extraction of marc with water was carried out totally three times. The liquid aqueous extracts were drained from the extractor, combined and passed into a concentrator and were subjected to distillation under vacuum (at <75°C) until the total solid content in the liquid reached about 15–20% (w/v). The concentrated liquid was then spray dried to get the successive water extract of O. sanctum (around 45 kg).
The alcohol and water extracts were then analysed for the content of active constituents and blended to get OciBest™ with the required levels of active constituents. OciBest™ was ensured to comply with phytochemical specifications, namely, ociglycoside-I (>0.1%w/w) (1), rosmarinic acid (>0.2%w/w) (2) and triterpene acids (>2.5%w/w) (Oleanolic acid (OA) (3) and UA (4)). 7
Analysis method
Standards (1–4) and OciBest™ were prepared in methanol (high-performance liquid chromatography (HPLC) grade, Qualigens, Mumbai, India). The analytical method was validated for specificity, linearity, precision, accuracy and range of quantification. 23 Standards and OciBest™ solutions were injected to the HPLC system (Model LC 2010 A; Shimadzu, Kyoto Japan) consisting of quaternary pump with ultraviolet detector, auto injector and column oven with class LC software. The stationary phase used for standards 1 and 2 was phenomenex Luna column (C18, 5 μm, 250 × 4.6 mm) and phenomenex Luna column (C18, 2.5 μm, 100 × 3 mm) for standards 3 and 4. The mobile phase used for the detection of standards 1 and 2 was a gradient mixture of acetonitrile (solvent B) and 0.001 N monopotassium phosphate in HPLC grade water (solvent A). Solvents A and B were mixed in such a manner that the concentration of solvent B was increased from 10% to 30% as linear gradient in the first 18 min. From 18 min to 25 min, the concentration of solvent B was increased from 30% to 85% as a linear gradient at a flow rate of 1.5 mL/min. The detection wavelength was set at 278 nm. The mobile phase used for standards 3 and 4 was a degassed mixture of 67 volumes of acetonitrile and 33 volumes of water containing 0.25% ammonium acetate at a flow rate of 0.3 mL/min. The detection wavelength was set at 205 nm. HPLC chromatograms of OciBest™ were recorded (Figure 1(a) and (b)) and the quantification of standards 1–4 was achieved by external standard method.

HPLC chromatogram of OciBest™. (a) Identification of ociglycoside I and rosmarinic acid in OciBest™. (b) Identification of oleanolic acid and ursolic acid in OciBest™.
Bacterial reverse mutation test
The bacterial reverse mutation test was used to evaluate the ability of test substance to induce reverse mutation at histidine loci in TA98 and TAMix (mixture of six base pair mutant strains TA7001-7006). This mutagenicity test was conducted according to standard procedure. 19,24 –28 Briefly, S. typhimurium tester strains were exposed to OciBest™ via the fluctuation method with and without S9. Dimethyl sulphoxide (DMSO; 4%) was used as vehicle control. OciBest™ at a maximum concentration of 2500 µg/mL, which did not produce bacterial cytotoxicity, was used and subsequent concentrations (791.13 µg/mL, 250.36 µg/mL, 79.23 µg/mL, 25.07 µg/mL and 7.93 µg/mL) were determined by dividing the previous concentrations with square root of 10. OciBest™ at the concentration range of 7.9–2500.0 µg/mL, tester strain and S9 mix (when required) were added along with reversion indicator media into the wells of 384-well plate and incubated for 48 h at 37°C. All doses of the OciBest™, negative control (DPBS—0.96%) and positive controls were plated in triplicate. 2-NF (2 µg/mL) + 4-NQO (0.5 µg/mL) and 2-AA (5.0 µg/mL) were used as positive controls in the absence and presence of metabolic activation, respectively. After incubation, the revertant colonies were counted and positive response was determined by significant increase in the mean revertant per plate of at least one of the tester strains as compared to negative control.
Chromosome aberrations in CHO-K1 cells
Chromosome aberration (CA) test was performed as per Organisation for Economic Co-operation and Development (OECD) guideline no. 473.
20
CHO-K1 cells were seeded at a density of 7.5 × 105 cells per plate and allowed for overnight growth. Post-incubation, the cells were treated with different dose levels of OciBest™ (10.0–100.0 µg/mL) in the presence and absence of S9. OciBest™ at a maximum concentration of 100 µg/mL, which did not produce cytotoxicity to CHO-K1 cells, was used and subsequent concentrations (31.6 µg/mL and 10.0 µg/mL) were determined by dividing the previous concentration with square root of 10. DMSO (0.5%) and sodium chloride (0.96%) were used as vehicle control and negative control, respectively. MMC (0.2 µg/mL) was used as positive control for both short- (4 h) and long-term (18 h) exposure studies in the absence of metabolic activation, whereas B[a]P (20.0 µg/mL) was used as positive control for short-term exposure study only with metabolic activation. After treatment (4 h and 18 h for short- and long-terms, respectively), the cells were washed with DPBS and then colchicine (0.3 µg/mL) was added to block the cells at the metaphase stage before harvesting. At the end of incubation period, the cells were trypsinized and centrifuged. The cells were resuspended in hypotonic medium (75 mM potassium chloride (KCl)) and fixed (prechilled, three parts of ethanol and one part of glacial acetic acid). Approximately 3–4 drops of the fixed cell suspension from the height of 30 cm was transferred onto a clean microscope slide and stained with 5% Giemsa solution for 10 min. For scoring, 200 cells arrested at metaphase stage containing around 19–23 chromosomes were examined for chromosome break, chromatid break, deletion, ring, dicentric and rearrangements as indicated by Savage.
29
Experiments were performed in duplicates. Cytotoxicity was determined by calculating mitotic index (MI) according to the formula
In vitro MN test in CHO-K1 cells
MN test was carried out according to the OECD guideline no. 487.
21
CHO-K1 cells were incubated with OciBest™ (10.0–100.0 µg/mL) in both short- and long-term exposure studies. DMSO (0.5%) and sodium chloride (0.96%) were used as vehicle control and negative control, respectively. MMC (0.2 µg/mL) was used as positive control in the absence of metabolic activation for both short- and long-term exposure studies. B[a]P (20.0 µg/mL), on the other hand, was used only with metabolic activation in a short-term exposure study. Post-incubation, the cells were washed with DPBS, then cytochalasin B (3.0 µg/mL) was added to arrest cytoplasmic division of the cells. After trypsinization, cells were later suspended in 75 mM KCl and fixed using precooled fixative (ethanol and glacial acetic acid (3:1)). Approximately 3–4 drops of the cell suspension from the height of 5 cm was transferred onto a clean microscope slide. The slides were air dried overnight and stained with 10% Giemsa for 10 min. Experiments were performed in duplicates. The criteria for selecting the binucleated (BN) cells were based on the report by Fenech.
30
Per treatment, 2000 cytochalasin B-arrested cells were examined for the presence of MN. Cytokinesis block proliferation index (CBPI) was determined according to the formula
CBPI value of 1 was considered 100% cytotoxicity. 30
Acute oral toxicity
The animal experiment was conducted taking into consideration the Committee for the Purpose of Control and Supervision of Experiments on Animals guidelines and after approval by the Institutional Animal Ethics Committee. Female albino Wistar rats (8–12 weeks) were housed in polypropylene cages with stainless steel grill tops and provided with bedding of clean paddy husk. Temperature was maintained between 25 ± 2°C with 12 h each of dark and light cycle. The animals were acclimatized to laboratory conditions for 1 week prior to treatment. The rats were fed with standard laboratory pelleted feed (M/s Gold Mohur Foods and Feeds Ltd, Bangalore, Karnataka, India). The rats were deprived of feed overnight before and 3 h after the administration of OciBest™. Acute oral toxicity study was performed as per the OECD Guideline for the Testing of Chemicals (No. 420, Section 4: Health Effects) Acute Oral Toxicity-Fixed Dose Procedure. 22 OciBest™ suspended in demineralized water was administered by oral route to rats at a limited dose of 5 g/kg body weight to prove its safety under section ‘unclassified’ in the hazard category according to Globally Harmonized System. 22 On the day of dosing, all the animals were observed for mortality and clinical signs for first 10 min, 30 min, 1 h, 2 h, 4 h and 6 h after dosing and thereafter twice daily for mortality and once a day for clinical signs, for 14 days. Macroscopic examination was performed on animals found dead. Animals were sacrificed at the end of the study period of 14 days.
Data analysis
Data are expressed as mean ± SEM. One-way analysis of variance was performed on the results followed by a Dunnett’s test for multiple comparisons using GraphPad Prism 5.0 (GraphPad Software, Inc., San Diego, California, USA) statistical software package. The significance level was chosen at p < 0.05 for all statistical analyses in comparison with the respective negative control. 31
Results
Bacterial reverse mutation test
Histidine-requiring mutants of S. typhimurium TA98 and TAMix strains with and without metabolic activation were used to detect point mutations. No significant bacterial cell toxicity was observed upon treatment with OciBest™ up to a maximum concentration of 2500 µg/mL. The frequencies of spontaneous reversions agree with those reported for such strains. 26 –28 Addition of OciBest™ at concentrations 7.93 µg/mL, 25.07 µg/mL, 79.23 µg/mL, 250.36 µg/mL, 791.13 µg/mL and 2500.00 µg/mL did not show any significant increase in the number of revertant colonies in both the strains with and without exogenous metabolic activation. However, the positive controls (2-NF + 4-NQO, 2-AA) demonstrated a significant increase in the number of revertant colonies both in the absence and presence of metabolic activation (Tables 1 and 2). These results showed that OciBest™ was not able to induce mutagenesis in TA98 and TAMix strains of S. typhimurium.
Mutagenicity testing of OciBest™ in Salmonella typhimurium strain TA98.
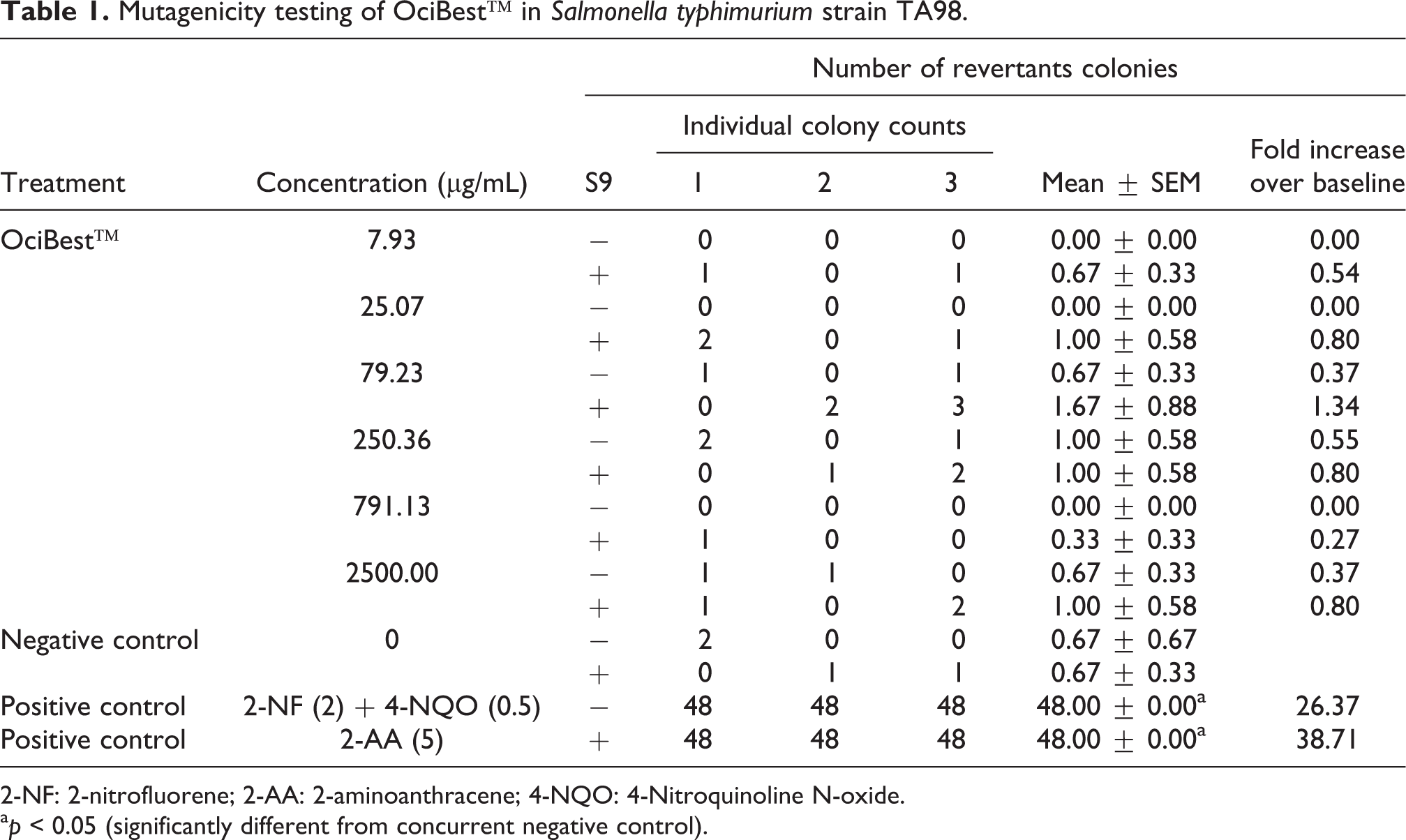
2-NF: 2-nitrofluorene; 2-AA: 2-aminoanthracene; 4-NQO: 4-Nitroquinoline N-oxide.
a p < 0.05 (significantly different from concurrent negative control).
Mutagenicity testing of OciBest™ in Salmonella typhimurium strain TAMix.
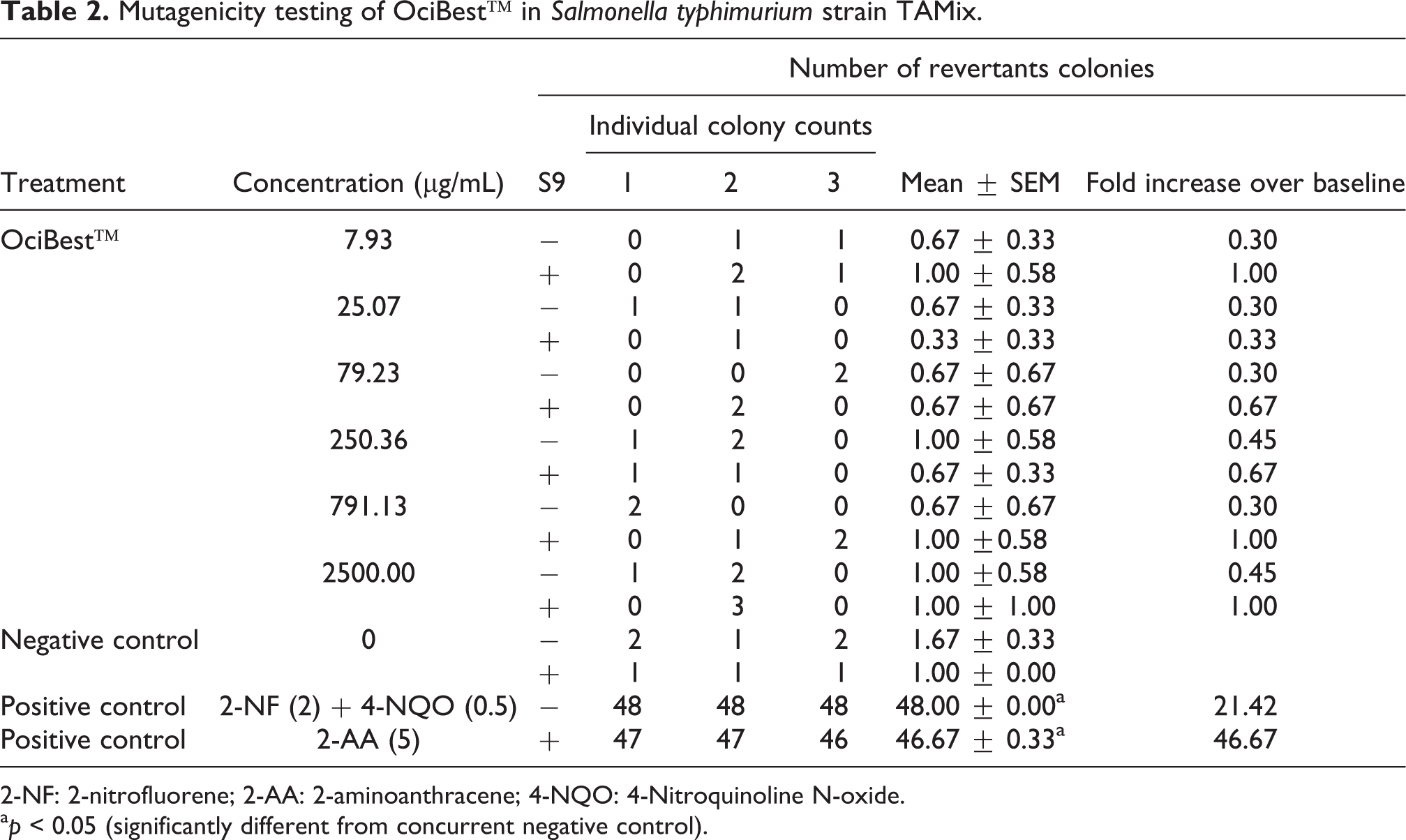
2-NF: 2-nitrofluorene; 2-AA: 2-aminoanthracene; 4-NQO: 4-Nitroquinoline N-oxide.
a p < 0.05 (significantly different from concurrent negative control).
In vitro CA analysis in CHO-K1 cells
MI values indicated that OciBest™ at the indicated concentrations did not produce cell toxicity in both short- and long-term exposure with and without exogenous metabolic activation. Cells were arrested at metaphase stage and were analysed for structural chromosomal aberrations. Treatment with OciBest™ did not induce any significant number of structural chromosomal aberrations at the tested concentrations (10.0 µg/mL, 31.6 µg/mL and 100.0 µg/mL) in short- and long-term exposure studies with and without S9. The negative control cultures had insignificant number of structural chromosomal aberrations, which were within the limit of published data. 26,28 The positive controls (MMC and B[a]P) were found to induce statistically significant number of structural chromosomal aberrations, namley, chromosome break, chromatid break and deletion in CHO-K1 cells (Tables 3 to 5).
Clastogenicity study upon short-term treatment of OciBest™ in the absence of metabolic activation in CHO-K1 cells.

CHO-K1: Chinese hamster ovary K1; ND: not detected.
aTotal aberrations (mean ± standard error of the mean from 100 metaphases per replicate per experiment; 200 total metaphases per treatment for the study).
b p < 0.05 (significantly different from concurrent negative control).
cIncreased compared to the concurrent negative control.
dMitotic index (mean ± standard error of the mean from 1000 cells per replicate per experiment; 2000 total cells per treatment for the study).
eDecreased compared to the concurrent negative control.
Clastogenicity study upon short-term treatment of OciBest™ in the presence of metabolic activation in CHO-K1 cells.

CHO-K1: Chinese hamster ovary K1; ND: not detected.
aTotal aberrations (mean ± standard error of the mean from 100 metaphases per replicate per experiment; 200 total metaphases per treatment for the study).
b p < 0.05 (significantly different from concurrent negative control).
cIncreased compared to the concurrent negative control.
dMitotic index (mean ± standard error of the mean from 1000 cells per replicate per experiment; 2000 total cells per treatment for the study).
eDecreased compared to the concurrent negative control.
Clastogenicity study upon long-term treatment of OciBest™ in the absence of metabolic activation in CHO-K1 cells.

CHO-K1: Chinese hamster ovary K1; ND: not detected.
aTotal aberrations (mean ± standard error of the mean from 100 metaphases per replicate per experiment; 200 total metaphases per treatment for the study).
b p < 0.05 (significantly different from concurrent negative control).
cIncreased compared to the concurrent negative control.
dMitotic index (mean ± standard error of the mean from 1000 cells per replicate per experiment; 2000 total cells per treatment for the study).
eDecreased compared to the concurrent negative control.
In vitro MN analysis in CHO-K1 cells
CBPI values indicated that none of the tested concentrations of OciBest™ significantly reduced the cell viability of CHO-K1 cells. MMC treatment showed significant decrease in CHO-K1 cell viability in both short- and long-term exposure cultures. In the MN test, percentage micronuclei (MNs) were not significantly altered upon treatment with OciBest™ at the tested concentrations (10.0 µg/mL, 31.6 µg/mL and 100.0 µg/mL) with and without S9 in short- (4 h) and long-term (18 h) exposure cultures. Both the positive mutagens (MMC and B[a]P) significantly increased the percentage of MNs. Percentage micronucleated BN cells and CBPI values for all treatment cultures are presented in Table 6.
Effect of OciBest™ on MN induction in CHO-K1 cells.

MN: micronucleus; BN: binucleated; MMC: mitomycin C; CHO-K1: Chinese hamster ovary K1; CBPI: cytokinesis block proliferation index.
aPercent MN-BN cells (mean ± standard error of the mean from 1000 cells per treatment per experiment; 2000 total cells per treatment for the study).
bCBPI (mean ± standard error of the mean from 1000 cells per treatment per experiment; 2000 total cells per treatment for the study).
c p < 0.05 (significantly different from concurrent negative control).
dIncreased compared to the concurrent negative control.
eDecreased compared to the concurrent negative control.
Acute oral toxicity
The rats treated with OciBest™ survived throughout the study period and did not show any adverse clinical signs immediately following dosing and during the observation period of 14 days (Table 7). OciBest™ did not reveal any major adverse effect on the body weight gain both during the first and second week of 14-day observation period. Overall, the percentage body weight gain during the complete 14-day observation period was found to be normal in all the animals (Table 8). On necropsy, no major gross pathological changes were observed in any of the treated rats (Table 7). Based on the findings of this study, OciBest™ was found to be safe up to 5 g/kg body weight after oral administration as a single dose to female albino Wistar rats.
Clinical signs and gross pathology findings in rats after treatment with OciBest™.

n = no. of animals.
Effect of OciBest™ on body weight and percent body weight gain in rats.

n = no. of animals.
Discussion
O. sanctum known as Tulsi is being used as medicinal herb for thousands of years without any known adverse effects. There had been number of studies conducted to establish the safety of this plant. 32 There are many reports highlighting the efficacy and safety of O. sanctum, but limited scientific reports have been published about its genotoxicity risk. Thus, this study was conducted to investigate the possible genotoxic potential of O. sanctum extract (OciBest™) using bacterial reverse mutation, chromosomal aberration and MN tests.
OciBest™ did not increase the number of histidine revertant colonies at any of the tested concentration in TA98 and TAMix strains of S. typhimurium, regardless of the metabolic activation system. A similar finding, but on another species of Ocimum, Ocimum selloi oil was evaluated in the Salmonella/microsome assay without and with S9 mixture. The oil, tested up to the toxicity limits (500.0–700.0 µg/plate), was not mutagenic to tester strains TA97a, TA98 and TA100. 33 One another species of Ocimum, Ocimum bascillicum and its major phytoconstituents were found to be not mutagenic and interestingly produced antimutagenic effect in a standard mutagenicity tests. 34,35 In chromosomal aberration and MN tests, OciBest™ neither induced structural chromosomal aberrations nor increased the frequency of MN formation at the tested concentrations in short- and long-term cultures, regardless of S9 activation. Similar to our findings, effect of O. sanctum water extract was studied on genotoxic effect induced by a synthetic progestin cyproterone acetate, on human lymphocytes using chromosomal aberrations, MI, sister chromatid exchanges and replication index as a parameters. The results indicated that the plant infusion per se does not have genotoxic potential but significantly reduced the genotoxicity of cyproterone acetate on human lymphocytes in vitro. 36 Administration of ethanolic extract of O. sanctum (OE) at a dose 5 g/kg body weight did not cause mutagenicity in the rats in a rodent bone marrow MN test. On the other hand, Repeat oral administration of OE at a dose level of 5g/kg for 7- and 21- day showed antimutagenic property by inhibiting MN formation induced by cyclophosphamide. The antimutagenicity of OE suggested an enhancement of detoxification enzymes against cyclophosphamide as shown in the reduction of MN formation. 37 O. sanctum has been reported to have anticlastogenic effect against MMC- and chromium-induced genotoxicity in human peripheral blood lymphocytes in vitro. 38 O. sanctum offered significant protection against DMPA (7,12-dimethylbenz (a) anthracene)-induced genotoxicity and oxidative stress by modulating xenobiotic metabolizing enzymes, reducing the extent of protein and lipid oxidation and upregulating antioxidant defense. 39
In an in vivo chromosomal aberration study, leaves of O. sanctum exhibited protective effect on chromium- and mercury-induced structural chromosomal aberrations. 40 The radioprotective effect of the leaf extract of O. sanctum was demonstrated against γ-ray-induced cytogenetic damage, and authors hypothesized that free radical scavenging is a likely mechanism of its protection. 15,41 Immu-21, a polyherbal formulation that contains O. sanctum as one of the ingredient, was given at 100 mg/kg, daily, over 7 days and 30 mg/kg daily over 14 days significantly inhibited cyclophosphamide-induced chromosomal aberrations. 42 The major phytoconstituent of O. sanctum, eugenol, did not show significant MN induction in an in vivo MN test. 43 UA and OA are triterpenoid compounds found in O. sanctum exerted antimutagenic property against doxorubicin-induced MN formation in peripheral blood and bone marrow of Balb/c mice. 44
Acute oral toxicity data on OciBest™ is used to satisfy hazard classification and labelling requirements for its risk assessment in human health and environment. 45 In this study, treatment of OciBest™ did not cause mortality, abnormal clinical signs or any significant pathological changes up to the dose level of 5 g/kg body weight. Also, the overall body weight gain was found to be normal in all the treated rats and hence resulted in labelling the OciBest™ as unclassified in the hazard category according to Globally Harmonized System. 22 Moreover, the administration of water and alcoholic extracts of O. sanctum did not present any acute toxic symptoms and well tolerated by albino mice up to 5 g/kg and 4 g/kg bodyweight, respectively. The acute lethal dosage (LD50) values for aqueous and alcoholic extracts were determined to be 6.2 g/kg and 4.6 g/kg body weight, respectively, in albino mice. 46 As described in OECD guidelines, Lagarto et al. 47 conducted oral acute and 90-day subchronic toxicity in Wistar rats of both sexes using lyophilized aqueous extracts of O. sanctum. Oral acute and subchronic toxicity tests showed no significant toxic effects attributable to the test substance. Extracts of O. sanctum were well tolerated, and no adverse changes were observed in Charles Foster rats in acute and subacute toxicity studies. No mortality, morbidity, gross pathological, body weight and biochemical parameters changes observed upon administration of O. sanctum. 48 The toxicity of fixed seed oil of O. sanctum in rats at a dose level of 30 mL/kg body weight was studied by intraperitoneal administration and the results indicated no mortality. In a subacute administration of Tulsi seed oil at a dose of 3 mL/kg exhibited no abnormal behavioural as well as histological changes in the brain, lungs, liver and kidneys. 49 In a detailed toxicological study, the aqueous extract of O. suave, another species of Ocimum, was administered to Swiss mouse and evaluated for acute, subchronic toxicity and teratogenic effects. In an acute toxicity study, intake of extract up to 8000 mg/kg did not show mortality and significant changes in general behaviour. Subchronic treatment did not reveal any change in body and organ weights, feeding habits or behaviour in treated groups of both sexes. Haematological analysis and blood biochemistry revealed no toxicity effects of the extract. No gross abnormalities or histological changes in vital organs were noticed. Teratogenic and fertility studies did not reveal any significant toxic manifestations or fetal abnormalities. 50
Beside its strong data on safety, there are few reports published on antifertility effect of Tulsi. The leaves of O. sanctum reported to produce antizycotic, anti-implantation and early abortifacient effects in women and experimental animals. The benzene and petroleum ether extracts of the leaves of O. sanctum have been reported to have antifertility activity in female rats. 14 Benzene extract of Tulsi had been reported to reduce the spermatogenesis by impairing the sertoli cell activity without affecting germ cells in male rats. 51 The major phytoconstituents of O. sanctum, such as UA 52 and OA, 53 have shown antifertility activity in male rats. Antifertility effects including abortifacient and antispermatogenic effects have been described in rodents using O. sanctum in high-dose levels. 54 –57 Effective clinical dose of OciBest™ for alleviating stress symptoms in humans is 1200 mg/day, 7 which extrapolated to a rat effective dose of around 107 mg/kg. In a rodent study, OciBest™ alleviated stress symptoms at a dose level of 100 mg/kg. 58 In this study, we have assessed the safety (acute oral toxicity) of OciBest™ at a dose of 5 g/kg, which is approximately 46 times more than effective dose. The in vitro effective concentrations for antistress activity of OciBest™ were found to be ranging from 6.25 µg/mL to 100 µg/mL (unpublished data). In this study, we have assessed the possible mutagenic effect at maximum concentrations of 100 µg/mL (chromosomal aberration and MN tests) and 2500 µg/mL (Ames II test), which is 1–25 times higher than the effective concentration. Hans Wohlmuth, Founding Editor of Botanical Pathways (issue 11), stated that the doses of Basil producing these effects are in the order of 1 g/kg body weight or more daily in rodents, which is equivalent to a daily dose of 50 g or more in humans. Based on the available scientific reports, our study results and a long history of safe traditional use, O. sanctum could be considered as safe ingredient.
In conclusion, OciBest™ was found to be nonmutagenic to TA98 and TAMix strains of S. typhimurium. OciBest™ did not induce or increase the occurrence of structural chromosomal aberrations or MN formation in CHO-K1 cells. Also the OciBest™ was found to be safe after oral administration as a single dose to female albino Wistar rats up to 5 g/kg body weight. Therefore, OciBest™ is not mutagenic in the tested standard battery of genotoxicity tests and found to be safe in an acute oral toxicity study.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of Conflicting Interests
Trade mark of the product under study is owned by the company by which all the authors work.


