Abstract
The Globally Harmonized System for Classification and Labelling of Chemicals (GHS) considers metallic alloys, such as nickel (Ni)-containing stainless steel (SS), as mixtures of substances, without considering that alloys behave differently compared to their constituent metals. This study presents an approach using metal release, explained by surface compositional data, for the prediction of inhalation toxicity of SS AISI 316L. The release of Ni into synthetic biological fluids is >1000-fold lower from the SS powder than from Ni metal, due to the chromium(III)-rich surface oxide of SS. Thus, it was hypothesized that the inhalation toxicity of SS is significantly lower than what could be predicted based on Ni metal content. A 28-day inhalation study with rats exposed to SS 316L powder (<4 µm, mass median aerodynamic diameter 2.5–3.0 µm) at concentrations up to 1.0 mg/L showed accumulation of metal particles in the lung lobes, but no signs of inflammation, although Ni metal caused lung toxicity in a similar published study at significantly lower concentrations. It was concluded that the bioaccessible (released) fraction, rather than the elemental nominal composition, predicts the toxicity of SS powder. The study provides a basis for an approach for future validation, standardization and risk assessment of metal alloys.
Introduction
Stainless steel (SS) is an iron (Fe)-based alloy that contains at least 10.5% chromium (Cr) to enable the formation of a highly corrosion resistant passive surface oxide. This very thin (10−9 m) oxide layer is spontaneously formed under the influence of oxygen from air or water and is mainly composed of Fe(III)- and Cr(III)-oxides. 1 If locally damaged by, for example, exposure to chlorides or mechanical scratching, it repassivates and regains its protective capacity. By increasing the Cr alloy content to 17–20%, the corrosion resistance improves significantly. 1 The resistance can be enhanced even further by alloying with elements such as manganese (Mn), molybdenum (Mo) and nickel (Ni; <1–38%). The latter elements are not the typical constituents of the passive oxide but are rather enriched in an alloy surface layer adjacent to the surface oxide to improve repassivation. 2,3
The passive surface oxide is the first line of defence reducing the corrosion rate to very low levels (high corrosion resistance) and it also hinders to a large extent the release of ions of elements such as Fe, Cr, Ni and other alloy constituents into the surroundings, for example, into different biological fluids. 4 –13 The case reports on the adverse health effects, like skin allergies, related to SS appliances are very rare compared with their widespread use. Allergic reactions of persons with Ni allergy exposed to SS are also extremely uncommon. 14 –18 Compared with the normal dietary intake of Ni and Cr, the release of these metals from cookware is at non-significant levels. 19,20 Low release of alloy components suggests that SS is unlikely to cause local or systemic toxic effects.
The Globally Harmonized System for Classification and Labelling of Chemicals (GHS) 21 considers metal alloys, such as SS, as the mixture of substances. The same interpretation is applied under the European Chemicals Regulation (REACH) 22 and the Classification, Labelling and Packaging (CLP) regulation related to it. 22 Ni metal is classified for repeated dose toxicity (STOT RE 1, H372) following inhalation, as a skin sensitizer (Skin Sens. 1, H317) and also as suspected of causing cancer (Carc. 2, H351) in the European Union (EU). 23 As a consequence, SSs alloyed with more than 1 wt% Ni have to be classified for repeated dose inhalation toxicity under the CLP regulation. Elemental Fe and Cr, the other main alloy components, are not classified as toxic and are hence not regulated.
Triggered by the lack of quantitative metal release data from SSs with relevance for inhalation exposure (welding fumes excluded), and the lack of understanding between surface characteristics and metal release processes, a stepwise iterative research programme was initiated by the research team at the Royal Institute of Technology (KTH) in 1990 and still continues. A reliable and reproducible in vitro testing methodology was designed and employed to quantitatively assess the release of metals (Cr, Fe, Ni, Mo and Mn) from a range of massive forms of SS grades, screening different ferritic, austenitic and duplex microstructures, all in relation to changes in surface composition. 24 –27 The methodology was later applied to micron-sized SS powders 6 –10,28 and other alloys.
In this study, we hypothesized that in the case of metals and alloys, the fraction of the metal ionic species released from the material in relevant biological fluids (i.e. the bioaccessible fraction) can be considered as the effective concentration, which has a potential to cause either local or systemic toxicity. Since Ni has shown significant pulmonary toxicity in rats in repeated dose inhalation tests at concentrations of 0.1 mg/m3 and higher 29,30 , as reviewed in the study by Santonen et al., 31 it was relevant to study whether any differences in pulmonary toxicity between SS and Ni metal would be predicted by the Ni content in the alloy or rather by the Ni release observed in in vitro studies. The aim of this study was to investigate whether the in vitro metal release data, explained by alloy surface characteristics, can predict the toxicity of alloys using SS as an example. This was achieved by performing a series of studies and comparisons, including (1) comparison to existing data on surface characterization and in vitro metal release studies with inert gas-atomized 316L micron-sized SS (powder <4 µm, mass median aerodynamic diameter (MMAD): 2.5–3.0 µm) and pure metal powders of Fe, Cr and Ni; (2) an in vivo 28-day repeated dose SS 316L inhalation study in rats and (3) comparison of the results from these studies with existing data on the toxicity of the individual metal constituents of SS, especially of Ni metal.
Methods
Selection of SS grade
Previous metal release studies screening SSs with different microstructures in artificial biological fluids of relevance for inhalation (artificial lysosomal fluid (ALF), pH 4.5 and Gamble’s solution (GMB), pH 7.4) 23 have shown that the release of metals is very low with relatively small differences between different grades, even in fairly aggressive media. Of the SS grades examined, AISI 316L is the one that releases highest amounts of Ni, although its bulk Ni content is lower (10.7%) than that of grade 310 (19.1%), which was also tested. The release of metals from SS alloys is non-proportional to the alloy composition. 4,24 As Ni is the main constituent in SSs causing concern for human health, AISI 316L was selected as a representative grade for SS alloys for further in vitro metal release and surface reactivity studies, and for a 28-day repeated dose inhalation study in rats.
SS welding fume powder particles are significantly different from the inert gas-atomized SS powder. The most important difference from a toxicological perspective is that the gas-atomized SS particles have a thin (a few nanometre) 32 surface oxide composed only of trivalent Cr and Fe, 10 whereas welding fume particles reveal surface oxides that are at least >10-fold thicker 33 compared with the particles investigated here and enriched in, for instance, chlorine, fluorine and potassium. 33 It has, in addition, been shown in numerous publications that welding fume particles are composed of a large amount of soluble chromate 34 –36 or even only chromate (no trivalent Cr), 36 which is dissolved as chromates into solution 33 at concentrations about 30,000-fold higher 37 in comparable solutions 38 as in this study for inert gas-atomized particles. Their release is however strongly depending on welding conditions. 37 The welding fume particles cause major health concerns due to their Cr(VI)-contents. No Cr(IV) is actually present in the AISI 316L SS powder selected for this study or on massive SS. 10 Welding fumes can be considered as separate end use problems and are not directly related to health hazard assessments of metals or alloys as such. Therefore, welding fume particles are not in focus or relevant for comparison in this article.
Surface characteristics of SS 316L powder particles
For the inhalation study and the in vitro release tests, fine SS 316L powder (geometrical diameter: 10% sized <1.1 µm, 50% sized <1.8 µm and 90% sized <3.6 µm, based on supplier powder sizing data, here referred to as <4 µm) was used. The specific surface area of the powder was 0.700 m2/g, as determined by Brunauer–Emmet–Teller (BET) measurement. 8 The particles were produced by inert gas-atomization by Osprey Metals Ltd (Neath, UK). Characteristics of fine 316L powder are illustrated in Figure 1. The individual particles were mainly spherical (Figure 1, top) as a result of the gas-atomization production process. Despite a chemical composition suggesting an austenitic (non-magnetic) microstructure, most of the particles actually have a ferritic structure (ferromagnetic). 39 The reason is connected to an extremely high cooling rate during the production of fine powder. 39 In addition, the surface oxide of the powder was significantly enriched in Mn (47 wt%: Mn/(Cr + Mn + Fe); compare Figure 1, bottom left), 10,32,40 despite a very low bulk alloy content (1.4 wt%). This is also an effect from the production process of fine particles and differs from coarser particles (<45 µm investigated) and from particles formed, for example, by grinding of massive sheets. 32 However, the bulk composition of the majority of the fine particles was identical with that of massive 316L. 32,39 –41 When the particles were immersed in solution, some agglomerates (50–300 µm) were formed, but most particles were sized less than 3 µm8 (compare Figure 1, bottom right). After exposure to citric acid or ALF (high content of citric acid), metallic Ni has been detected on the outermost surface, most probably as a result of a reduced oxide thickness. 10
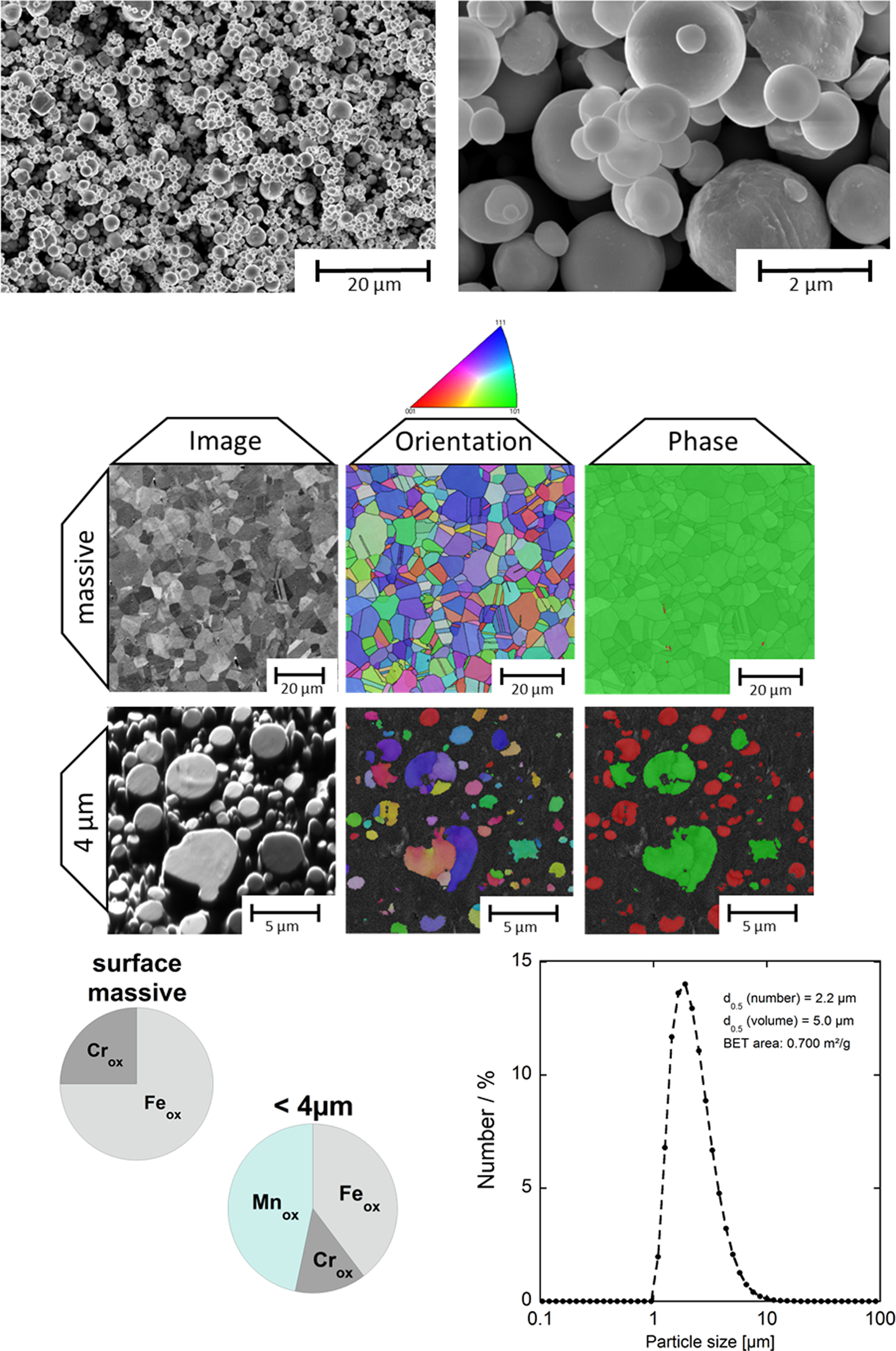
Characterization of the 316L powder (<4 µm) compared with massive 316L. Top: FEG-SEM images at different magnifications (left: ×5000, right: ×50,000); middle: FEG-SEM and electron backscatter diffraction measurements of massive 316L and individual particles 39 ; bottom left: x-ray photoelectron results of the relative composition of Cr, Fe and Mn (in wt%) in the outermost surface oxide 10 ; bottom right: particle size distribution (in number %) and median size (by number and volume %) and BET area, measured by means of laser diffraction in PBS. 8 BET: Brunauer–Emmet–Teller; Cr: chromium; Mn: Manganese; FEG-SEM: field emission gun-scanning electron microscopic; PBS: phosphate-buffered saline; Fe: iron.
In vitro release tests
Experimental details are given elsewhere, 7 –10 where metal release data of powder particles of SS 316L, Ni, Cr and Fe metals are published. The nominal compositions of the powders were Fe (>99.96 wt%, Höganäs, Höganäs, Sweden) for Fe metal, Cr (>99.76 wt%, Delachaux S.A. (Gennevilliers Cedex, France)) for Cr metal, Ni (>99.74 wt%, Inco 123 powder, NiPERA, Inc. (Durham, North Carolina, USA)) for Ni metal and 68.9 wt% Fe, 16.8 wt% Cr, 10.3 wt% Ni, 2.1 wt% Mo, 1.4 wt% Mn, 0.5 wt% Si, 0.03 wt% C and 0.01 wt% S for SS 316L (Osprey Metals Ltd). In summary, metal release was studied by immersing a powder (100 mg/L) for different periods into various synthetic biological fluids. The temperature was 37°C. The immersion studies were conducted under dark conditions in acid-cleaned polymethylpentene jars in incubators with controlled shaking. Time-dependent studies were conducted using triplicate powder samples and one blank sample without any powder. After the exposures, the particles were separated from the solution by centrifugation. The supernatant was then poured into polyethylene flasks and acidified to a pH <2 prior to the metal analysis with atomic absorption spectroscopy. The exposures of abraded massive sheet were conducted following the procedure described by Herting et al. 5
Information on the solution compositions is given in the respective referred articles. The proteins and phosphate-buffered saline (PBS, pH 7.4) used are identical as described in the study by Lundin et al. 41 ALF has been considered as a fairly good test fluid for simulation of lung inflammation related to inhalation of powder particles. 42 For comparative reasons, in addition to the previously published data, also manganese oxide (MnO2) particles (Sigma-Aldrich, Lot no. MKBC2119V, Stockholm, Sweden) were exposed to ALF, pH 4.5, for 168 h (1 week), with the procedure described by Hedberg et al. 8 The reason for using a MnO2 powder instead of Mn metal powders was based on previous findings, which showed that the total Mn content is predominantly present in its oxidized form at the surface of the 316L powder (1.4 wt%), a consequence of the atomization process. 10,32,40
The specific surface area of the MnO2 particles was determined to be 0.054 m2/g, measured by the BET method as described by Hedberg et al. 8 Metal release/dissolution tests into different solutions for most of the powders described here were previously made in gastric fluid (pH 1.5), 7 ALF (pH 4.5), 7,8,10 constituents of ALF (pH 4.5), 10 artificial sweat (pH 6.5), 9 fresh water solutions of different pH and salt concentrations, 44 PBS (pH 7.4) 7 and artificial tear fluid (pH 8.0). 9
28-Day repeated dose toxicity test
A 28-day repeated dose nose-only inhalation study with rats using SS powder (grade 316L) was carried out by SafePharm Laboratories (now Harlan Laboratories Ltd, Shardlow, UK). The study complies with the requirements of the OECD 412 guideline and was performed in compliance with UK Good Laboratory Practice (GLP). The study was designed and conducted to cause the minimum suffering or distress to the animals consistent with the scientific objectives and in accordance with the Harlan Laboratories Ltd, policy on animal welfare and the requirements of the United Kingdom's Animals (Scientific Procedures) Act 1986. This protocol was subject to review as part of the Harlan Laboratories Ltd, Ethical Review Process.
Animals
Five groups of five female and five male Wistar rats, obtained from Harlan Ltd (Oxon, UK), were used for the exposure studies. The animals were acclimatized for 11 days before the exposures. At the start of the treatment, the animals were aged 5–8 weeks. The animals were housed in groups of five by sex in solid-floor polypropylene cages with SS mesh lids and softwood flake bedding (Harlan Ltd). The animals were allowed free access to water and food (a pelleted diet; Rodent 2014C Teklad (Certified) Diet, Harlan Ltd).
Exposure
The animals were exposed to SS powder grade 316L (obtained from Osprey Metals Ltd) at target concentrations of 0.01, 0.10, 0.30 and 1.00 mg/L by inhalation (nose only). The particle size (MMAD) was ≤3 µm. A control group (five female and five male rats) was treated in the same way as the study groups, but exposed to air only, at the same air-flow rates as in the 1.00 mg/L group. The exposures were carried out for 6 h/day, 5 days/week, followed by 2 days of rest, for a period of 4 weeks (28 days). All animals were killed at day 29.
Atmosphere generation
For the low concentration (0.01 mg/L), a dust atmosphere was produced from the test article using a ‘Wrights Dust Feeder (BGI Inc., Waltham, Massachusetts, USA) located at the top of the exposure chamber and driven by a variable speed motor. The dust feed was connected to a metered compressed air supply. For the other concentrations, a dust atmosphere was produced from the test material using a SAG 410 Solid Aerosol Generator I (TOPAS GmbH, Dresden, Germany) located adjacent to the exposure chamber. The SAG 410 was connected to a metered compressed air supply. A particle separator was introduced before the aerosol entered the exposure chamber in order to remove large particles and thereby increasing the inhalable portion of the generated aerosol for all groups. Compressed air was supplied by means of an oil free compressor and passed through a water trap and respiratory quality filters before it was introduced to the dust feeder or SAG 410.
The cylindrical exposure chamber had a volume of approximately 30 L (dimensions: 28 cm diameter × 50 cm high). The concentration within the exposure chambers was controlled by adjusting the rate of the variable speed motor and/or the air flow rate through the chambers and test material feed rate from the SAG 410. The extract from the exposure chamber passed through a ‘scrubber’ trap and was connected with a high efficiency filter to a metered exhaust system. The chamber was maintained under negative pressure. Homogeneity of the test atmosphere within the chamber was not specifically determined during this study. Chambers of the same design (ADG Developments Ltd, Hitchin, Herts, UK) have been fully validated and shown to produce evenly distributed atmospheres in the animals’ breathing zone with a wide variety of test materials. 45 Prior to the start of the study, test material atmospheres were generated within the exposure chambers. During this characterization period, air flow settings, test material input rates and the generation systems were varied to achieve the required atmospheric concentrations.
Exposure procedure
Each rat was individually held in a tapered, polycarbonate restraining tube fitted onto a single tier of the exposure chamber and sealed by means of a rubber ‘O’ ring. Only the nose of each animal was exposed to the test atmosphere. A separate chamber was used for each group and the position of each animal around the chamber was rotated daily to minimize any possible exposure.
Exposure chamber atmosphere concentration
The test atmosphere from each exposure chamber was sampled at least three times per day (once in the first hour, third hour and final hour) and the actual concentration of the test material was calculated. The gravimetric method used glass fibre filters (Gelman type A/E 25 mm (PALL Corporation, Ann Arbor, Michigan, USA)) placed in a filter holder. The holder was temporarily sealed in vacant port in the exposure chamber in the animals’ breathing zone and a suitable, known volume of exposure chamber air was drawn through the filter using a vacuum pump. Each filter was weighed before and after sampling in order to calculate the weight of collected test material. The above procedures do not apply to the control group exposure. No test material was introduced into the chamber and therefore no determination of concentration was required.
Particle size distribution
The particle size of the generated atmospheres inside each exposure chamber was determined using Marple Personal Cascade Impactors (Westech IS Ltd, Beds., UK). These devices consisted of six impactor stages (9.8, 6.9, 3.9, 1.4, 0.88, 0.28 and <0.28 or 9.8, 6.3. 3.9, 1.7, 0.8, 0.27 and <0.27 µm cut points) with SS collection substrates and a back up glass fibre filter, housed in an aluminium sampler. The sampler was temporarily sealed in a sampling port in the animals’ breathing zone and a suitable, known volume of exposure chamber air was drawn through it using a vacuum pump. Each week, sampling was performed once during the first hour of the exposure and once during the last hour of the exposure.
Examinations
Individual clinical observations were performed prior to the start of each exposure, 3 h after the start of the exposures and as soon as possible after removal from the chamber at 6 h. Individual body weights were recorded on day 1 (prior to start of treatment) and on days 8, 15, 22 and 29. Food consumption was recorded for each cage group at weekly intervals throughout the study. Water intake was observed daily by visual inspection of the water bottles.
Haematological (haemoglobin, erythrocyte count, haematocrit, erythrocyte indices, total and differential leukocyte count, platelet count and reticulocyte count) and blood chemical (urea, calcium, glucose, inorganic phosphorus, total protein, aspartate aminotransferase, albumin, alanine aminotransferase, albumin/globulin (A/G) ratio, alkaline phosphatase, sodium, creatinine, potassium, total cholesterol, chloride and total bilirubin) investigations were performed on all animals from each test and control group at the end of the study (day 29). Blood samples were obtained from the lateral tail vein. All animals were subjected to gross necropsy examination. Weights were recorded for selected organs. Tissues were preserved/fixed in 10% formalin. Lungs were inflated to approximately normal inspiratory volume with buffered 10% formalin before immersion in fixative. Histopathological examination of selected tissues, including lung and nose, was performed.
Statistics
Haematological, blood chemical, organ weight (absolute and relative to terminal body weight), weekly body weight gain and quantitative functional performance and sensory reactivity data were assessed for dose–response relationships by linear regression analysis, followed by one-way analysis of variance (ANOVA) incorporating Levene’s test for homogeneity of variance. Where variances were shown to be homogenous, pairwise comparisons were conducted using Dunnett’s test. Where Levene’s test showed unequal variances, the data were analysed using non-parametric methods: Kruskal–Wallis ANOVA and Mann–Whitney U test. p < 0.05 was considered as statistically significant. Data were analysed using the SPSS software (Statistical Package Version 11.5).
Toxicological evaluation
The outcome of the repeated dose toxicity study with fine 316L powder was evaluated by comparing the results obtained in this study with reported toxicity findings observed in tests performed with the pure main constituent metals of SS (especially Ni). These results were compared with the in vitro data on the surface characteristics of SS and the differences in release rates of metal constituents from SS, Fe, Cr and Ni metals and Mn(IV) oxide powders.
Results
Metal release from the 316L powder – comparison with massive 316L and pure metal and MnO2 powders
The release of Fe, Cr and Ni from 316L powders has been thoroughly investigated as a function of particle size 6 and in different synthetic media of relevance for inhalation and dermal contact. 6,8,9 The total release of metals from 316L powders into the different test fluids is very low, although complexing solutions and proteins enhance the release of metals from the 316L powder (Figure 2). 11,42

Metal (Fe, Cr, Ni and Mn) release (metal released per amount of particles loaded (microgram per microgram)) from the 316L powder (<4 µm) into different solutions, ALF, citric acid, sodium chloride and water (all initially at pH 4.5) after 24 h. Data are adapted from Hedberg et al. 10 ALF: artificial lysosomal fluid; Cr: chromium; Mn: Manganese; Ni: nickel; Fe: iron.
Data on the release of constituent metals from SS 316L powder and from pure Cr, Fe and Ni metals and MnO2 into ALF are summarized in Figure 3 and Table 1.
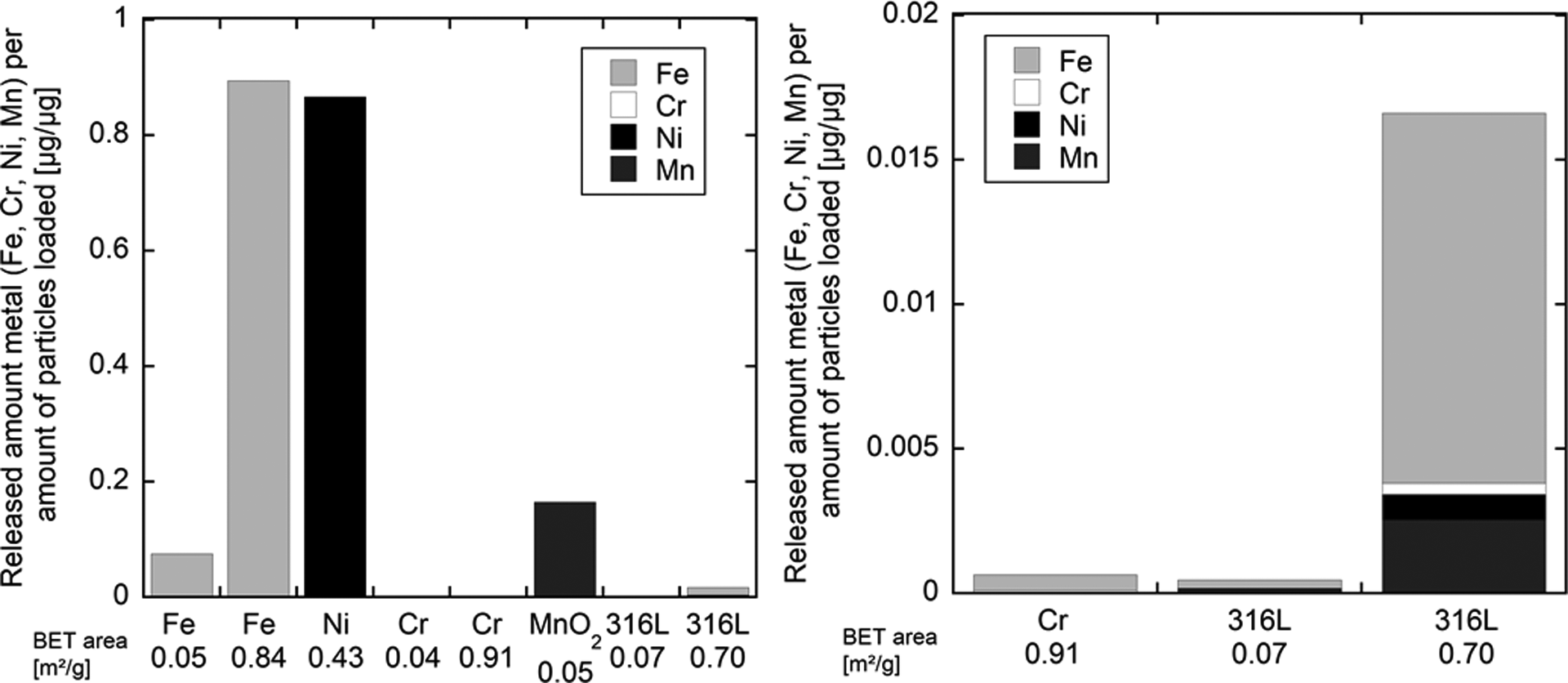
Released amount of metals (Fe, Cr, Ni and Mn) per amount of loaded powder (microgram per microgram) for differently sized metal powders of pure Fe, pure Ni, pure Cr, Mn dioxide and 316L powder, sized <4 µm (BET area 0.7 m2/g) and <45 µm (BET area 0.07 m2/g) into ALF (pH 4.5) after 168 h. The specific surface area (measured by the BET method 8 ) is given for comparison. The graph to the right is of higher magnification. Parts of the data on Fe, Ni and Cr and 316L powders are adapted from reports published by Hedberg et al. 8 , 10 ALF: artificial lysosomal fluid; BET: Brunauer–Emmet–Teller; Cr: chromium; Mn: Manganese; Ni: nickel; Fe: iron.
Released amount of metals (Fe, Cr, Ni, Mn) per amount of loaded powder for differently sized powders of Fe metal, Ni metal, Cr metal, Mn dioxide and 316L powder, sized <4 µm and <45 µm into ALF (pH 4.5) after 168 h.a

<LOD: below limit of detection; ALF: artificial lysosomal fluid; BET: Brunauer–Emmet–Teller; Cr: chromium; Fe: iron; Mn: Manganese; Ni: nickel.
The release of Ni from SS powder into ALF was more than 1000-fold lower than the release of Ni from Ni metal powder (Figure 3, Table 1 8 ). When normalized to the surface area, the release of Ni from the SS 316L powder compared with Ni powder was 1600-fold lower for the small powder particles (<4 µm) and 8400-fold lower for the <45 µm particles. 8 Similarly, significantly lower release of, for example, Fe from the fine 316L powder compared with an Fe powder has previously been shown for a variety of synthetic body fluids with solution pH values ranging from 1.5 to 8.0 (including gastric fluid, GMB, PBS, ALF, artificial sweat and artificial tear fluid). 7,9
Although the gas-atomization techniques used to produce the SS powders created a surface composition which was different from that of massive SS, the Ni release rate (normalized to the surface area) from SS powder was comparable with that of massive 316L (compare Figure 4). Ni release from the < 4-µm powder was similar to that of abraded massive 316L in solutions with and without proteins (Figure 4, left). In ALF, it was comparable to that of as-received 316L massive sheet and lower compared with abraded massive 316L.

Released amount of nickel (microgram per centimetre square) from massive (abraded) 316L and the <4 µm stainless steel 316L powder after 24 and 168 h of exposure to PBS (pH 7.4) with and without 10 g/L bovine serum albumin (PBS + BSA) or 2.2 g/L lysozyme (from chicken egg white; PBS + LYS; left) and after 168 h of exposure in ALF (pH 4.5; right). The data of the figure to the left were published in the study by Hedberg et al. 47 , the data marked with “*” were published in the study by Herting et al. 5 and the data marked with “#” published in the study by Midander et al. 6 . ALF: artificial lysosomal fluid; PBS: phosphate-buffered saline.
Recent studies have shown that the presence of proteins in solution can enhance the release of metals from massive 316L and Cr powders. 42 Although complexing solutions and proteins do enhance the relative release of metals from 316L (Figures 2 and 4), 10,42 the release is still very low compared with pure Fe or pure Ni (compare Figure 3 and Table 1).
In vivo 28-day repeated dose toxicity test on rats
Based on the particle size distribution determinations, the calculated MMAD of the particles in the exposure chamber was 2.87 ± 0.43 µm (0.01 mg/L group), 2.50 ± 0.57 μm (0.10 mg/L group), 2.53 ± 0.41 µm (0.30 mg/L group) and 3.04 ± 0.54 µm (1.00 mg/L group). The geometric standard deviation (GSD) was 2.16, 2.67, 2.63 and 2.32 for the 0.01, 0.10, 0.30 and 1.00 mg/L groups, respectively. The mean measured test atmosphere concentrations for the exposure groups were 0.015 mg/L (0.01 mg/L group), 0.141 mg/L (0.10 mg/L group), 0.349 mg/L (0.30 mg/L group) and 1.040 mg/L (1.00 mg/L group).
In the 28-day inhalation study 46 with SS powder at concentrations of up to 1.0 mg/L (1 g/m3), no deaths or clinical signs of toxicity were observed during the exposure period, and the treatment did not affect food or water consumption. No statistically significant changes in body weights were seen (Table 2; Figure 5). No treatment-related changes were observed for the haematological or blood chemical parameters investigated.

Body weights ± SD of (a) male (b) female rats, exposed to 0, 0.01, 0.1, 0.3 and 1 mg/L stainless steel 316L powder. Body weights were measured on days 1, 8, 15, 22 and 29 of the experiment.
Summary table of in-life phase of the 28-day repeated dose exposures inhalation (nose only) toxicity study in rats.

aStatistically significant from control p < 0.01.
bStatistically significant from control p < 0.001.
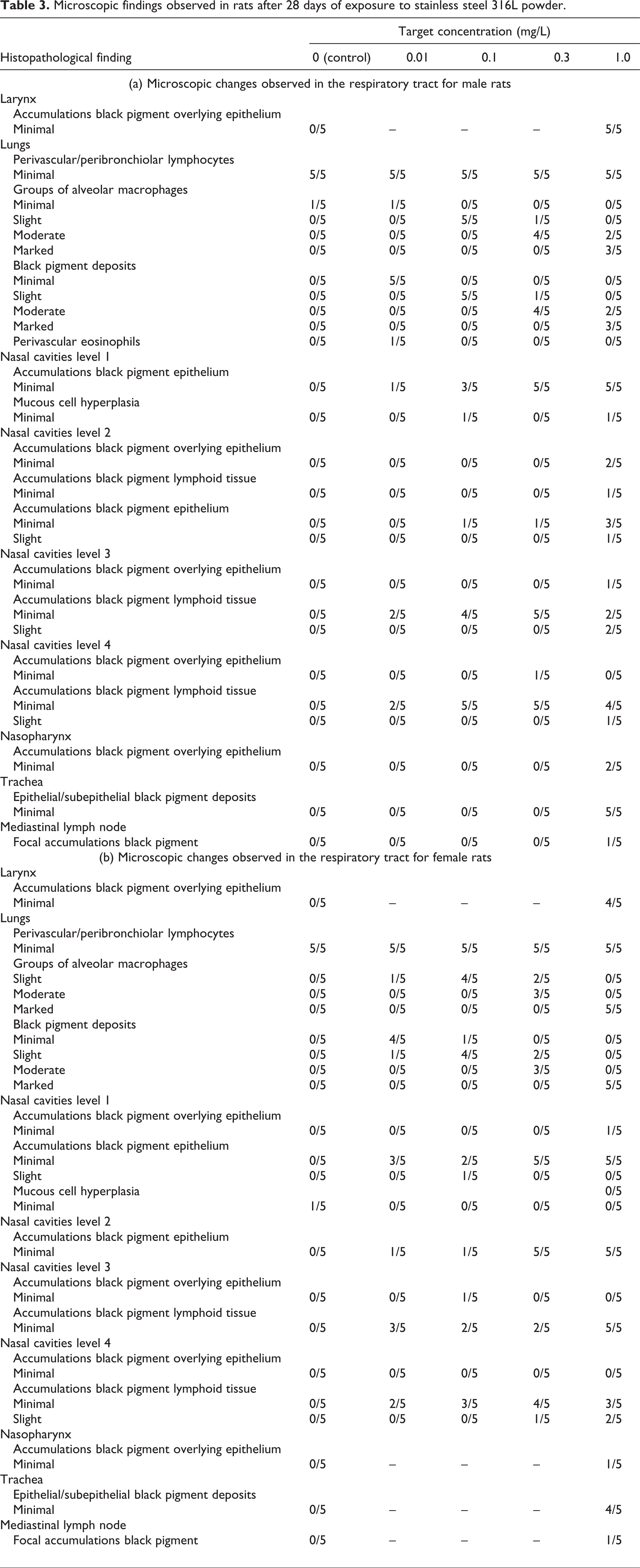
Significantly elevated relative lung weights were observed for male and female rats treated with 0.3 and 1.0 mg/L SS powder when compared with the controls. This is the result of slight increases in absolute lung weights together with slight decreases in overall body weights (Table 2). These changes were not found to be related to lung inflammation. Grey discolouration of the lobes of the lungs was observed for all animals treated with 1.0, 0.3 or 0.1 mg/L. Two males and two females treated with 0.01 mg/L also showed grey discolouration of the lungs. No further macroscopic abnormalities were detected at the end of the study period. Accumulation of black pigment in the lung lobes was observed for all treated animals, being more severe at the highest exposure level. Alveolar macrophages, phagocytizing pigment, were found in high amounts in the lungs, but no signs of inflammatory responses, tissue degeneration or necrosis could be observed (Figure 6). Small amounts of black pigment were found in the nasal cavities at all exposure levels. No signs of inflammation, tissue degeneration or necrosis could be observed. Accumulations of black pigment were also seen in the trachea, larynx, pharynx and mediastinal lymph nodes of animals exposed to the highest concentration of SS powder. However, there was in no instance any associated inflammatory response. A summary of respiratory tract findings is presented in Table 3. No treatment-related changes in other organs were observed (data not shown). Based on the lack of adverse effects of treatment in this study, the ‘no observed adverse effect concentration’ was therefore considered to be 1.0 mg/L.

Histopathological lung samples from the 28-day exposure study in rats. (a) Normal lung parenchyma of an unexposed male rat (control group); (b) lung tissue of a male rat in the highest concentration group (1.0 mg stainless steel/L): accumulation of stainless steel particles in macrophages and extracellular accumulations. ×400 magnifications.
Microscopic findings observed in rats after 28 days of exposure to stainless steel 316L powder.
Discussion
The total release of metals was significantly lower for SS 316L compared with what might be expected from the release of the individual pure metal constituents. The low Ni release from the tested SS powder is particularly relevant since Ni is the foremost health concern in the case of Ni-containing SSs. The Cr(III)-rich oxide present on the surface of Cr metal and SS powders is the reason for their high passivity and hence low release of metals. 1,4,5,8 –10,24 The very low release of Ni from SS can be explained by its absence within the outermost surface oxide of the SS powder. 8,10 The release of Fe from SS was significantly lower compared with pure Fe metal powder (Table 1). There were no indications that Fe would induce toxicity after inhalation, and as the release was much lower (70-fold) than from the fine Fe metal powder, Fe release is believed not to be of importance for the potential toxicity of SS. The Cr(III)-rich oxide is not able to inhibit the relatively high release of Mn induced by the strong surface enrichment of oxidized Mn in an outermost oxide of the inert gas-atomized 316L SS powder. 32 The release of Mn (microgram per microgram) in ALF was lower from the 316L powder than from the MnO2 powder (Table 1), but relatively higher if considering the total metal release and the nominal bulk composition of 316L. 10,47 This is most likely specific for the powder of this investigation (inert gas atomization) and not relevant for SS produced and cooled under normal conditions. The impact of specific surface properties of differently sized and produced 316L powders on the extent of metal release was recently discussed in detail. 32,46 Thus, the release of Mn is unlikely to be relevant, for example, for the occupational exposure to SS dusts, as significantly lower (non-detectable) amounts of Mn release were observed for massive 316L. 10,31,39,40,46 ALF is considered as a worst case, as it causes the highest complexation-induced metal release and surface changes 10,46 (Figures 2 to 4). Lower metal releases were determined in non-complexing solutions (e.g. sodium chloride). 10 Exposures to media of lower pH have resulted in a higher release of metals. 7 Recent findings have also shown a local presence of a stable Cr(VI) surface phase after exposure in ALF and citric acid. 10 No Cr(VI) was, however, found in solution, as it would be immediately complexed by citrate and reduced to trivalent Cr, as has been experimentally confirmed. 10 This surface reaction is unique for ALF, containing citric acid, 10 and was not observed in different protein solutions. 47 The presence of the Cr(VI) phase is believed to be a consequence of the known ability of MnO2 to oxidize Cr(III) to Cr(VI). 10,48
The Cr release from 316L was higher when compared with pure Cr metal (Table 1). This would be expected from a metallurgical point of view, because of the corrosion resistance and hence the inhibition of metal release decreases strongly with increasing Cr bulk content (above a certain threshold value of approximately 11 wt% Cr). 1 It should be noted that the extent of Cr released from the 316L powder is still very low and only 5.6-fold higher compared with the Cr release from pure Fe metal powder (Table 1), which contains approximately 0.1 wt% of Cr as an impurity. As Cr was the only alloying metal for which the release from SS 316L was similar to or higher than from the pure corresponding metal (Table 1), and since it was enriched in the surface oxide over time (while Fe and Mn were depleted and Ni was completely absent), it can be assumed that the potential hazardous or non-hazardous effects of SS 316L are most likely driven by Cr.
With the exception of welding, human exposure to SS via inhalation is mainly likely in occupational settings during handling of powders and grinding and polishing of massive SS. According to the GHS classification and labelling system, 20 SS 316L should be labelled for repeated dose toxicity following inhalation exposure because of its Ni content (10.7 wt%). Our results indicate that the potency of inert gas-atomized 316L SS powder to cause effects by subacute inhalation exposure is significantly lower and could be directly assumed from the relatively high content of Ni metal (10.7wt%) in the SS 316L grade used for the study. This is in agreement with the low amount of metals released from SS into different simulating body fluids, in comparison with Ni powder, as discussed above.
Ni metal powder particles (MMAD (with GSD in parenthesis) was 1.8 µm (2.40), 1.7 µm (2.16) and 1.8 µm (2.09) for 0.1, 0.4 and 1.0 mg/m3 groups, respectively) showed significant toxic effects in Wistar rat lungs after 2-year inhalation exposure at the concentrations of 0.1 mg Ni/m3 (0.0001 mg/L) and higher. 29 In addition, a 28-day range-finding study with Ni metal showed significant lung toxicity in Wistar rats at the concentrations of 4 mg/m3 (0.004 mg/L 30 ; as reviewed in the study by Santonen et al. 30 ). The latter study included the exposure of groups of five male and five female Wistar rats at 4, 8 and 24 mg/m3 (0.004, 0.008 and 0.024 mg/L) of Ni metal powder (90% of particles sized <1.84 µm; MMAD (with GSD in parenthesis) 1.5 µm (2.45), 1.8 µm (2.24) and 1.6 (2.26) for the 4, 8 and 24 mg/m3 groups, respectively). The exposure pattern was the same as in the 28-day SS study presented in this article: 6 h/day, 5 days/week, followed by 2 days of rest, for a period of 4 weeks, except that in the Ni metal study, the exposures were whole body and in the SS study they were nose only. Exposure-dependent increases in the absolute and relative lung weights and macroscopic effects in the lungs and lymph nodes were seen at all concentration levels. Microscopic examinations revealed granulomatous inflammation, proteinaceous and/or mucoid exudate and black pigment in the lungs and granulomatous inflammation, hyperplasia and black pigment in the lymph nodes at all Ni concentrations. Based on these results, the ‘lowest observed adverse effect level’ was 4 mg Ni/m3 for 28-day inhalation exposure. The concentrations used in the SS study presented in this article were markedly higher than those used in the Ni studies (maximum SS concentration 1 mg/L; maximum Ni concentration 0.024 mg/L 30 ; as reviewed in the study by Santonen et al. 31 ). No adverse effects were seen, even at the highest concentration of SS, whereas the lowest Ni concentration (0.004 mg/L) already resulted in clear signs of toxicity in the 28-day Ni range-finding study 30 , as reviewed in the study by Santonen et al. 31 The deposition, estimated by a dosimetric MPPD model (www.ara.com/products/mppd.htm), was about 260-fold higher in the 1 g/L-exposure group in the present 316L study compared with the 0.004 g/L-exposure in the Ni metal study 30 , as reviewed in the study by Santonen et al. 31 Thus, the toxicity observed after exposure to Ni is definitely not related to a relatively higher deposition.
The amount of Ni released from the 316L powder particles was more than 1000 times lesser when compared with the release from Ni powder when tested at identical conditions (see Figure 3). Based on the results from the 28-day study with Ni metal, 29 as reviewed in the study by Santonen et al., 30 and the Ni content of SS 316L, a ‘lowest observed adverse effect concentration’ of 40 mg SS 316 L/m3 (0.04 mg/L) would have been predicted by direct extrapolation. Yet the exposure to up to 1.0 g/m3 (1.0 mg/L) SS 316L had no adverse effect. The observed effects are thus different from the predicted effects based on the composition by over 25-fold.
No inhalation studies have been performed with Cr metal, but the results obtained with SS can, at a general level, be compared to those observed in a 90-day rat inhalation study with Cr(III) oxide, as Cr(III) oxide is present at the surface of SS. Derelanko et al. 49 showed that 90 days of exposure to Cr(III) oxide at concentrations of 4.4–44 mg/m3 (0.0044–0.044 mg/L) resulted only in mild inflammation in the lungs. Higher concentrations were not tested. The authors concluded that the effect should be considered as a non-specific particle effect rather than as inherent toxicity caused by the Cr(III)-ion. 49 Previous studies have shown lower Cr release from Cr2O3 powder than from Cr metal or SS 316L powders, when normalized to the surface area or expressed as microgram per microgram. 8,9,36 No 28-day study data are available on Cr2O3, and due to the shorter exposure period, it is not possible to make any direct comparison between doses and effects of the 90-day study with Cr2O3 49 and the present SS study.
It should be noted that a 28-day inhalation study gives data on subacute effects but does not necessarily fulfil all regulatory criteria for studies on repeated dose toxicity. This study was designed to match the Ni metal study, 30 as reviewed in the study by Santonen et al. 31 (mainly for comparative purposes). For this reason, the results from histopathological examinations were compared in both the studies. Bronchoalveolar lavage fluid (BALF) analyses might be more sensitive for the detection of lung inflammation, but results from BALF analyses were not available from the Ni metal study. The 28-day study protocol used in the present study did not look at the toxicokinetics. Other published data on the repeated dose toxicity of SS (with the exception of welding fume particles) are limited. Huvinen et al. 50,51 investigated long-term health effects among workers engaged in the manufacture of SS (average duration of exposure was 18 years in the first study, and the results were followed up 5 years later). The lung function in the workers of the SS melting shop did not differ from those of the cold-rolling mill. The study focused on health effects caused by Cr and the workers were grouped according to Cr exposure. In addition, the roles of exposures of other elements than Cr remain unclear, and the cold-rolling mill workers cannot be considered as true unexposed controls. The negative findings, however, did not reveal any adverse effects possibly caused by SS.
Hedberg et al. 8 studied the ability of a similar fine-sized SS powder as used in this study to induce cell death and red blood cell haemolysis in vitro and found SS powder particles non-toxic in these assays. In an in vitro comet assay, a statistically significant increase in comet tails was seen, 8 but the relevance of these results concerning potential genotoxicity is limited as the tests were not performed according to standard methods. No conclusions could be drawn regarding a dose–response relationship, as only one concentration was used. Other in vitro genotoxicity tests (Ames test, chromosomal aberration test and morphological transformation studies) were carried out mainly with SS powder showing negative results (reviewed in 31 ). Animal studies have focused on the ability of SS implants to induce local tumours. The weight of evidence from these studies supports a lack of carcinogenicity, and IARC has evaluated that SS implants are not classifiable as to their carcinogenicity to humans. 52 Ni metal has been classified as possibly carcinogenic to humans (category 2B) based on limited animal studies. 53 However, a recent inhalation carcinogenicity study with Ni metal did not show an increased incidence of pulmonary tumours in rats. 29 Ni is a well-known skin sensitizer. The possibility that SS could elicit reactions in previously Ni-sensitive persons has been investigated by patch testing in a number of studies. The results clearly show that allergic reactions are extremely rare and SS is regarded as safe, even in persons with Ni allergy. 12 ,15,17,54–56 This is consistent with the Nickel Directive, 56 since most SS powders release less than 0.5 μg Ni/cm2/week when tested in synthetic sweat, which is the limit value for sensitization classification of Ni-containing alloys according to the EU CLP regulation. 23
The results of our study show that the surface and metallurgical properties of an alloy play an extremely important role in its potential to elicit toxic effects. Relative bioaccessibility studies of alloys and their constituent elements, such as those reported here for SS grade 316L and Ni, Cr and Fe, allow the determination of the effective concentration of the elements in the alloy. Application of the GHS mixture rule 21 to the effective concentrations rather than to the bulk composition would have resulted in a more accurate prediction of pulmonary toxicity for SS 316L. This approach may also be applicable to other alloys and routes of exposure.
Conclusions
The release of alloy constituent metals from inert gas-atomized SS powder into artificial body fluids is very low, which is explained by the Cr(III)-rich passive surface oxide of SS. The release of Ni from the SS 316L powder was >1000-fold lower than from Ni metal powder.
An in vivo 28-day repeated dose inhalation test carried out with the same SS 316L powder showed no signs of adverse effects at exposure levels up to 1 mg/L, although repeated dose toxicity might be assumed based on the relatively high Ni content of the material. In the case of SS 316L, the potential toxicity or non-toxicity is likely to be driven by its Cr content and hence the surface composition, although the international rules for classification and labelling would require classification as hazardous due to the Ni in the bulk alloy composition.
These findings support the hypothesis that in the case of alloys, the surface composition and the bioaccessible fraction of the released alloy constituents, rather than the nominal composition, is crucial for the eventual toxicity or non-toxicity of the alloy.
Footnotes
Acknowledgements
Drs Tony Newson and Grant Darrie are thanked for their expert advice. Neda Mazinanian, Dr Gunilla Herting and Maria-Elisa Karlsson at KTH are all highly acknowledged for their experimental help.
Authors’ Note
This study is dedicated to the memory of Dr Pat Koundakjian.
Funding
This work was supported by the International Chromium Development Association and Cusanuswerk, Germany.
