Abstract
Environmental compounds may be important contributors to Parkinson’s disease etiology. Epidemiological and experimental evidence for the facilitation of parkinsonism by manganese is equivocal. This work addressed methodological concerns in the few studies of manganese modulation of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced toxicity in C57BL/6 mice. Male, retired breeder mice received 0 or 100 mg/kg of manganese chloride (MnCl2; subcutaneously on days 1, 4 and 7) and 0 or 20 mg/kg of MPTP (intraperitoneally on day 8) and survived up to day 15 or 22. On the day of sacrificing, horizontal (grid crossing) and vertical (rearing) open field movement, swimming, grip strength and grip fatigue were examined. Striata were analyzed for dopamine and 3,4-dihydroxyphenylacetic acid (DOPAC) using high-performance liquid chromatography. MPTP produced a main effect decrease in striatal dopamine (48.8%) and DOPAC (38.1%), but there was no main effect of MnCl2 or MnCl2 × MPTP interaction. However, modulatory interactions were observed between the effects of MnCl2 and MPTP for grid crossing, rearing and grip strength. Interestingly, these interactions reduced the severity of behavioral deficits attributable to either of these compounds alone. For rearing and grip strength, the MnCl2 × MPTP interaction was dependent upon survival time. The mechanistic nature of the MnCl2 × MPTP interaction upon these behaviors, in the absence of such an interaction for striatal dopamine and DOPAC, remains to be clarified.
Keywords
Introduction
The etiology of Parkinson’s disease (PD) remains elusive. Together, the epidemiological, genetic and laboratory evidence suggest that PD may have a multifactorial etiology. Repeated exposure to one or more exogenous compounds could interact with each other or with subthreshold genetic mutations to trigger or exacerbate the nigrostriatal degeneration characteristic of the disorder. 1,2 It is therefore important to continue to explore the potential of exogenous compounds to disrupt dopaminergic nigrostriatal function and integrity in normal as well as compromised nigrostriatal systems.
Manganese seems a logical candidate for the investigation of potential triggering or facilitation of PD because overexposure to the metal can produce a neurological disorder called manganism that can be difficult to distinguish from PD on the basis of clinical signs. 3 The similarity is not surprising because both PD and manganism have a focus of neuropathology within the basal ganglia, although PD targets the dopaminergic nigrostriatal pathway and manganese overexposure is thought to principally target the globus pallidus. 4 However, these two structures are disynaptically connected in a manner that suggests that manganese-induced toxicity within the globus pallidus could result in downstream glutamatergic excitotoxic influences upon dopaminergic nigrostriatal neurons. 5 There is experimental evidence that manganese intoxication is associated with changes in elements of the glutamate–glutamine metabolic cycle and with changes in glutamate receptor sensitivity. 6 –8 Effects such as these may not be sufficient to induce nigrostriatal damage in otherwise normal individuals. However, they could decrease the threshold for, or increase the severity of, such damage in individuals predisposed to PD because there is evidence for overactivity of glutamatergic structures within the aforementioned disynaptic pathway in patients with PD. 9 –11
Epidemiological evidence for the relationship between manganese exposure and PD is equivocal, arguing for related experimental studies. 12 –17 However, the body of experimental work examining the influence of manganese exposure upon experimental parkinsonism is fairly small and runs a gamut from potentiation 18,19 to protection. 20,21
The aged, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated C57BL/6 mouse is one of the most common and sensitive animal models for PD. 22,23 Baek et al. failed to observe an interaction between manganese chloride (MnCl2) and MPTP exposure in the basal ganglia using immunohistochemical and high-performance liquid chromatography (HPLC) measures of dopaminergic integrity. 24,25 Although they reported a significant interaction between MnCl2 and MPTP dosing upon glial fibrillary acidic protein immunoreactivity in the globus pallidus, they expressed doubt over this finding, given its small magnitude, and they failed to replicate the finding using an even higher dose of MnCl2. Baek and colleagues noted several limitations in their studies on manganese influence upon MPTP neurotoxicity, including small numbers of mice, possible masking of a manganese/MPTP interaction by a very large reduction in dopaminergic indicators produced by MPTP alone, and a lack of behavioral assessment that could be a more subtle indicator of the integrity of basal ganglia circuitry. Furthermore, although PD is usually seen in late middle age, the C57BL/6 mice used in these studies were only 8 weeks old. The median lifespan of this strain is in the vicinity of 27 months. 26
Given that exogenous compounds can modulate dopaminergic nigrostriatal function, variable findings for manganese facilitation of idiopathic and experimental PD, remaining questions from MPTP-treated C57BL/6 mice studies regarding manganese effects in the basal ganglia and its interaction with MPTP and the methodological concerns expressed by Baek and colleagues regarding their work, this study further explored the ability of manganese to modulate MPTP-induced toxicity in sensitive C57BL/6 mice, while addressing the aforementioned methodological concerns.
Methods
Animals
A total of 124 retired breeder, male, C57BL/6 mice were obtained from Jackson Laboratories (Bar Harbor, Maine, USA) and were 7–9 months old at the time of the experiment. They were cared for in an Association for the Assessment and Accreditation of Laboratory Animal Care, International-accredited facility in compliance with the National Research Council’s Guide for the Care and Use of Laboratory Animals, 8th edition. 27 All procedures were approved by the Virginia Tech Institutional Animal Care and Use Committee. The mice had free access to food and water, and lights were on from 6:00 to 18:00 h daily. Body weight was recorded on each experimental day.
Experimental design and dosing
Mice were randomly and unequally assigned to eight treatment groups in a 2 (MnCl2 dose level) × 2 (MPTP dose level) × 2 (survival time) factorial design. In brief, a random number was assigned to each mouse identification number using Microsoft Excel (Microsoft Corp., Redmond, Washington, USA). Upon sorting the column of random numbers from lowest to highest, the adjacent, linked, mouse identification numbers were randomly shuffled. Consecutive treatment group numbers from 1 to 8 were then assigned down the list in repeating cycles. The four mice from the incomplete last cycle were added to each of the four longer survival time treatment groups, respectively. MnCl2 dose levels were 0 or 100 mg/kg body weight in a water vehicle and were delivered subcutaneously in the scruff of the neck. MPTP dose levels were 0 or 20 mg/kg body weight in a 0.9% (w/v) saline vehicle and were delivered intraperitoneally (i.p.). Mice received MnCl2 or water vehicle on days 1, 4 and 7, MPTP or saline vehicle on day 8 and were sacrificed after 7 or 14 days (days 15 or 22, respectively; Figure 1). Our previously published MnCl2-dosing regimen 28 has been employed in published studies by others examining manganese neurotoxicity in mouse brain. 29 Behavioral assessment, as described below, was performed on the day of sacrificing between 9:00 and 15:00 h. Fresh 10% (w/v) MnCl2 tetrahydrate (Sigma-Aldrich, St Louis, Missouri, USA) solution was prepared on each day of injection. The 3% (w/v) MPTP hydrochloride (Sigma–Aldrich) solution was kept for no more than 25 h after preparation, at 4°C. Injection volumes of MnCl2 and MPTP were calculated from individual daily body weights with respective means of 150 µl and 20 µl. Injections of these compounds were made with a 27 gauge × 1/2 inch needle attached to a 1-ml (with 10 µl gradations; Becton-Dickinson, Franklin Lakes, New Jersey, USA) or a 100-µl (1 µl gradations; Hamilton, Reno, Nevada, USA) syringe. Due to limited availability from the supplier, there were 15 mice in each of the four 7-day postinjection survival groups, and there were 16 mice in each of the four 14-day postinjection survival groups. Temporal constraints on the number of mice that could undergo behavioral testing and brain tissue processing on a single day necessitated mice being partitioned into eight processing blocks. The matched representation of all the treatment groups within each processing block reduced systematic residual variation between treatment groups.
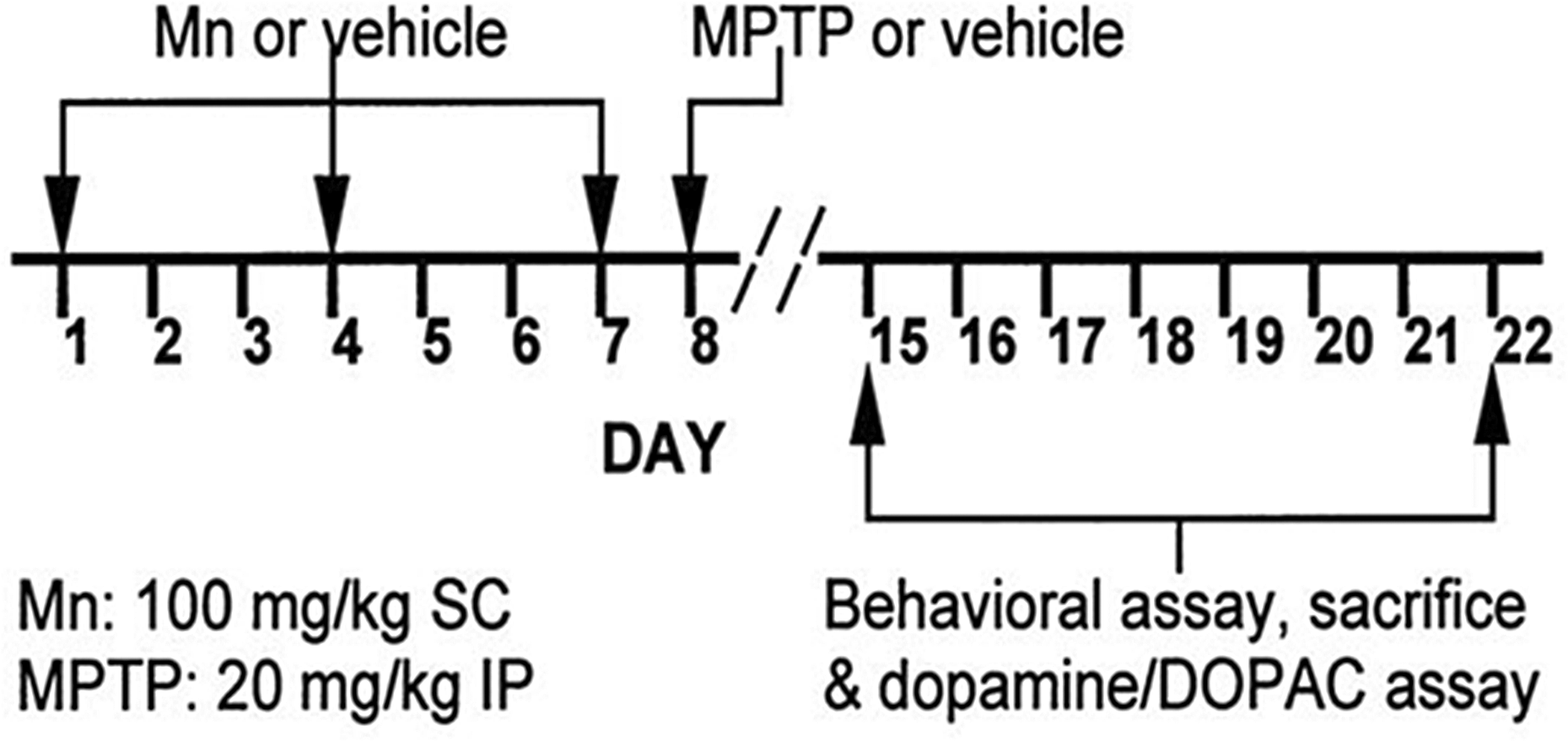
Timeline for the 2 (MnCl2 dose level) × 2 (MPTP dose level) × 2 (survival time) factorial design. The vehicles for MnCl2 and MPTP were water and saline, respectively. MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MnCl2: manganese chloride.
Behavioral assessment
Motor behavior assessment was performed according to previously published methods from this laboratory. 28 Five behaviors, requiring no special training or habituation, were examined once for each mouse. These behaviors were horizontal and vertical open field movements, swimming to a visible platform, grip strength and grip fatigue.
Open field movement was assessed in the horizontal and vertical planes using a single, empty, standard glass tank (46.5 cm long × 26 cm wide × 30 cm high). The floor was marked out into a grid of six equal 15.5 × 13 cm2 rectangles. The walls were covered with white opaque paper, on the outer side, to limit visual distractions. The tank was mounted on top of a wooden stand that allowed an unobstructed view of the tank floor by use of a mirror mounted at a 45° angle, 24 cm under the tank. The experimenter, not blinded to experimental condition, sat on the floor approximately 1 m from the apparatus and observed the movement for a period of 3 min after a mouse was placed in the apparatus. Movement in the horizontal plane was determined by counting the number of grid crossings. A grid crossing was defined as a complete movement of the forelimbs and hind limbs from one grid block to another. Movement in the vertical plane was determined by counting the number of times the mouse reared up on its hind limbs, lifting both forelimbs off the floor, followed by the exploratory mannerisms of head rotation and sniffing. A double counter was manually used to record the respective occurrence of the crossing and rearing behaviors. The inner side of the apparatus was cleaned with disinfectant spray and wiped dry before each mouse was tested.
Swimming was used to assess coordinated movement with a different muscle load and energy requirement compared with the open field tests. The procedure measured the time necessary for swimming from one end of a narrow, water-filled trough to a visible, raised platform at the other end. The single plastic trough (Dynamic Design, New York, New York, USA) was a 91.5-cm long × 18.5-cm wide × 17-cm high rectangular basin of opaque plastic that was filled with 27°C tap water to within 4.5 cm of the top edge. The water was changed at the beginning of each testing day. The platform was a 7.5-cm long × 3.3-cm wide × 1.5-cm thick piece of wood with a wire mesh grid attached to the top that extended an additional centimeter on all sides. The platform was attached to a weighted base by a wooden dowel, anchoring the platform to the floor of the trough, such that the top of the platform sat 14 cm above the floor and 1.5 cm above the water. The wire mesh edges of the platform were 4.5 cm away from the end-wall and sides of the trough. Swim duration was manually recorded on four consecutive trials by the nonblinded experimenter. In each trial, the mouse was held by the base of the tail and gently lowered into the water next to the end-wall that was opposite to the platform. Timing of swim duration began when the mouse was released, and the timing ended when the mouse gripped the raised platform with at least one forepaw. The maximum time limit for a trial was 4 min. At the end of a trial, the mouse was placed on a dry towel for about 30 s before the next trial was initiated.
The single grip strength apparatus consisted of a 12 × 12-cm2 wire mesh grid (0.5 × 0.5 cm2) attached to the end of, and oriented perpendicular to the long axis of, the armature of a tensile load cell (strain gauge; Instron Corporation, Canton, Massachusetts, USA). The load cell output was fed to a Measurements Group Conditioner/Amplifier (Model 2310) and was calibrated such that a 100-g load, suspended from the grid along the gripping axis, produced a signal of 0.5 V. The grip strength-dependent variable was therefore measured as weight pulled in grams. Every 50 ms, analog voltage signals were digitized by a National Instruments (Model PCI-MIO-16E-4, Austin, Texas) computer interface card. Labview (National Instruments) software automatically stored the voltage measurements that were later displayed as weight pulled as a function of time. For each trial, after the mouse gripped the grid, the nonblinded experimenter gently and steadily pulled the mouse away from the load cell by the base of the tail. The trial was terminated when the mouse released the wire mesh grid. The weight pulled immediately prior to the release of the grid, which was unambiguously recognizable on the plot as the sharp peak of a spike followed by an immediate drop to a continuous, flat baseline, was used as the value for a given trial. A mouse received four trials (typical trial duration 5–8 s), each separated by 20 s. Trial 1 was used as the grip strength measurement for a given mouse. The slope of the best fit line through an animal’s four trials was used to evaluate the decline in grip strength over time, referred to here as grip fatigue. Grip strength values observed in this study (see ‘Results’ section) were within the range reported for adult mice recorded with a commercial variation of the apparatus used here. 30 –32
Brain tissue processing and neurochemical measurement
Mice were sacrificed on day 15 (7-day postinjection survival) or 22 (14-day postinjection survival) upon completion of motor assessment. Processes of sacrificing, brain removal and striatal dissection were all performed according to the previously published methods from this laboratory. 28 Briefly, following rapid cervical dislocation, brains were immediately removed and placed atop an ice-chilled glass Petri dish for dissection. Following a midsagittal cut and entry through the septum pellucidum, the striatum was pinched from the wall of the lateral ventricle of both hemispheres. Combined striatal weight was determined after placement in a preweighed 1.5-ml microcentrifuge tube. A 6.8 × 10−6 M solution of 3,4-dihydroxybenzylamine hydrobromide (DHBA; Sigma-Aldrich) internal standard was added to the capsule at 10 μl/mg of tissue. The sample was then homogenized and frozen at −70°C for HPLC with electrochemical detection analysis at a later date.
On the day of HPLC analysis, samples were thawed over ice and centrifuged at 9300g for 5 min. The supernatant was removed and filtered through a Pall Gelman Acrodisc (Pall, East Hills, New York, USA) LC 13 mm, 0.2 μm filter and transferred to an HPLC vial. Dopamine and its metabolite 3,4-dihydroxyphenylacetic acid (DOPAC) were detected according to a modification in the method described by Jussofie et al. 33 using an Agilent Technologies (Santa Clara, California, USA) 1100 Series HPLC system connected to a Hewlett Packard 1049 electrochemical detector set at a potential of +0.35 V for the detection of catecholamines. A Nucleosil 100 C18 188 (reverse phase), 250 × 4.0 mm2, 3 μm column (Macherey-Nagel, Bethlehem, Pennsylvania, USA), was used for separation with a buffer flow rate of 0.6 ml/min. The mobile phase consisted of 0.1 M sodium acetate, 25 mM citric acid, 134 μM ethylene diaminetetraacetic acid, 230 μM octane sulfonic acid and 6% methanol (v/v) at pH 6.1. Calibration standards for DHBA, dopamine and DOPAC were run before and after every 15–20 samples. The injection volume for both standards and samples was 20 μl. Sample concentration of dopamine and DOPAC was expressed as picomole per milligram wet weight of striatal tissue. Percentage recovery of each sample was also determined.
Statistical analysis
An independent analysis of variance (ANOVA) was performed for each dependent variable using the MIXED procedure of SAS (SAS Institute, Cary, North Carolina, USA). Prior to ANOVA, plots of standardized residual errors were examined for every factor in the model to determine model adequacy. 34 Error plots suggested independence, equal variance and lack of skewness. Therefore, no transformations of the raw data appeared necessary. Although swimming trials had a maximum time limit of 4 min, procedures for censored data, which would have been very complex for our experimental design, were not used after considering that this limit applied to only a small percentage of mice (8.6%) that were fairly equally distributed across different experimental groups. Except for body weight, all analyses examined the fixed effects of MnCl2 dose, MPTP dose and postinjection survival time. Interactions examined in these three-way ANOVAs were MnCl2 dose × MPTP dose, MnCl2 dose × survival time, MPTP dose × survival time and MnCl2 dose × MPTP dose × survival time. The alpha level was 0.05 for each main effect and interaction examined using ANOVA. For each dependent variable, the results of the ANOVA, with respect to interactions, were used as a guide to determine the most appropriate a priori combination of post hoc, pairwise comparisons. This was carried out to ensure maximal statistical power for the post hoc analysis of means. The Bonferroni alpha was used for each pairwise comparison and was adjusted according to the number of comparisons (0.05 per number of comparisons). For two comparisons, the Bonferroni alpha was 0.0253, for four comparisons 0.0127 and for eight comparisons 0.0064.
Three analyses of body weight were performed: one during the MnCl2-dosing period (days 1–8, prior to MPTP), one during the 7-day post-MPTP-injection period (days 9–15) and one during the 14-day post-MPTP-injection period (days 9–22). For the first analysis, which was a two-way ANOVA, fixed effects examined were MnCl2 dose and experimental day, and the interaction examined was MnCl2 dose × day. For the latter two analyses, which were three-way ANOVAs, fixed effects examined were MnCl2 dose, MPTP dose and experimental day. Interactions examined were MnCl2 dose × MPTP dose, MnCl2 dose × day, MPTP dose × day and MnCl2 dose × MPTP dose × day. In the analyses of body weight, experimental day was treated as an independent variable along with MnCl2 dose and MPTP dose, and different days were treated as independent of each other. Because no significant three-way interactions were observed, post hoc analysis of multiway ANOVAs for weight was performed using the slice function of the MIXED procedure of SAS. This procedure performs a test of simple main effects of one independent variable at each level of another independent variable.
Given the statistical models used for the analysis of our experimental design, in conjunction with the number of dependent variables examined, it should be kept in mind that 58 p values were examined across the 10 ANOVAs that were performed when considering the results to be presented. Statistically controlling potential false positives of multiple testing, across ANOVAs, would likely have introduced a substantial type II error for the experiment, limiting the likelihood of detecting any real differences. 35 Therefore, as described above, we chose to focus our efforts on statistically controlling the type I errors of multiple group comparisons within each independent ANOVA.
Graphical representations of our data (Figures 2 to 6) present least squares means because those values are the most concordant with the models that were used to account for sources of variability in the statistical analyses. Standard errors are most appropriate to use in conjunction with the presentation of least squares means. However, we have also supplied the raw arithmetic means and SDs in the figure captions to provide a better idea of the data dispersion.

Main effect decreases in striatal dopamine (p < 0.001, solid line) and DOPAC (p < 0.001, dashed line) concentration induced by MPTP treatment. Data are collapsed across MnCl2 dose and survival time because there were no main effects of those variables, nor was there any interaction among the independent variables. p Values for statistically significant comparisons are also indicated in the figure key inset. In accord with the statistical model used, graphed values are least squares means ± SE. For a better idea of uncorrected data dispersion, raw arithmetic means ± SD follow. Dopamine concentration: no MPTP 86.1 ± 22.1, MPTP 44.8 ± 30.9. DOPAC concentration: no MPTP 5.9 ± 1.4, MPTP 3.7 ± 1.9. MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; DOPAC: 3,4-dihydroxyphenylacetic acid; MnCl2: manganese chloride; SE: standard error.
Results
Dopamine and DOPAC
ANOVA revealed a significant 48.9% main effect decrease in striatal dopamine concentration (df = 1, 90.8; p < 0.0001; Figure 2) and a 38.1% main effect decrease in striatal DOPAC concentration (df = 1, 89.1; p < 0.0001) in mice treated with MPTP, compared with MPTP vehicle. These data are collapsed across MnCl2 dose and postinjection survival time because there was no main effect of either of the latter two variables, nor could interactions among the three independent variables be statistically validated.
Horizontal open field movement
No significant main effect of MPTP dose or postinjection survival time on open field horizontal movement (grid crossings) was revealed by ANOVA. However, a MnCl2-induced 14.2% decrease (collapsed across MPTP dose and survival time) in this measure represented a strong numerical trend toward a main effect of MnCl2 dose (df = 1, 90.8; p = 0.0547). Upon further exploration of a significant interaction between MnCl2 dose and survival time (df = 1, 90.5; p = 0.0167) with respect to their effects upon open field horizontal movement (grid crossing), pairwise comparison revealed no significant MnCl2-induced change in horizontal movement on day 15, but there was a significant 29.7% decrease on day 22 (p = 0.0035; Bonferroni α = 0.025; Figure 3(a)). Post hoc analysis of a significant interaction between MPTP dose and postinjection survival time (df = 1, 90.2; p = 0.0192) revealed that MPTP produced a numeric decrease in grid crossing on day 15 and a numerical increase on day 22. However, neither of these changes by themselves was statistically significant. ANOVA also revealed a strong numeric trend toward a MnCl2 dose × MPTP dose interaction (df = 1, 90.8; p = 0.0546, α = 0.05) with respect to horizontal movement. Further exploration of this relationship, using pairwise comparisons, revealed a significant 26.6% reduction in movement induced by MnCl2 in the absence of MPTP (p = 0.0101, Bonferroni α = 0.0127), but there was no significant MnCl2-induced change in the presence of MPTP (Figure 3(b)). It therefore appeared that the MnCl2-induced reductions in horizontal movement were dependent upon both postinjection survival time and the presence or absence of MPTP.
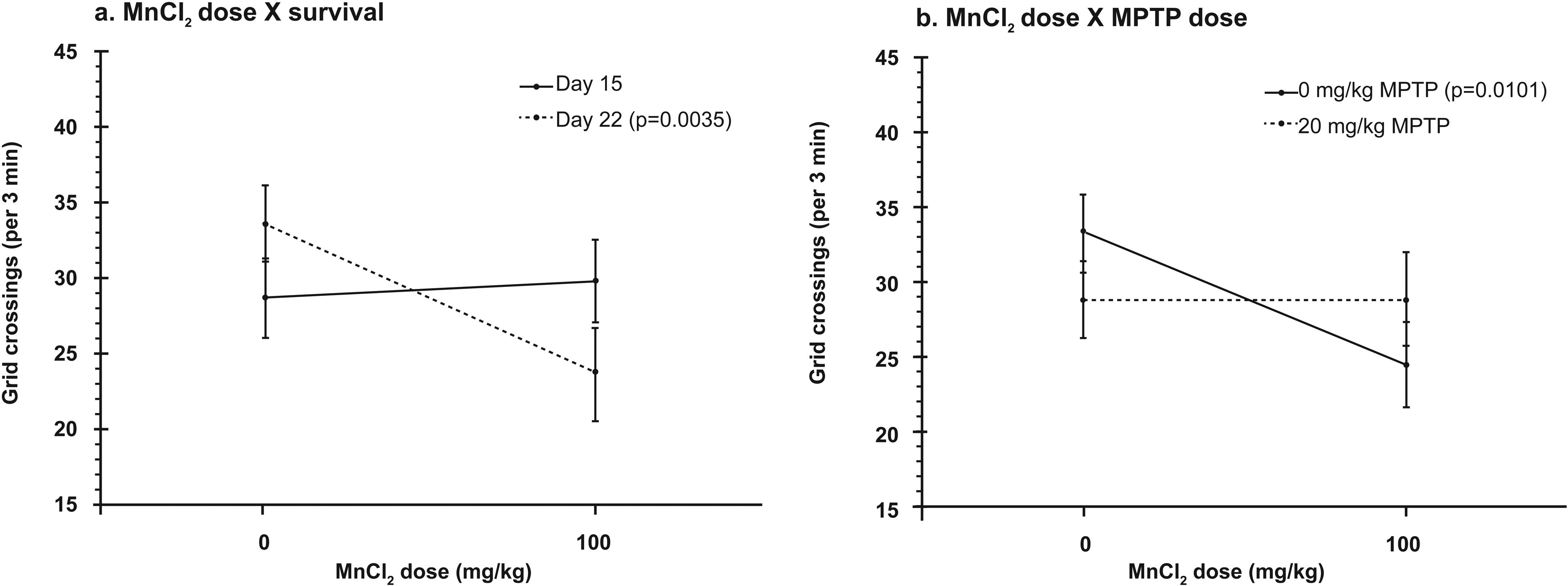
(a) Significant interaction of MnCl2 dose with survival time, for horizontal movement in the open field. MnCl2 produced a significant decrease in grid crossings 14 days after dosing (p = 0.0035, dashed line), but not 7 days after dosing. Data are collapsed across MPTP dose. p Values for any statistically significant comparisons are also indicated in figure key insets. In accord with the statistical model used, values in all graphs are least squares means ± SE. For a better idea of uncorrected data dispersion, raw arithmetic means ± SD follow. Survival to day 15: no MnCl2 28.7 ± 13.1, MnCl2 29.1 ± 9.2. Survival to day 22: no MnCl2 33.6 ± 14.2, MnCl2 25.0 ± 11.6. (b) Significant interaction of MnCl2 dose with MPTP dose, for horizontal open field movement. MnCl2 produced a significant decrease in grid crossing in the absence of MPTP (p = 0.0101, solid line), but not in its presence. Data are collapsed across survival time. Raw arithmetic means ± SD follow. No MPTP: no MnCl2 33.3 ± 12.0, MnCl2 26.0 ± 7.6. MPTP: no MnCl2 29.2 ± 15.4, MnCl2 28.2 ± 12.2. MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MnCl2: manganese chloride; SE: standard error.
Vertical open field movement
No significant main effects of MnCl2 dose, MPTP dose or postinjection survival time upon vertical open field movement (rearing) were revealed by ANOVA, nor were any statistically significant two-way interactions were observed among these independent variables. However, a significant three-way interaction was observed between these three independent variables (df = 1, 90.7; p = 0.0412) with respect to their effects upon rearing. Post hoc exploration of this interaction revealed that on day 15, a significant 39.8% decrease in rearing was observed for mice that had received MPTP in the absence of prior MnCl2 exposure (p = 0.0053, Bonferroni α = 0.00639), but no significant MPTP-induced change was seen in the presence of prior MnCl2 exposure (Figure 4(a)). Alternatively, on day 22, no significant change in rearing was observed for mice that had been exposed to either MnCl2 or MPTP (Figure 4(b)). Therefore, the interaction between MnCl2 dosing and MPTP dosing upon vertical open field movement was dependent upon survival time.

Significant interaction between MnCl2 dose, MPTP dose and survival time, for vertical open field movement. (a) At 7 days after dosing, a significant interaction of MnCl2 dose with MPTP dose was observed. MPTP produced a significant decrease in rearing in the absence of MnCl2 (p = 0.0053, solid line), but not in its presence. p Values for any statistically significant comparisons are also indicated in figure key insets. In accord with the statistical model used, values in all graphs are least squares means ± SE. For a better idea of uncorrected data dispersion, raw arithmetic means ± SD follow. No MnCl2: no MPTP 11.5 ± 5.7, MPTP 6.9 ± 4.8. MnCl2: no MPTP 8.0 ± 3.2, MPTP 11.6 ± 4.5. (b) At 14 days after dosing, no significant interaction was observed between MnCl2 dose and MPTP dose, with neither compound affecting rearing. Raw arithmetic means ± SD follow. No MnCl2: no MPTP 8.0 ± 3.3, MPTP 9.4 ± 4.9. MnCl2: no MPTP 5.9 ± 3.3, MPTP 8.2 ± 4.2. MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MnCl2: manganese chloride; SE: standard error.
Swimming
MnCl2 treatment resulted in a 26.5% main effect decrease (collapsed across both MPTP dose and postinjection survival time) in swim time (df = 1, 85; p = 0.0290). No significant main effects on swim time were observed for MPTP dosing or postinjection survival time, nor were there any significant interactions among the independent variables.
Grip strength and grip fatigue
ANOVA for grip strength revealed a significant main effect of survival time (df = 1, 89; p < 0.0001). Mice surviving to day 22 pulled 29.4% more weight than mice surviving to day 15. Although no two-way interactions between independent variables achieved statistical significance, ANOVA revealed a significant three-way interaction between MnCl2 dose, MPTP dose and postinjection survival time (df = 1, 89; p = 0.0325) with respect to their effects upon grip strength. Post hoc analysis showed that on day 15, no significant change in grip strength was observed for mice that had been dosed with either MnCl2 or MPTP (Figure 5(a)). On day 22, a significant 28.4% decrease in grip strength was observed for mice that had received MPTP in the absence of prior MnCl2 dosing (p = 0.0030; Bonferroni α = 0.00639), but no significant MPTP-induced change was seen in the presence of prior MnCl2 exposure (Figure 5(b)). Therefore, the interaction between MnCl2 dosing and MPTP dosing upon grip strength was dependent upon survival time. For grip fatigue, no significant main effects of any of the independent variables were revealed by ANOVA, nor were any significant two-way interactions observed. Although a significant three-way interaction was seen between the three independent variables for their effects on grip fatigue (df = 1, 89; p = 0.0192, α = 0.05), post hoc pairwise comparisons failed to support this result.

Significant interaction between MnCl2 dose, MPTP dose and survival time, for grip strength. (a) At 7 days after dosing, no significant interaction was observed between MnCl2 dose and MPTP dose, with neither compound affecting grip strength. In accord with the statistical model used, values in all graphs are least squares means ± SE. For a better idea of uncorrected data dispersion, raw arithmetic means ± SD follow. No MnCl2: no MPTP 39.1 ± 24.1, MPTP 47.0 ± 29.0. MnCl2: no MPTP 48.3 ± 21.6, MPTP 47.0 ± 13.8. (b) At 14 days after dosing, a significant interaction of MnCl2 dose with MPTP dose was observed. MPTP produced a significant decrease in grip strength in the absence of MnCl2 (p = 0.0030, solid line), but not in its presence. p Values for any statistically significant comparisons are also indicated in figure key insets. Raw arithmetic means ± SD follow. No MnCl2: no MPTP 82.2 ± 22.7, MPTP 58.9 ± 23.1. MnCl2: no MPTP 58.9 ± 29.7, MPTP 62.1 ± 17.5. MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MnCl2: manganese chloride; SE: standard error.
Weight
ANOVA for body weight during the MnCl2-dosing period (MnCl2 on days 1, 4 and 7; day 1 weight is predose and day 8 weight is pre-MPTP dosing) revealed a significant main effect of MnCl2 dosing (df = 1, 115; p < 0.0001) and of experimental day (df = 7, 788; p < 0.0001) as well as an interaction between the two variables (df = 7, 788; p < 0.0001). Post hoc analysis showed that mice receiving MnCl2 had significantly lower weights on days 2–8, compared with vehicle controls (df = 1, 788; all p < 0.0001), with a 7.2% difference 1 day after the first MnCl2 injection (day 2) and a 9.4% difference 1 day after the last MnCl2 injection (day 8; Figure 6(a)). A numerical pattern was observed with each MnCl2 injection in which weight decreased 1 day after injection, then began a monotonic recovery over the next two consecutive days, until 1 day after the next MnCl2 injection.
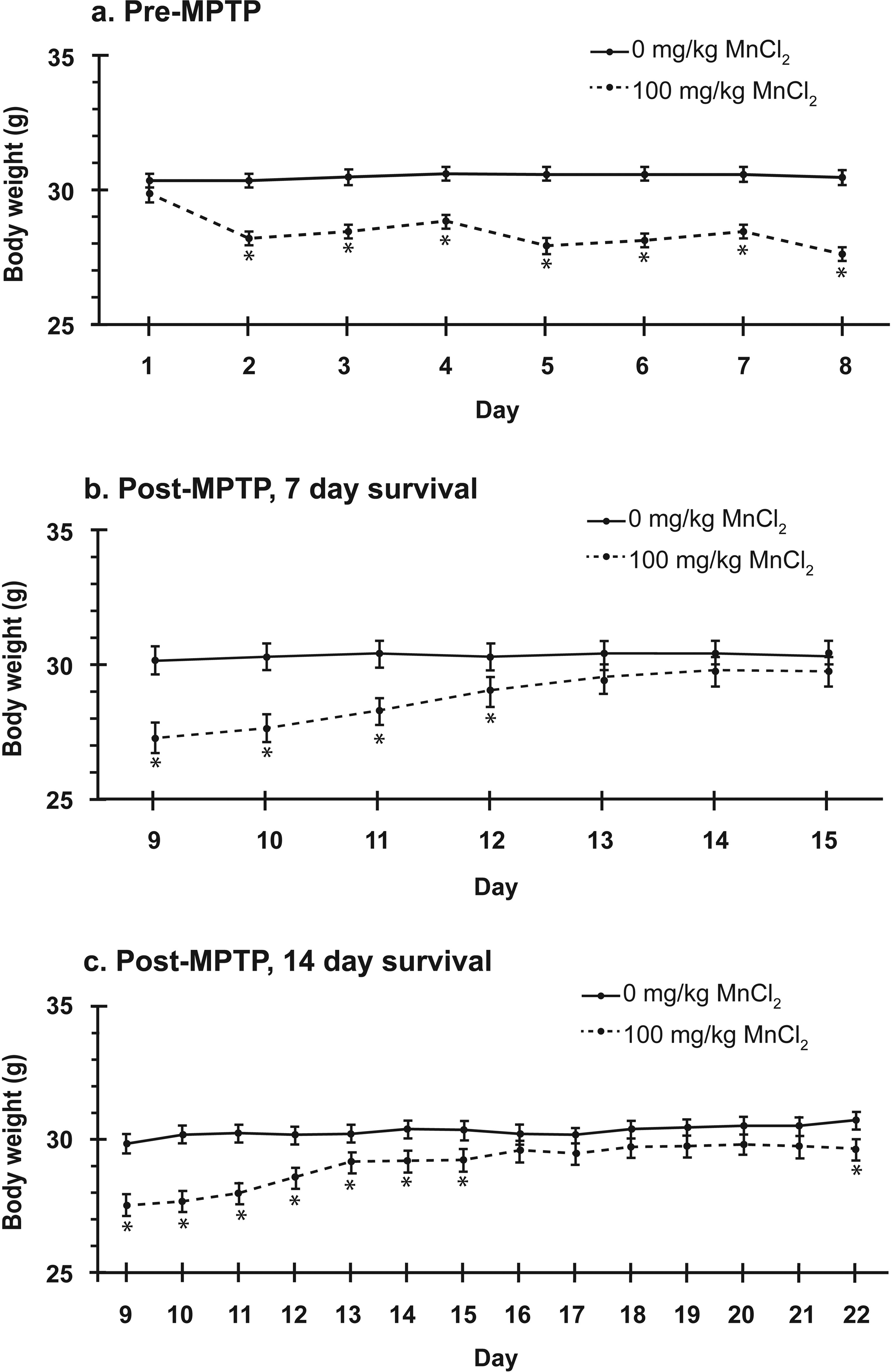
(a) The MnCl2-dosing regimen produced a significant decrease in weight on days 2–8, compared with vehicle, prior to any groups receiving MPTP. For all figure portions, asterisks indicate a statistically significant difference (p < 0.048) on a given day. In accord with the statistical model used, values in all graphs are least squares means ± SE. For a better idea of uncorrected data dispersion, selected raw arithmetic means ± SD follow. Day 3: no MnCl2 30.5 ± 2.7, MnCl2 28.5 ± 1.2. Day 7: no MnCl2 30.6 ± 2.8, MnCl2 28.3 ± 1.3. (b) Upon adding MPTP or vehicle, for groups surviving seven more days (up to day 15), there was no significant effect of MPTP on body weight nor was there an interaction between MnCl2 and MPTP dosing. For mice that had received MnCl2, weight recovered by day 13. Data are collapsed across MPTP dose. Selected raw arithmetic means ± SD follow. Day 11: no MnCl2 30.3 ± 2.9, MnCl2 28.1 ± 1.6. Day 14: no MnCl2 30.4 ± 3.0, MnCl2 29.6 ± 1.5. (c) Results for groups that survived 14 days (up to day 22) after adding MPTP or vehicle were similar to those for 7 day survival except that, for MnCl2-dosed mice, weight did not recover until day 15, and a small significant difference (3.4%) reappeared for those mice on day 22. Selected raw arithmetic means ± SD follow. Day 18: no MnCl2 30.4 ± 1.5, MnCl2 29.8 ± 1.3. Day 21: no MnCl2 30.5 ± 1.6, MnCl2 29.9 ± 1.3. MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; MnCl2: manganese chloride; SE: standard error.
During the post-MPTP-dosing period, for mice surviving 7 days (days 9–15), ANOVA for body weight revealed a significant main effect of prior MnCl2-dosing status (df = 1, 31; p = 0.0143) and of experimental day (df = 6, 222; p < 0.0001) as well as a significant interaction between the two variables (df = 6, 222; p < 0.0001, α = 0.05). Post hoc analysis showed that mice that had received MnCl2 had significantly lower weights on days 9–12 compared with mice that did not receive MnCl2 (df = 1, 35; p values from <0.001 to 0.048), and there were no significant differences on days 13–15 (Figure 6(b)). The percentage difference on day 9 was 9.67% and was followed by a monotonic recovery to as little as a 2.27% numerical, nonsignificant difference on day 15. There was no main effect of MPTP dosing on body weight, nor was there an interaction of MPTP dosing with MnCl2 dosing. Although a statistically significant interaction of MPTP dosing with experimental day was calculated by ANOVA (df = 6, 222; p = 0.0106), post hoc exploration of this relationship failed to validate a significant effect of MPTP dosing on any individual experimental day. Finally, no three-way interaction among the variables was revealed by ANOVA. Therefore, the effect of MnCl2 exposure on weight was not affected by the subsequent MPTP dose over the course of the 7-day post-MPTP survival period.
For mice surviving 14 days during the post-MPTP dose period (days 9–22), ANOVA revealed a significant main effect of prior MnCl2-dosing status (df = 1, 45; p = 0.0037) and of experimental day (df = 13, 532; p < 0.0001) as well as a significant interaction between the two variables (df = 13, 532; p < 0.0001, α = 0.05). Post hoc analysis showed that significant MnCl2-induced decreases in weight, relative to vehicle control, persisted from day 9 (7.8% difference) to day 15 (3.8% difference; df = 61.9–64.2; p values from <0.001 to 0.011; Figure 6(c)). The significant differences in weight then disappeared on days 16–21, but the 3.4% difference observed on day 22 was again significant (df = 64.2; p = 0.019). No significant main effect of MPTP was observed nor any other significant two- or three-way interactions were observed.
Discussion
In general, the results of this experiment showed that a dosing regimen of MnCl2, previously shown to produce a significant increase in basal ganglia manganese, can interact with MPTP to modulate motor deficits induced by either of these compounds alone, but fails to modulate MPTP-induced dopaminergic toxicity in the nigrostriatal pathway. Surprisingly, in this paradigm, there appeared to be mitigation, as opposed to facilitation, of motor deficits.
The absence of a modulatory effect of MnCl2 exposure upon MPTP-induced decreases in striatal dopamine and DOPAC support similar findings in the C57BL/6 mouse by Baek et al. 24,25 This absence of effect occurred even though the present study delivered a higher cumulative dose of MnCl2 (3 × 100 mg/kg vs. 21 × 8 mg/kg) that we have previously shown to produce a higher proportional increase in basal ganglia manganese compared with that reported by Baek et al. 24,25,28 The smaller total dose of MPTP used in the present study (1 × 20 mg/kg vs. 5 × 30 mg/kg) had the desired effect of producing smaller decreases in dopamine and DOPAC, at the time of sacrificing (49 and 38% vs. 88 and 60%), compared with Baek et al. 24 This minimized the likelihood that a potential facilitation of dopaminergic toxicity by MnCl2 was masked by too large of an MPTP-induced reduction in dopamine and DOPAC. However, using this single MPTP dose may have reduced the external validity, somewhat, in relation to a PD model. Nevertheless, it should also be recalled that the present study used mice that were 7–9-month-old retired breeders and thus were more concordant with the stage of life for human PD, compared with the young, 8-week-old mice used by Baek et al. 24 Our study also used higher sample sizes (15–16 per group vs. 7–9 per group). Despite these improvements in design, there was still a lack of support for the modulation of MPTP-induced nigrostriatal dopaminergic damage by MnCl2 exposure. At this time, we are not aware of any other studies in this strain that have examined the effect of an inorganic manganese compound on MPTP-induced dopaminergic neurotoxicity. The form of inorganic manganese employed in our study was principally chosen to be concordant with that used by Baek and colleagues, whose studies we were revisiting. Soluble forms of manganese have been shown to produce greater increases in brain manganese concentration than insoluble forms, for several dosing routes. 36 –39 As noted, we have previously confirmed that the MnCl2 dosing regimen and route used here produce a considerable increase in basal ganglia manganese, in our hands, by the day that MPTP exposure occurred in this experiment. 28 Although the most common environmental exposure to manganese may be through inhalation of nonchloride forms, MnCl2 is often a component of total parenteral nutrition (TPN), which bypasses the alimentary and respiratory routes, and signs of manganism have been reported in some individuals receiving TPN. 40 –42
Significant modulatory interactions were observed between the effects of MnCl2 and MPTP for three of our five behavioral dependent variables, despite the lack of interaction effects upon striatal dopamine and DOPAC concentrations. Interestingly, for each of these behavioral measures, a deficit induced by one compound was ameliorated by the presence of the other compound. To date, we are aware of only two other studies that examined the effects of a manganese-containing compound on behavior in an MPTP-induced parkinsonism paradigm. In one study, the manganese-containing fungicide maneb was reported to facilitate MPTP-induced catalepsy and deficits in locomotion in Swiss mice. 19 In another mouse study, using the C57BL/6 strain, MPTP triggered locomotor deficits in maneb-exposed mice that were not seen with either maneb or MPTP alone. 43 However, the relevance of manganese in these effects is unclear because maneb is a dithiocarbamate compound, and nonmanganese-containing diethyldithiocarbamate has previously been reported to exacerbate the behavioral effects of MPTP alone, in the mouse (C57BL/6 strain). 44 –46 Neither of these studies examined corresponding effects on brain neurochemistry. A novel manganese porphyrin was shown to attenuate paraquat-induced decreases in pole climbing and swimming behavior that accompanied an attenuation of paraquat-induced decreases in striatal dopamine content. 47 In the 6-hydroxydopamine (6-OHDA) rat model of PD, combined administration of MnCl2 (4.8 mg/kg i.p., three times per week for 5 weeks) exacerbated deficits in 2 of 10 motor behaviors measured (reactivity to handling and limb hopping) despite a lack of facilitation of striatal dopamine depletion. 18 For the 6-OHDA rat model induced in pups 3 days postnatally, MnCl2 in the maternal drinking water, from conception to weaning, facilitated changes in some dopamine agonist-induced motor behaviors (increased chewing and decreased yawning), but eliminated changes in agonist-induced catalepsy, all in the absence of a facilitated striatal dopamine depletion. 48 In an iron-induced rat model of parkinsonism, adding intranigral MnCl2 suppresed contralateral turning behavior induced by intranigral iron alone. 21 This behavioral effect accompanied a protection against iron-induced, striatal dopamine depletion.
It is apparent from the aforementioned manganese–parkinsonism behavioral interaction findings that the nature of the interaction can be quite complex and depend upon the particular manganese compound, the particular parkinsonism model, the species, and even the particular behaviors examined within a specific experiment. It is also apparent that an interaction effect upon striatal dopamine or DOPAC is not a necessary substrate for observing a manganese–parkinsonism interaction at the behavioral level. There is evidence that immunohistochemical indicators of nigrostriatal damage, such as decreased tyrosine hydroxylase immunopositive somata in the substantia nigra pars compacta, can be observed in the absence of a decrease in striatal dopamine in the maneb–paraquat C57BL/6 model. 43,49 An analogous manganese–MPTP interaction may have been missed in the present study because the substantia nigra was not histologically examined. More subtly, even in the absence of evidence for decreases in striatal tyrosine hydroxylase, alterations of striatal D2 receptors were observed that accompanied manganese-associated changes in motor performance in C57BL/6 mice and in rats. 50,51 Subtle manganese-associated dopaminergic changes that affected primate fine motor behavior, such as a decrease in dopamine release, have also been observed in the absence of a change in striatal dopamine or DOPAC. 52 It is also possible that interaction effects on behavior, in the absence of interaction effects on striatal dopamine, could be mediated by effects on other neurotransmitter systems or by changes in areas other than the basal ganglia. In the C57BL/6 mouse, manganese-associated decreases in the γ-aminobutyric acid (GABA) synthesizing enzyme glutamate decarboxylase 67 were observed in the striatum. 53 In the rat 6-OHDA model of parkinsonism, where MnCl2 exacerbated deficits in motor function, the metal compound alone failed to affect striatal dopamine, but significantly increased striatal GABA, as well as increasing striatal aspartate, when combined with 6-OHDA. 54 Manganese sulfate inhalation in the rhesus monkey has been shown to decrease GABA concentration and the serotonin metabolite 5-hydroxyindoleacetic acid in the globus pallidus as well as norepinephrine concentration in the striatum. 55 Depending on the specific MPTP dose regimen, changes in striatal serotonin concentration and/or turnover have been reported along with the changes in frontal cortical norepinephrine, which accompanied changes in particular motor behaviors. 56 In addition, the glutamatergic system is thought to play a significant role in dopaminergic cell death in PD and in experimental PD models. 8,57,58 Furthermore, it is apparent that following systemic exposure to manganese compounds, the metal can accumulate in motor-related regions other than the basal ganglia, such as the cerebral cortex and cerebellum. 59 –61
As cited above, there is evidence that the particular behavior that was examined can be relevant to whether one observes a manganese facilitation or attenuation of the effects of compounds that induce experimental parkinsonism. However, metabolic interactions of these compounds, both peripherally and centrally, could have accounted for the attenuation of behavioral effects observed in this experiment. In Swiss mice, systemic administration of MPTP was shown to significantly reduce manganese content in the striatum. 62 Interestingly, 6-OHDA has been reported to produce the opposite effect in rats. 63 It has recently been demonstrated that MnCl2 can compromise the function of rat’s immortalized endothelial cells in an in vitro model of the blood–brain barrier. 64 Such changes could affect the integrity of the barrier and the passage of MPTP to the brain. Several studies have suggested the involvement of the dopamine transporter (DAT) upon manganese accumulation in the basal ganglia. DAT-knockout mice were found to have decreased manganese accumulation in the striatum, and treatment with the DAT inhibitor GBR12909 significantly decreased manganese accumulation in the globus pallidus. 65,66 Given that the DAT is the principal means by which MPTP’s neurotoxic metabolite 1-methyl-4-phenylpyridinium (MPP+) gains access to dopaminergic neurons, if manganese is being carried by the DAT, it is possible that such transport could reduce MPP+ access to nigrostriatal neurons following manganese exposure in the MPTP model. In addition, postnatal, high-dose exposure to MnCl2 caused a decline in DAT protein expression and consequently decreased dopamine uptake in adult rats. 67 Such an effect could also attenuate MPP+ uptake and subsequent insult to dopaminergic basal ganglia neurons. Conversely, MPP+ derived from MPTP dosing could interfere with DAT-mediated manganese accumulation in the basal ganglia.
We have previously reported a significant main effect decrease in horizontal open field movement 24 h after the same dosing regimen used in the present study, but the decrease (43.2%) was larger in the previous experiment. 28 The smaller magnitude decrease observed in the present work (26.6%), in the absence of MPTP, could be attributable to recovery over a much longer survival time. However, it is puzzling why a significant decrease in horizontal movement was seen here at 14-day postinjection, but not at 7 days. We are aware of only a handful of other reports regarding the effects of manganese on behavior of the adult C57BL/6 mouse. A 3 × 60 mg/kg MnCl2 i.p. dosing regimen (days 1, 3 and 5) produced a significant decrease in horizontal locomotion less than 24 h after the last dose, 68 but 63 × 5 mg/kg daily i.p. doses 69 and 56 × 30 mg/kg daily oral doses 70 failed to change locomotion. The decreased swim time to platform that was observed in the present study was not seen 24 h after the same dosing regimen in our previous work. 28 This could be related to differences in survival time. Nevertheless, the direction of the change, facilitation rather than poverty of movement, was unexpected. It is interesting to note, however, that C57BL/6 mice exposed as juveniles have been reported to show increased novelty-seeking and hyperactive behaviors, and similar findings have been reported in preweaning-exposed rats. 70,71 It appears that even within strain, and employing a consistent chemical form of manganese, effects on behavior as well as on striatal dopamine content are sensitive to variation in dosing route, dosing duration and postdosing survival time. Furthermore, whether manganese induces a decrease or facilitation in motor behavior can also depend on the age of exposure and on the specific behavior examined.
Although a significant MnCl2-induced weight loss of up to 9.7% was observed 2 days after the final dose, it is unlikely that this had a notable impact on the behavioral findings because this difference was gone 2 days before behavioral testing in the 7-day postdosing survival group, and was only 3.4% (after six prior days of no difference) at testing in the 14-day survival group. Furthermore, it is unlikely that weight loss was responsible for a general morbidity at behavioral testing because a manganese-associated increase in swimming performance was observed. Moreover, if weight loss-induced morbidity were occurring at behavioral testing in MnCl2-treated mice, we would not have expected to see the amelioration of MPTP-associated motor deficits in the presence of MnCl2. No previous studies of effects of body weight on brain dopamine concentration could be found.
As noted above, the emerging picture of PD is one of multifactorial etiology. It is a daunting task to identify those contributing factors, as well as potential interactions among them, in determining the onset or severity of the disease. The number of literature-relevant, independent and dependent variables that can be, respectively, chosen for manipulation and measurement in studying such a disorder can be overwhelming, particularly in the laboratory setting. The overall body of investigation needs to cover a variety of experimental strategies, thoroughly and systematically focusing on a small number of relevant variables in well-defined experimental models, yet have the breadth to incorporate a range of relevant models and sets of variables. The present work followed the spirit of the former strategy. It revisited the potential interaction of manganese with the Parkinson’s-like-inducing compound MPTP in determining dopamine concentration, keeping the strain, chemical form of manganese, brain region examined and measurement technique the same as specific previous studies. 24,25 However, improvements were made in sample size, external validity (older mice), induced striatal manganese level and in the reduction of masking of potential manganese–MPTP interactions (smaller MPTP-induced dopamine reductions). We therefore have stronger confidence in the previously reported finding that MnCl2 exposure fails to modulate MPTP-induced decreases in striatal dopamine and DOPAC in the C57BL/6 mouse. 24,25 Our extension of dependent variables into the behavioral realm allowed us to uncover an interaction of MnCl2 with MPTP exposure, previously unreported in this specific paradigm, that reduced the severity of behavioral deficits attributable to either of these compounds alone. This finding suggests that some environmental compounds may influence MPTP-induced toxicity in ways more subtle than decreased striatal dopamine concentration. The results also extend the handful of literature regarding the effects of manganese on behavior of the adult C57BL/6 mouse.
Our behavioral findings warrant further exploration to verify their reliability and to identify their mechanistic substrates. In looking back on the present work, a dose–response design, with at least three dose levels, would have been desirable and simple to implement to provide a clearer, more reliable, causal picture of the manganese–MPTP interaction upon behavior. Another simple modification would have been to include the measurement of other monoamine neurotransmitters, and their metabolites, in the HPLC analysis of our samples because changes in these neurochemicals are potential alternative substrates for the type of interaction described. However, adding these to the present experiment would have resulted in quite an unwieldy statistical analysis.
With a prospective eye toward systematically uncovering the substrates of the manganese–MPTP interaction identified in the present experiment, it would initially be of value to assess whether each of these compounds can affect the brain accumulation, metabolism or clearance of the other, within the same treatment paradigm used in the present experiment. This would shed light on whether pharmacokinetic interactions between the compounds could be significantly contributing to the reduced severity of behavioral deficits by affecting access to neural tissue, in contrast to physiological or anatomical effects upon that tissue. If so, future use of a Parkinson’s-like-inducing compound, other than MPTP, might be warranted to more clearly assess the influence of MnCl2 exposure upon Parkinson’s-like phenomena in the C57BL/6 mouse.
Footnotes
Acknowledgments
The authors thank Drs Daniel Ward and Stephen Werre for their thoughtful assistance with the statistical analysis; Robert Simonds for providing access to the load cell and for help with digitizing and recording its output, in the assessment of grip strength and grip fatigue; and the Teaching and Research Animal Care Support Service (TRACSS) of the Virginia-Maryland Regional College of Veterinary Medicine for excellent animal care.
Funding
This work was funded by the Alzheimer’s and Related Diseases Research Award Fund of the Virginia Center on Aging (05-1863-03).
