Abstract
A method of extracting membranes from red blood cells (RBCs) is described, which were in turn used to assay acetylcholinesterase (AChE) activity. The evidence for the enzyme activity was established by selective inhibition using 1,5-bis(4-allyldimethylammoniumphenyl) pentan-3-one dibromide, tetraisopropyl pyrophosphoramide and neostigmine. Blood samples were exposed to three organophosphorus (dichlorvos, chlorpyrifos and diazinon) and two carbamate (carbaryl and carbofuran) pesticides. Afterwards AChE activities in RBC membranes were determined. The concentrations capable to inhibit the enzyme activity by 50% (IC50) for the pesticides were 10.66 µM (dichlorvos), 21.42 µM (chlorpyrifos), 109.98 µM (carbaryl) and 5.44 µM (carbofuran). The results related to 20% enzyme inhibition (level used in the estimation of threshold limits for anticholinesterase compounds) were below those acceptable daily intake values enacted by relevant national and international regulations. These results suggest that the proposed AChE extraction from RBC and assay could be a suitable method for monitoring occupational exposure to pesticides.
Introduction
Although pesticides have provided an increase in agricultural productivity, enabling high-quality food at lower costs, the improper use of these chemicals can bring harm to human health and negative impact to environment. 1 It is estimated that only 0.1% of the applied pesticides in fact reach the target animals, whereas the rest spreads throughout the environment. The financial costs saved by pest control are partially wasted through the environmental and public health problems caused by pesticides. 2 Agricultural workers are exposed to high risks of poisoning due to intense contact with pesticides. Such compounds can cause adverse effects on central and peripheral nervous system, immune system and are carcinogenic. The most used classes of insecticides and source of occupational poisoning are the organophosphates (OPs) and carbamates (CBs). In 2007, OPs accounted for 35% of all insecticides used in the United States. 3
OPs and CBs are typical inhibitors of the cholinesterases (ChEs), and there are two accepted types of these enzymes. First, the enzyme acetylcholinesterase (AChE; EC 3.1.1.7), that occurs in brain, ganglia of the autonomic nervous system and motor end plates, is produced by the neurons and it plays the primary function in the nerve impulse modulation at the synaptic clefts. Another form of AChE is observed in the plasma membrane of red blood cells (RBCs), which is synthesized during the process of bone marrow hematopoiesis and has a half-life of approximately 120 days, the same as that of RBCs. Second, the enzyme butyrylcholinesterase (BChE; EC 3.1.1.8) is synthesized in liver and predominates in plasma, glial cells, pancreas and the walls of digestive tract and presents an in vivo half-life of 7 days. 4 –6
OPs and CBs are widely used in developing countries, predominantly in agriculture, for pest control and public health campaigns to eradicate disease vectors. 7 –9 In the developing world, the negative consequences of pesticide usage are conditioned by factors closely related, such as the mishandling of these substances, the high toxicity of some products, the lack of protective equipment and the poor surveillance. This situation is aggravated by low cultural and socioeconomic status for most of these workers. Markedly in these countries, the monitoring of occupational exposure to such compounds presents problems in rural locations, especially the distance of testing laboratories where there is no appropriate infrastructure for the analysis. 10 –12 Here, we developed a method for the extraction and assay of human RBC AChE and evaluated the sensitivity and the specificity of the method to measure the exposure to pesticides, comparing the results of inhibition with relevant national and international regulations in force.
Materials and methods
Reagents and materials
Acetylthiocholine iodide, tetraisopropyl pyrophosphoramide (Iso-OMPA), 1,5-bis(4-allyldimethylammoniumphenyl) pentan-3-one dibromide (BW284c51), neostigmine bromide, bovine serum albumin, 5,5′-dithiobis(2-nitrobenzoic) acid (DTNB), tris (hydroxymethyl) aminomethane and dimethyl sulfoxide (DMSO) were purchased from Sigma-Aldrich (St Louis, Missouri, USA). Disodium ethylenediamine tetraacetic acid (EDTA) was obtained from Merck (Darmstadt, Germany). Analytical grade dichlorvos (98.8%), diazinon (99.0%), chlorpyrifos (99.5%), carbofuran (99.9%) and carbaryl (99.8%) were obtained from Riedel-de-Haën, Pestanal® (Seelze, Germany). A Bio-Rad xMark (Hercules, California, USA) microplate spectrophotometer was used.
Methods
Blood samples
The blood samples were collected (9 mL) from healthy students (n = 22), who were not exposed to pesticides by venepuncture and gentle hand shaking and the blood samples were homogenized with 10% EDTA (100 µL). These individuals were previously interviewed regarding the occurrence of diseases that could interfere with the activity of the enzyme: pregnancy, anemia, hemorrhagic events and reticulocytosis. Furthermore, they previously agreed to participate in this research approved by the Ethical Committee of The Health Science Centre of the Federal University of Pernambuco, Brazil (protocol number 0158.0.172.000-09).
Sample processing and enzyme extraction
Aliquots (1.8 mL) of whole blood were incubated for 60 min with 200 µL solutions of each pesticide prepared with 0.01 M Tris-HCl buffer, pH 7.6, containing 0.15 M sodium chloride (Tris-buffered saline (TBS) buffer). The pesticides used included the OPs (dichlorvos, diazinon and chlorpyrifos) and the CB (carbaryl and carbofuran). The pesticides were first dissolved in DMSO and diluted with distilled water to attain 13 solutions at final concentrations ranging from 0.01 to 100 µg/mL in 2% (v/v) DMSO.
Modifying Oliveira-Silva et al. 11 method, the whole blood samples were also incubated with 200 µL of TBS only and TBS + 2% DMSO instead of the pesticides as controls. The samples were then centrifuged at 2,000g for 4 min to obtain plasma and erythrocyte fractions. They were separated and the aliquots of 500 µL of RBC were resuspended in 4.5 mL of lysis buffer (0.01 m Tris-HCl pH 7.6 without salt). The samples were kept at −20°C for 24 h for RBC lysis. After lysis, the RBC samples were centrifuged three times at 4,000g for 15 min, discarding the supernatant after each centrifugation. Afterwards the pellet (RBC membranes or ‘ghost’ suspension) was resuspended in 500 µL of enzymatic assay buffer (0.5 M Tris-HCl pH 7.4). Aliquots of the RBC membrane controls were also used to characterize RBC AChE selective inhibition.
Enzyme activity for selective inhibition
Samples of the RBC membranes from controls were exposed to selective inhibitors: BW284c51 for AChE, Iso-OMPA for BChE and neostigmine bromide as total ChEs inhibitor. The samples (10 µL) were kept inside the microplate wells and exposed to each inhibitor (10 µL) at five different concentrations ranging from 0.001 to 10 mM for 60 min. After incubation, the enzyme activity was determined using Ellman method 13 with modification by Assis. 14 Briefly, 0.25 mM DTNB (200 μL) prepared in 0.5 M Tris-HCl buffer, pH 7.4 was added to the incubated mixture and the reaction was started by the addition of 62 mM acetylthiocholine iodide (20 μL). Enzyme activity was determined by reading the absorbance increase at 405 nm for 180s. A unit of activity (U) was defined as the amount of enzyme capable of converting 1 μM of substrate per minute. A blank was prepared with the assay buffer instead of ghost suspension sample. All these assays were carried out in quadruplicates.
Enzyme activity in blood samples exposed to pesticides
The activity of membrane RBC AChE extracted from whole blood samples incubated with pesticides and controls were determined by mixing 200 µL of DTNB with 20 µL of ghost samples and finally adding 20 µL of the substrate acetylthiocholine iodide. The reaction was carried out and followed spectrophotometrically, similarly as described in the section on selective inhibition. All these assays were carried out in quadruplicates.
Protein determination
Protein content in the RBC membrane preparations was estimated according to Sedmak and Grossberg, 15 using bovine serum albumin as a standard. All tests were performed at room temperature (25°C) in triplicates.
Estimation of IC20, IC50 and Ki
The enzymatic activity values obtained from selective inhibitors and pesticide exposition were plotted against selective inhibitor or insecticide concentration. From the curves generated by nonlinear regression fitting (using MicroCal™ Origin® Version 8.0), the IC50 and IC20 (concentrations capable of inhibiting the enzyme activity by 50% and 20%, respectively) were estimated for each selective inhibitor or pesticide. Their respective inhibition constants (K i) were calculated using the Cheng and Prusoff equation. 16
Comparative study of enzyme inhibition in accordance with current regulations
The IC20 found for the pesticides was converted from microgram per milliliter to milligram per kilogram body weight per day (acceptable daily intake (ADI) unit) for comparison with the results reported by specialized agencies.
Sensitivity and specificity of the method
The ability to correctly detect whether the samples were exposed to each pesticide was analyzed by measuring the sensitivity and specificity of the test according to Glaser 17 and using 20% of enzymatic inhibition as a cutoff. 18
Results
AChE present in the RBC membrane preparation was strongly inhibited (about 50%) by its specific inhibitor BW284c51 at 0.001 mM (Figure 1(a)), whereas under Iso-OMPA exposure (BChE specific inhibitor) its activity was only 19% reduced at 1 mM (Figure 1(b)). On the other hand, neostigmine, a very potent inhibitor of total ChEs, abolished the enzyme activity even at 0.001 mM (Figure 1(c)). These results show the ability of the enzyme extraction method in getting just the fraction of AChE from RBC. The IC50 and K i values for BW284c51 and neostigmine were 0.92 µM and 0.0054 µM; and 0.30 µM and 0.0018 µM, respectively. It was not possible to estimate these parameters for Iso-OMPA.

Effect of (a) BW284c51, (b) Iso-OMPA and (c) neostigmine bromide from 0.001 to 10 mM in frozen blood samples of human RBC AChE activity. RBC: red blood cell; AChE: acetylcholinesterase; Iso-OMPA: tetraisopropyl pyrophosphoramide; BW284c51: 1,5-bis(4-allyldimethylammoniumphenyl) pentan-3-one dibromide.
The effects of dichlorvos, diazinon and chlorpyrifos (OPs) as well as carbaryl and carbofuran (CBs) on RBC membrane AChE are displayed in Figures 2 and 3, respectively. The results show that diazinon is a less effective inhibitor among the five investigated pesticides, whereas dichlorvos and carbofuran were the most powerful inhibitors. These findings are corroborated by their IC20, IC50 and K i (Table 1) estimated from the curves. The parameter IC20 converted to milligram per Kg body weight per day was confronted with the values of ADI found in national and international specific legislation in Table 1.
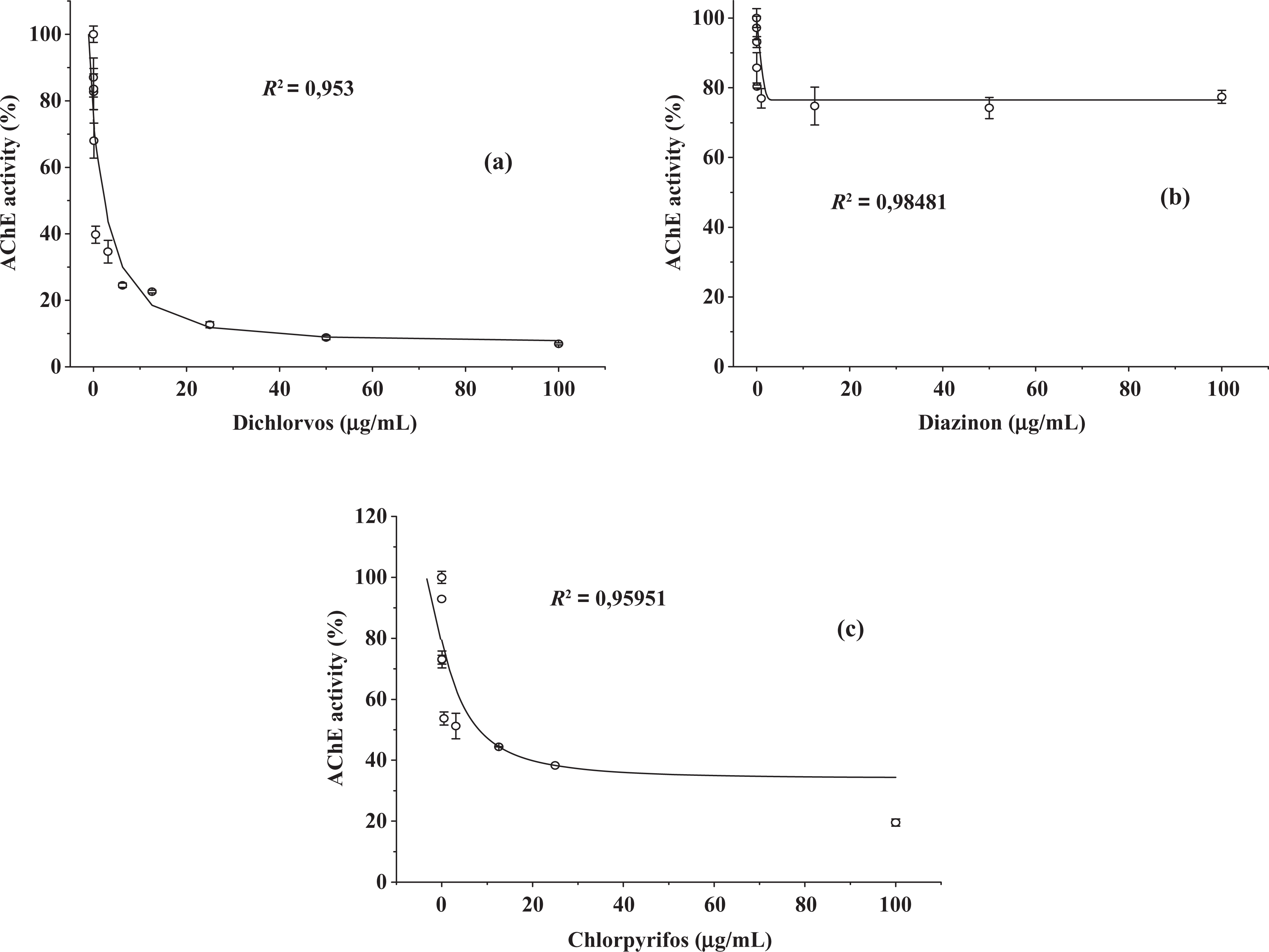
Effect of (a) dichlorvos, (b) diazinon and (c) chlorpyrifos from 0.01 to 100 µg/mL on frozen blood samples of human RBC AChE activity. RBC: red blood cell; AChE: acetylcholinesterase.
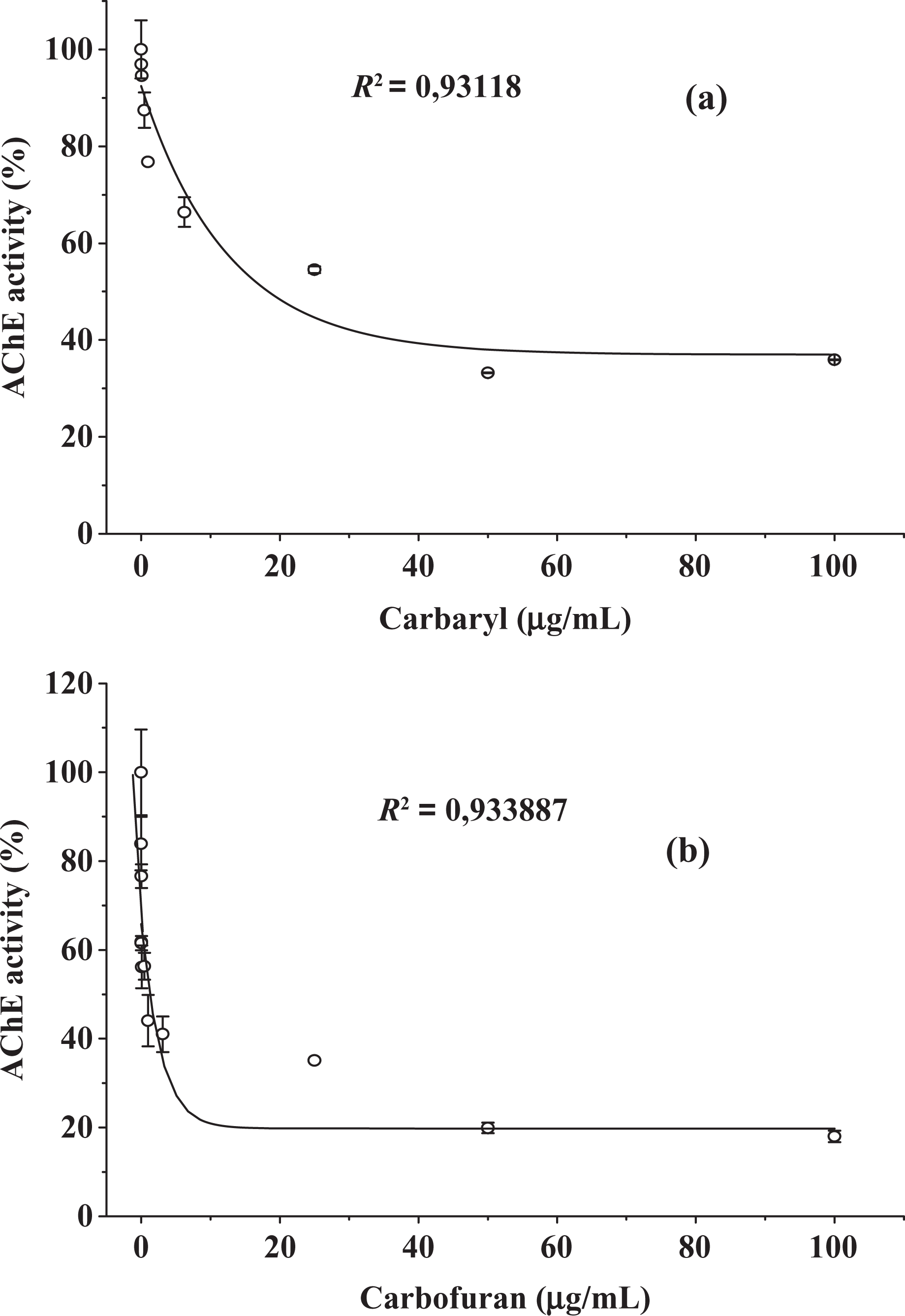
Effect of (a) carbaryl and (b) carbofuran from 0.01 to 100 µg/mL on frozen blood samples of human RBC AChE activity. RBC: red blood cell; AChE: acetylcholinesterase.
ADI, IC20 and IC50 for human RBC AChE referring to pesticides under study

RBC: red blood cell; AChE: acetylcholinesterase; IC20 and IC50: concentrations capable to inhibit the enzyme activity by 20% and 50%, respectively; ADI: acceptable daily intake.
The results of sensitivity and specificity for each pesticide are listed in Table 2. Sensitivity ranged from 75% (diazinon) to 100% (chlorpyrifos and carbaryl), and specificity from 63.6% (carbofuran) to 83.3% (dichlorvos).
Sensitivity and specificitya of the human erythrocyte AChE and pesticide inhibition method in relation to pesticides under study

AChE: acetylcholinesterase.
aAccording to Glaser. 17
Discussion
The mechanism of action of OPs and CBs occurs through binding to the esteratic site of enzymes with phosphorylation and carbamoylation, respectively. 19,20 Inhibition by the organophosphorus compounds tends to be irreversible if untreated. 7 The inhibition by CBs is reversible and the recovery of the enzyme may take several minutes to hours. 8
The monitoring of pesticides based on blood ChE inhibition recommended by World Health Organization (WHO) 21,22 requires fresh blood samples of total ChE using the methods of Edson 23 and Ellman 13 (adapted by WHO) 22 or RBC AChE by George and Abernethy. 24 The disadvantages of these methods are the nondiscriminating use of both blood enzyme activities in case of using total ChE samples and the less accuracy by hemoglobin interference in the case of those which use RBC AChE samples without complete red cell lysis and centrifugation. The method proposed by Oliveira-Silva et al. 11 not only uses RBC AChE but also allows the freezing of samples for further analysis in an appropriate place without colorimetric interactions with hemoglobin.
Several works chose RBC AChE instead of BChE for many reasons, as follows: (1) determination of inter- and intraindividual variation in both ChE activities, which was considered critical for a blood esterase monitoring program. The class of AChEs is more homogeneous in terms of their primary structure than the class of BChEs, 25 and RBC AChE activity was pointed to be less variable than BChE 26,27 ; (2) the first one is more closely correlated with the AChE activity from nervous system 28 ; (3) more stability in frozen blood samples: 7 days against 3 days for BChE 11 ; (4) BChE spontaneous recovery of inhibited forms are faster than AChE. 26 In order to perform a successful monitoring program, it should be taken into account that some conditions other than pesticide exposure can change blood ChEs activities, hindering the evaluation and interpretation of results from studies. BChE activity can be decreased by liver diseases, malnutrition, alcoholism, nephritic syndrome, pregnancy, contraceptive pills and metoclopramide, whereas RBC AChE activity is altered by pregnancy, anemia, hemorrhagic events and reticulocytosis. Other factors that may result in misinterpretation of ChE levels are collection, improper processing and transportation of samples and laboratory errors. 29,30 In addition, considering each person as his or her own control, measures should be taken to minimize intraindividual variation by collecting samples in periods of growing seasons before and after pesticide applications.
The efficiency of the proposed method to extract the membrane RBC AChE was shown by selective inhibition by BW284c51, whereas Iso-OMPA (BChE specific inhibitor) did not impair significantly the activity at low concentrations. Also, regarding the inhibition effect of BW284c51 and neostigmine on membrane RBC AChE is worthwhile to register that the dissociation rate (K i) of neostigmine was threefold slower than that of BW284c51. One of the more remarkable differences between AChE and BChE is the smaller cavity of AChE active site, lined by six aromatic amino acid residues that prevent the entrance of the selective BChE inhibitors and substrates. 31
Once established that membrane RBC AChE was properly extracted the whole blood was exposed to pesticides, simulating intoxication by them in order to evaluate the enzyme activity reduction as measure of this contact. The results showed the highest inhibitory action on the enzyme by dichlorvos compared with diazinon and chlorpyrifos. Dichlorvos is already bioactive as an oxon, whereas the thion form (diazinon and chlorpyrifos form) needs biotransformation to enhance its toxic action. 7,32 However, another feature that interferes in the toxicokinetic of OP pesticides and that was decisive in the results is their specific lipophilicity. The most lipophilic compounds are rapidly absorbed and accumulated in fat and this contributes to the reduction in the primary effects of these pesticides (ChEs inhibition) while increasing secondary effects in other biomolecules. Phosphorothioates (diazinon and chlorpyrifos) are more lipophilic than phosphates (dichlorvos). 32 In addition, serum oxonases seem to be more effective in the hydrolysis of diazinon and chlorpyrifos. 33 –35 Moreover, according to Rosenberry, 25 AChE is more sensitive to small acylchain size condition fulfilled by dichlorvos compared with diazinon and chlorpyrifos.
Between the investigated CBs, carbofuran showed to be more toxic to membrane RBC AChE than carbaryl. This difference may be attributed to the 2,2-dimethylbenzofuranyl ring (carbofuran) that is more reactive than the naphthyl ring (carbaryl) when interacting with residues in the AChE active center. 36 The controls with and without DMSO did not present significant difference.
The IC20 is the threshold limit to consider the presence of an anticholinesterasic compound, whereas the IC50 represents the point at which clinical signs and symptoms appear and death occurs after 90% inhibition. 18 Here, the IC20 values were converted into ADI unit that stands for the highest concentration causing no effect (no-observed-adverse-effect-level) on the most susceptible species of mammal in long-term studies (chronic exposure).
To verify the possibility of using membrane RBC AChE extracted by the method proposed here as a biomarker for pesticides exposure, the IC20 values of each one (Table 1) should be below their respective ADIs. Thus, the values of IC20 for all the studied pesticides were lower than those recommended by WHO, 42,46 Food and Agriculture Organization, 38,48,50 Agency for Toxic Substances and Disease Registry, 9,43,47 European Food Safety Agency, 40,44,51,54 United States Environmental Protection Agency 39,53 and ANVISA (Brazilian Sanitary Surveillance Agency) 37,41,45,49,52 except diazinon. It is noteworthy to mention that carbofuran presented low IC20 value and has been outlawed nowadays in the United States for use in food farming. 55
The cutoff of 20% inhibition of AChE activity was chosen to evaluate the sensitivity and specificity, according to several institution recommendations as mentioned above. Sensitivity values for the five investigated pesticides were above 80%, except for diazinon, whereas all the specificity values were above 60% (Table 2). Higher values would be found if the used cutoff was 50% inhibition but at this point clinical signs of poisoning by anti-ChE agents appear 18 and the present proposed procedure would lose its usefulness.
Conclusions
The results of the extraction method can be ascribed to RBC AChE according to selective inhibition. The used enzymatic assay allowed relevant levels of inhibition to be achieved, and they were at pesticide concentrations below the majority of ADIs adopted for the analyzed OPs and CB insecticides in foods by national and international regulations. Regarding this, the method showed sufficient in vitro accuracy to be a promising tool in human monitoring programs for occupational exposure to such pesticides and can be useful for sample collections in locations far from the laboratories.
Footnotes
Acknowledgments
The authors would like to thank Fundação de Apoio à Ciência e Tecnologia do Estado de Pernambuco (FACEPE), Empresa Brasileira de Pesquisa AgropecuÃria (EMBRAPA) Financiadora de Estudos e Projetos (FINEP/RECARCINE), Petróleo do Brasil S/A (PETROBRAS), Secretaria Especial de Aqüicultura e Pesca (SEAP/PR), Conselho Nacional de Pesquisa e Desenvolvimento Científico (CNPq).
Funding
Fundação de Apoio à Ciência e Tecnologia do Estado de Pernambuco (FACEPE) for financial support (Grant number: IBPG-0523-2.08/11 FACEPE).
