Abstract
The purpose of this study was to investigate the protective effects of selenium (Se) against genotoxicity induced by diazinon (DZN) in rat peripheral blood lymphocytes by micronucleus (MN) test. Animals were concurrently administered intraperitoneally with DZN in proper solvent (20 mg/kg body weight (b.w.)) and Se at three different doses (0.5, 1, and 2 mg/kg b.w.) for 30 consecutive days. The positive control group received DZN at the same dose without Se. After 24 h of last injection, 0.5 ml blood of each rat was received and cultured in culture medium for 44 h. The lymphocyte cultures were mitogenically stimulated with cytochalasin B to allow the evaluation of number of MNs in cytokinesis-blocked binucleated cells. Incubation of lymphocytes with DZN induced additional genotoxicity and is shown by increase in MNs frequency in human lymphocytes. Se at low dose of 0.5 mg/kg had a maximum effect and significantly reduced the MNs frequency in cultured lymphocytes (p < 0.0001) that reduced the frequency of MN from 12.78 ± 0.24% for DZN group to 4.40 ± 0.36. The present study revealed that Se particularly at low doses has a potent antigenotoxic effect against DZN -induced toxicity in rats, which may be due to the scavenging of free radicals and increased antioxidant status.
Introduction
People are encountered everyday with many chemical hazardous compounds such as cigarette smoke, pollutants, toxins, heavy metals, ionizing radiation and drugs. These toxic substances may induce increased oxidative stress and side effects in exposed biological system. The increase in the oxidative stress has been established to be the main reason for genotoxicity and may be the cause of cancer. However, human body is equipped with self-defense mechanisms such as detoxification process through various enzymes, and serious exposure to dangerous chemicals can lead to mutagenic and carcinogenic events. 1,2
Diazinon (DZN) is a commonly used organophosphorous (OP) pesticide (diethoxy-[(2-isoprophyl-6-methyl-4-pyrimidinyl) oxy]-thioxophosphorane). It is a synthetic chemical substance with broad spectrum insecticide activity. 3 Toxic effects of DZN are due to the inhibition of acetylcholinesterase activity, an enzyme needed for proper nervous system function. It has been widely used throughout the world with applications in agriculture and horticulture for controlling insects in crops, ornamentals, lawns, fruit, vegetables and other food products. 4,5 Some reports have been published with respect to DZN and its effects on hematological and biochemical parameters of rat, rabbits and mice. 6 –10 Toxicities of OP insecticide DZN cause adverse effects on many organs. 7 DZN’s mutagenicity studies and its ability to cause genetic damage showed that DZN in fact can damage DNA in human blood cells, in cells from laboratory animals and in bacteria. 11 DZN exposure was found to increase the occurrence of a type of genetic damage called micronuclei (MNs). MN may be induced by strand breaks in DNA due to oxidative stress. 12 The MN test using peripheral blood cells is used to detect chromosome breaks that are derive from DNA damage. 13
Systems that protect the living body from oxidative stress-induced damage include enzymes superoxide dismutase, catalase and glutathione peroxidase (GSH-Px) that removes reactive oxygen species (ROS); proteins ferritin and transferrin that mask the heavy metal ions; antioxidant system including vitamins E and C; enzyme repair systems for damaged DNA; and enzyme systems ubiquitin-proteasome that remove damaged proteins. 14,15 Antioxidant micronutrients are being widely studied for their alleged beneficial properties in the prevention of human diseases, cancer, arthritis and cardiovascular diseases. 16 Selenium (Se) is a structural component of several enzymes with physiological antioxidant properties, including GSH-Px and thioredoxine. 17
Se is an essential dietary trace element that plays an important role in a number of biological processes. 18 There is a long-standing association between Se compounds and cancer chemoprevention. 19 Many experimental studies in animals have demonstrated the ability of Se to prevent carcinogenesis, and epidemiological studies have suggested that a decreased Se status in humans is associated with an increased risk of cancer. 20 A recent widely publicized chemoprevention study has shown that Se supplements can decrease the incidence of certain types of cancer. 19
In the last decade, there is an increasing interest in Se compounds because of their environmental, biological and toxicological importance and, in particular, because of their anticancer activities. 21,22 Se compounds are considered as ‘Janus compounds’, that is, products with a double face, due to their contrasting behavior, which depend on the concentration used. Se in low concentrations may have anticarcinogenic effect, where as in high concentrations, it can be a genotoxic and carcinogenic agent. Se appears to function as an antimutagenic agent, preventing the malignant transformation of normal cells. These protective effects of Se (as co-antioxidant) seem to be primarily associated with its presence in the seleno-enzymes, which are known to protect DNA and other cellular components from oxidative damage. 23
The present study aims at disclosing the capability of DZN in inducing genotoxic effect in rat peripheral blood lymphocyte and the role of Se as anticlastogenic and/or antimutagenic agent against the DNA damage induced by DZN.
Materials and methods
Animals
Male Wistar Albino rats (n = 40) of 5–7 months old, weighing 150–200 g, were purchased from the Pasteur Institute of Iran (North Branch, Amol, Iran) and kept in a good condition at the university animal section and given standard food pellets and water ad libitum. All of the animals were maintained under controlled conditions of light (12/24 h) and temperature (23 ± 1°C). Their use and the experimental protocol used in this study were approved by the Ethical Committee of the medical Sciences, University of Mazandaran.
Experimental design
In the experiment, a total of 40 rats were used. The rats were randomly divided into eight groups (groups 1–8), with five animals in each group: Group 1: normal control; received normal saline (10 ml/kg body weight (b.w.)) intraperitoneally (i.p.) for 30 consecutive days. Group 2: vehicle control, received soybean oil (10 ml/kg b.w.) i.p. for 30 consecutive days. Group 2: positive control; received DZN (20 mg/kg b.w.) in soybean oil (10 ml/kg b.w.) i.p. for 30 consecutive days. Group 3: treated with Se (0.5 mg/kg b.w.) in normal saline (10 ml/kg b.w.) i.p. for 30 consecutive days. Group 4: treated with Se (1 mg/kg b.w.) in normal saline (10 ml/kg b.w.) i.p. for 30 consecutive days. Group 5: treated with Se (2 mg/kg b.w.) in normal saline (10 ml/kg b.w.) i.p. for 30 consecutive days. Group 6: concomitantly treated with Se (0.5 mg/kg b.w.) and DZN (20 mg/kg b.w.) i.p. for 30 consecutive days. Group 7: concomitantly treated with Se (1 mg/kg b.w.) and DZN (20 mg/kg b.w.) i.p. for 30 consecutive days. Group 8: concomitantly treated with Se (2 mg/kg b.w.) and DZN (20 mg/kg b.w.) i.p. for 30 consecutive days.
MNs assay
After 24 h of last injection, 0.5 ml blood of each rats was added to 4.5 ml of RPMI 1640 culture medium (Gibco, NY, USA), which contained a mixture of 20% fetal calf serum, 20 µl/ml phytohemagglutinin (Gibco, NY, USA), 50 U/ml penicillin, 50 µg/ml streptomycin and 2 mM glutamine (Cytochalasin B (Sigma, MO, USA)) at final concentration. All cultures were incubated at 37 ± 1°C in a humidified atmosphere of 5% CO2 and 95% air. (Sigma, MO, USA, final concentration: 6 µl/ml) was added after 21 h of culture. After 44 h of incubation, the cells were collected by centrifugation for 8 min at 1000 r/min, resuspended in 0.075 M cold potassium chloride and immediately fixed in a fixative solution (methanol:acetic acid, 6:1) for three times. The fixed cells were dropped onto clean microscopic slides, air-dried and stained with Giemsa solution. All slides were evaluated at 40× magnification to determine the frequency of MNs in the cytokinesis-blocked binucleated cell with a well-preserved cytoplasm. A typical image of binucleated cells with and without MNs is shown in Figure 1. The criteria for scoring MNs were a diameter between 1/16th and 1/3rd of the main nuclei, non-refractile, not linked to the main nuclei and not overlapping the main nuclei. 24 For each rat, three slides were prepared, and for each slide, a total of 1000 binucleated cells were scored to record the percentage of frequency of MNs.

A typical binucleated lymphocyte with MNs (left) and without MNs (right). MNs: micronuclei
Statistical analysis
For each rat, in each group, the incidence of DZN-induced MNs per 3000 binucleated lymphocyte was recorded. The data are presented as mean ± SD. Data were analyzed with Student’s t test. A p-value of less than 0.05 was considered to be significant.
Results
The data presented in Figure 2 show a significant difference in the percentage of micronucleated binucleated cells in lymphocytes treated with DZN when compared with those in control cells (p < 0.0001). The percentage of MNs in the lymphocytes of rats treated with 20 mg/kg of DZN for 30 consecutive days through i.p. injection was 12.78 ± 0.24%, while the percentage in non-treated control lymphocytes was 0.95 ± 0.12% (Table 1). The frequency of MNs in rats received concomitant treatment with Se at doses of 0.5, 1 and 2 mg/kg and DZN was 4.4 ± 0.36, 7.1 ± 0.36 and 9.2 ± 0.41%, respectively (Table 1). These results were significantly lower than the rats that received DZN alone (p < 0.0001–0.05) (Figure 2). The total micronucleated binucleated cell values were 34, 55, and 72% in the 0.5, 1 and 2 mg/kg doses of Se. It was unusually lower at 0.5 mg/kg compared with those at 1 and 2 mg/kg doses that are in agreement with previous studies. Se alone causes light genotoxicity at high dose of 2 mg/kg in rats without DZN (p < 0.05).
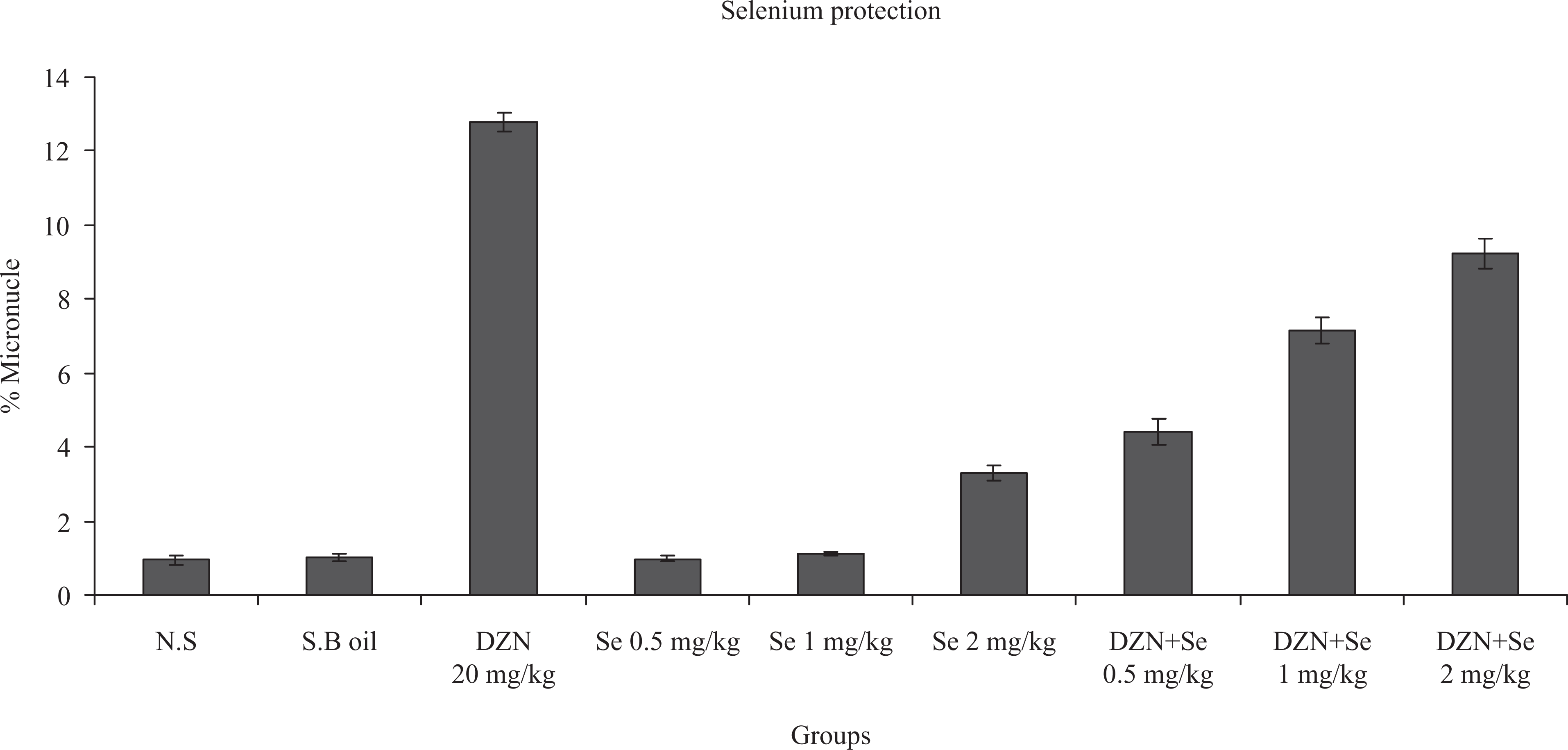
In vivo protection by Se at different doses (milligram per kilogram) against DZN-induced genetic damage in rat blood lymphocyte. The frequency of MN is represented as the percentage of the mean ± SD of five rats. Three slides were prepared for each rat and a total of 3000 binucleated lymphocyte cells were scored to determine the number of MNs for each rat. p < 0.0001: control sample compared with similarly administrated lymphocytes from the blood sample treated with DZN. p < 0.0001: DZN sample compared to DZN + Se 0.5 mg/kg and DZN + Se 1 mg/kg p < 0.05: DZN sample compared to DZN + Se 2 mg/kg and p < 0.05: control sample compared with similarly administrated lymphocytes from the blood sample treated with Se 2 mg/kg. DZN: diazinon; MN micronucleus.
The total number of MNs per 1000 binicleated lymphocytea
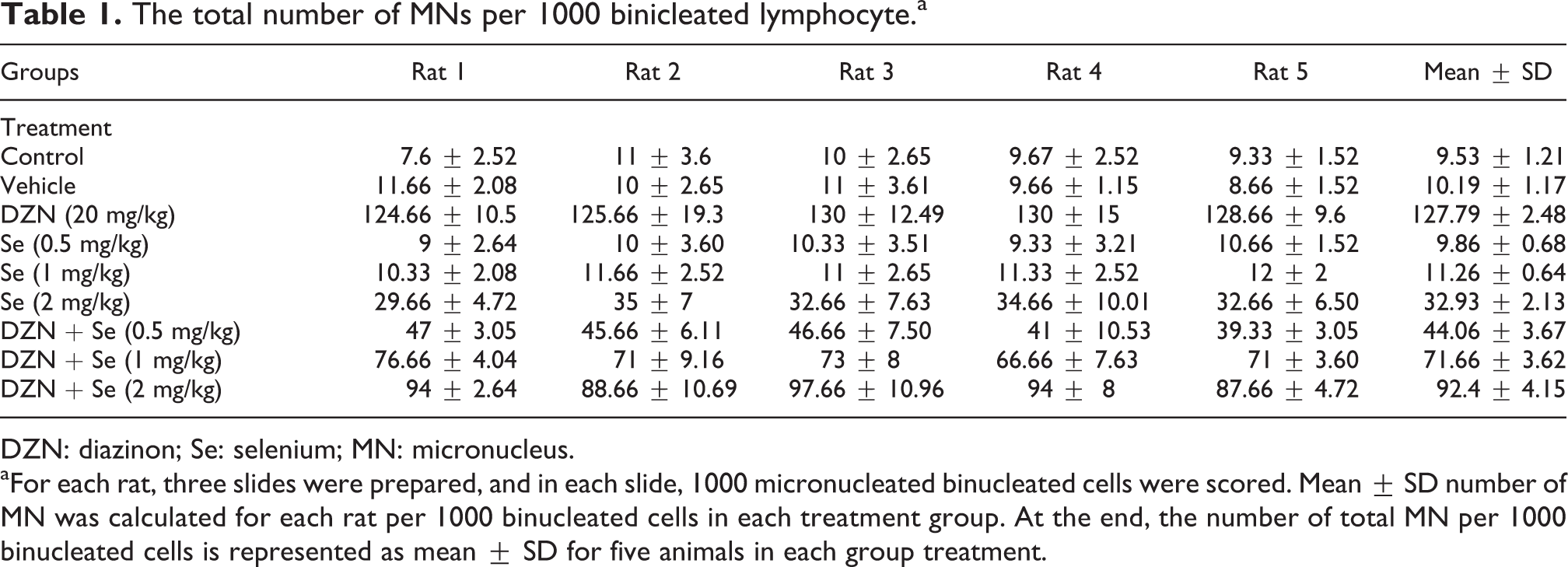
DZN: diazinon; Se: selenium; MN: micronucleus.
aFor each rat, three slides were prepared, and in each slide, 1000 micronucleated binucleated cells were scored. Mean ± SD number of MN was calculated for each rat per 1000 binucleated cells in each treatment group. At the end, the number of total MN per 1000 binucleated cells is represented as mean ± SD for five animals in each group treatment.
Discussion
Our results indicated that Se treatment mitigated the genotoxicity induced by DZN in rat lymphocytes. DZN is one of the most commonly used OP that is used to control a range of crop pests and also as a veterinary ectoparasiticide in many countries including Iran. It degrades easily, but under condition of low temperature, low moisture, high alkalinity and lack of suitable microbiological degraders, it may remain biologically active in soils for 6 months or longer. 25 DZN causes changes in liver enzymes and biochemical indices and swelling of mitochondria in hepatocytes. 26 It also causes toxic effects on blood cells, spleen, thymus and lymph nodes of rats 27 and on other organisms too. 28 Metabolic processes produce free radicals that are highly reactive and unstable and also can cause oxidative damage to the cells and tissues. 29 Increase in the intracellular level of ROS, frequently referred to as oxidative stress, represented a potentially toxic insult, which interact with macromolecules to induce cell membrane dysfunction, lipid peroxidation and DNA damage. 30 The results of many studies confirmed that DZN exposure induced DNA damage. Using MN assay, DZN treatment to human blood cells (lymphocytes and erythrocytes) and skin fibroblasts has been shown to results in a significant increase in the number of MN via clastogenic mode of action, inducing single and double strand breaks on DNA molecule. 31
In the last years, the MN assay has been extensively used to assess the cytogenetic damage induced by chemicals and radiation. This assay after the improvements introduced by Fenech and Morley appears to be sensitive, simple and fast enough to detect agents that induce chromosome damage. 32 This method is now one of the best established in vivo and in vitro cytogenetic assays in the field of genotoxic biomonitoring. 33 In cytokinesis-block MN assay, cells that have completed one nuclear division are blocked from performing cytokinesis using cytochalasin-B and are consequently readily identified by their binucleated appearance. MN are scored in binucleated cells only, which enables reliable comparison of chromosome damage between cell populations that may differ in their cell division. 32
DZN is capable of inducing chromosomal aberrations such as sister chromatid exchanges (SCEs) and MN. In the present study, DNA assay damage was evaluated by MN test. Administration of DZN resulted in DNA damage and induced MN in blood lymphocyte of rats. It is evident that exposure to DZN resulted in DNA damage when compared with control, and administration of Se for 30 consecutive days reduced this DNA damages in rats. Se at a low dose reduced DZN-induced MN as a DNA damage index. These results suggest that Se has a protective effect on DZN-induced genotoxicity.
Many in vivo and in vitro studies have demonstrated the role of Se in protecting against oxidative damage. 34 This protection occurs because Se is a component of important antioxidant enzymes such as GSH-Px. 34 The Se-dependent GSH-Px could detoxify both hydrogen peroxide and lipid hydroperoxides. 35,36 It has been proposed that Se exerts its anticarcinogenic actions by multiple mechanisms. Besides the selective killing of transformed cells mentioned above, Se compounds appear to function as antimutagenic agents, preventing the initiation and progression steps in the malignant transformation of normal cells. These preventive effects of Se may be primarily associated with its presence in GSH-Pxs, which are known to protect DNA and other cellular components from damage by free radicals. 37
Research comprising clinical trials, animal experiments and in vitro studies concluded that Se compounds are capable of inhibiting carcinogen-induced covalent DNA-adduct formation, DNA oxidative damage, DNA methylation, induction of MNs, chromosomal aberrations and cancer. 38 Se also reduced the genetic damage (SCEs and MN) induced by chemical pollutants, for example, carbon tetrachloride in animal cells. 39,40 Moreover, it was found that sodium selenite and ebselen (2-phenly-1, 2-benzisoselenazol-3(2H)-one) reduced the genotoxic effect of azoxymethane, methylmethansulfonate and hydrogen peroxide in Chinese V79 hamster cells. Sodium selenite is also involved in the detoxification of free radicals by increased activities of GSH-Px and catalase. 41 On the other hand, it was reported that Se is able to prevent erythrocyte membrane damage induced by T-2 toxin. 42
Figure 2 shows that Se had a protective effect against DZN-induced DNA damage especially at a low dose. Based on the results, it is clear that DZN might be inducing DNA damage by increased generation of reactive free radicals which are scavenged by Se. Administration of DZN in combination with Se showed decrease in the percentage of MN when compared with DZN-treated rats. It is evident that Se might be reducing the MN formation by scavenging the DNA damaging free radicals generated following DZN exposure. Another possible mechanism of Se might involve selective removal of cells with DNA damage by apoptosis. These results demonstrate that administration of Se along with DZN decreased the DNA damage and thus protected the cells against genotoxic effect of DZN.
Conclusion
Our findings demonstrate that DZN is genotoxic as assessed by MN assays and had adverse effects on peripheral blood lymphocytes. Se, on the other hand, was observed to repair the genotoxicity and reduced the frequency of MN induced by DZN at a low dose. It can be concluded that Se, as an antioxidant, has protective effect against DZN adverse effects by inactivating (scavenging) free radicals generated following exposure to DZN and daily supplement of Se might be beneficial to pesticides exposed populations.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was approved and supported by a grant from the Mazandaran University of Medical Sciences, Sari, Iran.
