Abstract
Duloxetine is a serotonin and norepinephrine reuptake inhibitor, which is mainly used to treat depression. This retrospective study describes the demographic and clinical effects of duloxetine ingestions reported to the National Poison Data System (NPDS). NPDS data were searched for duloxetine exposures between 2004 and 2010. A total of 11,373 patients were included and exposures were divided into three groups of ages ≤6 years old, 7–12 years and >12 years. Neurological clinical effects occurred in 6.1% of the patients aged ≤6 years, 13.0% of the patients aged 7–12 years and 24.6% of the patients aged >12 years. Cardiovascular effects occurred in 1.4% of the patients aged ≤6 years old, 2.5% of the patients aged 7–12 years and 11.6% of the patients aged >12 years. Gastrointestinal effects occurred in 4.1% of the patients aged ≤6 years old, 16.6% of the patients aged 7–12 years and 13.8% of the patients aged >12 years. Tachycardia, nausea, vomiting, agitation/irritability, dizziness/vertigo and drowsiness were among the most common clinical effects in all three groups. Overall, 61.4% of the patients aged ≤6 years and 77.5% of the patients aged 7–12 years were managed in a non–health care facility, while 55.8% of the patients aged >12 years were referred to or already in a health care facility. We conclude that the majority of ingestions are benign in both pediatrics and adults. Most symptomatic patients have neurologic, gastrointestinal and cardiovascular effects. Most pediatric patients will be able to be managed in a non–health care facility.
Introduction
Duloxetine (Cymbalta, Eli Lilly, Inc., Indianapolis, Indiana, USA) is a serotonin and norepinephrine reuptake inhibitor, which was approved by the United States Food and Drug Administration (FDA) in August 2004. It was initially approved for use as an oral medication in the adult population for the treatment of major depressive disorder. Since then, the FDA has expanded its approved indications to include peripheral neuropathic pain associated with diabetes (September 2004), generalized anxiety disorder (February 2007) and fibromyalgia (June 2008). It is also marketed in Europe for the treatment of major depression, diabetic peripheral neuropathy and female stress urinary incontinence.
The most common adverse drug reactions (ADRs) reported in clinical trials/post-marketing data include nausea, dry mouth, headache, diarrhea, dizziness and fatigue. 1 –7 Despite many clinical trials with therapeutic dosing, the safety profile of single-agent duloxetine unintentional and intentional ingestions has never been studied systematically.
The objective for this study is to describe the demographics and clinical outcomes of single-agent duloxetine ingestions reported to the National Poison Data System (NPDS) of the American Association of Poison Control Centers (AAPCC).
Methods
This is a retrospective study describing single agent oral ingestions of duloxetine in children and adults reported to NPDS between January 2004 and December 2010. The NPDS is managed by the AAPCC and contains data from all poison exposure and information phone calls from laypersons and health care providers to all poison control centers across the United States. All reports of ADRs and cases with unknown age were excluded. The study was approved by our local institutional review board and informed consent was waived.
NPDS data are derived from a collection of case records documenting all calls made to the U.S. poison control centers. Data are collected and coded by trained pharmacists and nurse specialists in poison information (SPIs). Extracted variables were age, gender, year of exposure, exposed substance, amount ingested, clinical effects, patient disposition, treatment and poison control center outcome designation. We defined the severity of health effect in accordance with AAPCC designations. These outcome designations are defined by the AAPCC as follows: (i) no effect; (ii) not followed and judged as nontoxic exposure; (iii) minimal clinical effect possible, not followed; (iv) minor (the patient developed some symptoms and signs as a result of exposure); (v) moderate (the patient exhibited signs or symptoms as a result of the exposure that were more pronounced, more prolonged or more systemic in nature than minor symptoms); (vi) major (the patient exhibited signs or symptoms as a result of the exposure that were life-threatening or resulted in significant residual disability or disfigurement); (vii) death; (viii) unable to follow-up, potentially toxic effects and (ix) unrelated, probably not responsible. Specific definitions of each of these outcomes are available at the AAPCC website. 8
The NPDS data set was searched for all duloxetine exposures between January 1, 2004, and December 31, 2010. Cases were identified using NPDS codes: 6569044 (Cymbalta®), 6569052 (Cymbalta®), 7041744 (Cymbalta®), 6569060 (Cymbalta®) and 6047280 (Duloxetine).
Data were analyzed using SAS version 9.2 (SAS Inc., Cary, North Carolina, USA). Exposures were analyzed with descriptive statistics and confidence intervals were calculated. For the purposes of our study we have divided these exposures into three different groups of ages: ≤6 years old, 7–12 years old and >12 years old as the circumstances under which these ingestions occur (intentional vs. unintentional) and dose ingested can vary depending on the age group studied. 9 All the clinical events recorded in NPDS are coded as related to the ingestion or not. We included all of the clinical events whether they were coded as related or not in the analysis. We also examined how many of the patients in our study had clinical effects of agitation/irritability, tremor and diaphoresis, which are the three components of Hunter Serotonin Toxicity Criteria (a validated set of criteria used to diagnose serotonin syndrome) that are coded by NPDS. Other components of Hunter Serotonin Toxicity Criteria that are not coded by NPDS include spontaneous clonus, inducible clonus, ocular clonus, hyperreflexia, hypertonicity and temperature >38°C. Specific decision rules and combinations of these clinical features are used to determine the presence of serotonin toxicity. 10
Results
Between January 2004 and December 2010, there were 29,346 cases of duloxetine exposures reported to the U.S. poison control centers. Overall, 74 patients were of unknown age, 17,016 patients had multidrug ingestions and 883 patients were coded as ADRs and were all subsequently excluded from the study. A total of 11,373 patients were included in the study. The number of overall reported exposures reached its peak in 2008 in all three age groups and the majority of exposures in all three age groups occurred between 2006 and 2010 (Figure 1). Information regarding reported ingestions by gender is shown in Table 1.

Duloxetine exposures by year.
Duloxetine ingestions in the three age groups divided by gender.
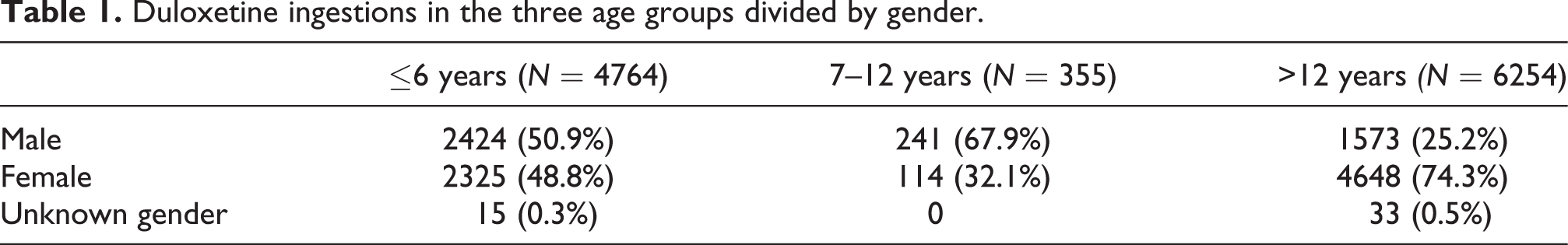
Age ≤6 years
From January 2004 to December 2010, there were 4764 cases of single-agent duloxetine exposures that were not ADRs in patients aged ≤6 years reported to the U.S. poison control centers. A total of 2424 (50.9%) of ingestions occurred in males. Overall, 4412 (92.6%) of cases were classified as no effect, minor effect, minimal effect and not followed but judged to have nontoxic exposure. Moderate outcomes occurred in 49 patients (1.0%), while 3 cases (0.06%) were judged to have a major outcome. A total of 251 cases (5.3%) were judged to be potentially toxic but unable to be followed and 49 (1.0%) were judged to have unrelated effects with the exposure deemed to be not likely responsible for the observed effect.
Clinical effects were documented in 740 (15.5%) of all cases. Of all patients studied, 291 (6.1%) had clinical effects that were neurologic. The most frequently encountered neurological clinical effects were drowsiness/lethargy in 216 patients (4.5%), agitation/irritability in 72 patients (1.5%) and ataxia in 12 patients (0.3%). A total of 194 patients (4.1%) had clinical effects that were gastrointestinal. The most common gastrointestinal clinical effects were vomiting in 161 patients (3.4%), nausea in 24 patients (0.5%) and abdominal pain in 10 patients (0.2%). Overall, 67 patients (1.4%) had clinical effects that were cardiovascular with the most frequent cardiovascular effects being tachycardia in 55 patients (1.2%) and hypertension in 20 patients (0.4%). Only 1 patient had multiple and discrete seizures and there were no patients with single seizure or status epilepticus. The most common clinical events in all age groups are listed in Table 2. There were 13 clinical effects in patients with major outcomes. These clinical events included vomiting, agitation/irritability, creatine phosphokinase elevations, fasciculations, fever/hyperthermia, hallucinations/delusions, muscle rigidity, nausea, nystagmus, seizures and tachycardia. Information regarding the location of management for all patients is listed in Table 4. A single dose of activated charcoal was administered in 747 (15.7%) of the patients, while 11 patients (0.2%) received multiple doses of activated charcoal.
Most common clinical events in all age groups.

Age 7–12 years
There were 355 cases of single-agent, non-ADR duloxetine exposures in patients aged 7–12 years reported to the U.S. poison control centers between January 2004 and December 2010. Overall, 241 (67.9%) ingestions occurred in males. A total of 326 (91.8%) cases were classified as no effect, minor effect, minimal effect and not followed but judged to have nontoxic exposure. Moderate outcomes occurred in 4 (1.1%) patients. None of the patients were judged to have a major outcome. Overall, 16 (4.5%) cases were judged to be potentially toxic but unable to be followed. A total of 9 (2.5%) were judged to have unrelated effects with the exposure deemed to be not likely responsible for the effect.
Clinical effects were documented in 129 (36.3%) cases. Of all patients studied, 46 (13.0%) had clinical effects that were neurologic. The most frequently occurring neurologic clinical effects were drowsiness/lethargy in 28 patients (7.9%), dizziness/vertigo in 10 patients (2.8%), agitation/irritability in 6 patients (1.7%) and headache in 6 patients (1.7%). Gastrointestinal effects occurred in 59 (16.6%) patients with the most common gastrointestinal effects being nausea in 29 patients (8.2%) and vomiting in 28 patients (7.9%). A total of 9 patients (2.5%) had clinical effects that were cardiovascular with the most frequent cardiovascular effect being tachycardia in 6 patients (1.7%). Seizures did not occur in any of the patients. Single-dose activated charcoal was administered in 19 (5.4%) patients.
Age >12 years
A total of 6254 cases of single-agent oral duloxetine exposures that were not coded as ADRs were reported to the U.S. poison control centers in patients aged >12 years. Totally, 4648 (74.3%) cases occurred in females. The median age was 33 years (inter quartile range: 20–48 years). In 3110 cases (49.7%), the ingestion was intentional and in 2985 cases (47.7%), the ingestion was unintentional. The intention was unknown in 159 (2.5%) cases. Overall, 4904 (78.4%) of cases were classified as no effect, minor effect, minimal effect and not followed but judged to have nontoxic exposure. A total of 454 cases (7.3%) were classified as moderate outcomes. Major outcomes occurred in 29 (0.5%) patients. Totally, 699 (11.2%) cases were judged to be potentially toxic but unable to be followed. A total of 168 cases (2.7%) were judged to have unrelated effects with the exposure deemed to not likely be responsible for the effect. One death was reported.
Clinical effects were documented in 4782 (76.5%) cases. Of all the patients studied, 1541 (24.6%) had clinical effects that were neurologic, 862 (13.8%) were gastrointestinal and 727 (11.6%) were cardiovascular. The most frequent neurological clinical effects were drowsiness/lethargy in 803 patients (12.8%), dizziness/vertigo in 291 patients (4.7%) and agitation/irritability in 243 patients (3.9%). The most common gastrointestinal clinical effects were 463 (7.4%) nausea and 453 (7.2%) vomiting. The most frequently seen cardiovascular effects were 516 (8.3%) tachycardia and 239 (3.8%) hypertension. A single seizure was reported in 14 (0.2%) patients and multiple/discrete seizures were reported in 18 (0.1%) patients. One patient had status epilepticus. The top clinical effects in patients with major outcomes in patients aged >12 years included tachycardia, coma, drowsiness/lethargy, hypertension, agitation/irritability and respiratory depression. A total of 1069 (17.1%) patients received a single dose of activated charcoal.
There were a number of notable serious clinical effects, including cardiac arrest, conduction disturbance, dysrhythmia (other), dysrhythmia (ventricular tachycardia/ventricular fibrillation), coma, seizure (single), seizures (multiple/discrete), seizures (status epilepticus), respiratory arrest and death, that affected a small subset of patients that are listed in Table 3. None of these effects occurred in the 7–12 age group. The majority of patients aged ≤6 years and those aged 7–12 years were managed on site at a non–health care facility, a site where a patient is not evaluated by a physician. Table 4 shows the location of management of the three different subgroups of patients.
Life-threatening effects/death reported to the U.S. poison centers following single-agent duloxetine exposure.

CI: confidence interval.
Location of management reported to the U.S. poison centers following single-agent duloxetine exposure.

95% CI: 95% confidence interval.
The incidence of patients with clinical effects of agitation/irritability, tremor and diaphoresis, which are components of the Hunter Serotonin Toxicity Criteria that are coded by NPDS are shown in Table 5. A total of 291 (2.56%) patients were agitated/irritable, 88 (0.77%) had tremor, 68 (0.60%) had diaphoresis and 5 (.04%) patients had all three components. We also attempted to look at dosing data but much of that information was missing.
Incidence of Hunter serotonin toxicity criteria components.a

aExcludes subjects with unknown age. Rows for each set of symptoms are mutually exclusive.
Discussion
The number of overall reported exposures reached its peak in 2008, which is also the year its indications were expanded to include fibromyalgia. The majority of exposures occurred between 2006 and 2010. The majority of duloxetine ingestions in all of the three subgroups we studied had favorable medical outcomes. Most patients in the ≤6 years and 7–12 age groups were also able to be managed on site, while the majority of those aged >12 years were referred to or already in a health care facility. Since its introduction in the U.S. market, exposures to duloxetine have steadily increased. Ingestions were fairly equally divided among both genders in the ≤6 years age group, while the majority of ingestions in the 7–12 years group occurred in males and the majority of events in the >12 years group occurred in females. Most patients did not receive activated charcoal.
The most common clinical effects in all of the three age subgroups were very similar to each other. The top 10 most common clinical effects in all of the three subgroups of patients included tachycardia, nausea, vomiting, agitation/irritability, drowsiness/lethargy and dizziness/vertigo. There were no major outcomes in the 7–12 age group and were minimal in the ≤6 years age group.
In those patients that became ill, there were a number of clinical effects that are linked to greater morbidity/mortality including seizures, coma, conduction disturbances, dysrhythmias, cardiac arrest and respiratory compromise. Most of these clinical effects occurred in the >12 years subgroup. There was also one death reported in the >12 years age group. These clinical effects may be a function of dose ingested and patient comorbidities but these associations were unable to be assessed.
The incidence of seizures in our study was lower than what is reported with overdose of other commonly used antidepressants. 11 –16 By comparison, a poison center-based review of 1313 adult and pediatric patients with tricyclic antidepressant poisoning found that overall 3% of all tricyclic antidepressant ingestions had convulsions; the rate was particularly high for desipramine where 18% of patients had seizures. 11 Bupropion is a commonly used antidepressant that has been reported to cause seizures with therapeutic use as well as overdose. One previous poison center study that examined ingestions of bupropion in 117 children and adults reported seizure rates of 31.6%, while a pediatric bupropion poison center study of 5964 cases found a seizure rate of 1.4%. 12,13 Another retrospective study of 469 cases from a single toxicology unit found an overall seizure rate of 1.9% for selective serotonin reuptake inhibitor (SSRIs) overall, while a prospective cohort study of 538 hospital admissions found a seizure rate of 14% in all venlafaxine overdoses. 14,15 Citalopram, a SSRI known to cause seizures, was found to have a seizure rate of 13.5% in a retrospective poison center study of 316 citalopram cases. 16 We found that only 1 patient in the ≤6 years group had seizures (multiple/discrete). In the >12 years age group, 14 (0.2%) patients had a single seizure and 8 patients (0.1%) had multiple/discrete seizures.
A second common concern following the overdose of other commonly used antidepressants is cardiotoxicity, especially abnormal ventricular repolarization. Conduction abnormalities such as QT prolongation are another effect that has been described in overdose with bupropion and citalopram. 17 –21 QT prolongation is unable to be assessed from NPDS data but the clinical event of conduction disturbance are collected. There was 1 patient in the ≤6 years age group that had a conduction disturbance and 28 (0.4%) patients in the >12 years age group that had a conduction disturbance reported. Our study was not adequately designed to examine the clinical effect of QT prolongation in duloxetine overdose. Although our study suggests that QT prolongation is not common, this manifestation is not always reported to poison centers and further studies are needed to better assess the prevalence of QT prolongation in duloxetine overdoses. However, it is likely that the rate of clinically significant dysrhythmias (which are generally well captured in poison center data) appears to be low.
The risk associated with the development of serotonin syndrome after ingestion of a single tablet is of particular importance especially in light of our data that shows 41% of all reported exposures occurred in children ≤6 years old, who tend to have accidental single-pill ingestions. 9 Gelener et al. has reported a case of serotonin syndrome in a 29-year-old woman after self-administration of a single duloxetine 30 mg tablet. 22 Hunter Serotonin Toxicity criteria is a validated instrument used to diagnose serotonin syndrome. NPDS data only code for 3/9 of the components of Hunter Serotonin Toxicity criteria. These criteria include agitation/irritability, tremors and diaphoresis. 10 Even in our study, which included a large subset of patients with intentional ingestions, the number of patients that exhibited all three components of Hunter Serotonin criteria that are coded by NPDS was minimal. Although we could not fully assess for serotonin syndrome in our study, the incidence of co-occurrence of the three clinical manifestations suggest that the likelihood of serotonin syndrome after duloxetine ingestions alone may be low.
Strengths of our study include the ability to collect data reported to all the U.S. poison centers over a 6-year span and the large sample size of our study population. Our study also has limitations. As with all studies involving NPDS data, there may be a substantial amount of miscoding and information that is missing. Information is typically reported by telephone in a typically strained environment and laboratory confirmation of exposure is not available. There is also reporting bias that would be expected and the potential of underreporting of nontoxic exposures. This impacted our evaluation as we were not able to collect all the data points we had initially planned to collect such as dosing data.
We conclude that the majority of ingestions are generally benign in both the pediatric population and in the adults. Most pediatric patients will be able to be managed on site in a non-health care facility. The most common clinical effects include drowsiness/lethargy, agitation/irritability, dizziness/vertigo, tachycardia, nausea and vomiting. There is a subgroup of patients with effects linked to greater morbidity/mortality and this may be a function of dose ingested and patient comorbidities but this is unable to be assessed from our data. The incidence of seizures in single-agent duloxetine ingestions is less than what is reported with other commonly used antidepressants such as tricyclics, bupropion, venlafaxine and citalopram. Future prospective studies may be needed to more fully characterize the demographics and clinical effects of duloxetine ingestions.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
The research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
