Abstract
This article describes antioxidative and cytoprotective property of andrographolide, a major active component of the plant Andrographis paniculata (A. paniculata). High yields (2.7%) of andrographolide was isolated from the aerial parts of this plant via silica column chromatography. The purity of the compound was determined by high-performance thin-layer chromatography (HPTLC) and reversed phase high-performance liquid chromatography (HPLC) analysis. The structure was elucidated using techniques such as UV-visible spectrophotometry, elemental analysis, Fourier transform infrared (FT-IR), 1H nuclear magnetic resonance (1H NMR), 13C nuclear magnetic resonance (13C NMR) and mass spectral analysis and the data obtained were comparable with reported results. It was observed that andrographolide exhibited significant antioxidative property (IC50 = 3.2 µg/ml) by its ability to scavenge a stable free radical 1,1-diphenyl-2-picrylhydrazyl (DPPH) as compared to known antioxidants like ascorbic acid, butylated hydroxy toluene (BHT) and the plant extract. The cytoprotective role of andrographolide against carbon tetrachloride (CCl4) toxicity in human hepatoma HepG2 cell line was assessed using trypan blue exclusion test, 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay, by estimation of various leakage enzymes and by measuring the glutathione levels. The recovery obtained for andrographolide treatment in the presence of CCl4 was two-fold compared to A. paniculata extract for all other related biochemical parameters investigated. The results of the study indicate that andrographolide is a potent inhibitor of CCl4-mediated lipid peroxidation.
Introduction
Antioxidants play a significant role in food processing industries. Traditionally these were utilized to prevent oxidative rancidity which may lead to unwanted changes in the flavor, texture, color, destruction of essential fatty acids and production of toxic end products. Many synthetic antioxidants are commercially available, but their use is limited because they enforce large number of side effects. 1,2 ‘Oxidative stress’ defined as imbalance between production of reactive oxygen species (ROS) and antioxidative defense mechanism is also encountered in number of pathological conditions and is a serious threat to human health. Mechanism of lipid peroxidation is of common concern to both food industrial personnel and health care practitioners. 3,4 Recently natural antioxidants derived from plants have attracted the attention of food manufacturers, because of their negligible side effects and potential to inhibit rancidity, along with additional capacity to overcome pathological conditions. Their use is recommended for controlling unwanted deleterious effects in food. 5,6 In addition their consumption may influence the unnecessary outbreak of pathological distress. The never ending quest to unravel nature’s resources by humans has resulted in discovery and subsequent utilization of many medicinal plants. The search is on for plant-based antioxidants that retard the process of oxidative damage but with low cytotoxicity.
Andrographis paniculata (family Acanthaceae) also known as kalmegh in Hindi is a well-known herb distributed throughout India, South East Asia and Southern part of China. A. paniculata has been used as a medicinal food, and it is accredited with health-promoting and disease-preventing properties. In India it has considerable medical reputation, being part of 26 ayurvedic formulations as listed in Indian pharmacopoeia. 7 Number of extensive and in-depth research and clinical trials carried out in the past has proven the broad spectrum of pharmacological activities exerted by this herb. It has been reported to have antidiabetic, 8 antiedemic and antioxidative, 9 antimicrobial, 10 antihepatotoxic, 11 immunostimulatory activity 12 and as an inhibitor of common cold virus. 13 Tripathi et al. 14 have reported that this plant has an edge over synthetic preservatives and possess significant potential to serve the food industry in the future.
The predominant active compound present in A. Paniculata, is a diterpenoid lactone andrographolide. 15 Significant effort toward isolation and characterization of this active component has been well documented in the literature. 16 –19 At the same time, notable work in the area of method development and validation has been reported for its determination from A. Paniculata by high-performance liquid chromatography (HPLC), 15,20 –22 and liquid chromatography–mass spectroscopy (LC-MS). 23
Carbon tetrachloride (CCl4) is conventionally employed to induce liver toxicity in vitro/in vivo models. 24 This model is very much in use for testing plant-based medications for their antioxidative and liver-protecting properties. This experimental hepatotoxin initiates lipid peroxidation detected as increase in malondialdehyde (MDA) concentration 25,26 via free radicals causing intracellular tissue damage. 27 The present study was an attempt to prove the cytoprotective ability of andrographolide against CCl4-induced oxidative damage in human hepatoma HepG2 cells. The antioxidative property of this compound was compared with known antioxidants like ascorbic acid and butylated hydroxy toluene (BHT) by means of 1, 1-diphenyl-2-picrylhydrazyl (DPPH) free radical scavenging assay.
Materials and methods
Chemicals
HPLC grade CCl4 (Cas No: 56-23-5), acetonitrile, ethyl acetate, chloroform, hexane and methanol were obtained from Merck Specialties Pvt. Ltd. (Mumbai, India). All other chemicals used were purchased from Sigma-Aldrich (St. Louis, MO, USA), unless specified. Andrographolide standard was procured from Southern Petrochemical Industries Corporation (SPIC), Chennai. Milli-Q water purification system from Millipore (Bangalore, India) was used to prepare water for the entire analysis.
Plant material
The aerial parts of A. paniculata were collected from botanical garden of Gujarat University, Ahmedabad, Gujarat, India. The plant material was authenticated by National Institute of Science Communication and Information Resources (Ref: NISCAIR/ RHMD/consult/-2007-08/938/122), New Delhi, India.
Isolation and purification of andrographolide from A. Paniculata
The aerial parts of A. Paniculata were dried in shade, pulverized (100 g) and extracted with ethanol:water (50:50, v/v, 3 × 1,000 mL) at room temperature. The solvent was removed by a rotary evaporator under reduced pressure at 40°C. The remaining solid residue (11.0 g) was mixed with silica gel (25 g) to form an admixture and chromatographed over silica gel column (500 mm length × 20 mm diameter, 63–200 μ particle size, 70–230 mesh size). Elution of analytes in the extract was carried out under gradient conditions in the following sequence, hexane–ethyl acetate (400 ml; 80:20, v/v) → hexane–ethyl acetate (400 ml; 60:40, v/v) → hexane–ethyl acetate (500 ml; 50:50, v/v) → hexane–ethyl acetate (700 ml; 25:75, v/v) → ethyl acetate (400 ml) → ethyl acetate–methanol (600 ml; 95:5) → ethyl acetate–methanol (500 ml; 90:10 v/v). Thin-layer chromatography (TLC) of the eluates (10 ml) was done using methanol:chloroform (15:85, v/v) as the mobile phase. The fractions (241–300) collected were combined, re-chromatographed and the solvent evaporated to give 3.5 g of crude andrographolide. The product was purified by crystallization (3 times) from ethanol to afford pure andrographolide (yield, 2.7%).
Instrumentation and conditions
Melting points were taken on a Toshniwal melting point apparatus (Mumbai, India) and are uncorrected. Elemental analysis was carried out on Heraeus Carlo Erba 1108 elemental analyzer (Milan, Italy). UV absorption studies were conducted on a Varian Cary 400 UV-Visible spectrophotometer (Palo Alto, CA, USA) with 10 mm matched quartz cells. Fourier transform infrared (FT-IR) spectra were recorded on Bruker Tensor 27 Infrared Spectrophotometer (Billerica, MA, USA) as KBr pellets. TLC was performed on precoated silica gel 60 F254 plates from E. Merck (Darmstadt, Germany) with a Linomat V band spotter from CAMAG (Switzerland). The plate was developed to a height of 10 cm with 20 ml of mobile phase consisting of methanol:chloroform (15:85, v/v) using Camag twin trough glass tank. Band scanning was done with CAMAG TLC scanner 3 using win-CATS software at 254 nm. 1 H nuclear magnetic resonance ( 1 H NMR; 500.13 MHz) and 13 C nuclear magnetic resonance ( 13 C NMR; 125.75 MHz) spectra were recorded on Bruker DPX-500 AVANCE in CD3OD with tetramethylsilane as internal standard. A Shimadzu LC-VP HPLC-UV system (Kyoto, Japan) consisting of LC-10AD prominence pump, SIL-HTc autosampler, CTO 10 ASvp column oven and a DGU-14A degasser was used for setting the reverse-phase liquid chromatographic conditions. Chromatographic separation was achieved under isocratic conditions on a Phenomenex analytical column, type Gemini C18 (250 mm × 4.6 mm, 5 μm particle size) analytical column, maintained at 40°C in a column oven. The mobile phase consisting of acetonitrile:water (60:40, v/v) was filtered through 0.2 µ membrane filter and pumped at a flow rate of 0.5 mL/min. The injection volume was kept at 20 µl and the wavelength of detector was set at 215 nm. Mass measurements were done on a JEOL GCmate II GC-MS (Peabody, MA, USA) bench top double-focusing magnetic sector mass spectrometer operating in electron ionization (EI) mode with TSS-20001 software.
Structural data for andrographolide
Color: white crystals, mp: 238°C–239°C (uncorrected). UV λmax/nm (methanol): 228 (molar absorptivity, log ∊ 4.1); IR ν/cm−1 (KBr): 3,398 (O-H stretch), 2,958, 2,927, 2,868, 2,849 (C-H stretch), 1,727 (-COOR stretch, α, β-unsaturated γ-lactone), 1,675, 1,464 (C=C stretch), 907 (exocyclic methylene). 1 H NMR (500 MHz, CD3OD, TMS): δ 6.85 [12-H, t, J = 6.2, 2.0], 5.01 [14-H, t, J = 5.4], 4.85, 4.75 [17-H, s], 4.4-4.5 [15-H, dd, 10.1, 2.6], 3.6 [3-H,19-H, m], 2.69 [11-H, t, J= 6.4], 2.05 [H-9, m], 1.85 [H-6,7, m], 1.3-1.4 [H-1,2, m], 1.24 [18-H, s], 0.65 [20-H, s]. 13 C NMR (125 MHz, CD3OD) δ 171.0 [C-16], 148.0 [C-8], 147.2 [C-12], 130.1 [C-13], 108.6 [C-17], 79.3 [C-3], 75.5 [C-15], 65.8 [C-14], 65.3 [C-19], 56.1 [C-9], 55.1 [C-5], 43.5 [C-4], 39.3 [C-10], 37.9 [C-7], 37.1 [C-1], 28.7 [C-2], 24.9 [C-11], 23.9 [C-6], 23.6 [C-18], 15.0 [C-20]. EI-MS, m/z (relative intensity, %): [M]+ 350 (15), 332 (25), 314 (68), 302 (91), 274 (95), 257 (65), 187 (26), 119 (100), 105 (75), 91(60), 79 (73). Anal. calcd. for C20H30O5: C, 68.54%; H, 8.65%, Found: C, 68.68%; H, 8.56%.
DPPH radical scavenging assay
The antioxidative ability of the ascorbic acid, BHT, plant extract and andrographolide was measured using DPPH, a stable free radical. 28 The reaction mixture comprised 0.1 ml DPPH (1 mM), 0.8 ml of ethanol (99%) and 0.1 ml of ascorbic acid (2.5–50 µmol/ml)/BHT (2.5–50 µmol/ml)/A. paniculata extract (50–500 µg/ml)/andrographolide (2.5–25 µmol/ml). The solution was left to stand for 30 min in dark. The decrease in absorbance at 517 nm was noted to measure the scavenging activity and was compared with DPPH control. The free radical scavenging property of ascorbic acid, BHT, tested plant extract and isolate were calculated and expressed as IC50, which is defined as the concentration required for inhibition of DPPH radical by 50%.
Cell line and culture
Cultures of Human hepatoma cell line HepG2 were obtained from ATCC, USA and grown on Eagle’s minimum essential medium (EMEM, GIBCO, USA). It was supplemented with 10% (v/v) heat-inactivated fetal calf serum, 2 mM
Treatment with different agents
Cells were plated in 24-well plate (3 × 104 cells/well/ml of EMEM) and incubated at 37°C, under 5% CO2 and incubated for 48 h to attain confluency. Then the cells were treated with 0.5 ml each of 0.4% (v/v) CCl4 dissolved in 0.25% DMSO in serum-free medium; 0.4% (v/v) CCl4 + A. paniculata extract (200, 400, 600 µg/mL); 0.4% (v/v) CCl4 + andrographolide [10 (3.5 µg/ml), 20 (7.0 µg/ml), 30 (10.5 µg/ml) µmol] for a period of 24 h.
Trypan blue exclusion test
Trypan blue exclusion assay was carried out according to a previously reported method with minor modifications.
30
After 24 h of treatment the cells were trypsinized from culture plates, pooled with floating cells from medium and centrifuged at 1000g for 10 min at 4°C. It was resuspended in 1 mL phosphate-buffered saline (PBS) and 0.1 ml of 0.2% trypan blue was added. The number of live cells was counted using hemocytometer under a light microscope. The results were expressed as percentage of live cells compared to control using the following formula
MTT assay
The ability of the viable cells to reduce MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide) was tested using this assay.
31
After 24 h of treatment as above with various agents, 100 µl of MTT (5 mg/mL) was added to each well and the cells were incubated at 37°C in dark. The medium was removed after 4 h and formazan crystals were dissolved in 100 µl of DMSO. Absorbance was assessed using a microplate reader at 570/630 nm. The percentage viability of cells was calculated by the formula
Lactate dehydrogenase (LDH), alanine transaminase (ALT) and aspartate transaminase (AST) leakage
After 24 h of treatment with various additives, the culture medium and cells were collected separately. The transaminases activities in medium (ALT and AST) was measured by International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) method as kinetic reaction at 340 nm using kits procured from Agappe Diagnostics, Ernakulam, Kerala, India. The cells were suspended in 0.5 ml PBS and sonicated for 5 s in an ice bath. The LDH activity in 0.2 ml of medium and cell lysate was estimated based on Scandinavian Committee on Enzymes (SCE) kinetic method using the kit obtained from the same company. The cytotoxicity index based on the LDH assay was expressed as the ratio of LDH medium/LDH lysate+medium × 100%.
Lipid peroxidation and glutathione levels
The lipid peroxidation (LPO) and GSH levels were measured using standard methods. 30 HepG2 cells were seeded in 24-well plates at a concentration of 3 × 104 /ml EMEM per well and treated with different agents as mentioned above for 24 h. The cells were scraped and harvested by trypsinization and resuspended in 0.5 ml PBS and sonicated for 10 s. The lipid peroxidation was quantified by estimating the levels of MDA by adding 0.5 mL of trichloroacetic acid (TCA) thiobarbituric acid (TBA) reagent and subsequent heating at 100°C for 1 h. The absorbance was measured at 535 nm and the formation of TBA reactive substances was calculated using a molar extinction coefficient of 1.56 × 105 M−1cm−1.
The glutathione content was estimated by adding 0.5 ml of 10% TCA to cells resuspended in 0.5 ml PBS. After centrifugation the supernatant was mixed with buffer A (0.1 M sodium phosphate, 5 μM EDTA, 0.6 mM 5,5'-dithio-bis 2-nitrobenzoic acid, 0.2 mM NADPH) and buffer B (10 U/mL glutathione reductase in 0.1 M sodium phosphate buffer) and absorbance was read at 412 nm. The glutathione levels in these cells were measured by plotting the values in a standard graph constructed using reduced glutathione as the internal standard. The protein content was also determined using bovine serum albumin as standard. 32
Percentage recovery
The percentage recovery
33
seen after A. paniculata extract/andrographolide treatment along with CCl4 in three different concentrations was calculated from the mean value of the respective groups using the formula
Statistical analysis
The result of the DPPH free radical assay (n = 3) was expressed as mean ± SEM. The IC50 values of ascorbic acid, BHT, A. paniculata extract and andrographolide were calculated by regression analysis. The HepG2 in vitro data were statistically analyzed using SPSS software, version 10. The results were expressed as mean ± SEM. Hypothesis testing method included one-way analysis of variance (ANOVA) followed by least significant difference (LSD) multiple comparison tests. The level of significance was accepted with p < 0.001.
Results and discussion
Isolation, purification and structural confirmation of andrographolide
Andrographolide was extracted from the plant material in considerable yields (2.7%) compared to reported procedures 34 from ethyl acetate–methanol (95:5, v/v) solvent fractions in silica column chromatography. Preliminary HPTLC evaluation of the plant extract in ethanol:water (50:50, v/v) showed seven spots with retention factors (Rf) of 0.12, 0.26, 0.32, 0.41, 0.60, 0.73 and 0.83 using methanol–chloroform (15:85, v/v) as the mobile phase. The major peak at Rf 0.6 corresponding to andrographolide was evident from the HPTLC (Figure 1) and reversed phase HPLC (Figure 2) analysis of plant extract and standard andrographolide. The retention time for andrographolide on the reversed phase Phenomenex C18 column was 7.1 min in a total run time of 30 min.

High-performance thin-layer chromatography (HPTLC) of (a) Andrographis paniculata extract and (b) standard andrographolide.

Reversed phase high-performance liquid chromatography (HPLC) analysis of (a) Andrographis paniculata extract and (b) standard andrographolide.
The structure of the isolated compound was established from FT-IR (Figure 3), 1 H NMR (Figure 4(a)), 13 C NMR (Figure 4(b)), mass (Figure 5) and elemental analysis. Considerable work has been done toward structural confirmation of andrographolide and other active components of A. paniculata. 16 –19 Fujita et al. 35 have determined complete stereochemistry of andrographolide by X-ray crystallography. The structural data obtained in the present study were in good agreement with published reports. Andrographolide showed a strong UV absorption at 228 nm and FT-IR bands at 3398 cm−1 due to hydroxyl group, two sharp bands at 1727 cm−1 and 1625 cm−1 corresponding to α,β-unsaturated γ-lactone system and 907 cm−1 for exocyclic methylene group. 1 H NMR and 13 C NMR gave complete assignment for all the protons and carbon atoms. The 1 H NMR spectra showed a triplet at δ 6.85 due to olefinic proton in an α,β-unsaturated lactone. Another triplet at δ 5.0 corresponded to 14-H, while double doublets at δ 4.4–4.5 accounted for 15-H protons. Exocyclic methylene protons, 17-H resonated at δ 4.85 and 4.75 as singlets. The two tertiary methyl groups showed signals at δ 1.24 and 0.65. The mass spectrum shows the molecular ion peak and a base peak at m/z 350 and 119, respectively. The probable fragmentation pathway reveals ions at m/z 332 and 314 due to successive elimination of water molecules.
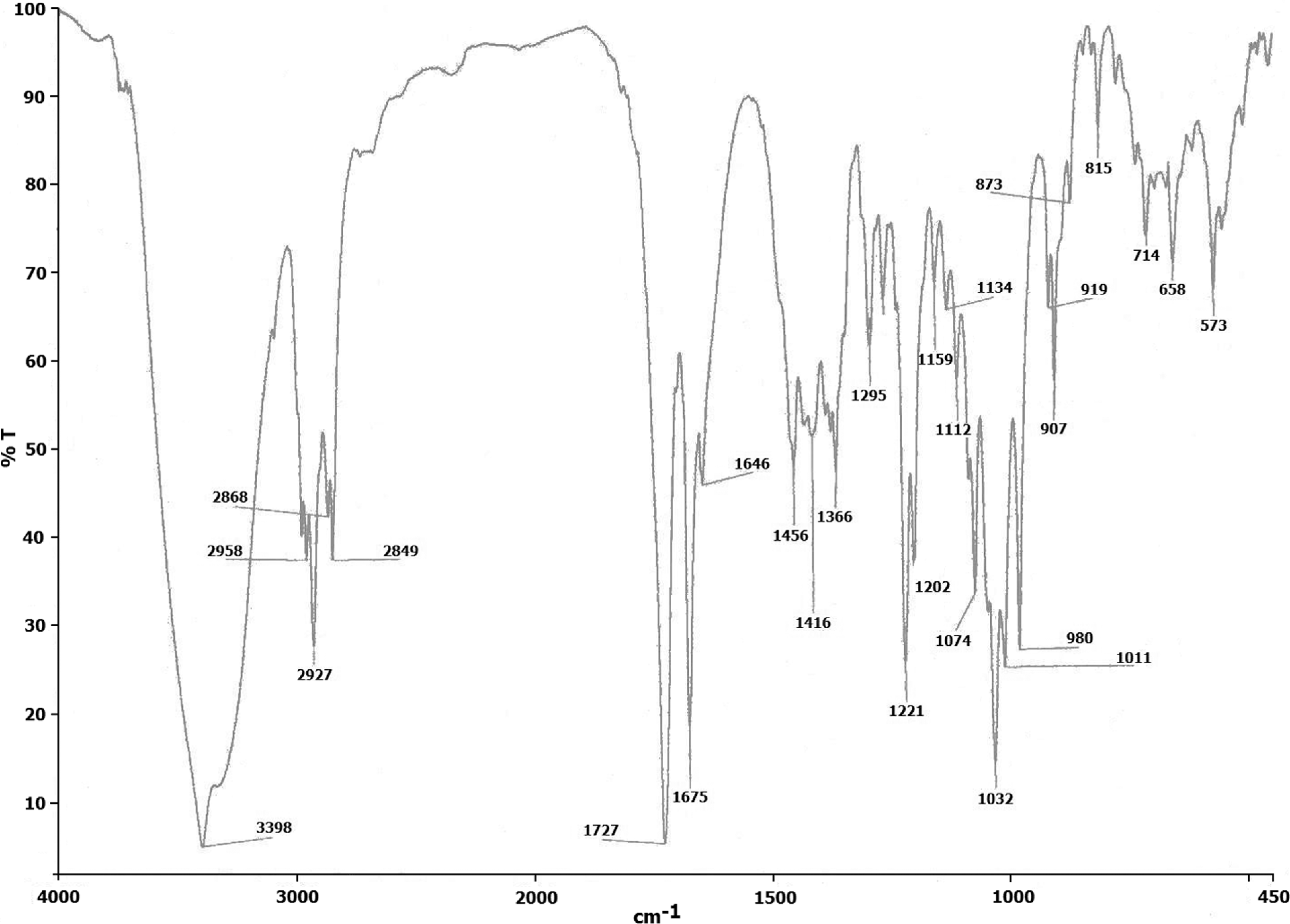
Fourier transform infrared (FT-IR) spectra of isolated andrographolide (4000–450 cm−1).

(a) 1H nuclear magnetic resonance (1H NMR; 500 MHz in CD3OD with TMS as internal standard) and (b) 13C nuclear magnetic resonance (13C NMR; 125 MHz in CD3OD) spectra of isolated andrographolide.

Electron impact mass spectra of isolated andrographolide with probable fragments.
Antioxidative property of ascorbic acid, BHT, A. paniculata extract and andrographolide
The results of DPPH free radical-scavenging activity of ascorbic acid, BHT, extract and andrographolide is presented in Figure 6(a)-(d), respectively. The ability of the tested products to quench this stable radical to a yellow-colored product—diphenyl picryl hydrazine—is a measure of their antioxidative ability. The scavenging activity was concentration-dependent in all cases and reached a maximum at a concentration of 40 µmol/mL for ascorbic acid and BHT and 300µg/ml for A. paniculata extract and 15 µmol/ml for andrographolide, respectively. The IC50 values calculated for ascorbic acid, BHT, A. paniculata extract and andrographolide were 4.3, 5.8. 220.5 and 3.2µg/ml, respectively. Our results indicated that andrographolide exhibited highest antioxidative property as compared to the standard antioxidants and the crude extract with the lowest IC50 value.

1,1-diphenyl-2-picrylhydrazyl (DPPH) scavenging activity of (a) ascorbic acid, (b) BHT, (c) Andrographis paniculata extract and (D) andrographolide with increasing concentrations.
Protective effects of andrographolide against CCl4-induced liver damage in HepG2 cells
In the present study, the cytoprotective property of plant extract and andrographolide was ascertained in HepG2 cell line by inducing toxicity using CCl4. In many such studies, HepG2 cells have been used as substitutes to hepatocytes. Both share many morphological, biochemical, phenotypic and genotypic features. 36,37 This cell line has also been used in various studies related to medicinal plants for their liver protecting and antioxidative properties. 38
The mechanism underlying the experimental hepatotoxicity induced by CCl4 suggesting involvement of lipid peroxidation is well established and reported in the past. 38,39 However, membrane damage by virtue of its direct solvent effect cannot be ignored. 40 To rule out such possibility we have standardized the concentration (0.4%) and exposure time (24 h) of CCl4 initiating lipid peroxidation in HepG2 cells in one of our previous studies. 41
Figure 7(a) and (b) represent the results of trypan blue exclusion test and MTT metabolism assay, respectively. Treatment with CCl4 caused significant loss of viability of cells as measured by trypan blue assay. MTT test also predicted the survival rate of HepG2 cells expressed as percentage of MTT metabolism relative to control was measured after 24 h treatment with the hepatotoxin. A significant reduction in the metabolism of MTT was also noted when HepG2 cells were exposed to CCl4. Harries et al. 29 have standardized the concentration and exposure time for CCl4-induced cytotoxicity on HepG2 cells using the index of MTT metabolism. Many previous reports have also judged the functional capacity of the mitochondria using MTT assay. 42,43 Both A. paniculata extract and andrographolide treatments along with CCl4 significantly increased cell viability and MTT metabolism. The highest concentration of andrographolide (30 µmol/ml) was most effective as compared to A. paniculata extract.

Protective effect of Andrographis paniculata extract and andrographolide against CCl4-induced toxicity on HepG2 as measured by (a) Trypan blue exclusion test and (b) 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay. Results are expressed as mean ± SEM (n = 5). Values shown in parenthesis indicate percentage recovery. No significance difference was noted between groups 1, 2, 3 and 4. a Compared to vehicle control (group 2) and CCl4 treated (group 5); b Compared to CCl4-treated (group 5) and CCl4 + antidote treated (groups 6, 7, 8, 9,10 and 11); c compared to CCl4 + highest concentration of andrographolide (group 11) and CCl4 + highest concentration of A. paniculata extract (group 8). Level of significance p < 0.001. Group 1. Control; Group 2. DMSO control (0.25% (v/v); Group 3. A. Paniculata extract control (600 µg/ml); Group 4. Andrographolide control (30 µmol); Group 5. Carbon tetrachloride (0.4% v/v); Group 6. CCl4 (0.4% v/v) + A. Paniculata extract (200 µg/mL); Group 7. CCl4 (0.4% v/v) + A. Paniculata extract (400 µg/mL); Group 8. CCl4 (0.4% v/v) + A. Paniculata extract (600 µg/mL); Group 9. CCl4 (0.4% v/v) + Andrographolide (10 µmol); Group 10. CCl4 (0.4% v/v) + Andrographolide (20 µmol); Group 11. CCl4 (0.4% v/v) + Andrographolide (30 µmol).
The results obtained in all three leakage parameters, LPO and glutathione levels in the experimental groups are presented in Table 1. The increased leakage enzymes—ALT, AST and LDH observed after 24 h of exposure to CCl4 predicted membrane damage. The treatment with CCl4 initiated lipid peroxidation in HepG2 cells which was quantified based on rise in MDA levels. A concurrent decrease in glutathione content in HepG2 cells was noted. The CCl4-induced oxidative stress in HepG2 cells was significantly ameliorated by cotreatment of the toxin along with A. paniculata extract/andrographolide. It was observed that highest concentration of andrographolide (30 µmol/ml) exhibited the maximum protective effect toward the various parameters studied as compared to all three concentrations of A. paniculata extract.
Protective effect of Andrographis paniculata extract and andrographolide on CCl4 toxicity in HepG2 cell.a

a Results are expressed as mean ± SEM (n = 5). Values shown in parenthesis indicate percentage recovery. No significant difference was noted between groups 1, 2, 3 and 4.
b Compared to vehicle control (group 2) and CCl4 treated (group 5).
c Compared to CCl4 treated (group 5) and CCl4 + antidote treated (groups 6, 7, 8, 9, 10 and 11).
d Compared to CCl4 + highest dose of andrographolide (group 11) and CCl4 + highest dose of A. paniculata (group 8). Level of significance p < 0.001.
Numerous scientific studies published in the past on andrographolide have produced mixed outcomes. In one such report, andrographolide was portrayed to be highly cytotoxic to HepG2 cells, by mechanism of G2/M phase cell cycle arrest. 44 Contradictory to this, it was reported to be nontoxic as compared to many other isolates from the parent plant A. paniculata in a panel of cell lines. 45,46 Its cytoprotective role against paracetamol and CCl4-induced toxicity has also been well documented. 47,48 We observed that co-incubation of andrographolide along with CCl4 resulted in protection of HepG2 cells against this experimental hepatotoxin. Andrographolide treatment caused no adverse effects in these cells. Based on our study results we conclude that andrographolide is relatively safe for biological systems, and it can be used as an effective antioxidative and cytoprotective agent in the long run.
In general our experimental plan assessed the possible hepatoprotective behavior of A. paniculata extract and andrographolide against CCl4-induced toxicity. The cell viability which reflected the degree of toxicity was quantified based on trypan blue and MTT assays. The depleted glutathione levels were also revived by the addition of extract/andrographolide. Our results suggest that andrographolide is cytoprotective and effective inhibitor of lipid peroxidation by virtue of its strong antioxidative ability.
Conclusion
In the present study, the mitigatory effect of A. paniculata extract and andrographolide, on oxidative damage induced by a known pathological agent namely CCl4 was studied using HepG2 cell line as an in vitro model. The protective mechanism was unraveled by measuring leakage of enzymes, lipid peroxidation and intracellular glutathione levels. Results indicated that andrographolide exhibited strong antioxidative property, as evident by the results of DPPH free radical assay. It was found to be twice as effective as compared to the crude extract against lipid peroxidation which was initiated after the toxin treatment. The cytoprotective and antioxidative role of andrographolide as demonstrated in this study makes it a potential future target molecule. In addition, it can be used as a substitute for many synthetic antioxidants presently available in the market.
Footnotes
Acknowledgments
The fellowship assistance to Dr. Krithika Rajesh (Research Associate; File no: 45/ 37/ BMS-TRM) from Indian Council of Medical Research (ICMR), New Delhi, is acknowledged with thanks. The authors also gratefully acknowledge the instrumentation facility provided by IIT, Madras, and Zydus Cadila, Ahmedabad. The authors also extend their sincere thanks to Mrs. Aishwariya Kumaran, for her valuable support in literature collection.
Conflict of Interest
The authors have declared no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.



