Abstract
Over the past decade, regulatory authorities and water purveyors have become increasingly concerned with accidental or intentional adulteration of municipal drinking water. Emergency response guidelines, such as the ‘Do Not Consume’ or use concentration limits derived herein, can be used to notify the public in such cases. Potassium permanganate (KMnO4) is used to control iron concentrations and to reduce the levels of nuisance materials that affect odor or taste of finished drinking water. Manganese (Mn) is recognized an essential nutrient, permanganate (MnO4 −) and manganous (Mn+2) ions are caustic, and the acute toxicity of KMnO4 is defined by its oxidant/irritant properties and by the toxicity of Mn. Ingestion of small amounts (4–20 mg/kg) of aqueous KMnO4 solutions that are above 200 mg/L causes gastrointestinal distress, while bolus ingestion has caused respiratory arrest following coagulative necrosis and hemorrhage in the esophagus, stomach, or liver. Dilute KMnO4 solutions (1–100 mg/L) are used as a topical antiseptics and astringents, but >1:5000 (200 mg/L) dilutions can irritate or discolor sensitive mucous membranes and direct skin or ocular contact with concentrated KMnO4 can perforate tissues. Based on clinical experience with 200 mg/L KMnO4, a Do Not Consume concentration of 7 mg/L KMnO4 (equivalent to 2 mg Mn/L) is recommended. Recognizing limited empirical data from which to calculate an ocular reference value, a skin contact ‘Do Not Use’ concentration of 30 mg Mn/L is recommended based on the skin irritation in some patients after a 10-min contact with 100 mg KMnO4/L.
Introduction
Title V of the Public Health Security and Bioterrorism Preparedness and Response Act (United States Public Law 107-188) mandated vulnerability assessments by drinking water purveyors who service more than 3300 connections. These assessments are intended to identify procedures that can be brought to bear in the event of a terrorist or other intentional attack on municipal water systems. 1,2 Emergency response plans can be valuable given the consequences of accidents involving gross chemical contamination of drinking water supplies. 3 –10 The present study was conducted to define ‘Do Not Consume’ and ‘Do Not Use’ concentrations for potassium permanganate (KMnO4; measured as total manganese (Mn)) that may be used by regulatory agencies and water purveyors in emergency response plans. The Do Not Consume limit was based on ingestion toxicity and the Do Not Use limit was based on skin and mucous membrane toxicity to account for contact with KMnO4 during bathing and showering.
Experience indicates the most likely scenarios for an emergency during which the affected water that reaches the public involve short-term (possibly once-in-a-lifetime) exposure for at least one but less than 30 days. 5,8 –10 Nevertheless, the first 24 h are critical in preventing adverse health effects after which time constituent levels are generally reduced by dilution unless the chemical source persists. Since utility customers can be exposed as a result of ingestion and direct skin and eye contact during bathing and showering, it is important to consider oral, dermal, and ocular contact with excessive concentrations of substances present in drinking water.
The present evaluation of acute oral, dermal, and ocular exposures to KMnO4 is intended to aid regulatory agencies in decisions to avoid or prohibit direct human contact with unusually high concentrations of KMnO4 in potable water. Carefully documented accounts of short-term (acute) human experience with reliable exposure data are preferred studies, but where those data are lacking controlled studies in laboratory mammals were evaluated. Either a lowest acute no-observed-adverse effect level (NOAEL), an acute lowest-observed-adverse-effect-level (LOAEL), or an acute benchmark dose (where possible) were the values that could be used to calculate an acute reference dose (RfD). To locate those values, the literature search strategy employed here was based on the Chemical Abstract Services Registry Number and at least one common name. The following public access databanks were searched:
ChemID Plus
Registry of Toxic Effects of Chemical Substances (RTECS)
Hazardous Substances Data Bank (HSDB)
GENE-TOX
Environmental Mutagen Information Center (EMIC)
Developmental and Reproductive Toxicology (DART)
TOXLINE – Core and Special
TRI (Toxics Release Inventory)
Chemical Carcinogenesis Research Information System (CCRIS)
Medline (via PubMed)
Integrated Risk Information System (IRIS)
Syracuse Research Corporation Online Toxic Substance Control Act Database (TSCATS)
Reports considered most valuable are detailed and published in the peer-reviewed open literature. Case reports of acute exposures usually describe symptoms and their consequences and those accounts were noted here, but nearly all case reports were limited by small numbers of patients and a near-universal absence of dose; these difficulties preclude use of individual case reports to establish short-term Do Not Use and Do Not Consume drinking water limits. Detailed reports from short-term clinical trials and summaries of (uncontrolled, but often extensive) clinical experience with short-term exposure durations are valuable. Due to the relatively few animal data for most corrosive materials (like KMnO4), all published accounts (including reviews and summary reports) that could be located were compiled. Lethality data were collected, but derivation of public warning levels based on death was not considered health-protective; similarly, reports concerning mild sensory irritation and adverse taste, odor, or appearance of affected water were not the candidate key data. Acute studies were preferred, but repeat dose, subchronic, reproduction, and chronic duration studies can provide information on target organs and the results noted during initial exposure can be used to set or support derivation of protective short-term limits. Criteria to identify candidate key studies that document serious or irreversible adverse health consequences included: relevant route of exposure in aqueous solution, a reasonable number of subjects, concurrent controls (as available), and sufficient details to explain study design, execution, and evaluation. Although toxicity data from primary reference sources were preferred, in some instances, the older literature is such that often only summaries and secondary sources are available. Single dose or short-term kinetic studies can inform interspecies dose scaling, but no controlled acute dose kinetic study in any species with KMnO4 was identified. Data from standard short-term genotoxicity and long-term bioassays for potential carcinogenicity were reviewed and where reliable studies of not more than 30 days exposure by an appropriate route demonstrate a significant genotoxic carcinogenic response in animals, short-term theoretical excess cancer risk greater than 1:10,000 (1 × 10−4) could be used in calculating short-term exposure limits. As acute toxicity studies for most large commodity materials used in water treatment were conducted decades before the advent of standard operating procedures and adoption of defined regulatory protocols, adherence to those regulations was not critical to identification of potential key studies; the limited animal and human toxicity data for KMnO4 precluded rigorous consideration of statistical parameters. Given the relatively few published studies in humans and animals that met all these criteria, consistent results from different sources were important toz identification of potential key and supporting studies. Since KMnO4 is corrosive at higher concentrations, its concentration-dependent relationship to water pH was integral to identification of short-term Do Not Use and Do Not Consume levels.
To derive the acute RfD and reference value (RfV), interspecies and intraspecies uncertainty factors were applied to LOAEL, NOAEL, or benchmark dose values. Data from clinical studies that included susceptible individuals were preferred key observations, where data on potentially sensitive groups (e.g. extremes of age, the infirm, nutritional status, those with genetic predisposition) are not available, additional intraspecies uncertainty factors may be selected by analogy to similar chemicals but usually those factors are less than 10. The concentrations for short-term notification derived here are designed to protect nearly all consumers including ‘susceptible’ individuals, but these emergency concentrations are such that people with idiosyncratic or unique (‘hypersusceptible’) conditions may not be fully protected. The acute oral RfD was used to derive the drinking water Do Not Consume limit, and the lower acute dermal RfV and the acute ocular RfV were used to derive the drinking water Do Not Use limit. Following US Environmental Protection Agency (EPA) convention when calculating a 1-day drinking water Health Advisory, the Do Not Consume limit is defined as the acute RfD (mg/kg) multiplied by the 10 kg weight of a child assumed to drink 1 L of water per day. Due to the brief duration, a relative source contribution was not applied.
Chemical and physical characteristics
KMnO4 (also known as permanganate (MnO4 −) of potash and Condy’s crystals) is the salt of manganic acid (also known as permanganic acid). The Mn found in KMnO4 is present in the VII oxidation state (Table 1). 11 –15
To illustrate the influence of KMnO4 concentration on pH, measurements of pH were taken of reagent grade KMnO4 dissolved in distilled, deionized water at room temperature by the NSF laboratory (Table 2).
Physical and chemical properties of KMnO4.

KMnO4: potassium permanganate.
aThe water solubility of KMnO4 varies with temperature; at 0°C, a saturated 100 mL solution contains 2.75 g; at 10°C, a saturated 100 mL solution contains 4.07 g; at 40°C, a saturated 100 mL solution contains 11.13 g, and at 60°C, a saturated 100 mL solution contains 18.15 g.
15
In aqueous solution, permanganic acid equilibrates with the hydrogen ions in water as follows: 
Influence of reagent grade KMnO4 concentration on water pH.

KMnO4: potassium permanganate.
Organoleptic properties
Dilute aqueous solutions are odorless, but are said to have a ‘sweet with astringent aftertaste’. 11 A 0.01% aqueous solution has a pink cast, but higher concentrations tend toward purple. When present in water at concentration of >0.05 mg/L, complaints of an ‘off-color’, turbidity, foul taste, and orange-brown to brown staining of ceramics, porcelain, utensils, skin, nails, bedding, and other textiles are associated with dissolved manganese dioxide (MnO2) increase. 16 The black stains of MnO2 on carpet, clothing, and porous surfaces are permanent. At elevated concentrations not only does the water have a black tint, but Mn reacts with tannins in tea, coffee, and liquor (e.g. whiskey) to produce a black sludge that precipitates, and it imparts a bittersweet to metallic taste.
Elevated Mn in water can promote bacterial growth to produce mats of gelantinous black-brown slime that is collect in hot water tanks, dishwasher lines and pumps, toilet tanks, and bowls. This slime sloughs off in globs and it can obstruct water distribution systems – including those used in home heating.
Production and use
Mn ores (pyrolusite, psilomelane, cryptomelane, and manganite) are mined in Australia, Brazil, Gabon, India, and the Republic of South Africa. KMnO4 is manufactured from MnO2 by treatment with KOH to yield potassium manganate (K2MnO4). Electrolytic oxidation of the latter in alkali yields KMnO4. 15
As a naturally-occurring element, Mn is present in groundwater as a result of contact with Mn mineral and it is present in surface waters where anaerobic decay of organic matter is active in sediment. 17
More than 80% of the KMnO4 produced is used in drinking water treatment and purification, 18 where it is added at concentrations between 2.5 and 20 mg/L to oxidize ferric (Fe+3) iron to ferrous (Fe+2) iron and the latter precipitates as iron oxide. It is also used to control nuisance materials in water that impart adverse odor or taste. 19 The MnO4 − ion has applications in bleaching waxes, resins and fats, in leather tanning, in treatment of sewage sludge, 20 and in treatment of well and waste water 21 –24 to reduce emissions of volatile organic hydrocarbons and H2S. It is a general biocide that is added at concentrations up to 4 mg/L to control fungi and algae, and it is also used to control parasites and other disease on fish. 25,26 One advantage to KMnO4 used in drinking water treatment is the reduction in total trihalomethane levels in finished tap water. 27 –30
In dilute aqueous solution (e.g. 1:10,000 or 100 mg/L) KMnO4 is used as a topical antiseptic and astringent where that activity is associated with the reduction in MnO4
− to manganous (Mn+2) ion.
11
A deep-purple solution was used during the late 19th and early 20th Century to treat acute gonorrheal urethritis wherein 1:5000 (200 mg/L) or 1:2000 (500 mg/L) dilutions were used to irrigate the anterior urethra over several weeks. These procedures resulted in ‘slight’ mucosal irritation,
31
but solutions at 1:500 (2000 mg/L) to 1:100 (10,000 mg/L) have also been used.
32
In addition, a wet dressing or bath with a 0.01% (1000 mg/L) solution or a localized treatment with a 1.0% solution (10,000 mg/L) is used topically to treat infected eczema, kill ringworms, to treat
KMnO4 is Generally Regarded as Safe (GRAS) by the US Food and Drug Administration for use in food starch at concentrations ≤0.2%, where residual Mn may not exceed 50 ppm (21 CFR 172.892), as a component of food packaging adhesives (21 CFR 175.105) and in sanitizing solutions used on food-processing equipment and utensils as well as in sanitizing solutions used on food contact surfaces in public eating establishments (21 CFR 178.1010).
Hazard assessment
Effects on humans
Ingestion
The toxicology of Mn 36 – 38 and that of the MnO4 − ion has been reviewed. 39 , 40 Numerous reports of acute accidental or suicidal KMnO4 ingestion have appeared 41 – 50 and sufficiently high oral doses (~25 g) can be abortifacient. 51 Gawarammana et al. 52 found that a single oral dose of 1200 mg KMnO4 (either as the crystal or dissolved in an unknown quantity of water) consumed by 13 suicidal adults (ages 19–37 years) caused epigastric pain, nausea, and vomiting in 73%; there were no fatalities or any indication of laryngeal edema. Death (often within 1 h) after ingestion of concentrated KMnO4 results from acute coagulative necrosis, mucosal ulceration, edema of the glottis, and asphyxiation. 53 – 57 Initial signs generally include black-brown stains in the mouth, inflammation of the oral mucosa, restlessness, and a low-grade fever; methemoglobinemia and severe hemorrhagic pancreatitis have been reported after ingestion of concentrated solutions. 40 , 45 , 58 Fatalities after ingestion (usually more than 10 g) of concentrated KMnO4 have also been attributed to hepatic necrosis and progressive cardiovascular collapse 45 , 55 – 57 ; autopsy revealed fatty degeneration and necrosis in the liver, hemorrhage into the subcortical and papillary regions of the kidney, and elevated serum Mn (756–1629 µmol/L). 59 – 61
Oral KMnO4 has been used to combat acute oral opiate intoxication; to some extent, outcome appears to depend on whether the person was fed or fasted and the quantity of morphine or opium consumed. 62 – 64 Raw 62 treated three women and one man with dram (11 mL) quantities of Condy’s fluid dissolved in one pint of water (~330 mg/550 mL or 600 mg/L KMnO4) by repeated gastric lavage every hour for 3–4 h. There were no adverse effects attributed to KMnO4 at ~22–28 mg/kg over 4–5 h, but the man died of acute opium-induced respiratory arrest. Luff 63 referenced 35 patients treated with KMnO4 in water at a pH-adjusted doses of 4–10 grains (1 grain = 0.0648 g) or 260–650 mg, and Maynard 64 described 19 adult males and females treated with repeated KMnO4 gastric lavage. Luff 63 recommended ‘8 or 10 grains of KMnO4 dissolved in water should be administered at once and repeated at intervals of 30 sec once or twice’ to be followed by ‘a weak solution of the antidote about 1 grain to a tumblerful of water should be administered by mouth from time to time even after all the opium or morphine in the stomach is supposed to have been rendered inert’. Patient outcomes varied, but five grains (320 mg) dissolved in one cup (~8 ounces or 237 mL equivalent to 1350 mg/L) of water given to an adult (4.6 mg/kg apparently on an empty stomach) caused local irritation, inflammation and vomiting at 30 min after each dose. Consumption of 4 or 8 grains (up to 520 mg or 7.5 mg/kg of a 2200-mg/L aqueous solution) on a full stomach elicited no acute adverse effect. 63
Ingestion of dilute KMnO4 solutions is benign and presents with mild local irritation of the mouth that is often self-limiting; spontaneous emesis or diarrhea may result if a large volume is ingested. 40 In a study with 171 adult male and 88 adult female patients (36.9 ± 16.2 years) hospitalized in an intensive care unit, participants received oral cleansing with 10 mL of a 0.01% (100 mg/L) KMnO4 solution twice daily. 65 Inspection of their oral cavity, palate, buccal spaces, posterior pharyngeal wall, and hypopharynx every 12 h made no mention of objective signs of irritation. While assessment of local oral irritation was not a specific objective of the Panchabhai et al. protocol, 65 routine use of KMnO4 as an oral antiseptic points to the conclusion that critically ill patients tolerate repeated dilute oral KMnO4 solutions without adverse effects.
Repeated ingestion (125 mL of 8% KMnO4 (80,000 mg/L) solution for a total dose of 10 g over 4 weeks) by an adult male induced manganism and classical signs (similar to those of Parkinsonism) were evident at 9 months. The first signs of psychological disturbance were evident within 2 weeks of the initial exposure. 66 In a similar case, an 80-year-old man presented with polyneuropathy and elevated hair Mn at 14 years after he accidentally ingested KMnO4 over a 4-week-period. 67 Manganism presents first as mania, increased libido, insomnia, and emotional liability and it then progresses to incoordination and fatigue and finally (in some cases many years after cessation of exposure) to involuntary, irregular muscle contractions (particularly noticeable upon walking) and skeletal muscle hyperflexion. The prolonged time course of the disease is a consequence of the slow rate of Mn uptake into and elimination from the brain. Although the clinical signs of manganism share some of the features of Parkinsonism, the neuropathology, 68 time course, therapeutic response, and lack of Mn accumulation in the brain of patients with Parkinsonism indicate manganism and idiopathic Parkinsonism have different etiologies. 69 – 71
Humans and other mammals adapt to increased Mn consumption by reducing gastrointestinal uptake and increasing biliary and pancreatic elimination (reviewed in Santamaria and Sulsky 72 ). Although prolonged daily ingestion of Mn salts can increase serum Mn significantly, healthy men and women tolerate repeated ingestion (60–124 days) of comparatively high Mn daily supplements (0.25–0.33 mg Mn/kg) without any discernible adverse effects 73 , 74 – including those on hematocrit, serum ferritin, serum transferrin, or serum iron in women 73 and performance on neurological tests. 75 The only change in note was an increase in the activity of lymphocyte superoxide dismutase after prolonged daily Mn supplementation (e.g. 89 days). 73
Kawamura et al. 76 recounted the experience of six Japanese families (some 25 people) who consumed manganese tetroxide (Mn3O4)-contaminated well water. All the members of the affected family developed manganism and at least two of the victims died. While the Agency for Toxic Substances and Disease Registry (ATSDR) 36 concluded that all these people were exposed to elevated Mn in their drinking water; ATSDR 36 questioned whether all the features described by Kawamura and associates 76 were attributable to Mn given the rapid onset of the disease; the US EPA 77 noted that some aspects of that description were inconsistent with clinical manganism. First, the onset of signs was rapid (2–3 weeks) and death occurred within 3 days. Second, some of the survivors recovered prior to reductions in Mn concentrations in their drinking water. The National Research Council (NRC) 38 stated that the well water contained 14 ppm Mn3O4 (7420 µg Mn/L) and calculated the daily dose as 20 mg Mn/day.
Kondakis et al. 78 used 30 different weighted measures (shown to have value in diagnosis of Parkinsonism) and examined older residents (mean age 67 years) who lived in three Greek towns with different levels of Mn in their drinking water (4.0–15.0, 80–250, and 1800–2300 µg/L). As the Mn concentration in drinking water increased, the average neurological score increased (2.7, 3.9, and 5.2, respectively). The authors 78 concluded that increased Mn in drinking water was associated with increased hair Mn levels and they were concerned with the significant association between use of groundwater containing 0.082–0.25 mg Mn/L and an increase in the prevalence of neurologic signs consistent with extrapyramidal dysfunction. The ATSDR 36 interpreted those results as suggestive that chronic consumption of drinking water with relatively high Mn concentrations ‘might contribute to an increased prevalence of neurological effects in the aged’, but qualified that conclusion with the caveat that Kondakis et al. 78 failed to verify that those who complained of neurologic symptoms actually ingested higher Mn levels than those who did not register such complaints.
Vieregge et al. 79 found a somewhat (but not significantly) higher blood Mn (8.5 ± 2.3 µg/L) in 41 people living in Northern Germany (mean age = 57.5 years) who consumed well water containing 300–2160 μg Mn/L for at least 10 years (and up to 40 years) compared with that in a referent group of 74 people (mean age = 56.9 years) who used water containing 7.7 ± 2.0 µg Mn/L. However, there was no increase in the prevalence of signs of manganism, and similar to the report of Kondakis et al., 78 the authors 79 failed to take into account dietary Mn contribution to total daily dose. Iwami et al. 80 correlated the incidence of motor neuron disease in a small Japanese town with elevated Mn in local rice. In this case, the concentrations of Mn in drinking water were low, but failure to document daily Mn exposure in light of other nutritional deficiencies compromised interpretation of those observations.
Bouchard and associates
81
measured hair Mn concentrations in 24 boys and 22 girls (6–15 years old) who lived in homes supplied with well water containing total Mn at a mean concentration 500 ± 129 µg/L with a maximum of 610 µg/L. Those who used water with higher Mn (
Bouchard et al. 82 published the results of follow-up cross-sectional study of 362 children (ages 6–13 years) from 251 families who lived in homes with well water (47%) or in homes with municipal water (53%) and who had lived in the same house for at least 3 months. Tap water Mn concentrations ranged from 1.0 to 2700 µg/L (arithmetic mean = 95 µg/L with an upper 95% concentration of 255 µg/L). Drinking water Mn was lower in homes with private wells (geometric mean = 8 µg/L) compared with those with drinking water supplied by public wells (55 µg/L). Mn intake from drinking water (including domestic water used in food preparation) ranged from 0 to 286 µg/kg/month with the highest recorded at 945 µg/kg/month. Bouchard et al. 82 made no mention of clinical abnormalities among any of the 362 participants, but reported that reduced performance on IQ tests was correlated with tap water Mn (but not with dietary Mn). Chen and Cope 83 questioned those conclusions since the participants’ dietary Mn consumption (2355 µg/kg-month) was far higher than that from water and 121 of the participants (33%) admitted they did not drink tap water at home.
This same group
84
–
86
conducted clinical evaluations, behavioral assessments, and evaluated children’s performance on academic achievement tests in relation to home tap water Mn concentrations. Clinical evaluation (body weight, head circumference, and blood and urine Mn concentrations) and classroom behavior (focusing on the disruptive) of 210 children who had home drinking water with Mn at 40–3442 µg/L made no mention of any sign of local or systemic Mn intoxication, but there was a weak (
The ATSDR 36 summarized two case reports 16 , 87 and three cross-sectional or comparative studies 88 – 90 that evaluated potential neurologic disease, impaired learning and reduced intellectual performance as a result of consuming drinking water with elevated Mn. The 6-year-old described by Sahni et al. 87 used well water containing 2200 µg Mn/L and she experienced progressive neurologic symptoms and became unable to stand alone; following chelation therapy, she recovered. The report by Woolf and coauthors 16 is of interest since the family used well water containing 1.2 mg Mn/L. The authors noted that the water was turbid and had a metallic taste; test results for learning, IQ, and cognition on a 10-year-old boy were normal, but his elevated serum Mn (0.90 μg/dL compared with the normal level of <0.26 μg/dL) and suggestive findings of impaired short-term verbal and visual memory lead the authors 16 to suggest that children may be more sensitive to excess Mn than adults. The He et al. 88 and Zhang et al. 90 reports concern the same group of Chinese children who were exposed to elevated Mn associated with sewage irrigation. He et al. 88 examined 92 children (ages 11–13 years) who consumed well water containing 240–350 µg Mn/L and concluded there were increased problems with short-term memory and manual dexterity compared with children who drank water containing 40 µg Mn/L. The study by Wasserman et al. 89 involved 142 children (ages 9.5–10.5 years) who used water from wells containing 103, 440, 801, 1923 and up to 3908 µg Mn/L (mean = 793 μg Mn/L); after adjusting sociodemographic factors, performance on the Wechsler Intelligence Scale for Children was inversely related to the Mn concentration in drinking water. Wasserman et al. 89 made no attempt to estimate dietary Mn intake for any of the study participants and there was no relationship between blood Mn and intellectual performance. Despite medical observation during venipuncture, there was no mention of any adverse clinical sign of acute or chronic manganism made in the report.
ATSDR 36 considered all these reports of limited value due to problems with selection of the referent groups, test administration, exposure assessment (including dietary Mn contribution), and confounding factors (including the presence of other metals in the water). Some authors have suggested that Mn levels are elevated in people with learning disabilities, amyotrophic lateral sclerosis or who exhibit violent behavior, but none of those suggestions have been verified. 36 In contrast to the reports of Mn exposure via drinking water, controlled trials with dietary Mn are robust; for example, when healthy young women consumed supplemental dietary Mn at 0.8 or 20 mg Mn/day for 8 weeks, there were no adverse effects on biliary function, activities of plasma glutathione peroxidase, catalase or superoxide dismutase, glucose, insulin, hemoglobin capacity or total whole blood iron-binding capacity, neurologic parameters or psychological measures of hostility, anger, negativism, positive attitude, and self-assurance. 75
Inhalation, dermal and other routes of exposure
Although inhaled dusts of MnO2 or Mn3O4 can produce pneumonitis, most of the concern regarding environmental Mn exposure is related to the symptoms of chronic manganism seen miners, welders, and those employed in battery and ferroalloy plants where inhaled dusts containing >0.2–1 mg Mn/m3 or fume that can result in progressive emotional, memory, and clinical motor disturbances. Somewhat lower exposures can alter visual reaction times, eye-hand coordination, hand steadiness, and libido and repeated Mn dust or fume exposure can increase fatigue and irritability. 36 , 91 , 92
While there are descriptions of dermal exposure to Mn compounds in humans, 92 , 93 uptake of ionized Mn across intact skin is expected to be limited. Elevated urinary Mn levels were observed in a man burned with a hot acid solution containing 6% Mn. 94 Ten children with atopic dermatitis aged 5 months to 9 years (mean age 35.5 months) received topical KMnO4 at 1:20,000 (80,000 µg/L) once daily for 7 days along with a topical corticosteroid. 33 Local skin irritation was noted in two of the 10 patients after 7 days of treatment, but no further details were reported. Although other concentrations (1:1,000; 1:10,000; and 1:50,000) were evaluated for bacteriocidal activity after 5 min of skin contact, irritation was not assessed for those concentrations.
Intentional intravaginal administration of aqueous KMnO4 solutions or tablets in criminal or self-inflicted efforts to induce abortion is notuncommon. 95 – 112 In some cases, the mother died. 112 In a review of 23 British cases, Obeng 107 noted that all of the women suffered caustic burns of the vaginal fornix, but none terminated their pregnancy and the vast majority delivered healthy offspring.
Direct ocular contact with crystals or concentrated KMnO4 solutions will burn and stain the affected area purple to brown-black, 113 and these materials can burn the eye 114 and mucous membranes leading to perforations. 95 – 97 Decreased visual acuity, corneal clouding, subconjunctival hemorrhages, and brownish conjunctival discoloration were described in a 22-year-old male subject who sustained burns to his eyes as a result of ocular contact with KMnO4 crystals. 114 There is one account by Sigg et al. 115 wherein a man splashed a concentrated (but unspecified) solution of KMnO4 into both eyes. He complained of a foreign-body type irritation and developed several hard eroded lesions that appeared as granular deposits on his conjunctiva.
Effects on laboratory animals
The literature concerning Mn toxicity in rodents is extensive and most of the studies are concerned with its neurotoxic mode of action or chronic exposure to inhaled MnSO4, MnO2 or the gasoline additive methylcyclopentadienyl manganese tricarbonyl. There are limited published reports concerning KMnO4 per se.
A number of studies considered ingested MnCl2 or mixed MnCl2/MnO in mice and rats, but many of those protocols examined only a single level of Mn exposure (reviewed by Gwiazda et al. 92 and US EPA 116 ). Studies in rodents report variable changes in neurotransmitters including increased striatal γ-aminobutyric acid, but repeated Mn injections (7–59 weeks) in nonhuman primates at 3–10 mg Mn/kg/week found normal markers of glutamatergic and GABAeric neurotransmission. 117 Very high cumulative parenteral doses (>260 mg Mn/kg) given to monkeys induce motor deficits and pathology (gliosis) in the frontal cortex, globus pallidus, caudate, and striatum. 92,117
With respect to neurological impairment, not only do the rodents not display the signs of extrapyramidal deficits characteristic of clinical manganism but also the species difference in dietary requirements are such that humans require only 1/100th of the daily dietary Mn on a body weight basis compared with rodents. 77,116 Furthermore, internal cumulative doses required to elicit Mn-induced toxicity in rodents are at least 100 × greater than those after which humans show impaired neurologic function. 92
Acute exposure
Oral
Acute oral median lethal dose (LD50) values for KMnO4 have been reported in rats (1090 mg/kg) 118 and mice (1449 mg/kg). 119 Single lethal dose values range from 400mg/kg in dogs, 600 mg/kg in rabbits, 750–2157 mg/kg in mice to 810–1151 mg/kg in guinea pigs. 120 The acute oral LD50 in male Swiss mice for KMnO4 referenced by Joardar and Sharma 121 was 1600 mg/kg, a value somewhat greater than the single dose oral LD50 values of MnCl2, MnSO4, and Mn3O4 in rats and mice that range from 225 to 1082 mg/kg. 118 , 121 – 125 Kostial et al. 123 observed that acute LD50 values of MnCl2 in fed albino rats varied with age and that the lowest single dose oral LD50 (619 mg/kg) was seen in older animals.
After Kojima et al. 126 gave a single oral gavage of 100, 200, or 400 mg/kg MnCl2 to fasted male Wistar rats, circulating and peripheral tissue Mn concentrations increased, but none of the animals died; furthermore, there was no effect on body weight. No rats died after daily oral MnCl2 intubations at 100 mg Mn were carried out over 90 days. Lipe et al. 127 stated that a single intraperitoneal injection of 10 mg/kg MnCl2 killed adult rats ‘within days’, but the single bolus oral and parenteral mortality results stand in contrast to repeated dietary administration where feeding 1300–2251 mg Mn/kg/day (as MnSO4 or MnCl2) to rats and mice failed to influence survival. 128 , 129
Dermal or ocular
Little information is available on the irritant and contact sensitivity properties of Mn compounds, 120 but Mn salts were negative in the murine local lymph node assay. 130 No quantitative studies with direct instillation of KMnO4 into the eye or application onto the intact skin of laboratory animals were located.
Repeat dose studies
No repeated dose studies with KMnO4 by any route in any species of laboratory animals were located other than one non-English report that identified behavioral changes in rats after (unspecified oral) the treatment with 7851 mg/kg, 120 and another report 121 that identified a dose-dependent increase in chromosome aberrations in the bone marrow of male Swiss mice were given an oral dose of KMnO4 at 65, 130, or 380 mg/kg for over 21 days. However, as the Russian oral dose was substantially greater than the rat oral LD50, 118 the validity of that report may be problematic. The oral (details of administration not provided) study of KMnO4 and bone marrow clastogenicity in mice 121 reported increased aberrations at the two lower doses, but not at the highest dose (representing 20% of a lethal dose). No information concerning clinical signs, food or drinking water consumption, body weight, gross necropsy, or histopathology was given. The numbers of scored chromosomal aberrations per cell were much higher than normally reported and it is possible that chromosome and chromatid gaps were counted as breaks. 121
While repeated oral dose studies with various Mn salts have been published (Table 3), 127,131 –144 the relevance of changes in neurotransmitter levels in rodents after Mn exposure to humans has been called into question. 72,145
Repeat dose manganese oral toxicity studies in rodents.

GABA: γ-aminobutyric acid; DA: dopamine; NE: norepinephrine; ♀: male; ♂: female; ↑: increase; ↓: decrease; HVA: homovanillic acid (a primary dopamine metabolite); DOPAC: 3,4-dihydroxyphenylacetic acid.
More than toxicity, it is Mn deficiency that is generally of greater concern. 145 A case in point is the fact that the offspring of pregnant rodents maintained on Mn-deficient diets were afflicted with axial skeletal malformations 146 and survivors developed ataxia. 147 The incoordination and loss of equilibrium induced by prenatal Mn deficiency cannot be mitigated with supplemental dietary Mn after birth as these signs result from malformations of the otoliths of the utricular and saccular maculae. 148 Defects of the vestibular portion of the inner ear arise from the same reductions in mucopolysaccharide production that are responsible for the deficiency-induced epiphyseal dysplasia and skeletal reductions. 149 Mn deficiency is teratogenic in rodents, but there are no reports of human congenital malformation attributed to low Mn status. 150
Ingestion of up to 1300 mg/kg/day as MnSO4 by F344 rats for 14 days reduced body weight gains, but feeding MnSO4 failed to influence their survival. 128 Studies with B6C3F1 mice found that feeding MnSO4 for 14 days failed to influence either body weight gain or survival. 128 Ingestion of up to 2251 mg/kg/day as MnCl2 for up to 6 months by male mice also failed to alter survival. 129
Dorman and coworkers 151 gave oral doses of MnCl2 to CD rats at 0, 25, or 50 mg/kg/day from birth to day 21. In a companion study, adult male CD rats were given identical doses over the same duration. Neonates that received the highest dose displayed reduced body weight gain, increase startle response and increased hypothalamic, striatal, hippocampal, hindbrain, and cerebellar Mn concentrations. Although there were no treatment-related changes in clinical signs, motor activity, passive avoidance, or neuropathology, neonates given the highest dose had increased striatal dopamine and 3, 4-dihydroxyphenylacetic acid. Adult rats given the highest dose accumulated Mn in the brain. Dorman et al. 151 suggested that newborn rats were more susceptible to that adverse effects of ingested Mn compared with adult rats fed with the same levels of MnCl2.
Long-term studies
No controlled chronic studies with KMnO4 in laboratory animals by any route were located. There are suggestions that Mn-deficient rodents tend to accumulate a higher Mn body burden and that feeding high dietary Mn3O4 levels over a prolonged period can reduce tissue iron (Fe) stores. 152 Feeding Mn (400–1100 ppm) to Long Evans rats in an Fe-deficient diet for 100–224 days induced microcytic anemia. 153
The NRC 38 summarized the older literature concerning long-term Mn intoxication and neurobehavioral alterations in nonhuman primates. Most primate studies used parenteral injections (intravenous, subcutaneous, and intramuscular) and examined MnSO4, 154 MnCl2, or MnO2, whereas other studies involved airborne MnSO4 or the phosphate or aerosols of manganese oxides (reviewed in ATSDR 36 ). Neurobehavioral evaluations of rats and mice failed to encompass the wide range of changes seen in manganism and ATSDR 38 considered the parenteral data of limited utility.
In the evaluation of Mn nutritional supplements, Golub et al. 155 gave groups of eight male infant Rhesus monkeys either a soy formula with 1.0 mg Mn/L (as MnCl2) or the soy formula with 0.30 mg Mn/L or a cow’s milk formula with 0.05 mg Mn/L to 4 months of age. The group given the highest total Mn dose (107.5 Mn/kg/day) was said to be ‘more active’ than those given the soy or cow’s milk alone.
In the only oral Mn toxicity (as contrast to nutritional) study conducted in nonhuman primates, Gupta et al. 156 gave 6.9 mg Mn/kg/day as MnCl2 to four male rhesus monkeys for 18 months. The monkeys developed weakness in skeletal muscles with rigidity of their lower limbs; necropsy identified depigmentation and neuronal degeneration within the substantia nigra. The WHO 157 considered the Gupta et al. 156 results to be of limited utility primarily because only one dose level was evaluated.
Absorption, distribution and elimination
After reviewing more than 100 publications, Gwiazda and associates 92 concluded that while systemic bioavailability of inhaled Mn was essentially equivalent to that after intravenous injection (100%), systemic uptake of ingested inorganic Mn (e.g. chloride and oxide) was not more than 3% (0.03). It is this marked difference in bioavailability as a function of different routes of exposure and the properties of different Mn chemical forms that account in large measure for the differences in dose required to elicit adverse effects. However, ingestion of the water soluble salts (e.g. MnCl2) can induce systemic toxicity at lower doses than ingestion of insoluble forms (e.g. MnO2).
In controlled studies with dietary Mn, women received a somewhat smaller daily Mn dose (3.2–4.6 mg/day) compared with men (3.9–6.3 mg/day) given the same foods, but women absorbed a greater percentage of ingested Mn (3.5%) compared with the men. Of note were the observations that there were no significant gender-related differences in plasma Mn and that women had a more rapid rate of whole body Mn elimination than men; this difference was reflected in the shorter mean half-time (34 days) compared with men (48 days). 74
Teeguarden et al. 158 –160 examined the kinetics of ingested Mn in rats and mice and provided explanations for the differences in metabolic handling between a single oral bolus and continuous high oral dosing. These differences account for the apparent inconsistencies between the single dose oral LD50 values (225–1082 mg/kg) compared with the fact that repeated consumption of higher daily doses (1300–2251 mg/kg) failed to cause death. Normally, tissue Mn levels are regulated by intestinal uptake and hepatic elimination and other than relative solubility of the various Mn forms, these are the primary physiological factors that dictate proportional control of systemic Mn bioavailability. First, as the dietary Mn exposure increased the rate constant for Mn uptake from the intestine declined (~23-fold). Second, as the dietary Mn exposure increased, biliary elimination of Mn increased (~7-fold). However, in case of the Mn acute oral bolus (1000–10,000 mg/kg), systemic exposure is controlled by the amount of Mn delivered to the liver as contrast to the relatively small control contribution provided by the intestine. Thus, a high oral Mn bolus delivered over minutes to hours overwhelms existing physiologic Mn control processes, whereas the same dose ingested over several hours can be handled without adverse consequence due to normal intestinal and hepatic homeostatic Mn regulation.
Genotoxicity
International Programme on Chemical Safety (IPCS) 93 and Assem et al. 161 reviewed the in vivo and in vitro genotoxicity data for KMnO4 and related Mn compounds, including the sulfate and chloride salts. KMnO4 has been associated with micronuclei or chromosomal aberrations in vivo 121 or in vitro 162 in some studies but not in others. 163 The IPCS 93 concluded that the results of in vitro studies show that some forms of Mn have mutagenic potential, but the results of in vivo studies in mammals are not consistent. Therefore, Assem et al. 161 concluded that Mn might be ‘weakly mutagenic in vitro and possibly clastogenic in vivo but with unknown genotoxic effects in humans’.
Risk characterization
Mn is an essential nutrient required for normal bone development, reproduction, and neurological function, 164 where it is necessary for the activity of succinic dehydrogenase, hydrolases, kinases, decarboxylases, and transferases. 38 Although isolated cases of Mn deficiency have been identified, there is no evidence that Mn deficiency is a public health problem in well-nourished populations and the usual dietary Mn intake (2–3 mg/day) appears adequate to meet nutritional requirements. 38
The MnO4 − and Mn+2 ions are oxidizing agents and direct contact with KMnO4 either as the crystal or in concentrated aqueous solution can induce chemical burns of mucous membranes, the skin, the cornea, and ocular membranes. 113,115,165 Direct contact with concentrated KMnO4 induced oropharyngeal, esophageal, and gastric ulceration, hemorrhage, and perforation with subsequent development of structures and pyloric stenosis. Acute ingestion of sufficient quantities has caused death as a consequence of acute glottic and laryngeal edema and respiratory obstruction. Death following acute KMnO4 ingestion is a consequence of gastrointestinal hemorrhage, cardiovascular collapse, profound hypotensive shock, and respiratory failure. 40 When used as a local antiseptic, contact with relatively dilute KMnO4 solutions is innocuous and the mild local irritation is said to be ‘self-limited’. 33
Guilarte et al. 154 presented data collected in macaques after repeated intravenous injections of 3.3–4.9 mg Mn/kg as MnSO4 that yielded whole blood Mn concentrations of 14–106 μg/L, and observed that those values were consistent with circulating Mn measured in some human populations, including residents of Mn-mining districts. 166 Guilarte et al. 154 suggested that the deficits in fine motor skills and certain aspects of macaque cognitive function at those Mn levels may also occur in human beings. The Golub et al. 155 study with Mn in soy formula found increased physical activity among Rhesus monkeys fed with 107.5 mg Mn/kg/day, but those subtle changes were within the normal range of the animal’s behavior and no adverse health outcomes were identified. The Gupta et al. 156 trial with chronic oral MnCl2 reported signs of central nervous system (CNS) toxicity, but the fact that only a single dose level was examined reduced the utility of that study. 157 Thus, in the absence of empirical data from primate drinking water studies relative to parenteral injection and identification of relative source contribution to determine at what level of Mn ingestion via drinking water might produce local tissue damage or elevate blood Mn concentrations after either acute or chronic exposure, the nonhuman primate data are not helpful in establishing Mn limits in drinking water.
In humans, the consequences of excessive Mn exposure result from its actions in the CNS after repeated inhalation of Mn dusts and fume. 37 Prolonged excessive Mn exposure is thought to increase substitution of Cu into normal CNS prion binding glycoproteins (PrPc) that are found on the surface of the neurons and glia. This substitution plays a role in conversion of those proteins to the pathogenic scrapie form (PrPSc). The PrPSc aggregates and serves as a template to convert more PrPc protein to the PrPSc and the altered Mn+2, Cu+2, and Zn+2 binding properties and increased Cu content reduce antioxidant capabilities and hasten the conversion of PrPC to PrPSc. Once absorbed into neuronal cells, Mn depletes glutathione, increases the generation of reactive oxygen species as a result of its accumulation in mitochondria and inhibition of electron transport, 167 activates caspase-3, and increases DNA fragmentation. Increased caspase-3 activity leading to DNA fragmentation and chromatin condensation are hallmarks of Mn-induced apoptotic cell death. 168
Chronic high Mn exposures are neurotoxic, but chronic poisoning is almost always the result of inhaled Mn dusts or fumes rather than as a result of high dietary levels due to the fact that gastrointestinal bioavailability of Mn is comparatively low (~3%). 38 However, in addition to the reports of acute KMnO4 poisoning by Degner et al. 67 and Holzgraefe et al., 66 there are accounts of repeated ingestion of Mn-contaminated water and development of manganism, but interpretation of those reports is problematic. 36
There is considerable uncertainty attendant to health risk assessment of Mn exposure, a situation complicated by the fact it is an essential nutrient. 72,145,164 Most of the studies in animals have been conducted in rodents where internal cumulative doses between 18 and 5300 mg/kg are associated with an increase in striatal γ-aminobutyric acid (GABA) and there are indications of a ‘variable’ effect on striatal dopamine at internal cumulative doses at 200 mg/kg or more. 91,92 Studies with nonhuman primates found mild deficits in working memory, 169 motor deficits and changes in the globus pallidus after inhalation or acute 154 or repeated parenteral exposure to MnSO4, MnCl2, or MnO2 after internal cumulative doses between 3 and 1000 mg Mn/kg (Table 3).
There have been suggestions that inhaled Mn from bathwater during showering may present a neurotoxic hazard, 170 but others 171 dismissed that as a speculation.
Cancer characterization
Information on the carcinogenic potential of Mn is limited. 93 Lifetime carcinogenicity/chronic toxicity bioassays have been conducted in mice and rats fed MnSO4 at up to 731 and 232 mg Mn/kg/day, respectively. There was no evidence for increased tumors at any site in rats, but based on a marginal increase in thyroid follicular adenomas and hyperplasia in the high-dose mice, the NTP 128 concluded that there was ‘equivocal’ evidence for MnSO4-induced carcinogenicity in mice.
The ATSDR 36 concluded, ‘There is no evidence that manganese causes cancer in humans. Although no firm conclusion can be drawn from the mixed results in animal studies, there are little data to suggest inorganic manganese is carcinogenic. The EPA has provided manganese with a weight-of-evidence classification D – not classifiable as to human carcinogenicity.” Assem et al. 161 considered the ‘rodent data do not provide any clear evidence that Mn exposure is carcinogenic and the epidemiologic evidence is at best equivocal’.
Susceptible populations
The ingestion hazards of elevated Mn in drinking water have been examined in children 16,81,89 and the elderly. 78 There are suggestions of possible Mn-induced changes in behavior, but the nature of those reports does not permit definitive conclusions. 36 Several studies concluded that people with chronic liver disease or those who are alcoholics may be at increased risk of Mn accumulation and manganism. 36,91,172 –175 There are reports 59 –61 that death after acute KMnO4 ingestion was associated with hepatic failure.
KMnO4 is an oxidizer and the corrosive hazards of KMnO4 are well known, but no specific groups have been identified as uniquely sensitive to the hazards of mucous membrane or skin contact with or ingestion of KMnO4 either as the solid or in solution.
Exposure assessment
Ritter et al. 17 compiled surface and groundwater concentration data and noted that not only is Mn an essential element but also the majority of human Mn exposure comes from food. Total dietary Mn consumption ranges from as low as 0.7–2 mg/day to as high as 8.8–10.9 mg/day (equivalent to 125–155 µg/kg/day for a 70-kg adult male) with the highest sources in nuts, tea, spices, cereals, fruits, and vegetables. Typical male American teenagers and adults consuming a 3900-cal diet ingest 3.8 and 3.7 mg/day, respectively.
After reviewing Mn concentrations in waters of North America, Ritter et al. 17 concluded that routine concentrations ‘do not present any significant hazards for human life’. Drinking water typically contributes 11% of daily Mn intake, assuming that an adult consumes 2 L/day of drinking water that contains the typical concentrations found in US tap water (mean = 17–32 µg/L); this equates to a drinking water total Mn contribution between 40 and 64 µg/day. If one were to consume drinking water with Mn at 1320 µg/L, the contribution from water to total daily Mn exposure would be equivalent to that from the diet. 38
Nevertheless, there are water systems and groundwater sources (typically from Mn-bearing minerals like pyrolusite and in situations where high carbonate, reducing conditions, low O2, and reduced pH predominate), where total Mn concentrations >1000 µg/L are routine. In some surface waters, total Mn concentrations up to 3230 µg/L have been detected 38 and in water with high organic content, Mn is adsorbed within suspended particulates. Loranger and Zayed 176 concluded that consumption of such high levels of total Mn might contribute up to 20% of one’s daily total Mn intake. Wasserman et al. 89 noted that 6% of US households use drinking water with Mn concentrations greater than the US EPA lifetime health advisory (300 µg Mn/L). 77
Do not consume level derivation
Key studies and critical effects
Recognizing that Mn is an essential nutrient and that ingestion of concentrated KMnO4 solutions can be lethal, review of the longer-term studies in rodents found that most were concerned with neurologic impairment (Table 3). Since rodents are poor surrogates for Mn toxicity in primates 72,77,92,116,145 and the primate data are limited to parenteral injection 92,117 or only single or relatively low dose levels 154 –156 that do not elicit serious acute or irreversible adverse health effects, the available animal data with various inorganic Mn salts are not informative to derivation of an acute oral RfD for KMnO4.
There are many case reports that describe damage to the gastrointestinal tract, liver, and pancreas after acute ingestion of either crystalline or concentrated KMnO4. These reports are consistent in that a single dose of 15–20 g causes destruction of tissue that ‘was so extensive as to suggest alkali corrosive. Even dilute solutions may cause irritation to the stomach while concentrated solutions inflame the mucous membrane and may even induce gastroenteritis’. 56 Study of people who use domestic tap water with comparatively high levels of Mn (610 µg/L, 81 1367 µg/L, 86 2100 µg/L, 79 2300 µg/L, 78 2700 µg/L, 82 3442 µg/L, 84 and 3908 µg/L 89 ) could provide indications of concentrations that are intolerable, but like the Bouchard et al. studies 81,82 where 33–91% of the participants did not drink tap water containing 500–2700 µg Mn/L, it is not clear that other groups actually consumed the affected water since Mn concentrations >50 µg/L have a pink to purple color and unpleasant taste. 16
Other than the absence of oropharyngeal irritation after rinsing the mouth twice daily with 100 mg/L aqueous 65 KMnO4 and death after consumption of >10 g, 45,58 –60,57 there are four reports that represent potential short-term key observations. Repeated (3–4 times at 1 h intervals) oral lavage of intoxicated (and apparently fasted) patients with ≥1350 mg/L of pH-adjusted aqueous KMnO4 caused local irritation, inflammation, and vomiting. 62 –64 Among 13 adults (ages 16–34 years) who ingested a single median KMnO4 dose of 1200 mg (17–20 mg/kg), 73% experienced epigastric pain, nausea, and vomiting and 27% complained of generalized weakness and headache; one developed skin burns, but none exhibited laryngeal edema and none died. Some 37% of these patients dissolved the material in water while the remainder ingested it as a solid. 52 Despite the caustic and irritant properties of concentrated KMnO4 in humans, the response can vary in that some apparently tolerate ingestion of 8% KMnO4 (80 mg/mL equivalent to ~10 g in a 125-mL water or 150 mg/kg), 66 whereas others who consumed aqueous KMnO4 at 300–500 mg (equivalent to 1300–2200 mg/L or/and 5–7.5 mg/kg) experienced local irritation and repetitive emesis (apparently on an empty stomach) or no ill effect (on a full stomach). 63,64
Considering the clinical reports together shows that humans vomit after consuming KMnO4 in water at >1000 mg/L but tolerate repeated direct oral contact with dilute (100 mg/L) solutions of KMnO4. Although oral rinsing with dilute KMnO4 is routine clinical practice in some hospitals, 65 no controlled concentration–response studies that assessed potential toxicity associated with oral KMnO4 rinsing were identified and no information was located to confirm that oral contact with 100 mg/L KMnO4 represents a definitive NOAEL. Since clinical experience with acute oral administration of aqueous KMnO4 best predicts the acute toxicity of KMnO4 in drinking water (as contrast to ingestion of crystalline KMnO4), 46,51,57 an acute oral RfD can be based on local irritation and inflammation 31,32,177 that precede the epigastric pain, nausea, and vomiting 52,62 –64 after ingestion of aqueous KMnO4 seen after oral doses at 4–20 mg/kg.
Dose-response
The acute ingestion hazard posed by KMnO4 in aqueous solution is primarily a function of its oxidant properties. 36,93 Permanganic acid is a mineral acid that after ingestion of sufficiently high concentrations can cause immediate, life-threatening local corrosive tissue damage and can cause death due to asphyxiation. However, it is difficult to determine the minimum human fatal dose as often the quantity is either not known, 50,59 speculative (e.g. 5–10 g), 46,60 or not reported at all. In other cases, prompt emesis occurs and at least some of the ingested material is eliminated thus reducing the amount and residence time in the gut. 51,52,62 –64 While KMnO4 concentrations greater than 1:5000 (200 mg/L) are irritating to mucous membranes, 31,32,177 the limited quantitative data in humans allow only approximations of dose–response for acute oral KMnO4. Direct mucous membrane contact with water containing KMnO4 at 100 mg/L appears innocuous, 65 direct contact with 200 mg/L causes mucosal irritation, 31,32,177 intubation of ~1350–2200 mg/L in water elicits oral irritation, inflammation, and vomiting (equivalent to 3.7–9.2 mg/kg), 62 –64 single ingestion of 17–20 mg/kg causes gastric pain, nausea, and vomiting 52 and ingestion of 20–25 g (~300–400 mg/kg) causes immediate tissue destruction, life-threatening symptoms, 51,56,58 and death. 45
Uncertainty factor selection
The following uncertainty factors were applied to the lowest concentration of KMnO4 (200 mg/L) above which mucous membrane irritation, 31,32,177 nausea and vomiting, 52 oropharyngeal, esophageal, and gastric injury 62 –64 occur.
Interspecies extrapolation (1×)
Since clinical experience 31,32,52,62 –64 serves as the basis for an acute oral RfD, an interspecies uncertainty factor of 1× was assigned.
Intraspecies extrapolation (10×)
Impaired hepatic function results in reduced capacity to eliminate Mn in bile. An intraspecies uncertainty factor of 10× was assigned based on reports that newborn rats are more susceptible to ingested Mn than adult rats 151 and reports linking chronic liver disease with increased Mn in blood and peripheral tissues. 36 People with preexisting liver disease and those deficient in iron are at increased risk of Mn accumulation and encephalopathy. 36,93,117,172 –175
LOAEL to NOAEL extrapolation (3×)
A LOAEL to NOAEL extrapolation factor of 3× was applied since the concentration associated with tissue irritation 166 and corrosive response (200 mg/L) 31,32 is twice the concentration that failed to elicit local irritation based on repeated oral applications. 65 The total uncertainty factor assigned for acute KMnO4 ingestion is, therefore, 30×.
Acute oral RfD derivation
An acute RfD can be calculated based on the concentration associated with tissue irritation (1: 5000 mL equivalent 1 mg/5 mL or 200 mg/L) 31 , 32 , 177 above which nausea, vomiting, and epigastric pain are seen
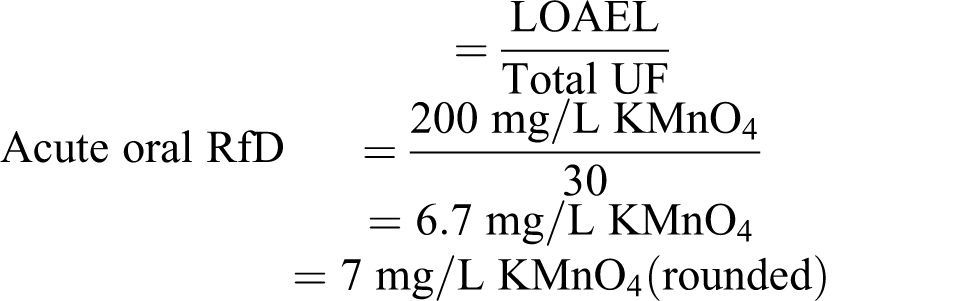
Using the ratio of Mn/KMnO4 molecular weights (55/158) applied to an acute oral RfD of 6.7 mg/L for KMnO4, the acute RfD expressed as total Mn is

The acute oral RfD calculated here can be compared with the observation of acute manganism that became evident in one man at 2 weeks after he consumed 10,000 mg Mn over the preceding 4 weeks 66 and to the report of chronic manganism associated with consuming well water containing 14 mg Mn3O4. 76 In the former case, the total dose was 360 mg/day (or 5.1 mg/kg/day as KMnO4 for a 70-kg adult); based on a 10-kg body weight and 1 L/day water consumption of a child, this equates ~51 mg/L KMnO4 or 18 mg/L Mn. In the latter case, the total daily dose (20 mg Mn3O4/day) was estimated 38 at ~0.3 mg Mn3O4/kg/day for a 70-kg adult or 0.2 mg Mn/kg/day.
Do not consume limit
The Do Not Consume Limit is equal to the acute oral RfD. The Do Not Consume Limit is expressed as a concentration rather than on a body weight basis since the critical effect was damage to mucous membranes and contact irritation as contrast to systemic toxicity.

Do Not Use level derivation: dermal
Key study and critical effect
An acute dermal RfV for KMnO4 can be based on the fact that KMnO4 is used at 100 mg/L as a direct soak antiseptic to treat various dermatological conditions. 33 –35 There are accounts in which the contact with 100 mg/L is not irritating to the skin or mucous membranes after a 10-min application to treat dermatitis, dermatomycoses, and snakebite. 177 Repeated irrigation of the urethra with 1:5000 or 1:2000 dilutions was said to elicit ‘slight’ mucosal irritation, 31 but contact with higher concentrations (>1:5000 equivalent to >200 mg/L) or prolonged or repeated exposures are irritating. 32
Dose-response
While there are many accounts of damage after direct KMnO4 contact with the skin and mucous membranes, rigorous dose–response relationships associated with topical exposure to KMnO4 are difficult to establish due to the lack of controlled trials examining duration, frequency, and magnitude of exposure. Data are extensive for the other common Mn salts, but the toxicological profile and chemical properties of permanganic acid are so different from those of the carbonate, chloride, oxide, tetroxide, and related congeners that little can be learned about acute KMnO4 toxicology from studies with other common Mn compounds. A robust database also exists concerning inhaled Mn dusts and fume, 36 but the clinical experience with KMnO4 indicates that reports of adverse health effects after chronic exposures to Mn dusts and fume offer little insight into potential health consequences of elevated Mn as a result of KMnO4 present in drinking water.
A great many reports show that direct contact with aqueous KMnO4 on mucous membranes caused caustic burns and at least in one instance caused death, 112 but others state that humans tolerated either repeat oral contact with an 8% solution 66 or repeated direct application of 0.05 to 0.5–1.0% solutions. 31,32 Other than those accounts and the fact that 0.01–1.0% KMnO4 is used as a topical astringent, there are no topical concentration–response data available in the open literature.
Uncertainty factor selection: dermal
The following uncertainty factors were applied to the clinical experience with 1:10,000 solutions (100 mg/L) KMnO4 used as a direct soak antiseptic to treat various dermatological conditions including those in children. 33 –35,113,177
Interspecies extrapolation (1×)
There is no evidence that percutaneous uptake of Mn through intact skin from KMnO4 dissolved in dilute aqueous solution can influence the total body Mn burden. Since clinical experience in humans 33 –35,113,177 serves as the basis of the dermal RfV, an interspecies uncertainty factor of unity was applied.
Intraspecies extrapolation (1×)
An intraspecies uncertainty factor of unity was applied since patients with dermatologic conditions served as the basis of the dermal RfV. People with disease or damaged skin were considered to represent a sensitive subpopulation and children were among those treated with direct soak applications. 33,113
LOAEL to NOAEL extrapolation (1×)
Because no signs of corrosion were observed during or after therapeutic application of KMnO4 solutions as a topical astringent and antiseptic, the 0.01% (100 mg/L) solution can be considered a NOAEL. This factor is supported by the 7-day repeat application study by Stalder et al. 33 in which repeated topical KMnO4 treatment at 1:20,000 (50 mg/L) for 7 days resulted in local irritation in 2 of 10 children with preexisting atopic dermatitis.
The total uncertainty factor for dermal exposure is, therefore, 1×.
Acute dermal RfV calculation
An acute dermal RfV based on the KMnO4 concentration used to treat dermatitis, ringworm, and other skin conditions is calculated as follows

Using the ratio molecular weight Mn/molecular weight KMnO4 (55/158), the acute dermal RfV = 34.8 mg/L for Mn and 30 mg/L for Mn (rounded)
Do Not Use limit: dermal
The Do Not Use limit – dermal is the same as the acute dermal RfV, since both are expressed as concentration.

Do Not Use limit derivation – ocular
Although direct contact with crystalline KMnO4 caused ocular damage 102,153 clinical accounts of ocular contact with aqueous KMnO4 solutions were limited to one case report 103 in which a man splashed a concentrated (but unspecified) KMnO4 solution into both eyes. Recognizing that the cornea may be more sensitive than mucous membranes to KMnO4, there are no data upon which to base an acute ocular RfV.
Other standards and guidelines
Assuming the 70-kg adult consumes 2 L of drinking water each day and the 10-kg child consumes 1 L/day, the daily Mn dose received at the 2 mg/L acute oral RfD calculated here is equivalent to 0.06 and 0.2 mg/kg/day, respectively. The US NRC 178 concluded the ‘estimated safe and adequate daily dietary intake’ of Mn was 0.3–0.6 mg for infants aged 6 months, 0.6–1.0 mg for infants aged 6–12 months, 1.0–1.5 mg for children aged 1–3 years, 1.5–2.0 mg for children aged 4–6 years, and 2.0–5.0 mg for adults, values equivalent to 0.15–0.2 mg/day for the 10-kg child and 0.03–0.08 mg/day for the 60-kg woman. In 2001, the Food and Nutrition Board, Institute of Medicine, 179 revised the value for newborns to 6 months of age as 0.003 mg/day. As age increased, the recommended intakes increased to 2.3 mg/day for adult males and up to 2.6 mg/day for lactating mothers. Thus, ingestion of drinking water at the 2 mg/L Do Not Consume level represents 100% of the child’s daily required Mn and 40–100% of the adult’s daily required Mn.
Taking into account consumption of grains, beverages (including tea), and vegetables, those sources provide only 70% of the Mn required each day for adult males and females, 180 yet no case of Mn deficiency in human beings has ever been described. 181 Nevertheless, the US EPA 77 adopted 0.14 mg/kg/day as an oral RfD for all forms of Mn and recommended adoption of a 10-day health advisory of 1 mg Mn/L as a 1-day exposure limit. The US EPA based the 10-day health advisory on ‘the upper end of the adequate intake for a 10 kg child (up to 1 mg/day)’ and considered the 1 mg/L as protective for adults (including the elderly and those with liver disease who may experience impaired biliary excretion). For infants younger than 6 months, the US EPA advised that the lifetime Mn health advisory for Mn of 0.3 mg/L be used even for acute exposures of 10 days ‘because of the concerns for differences in manganese content in human milk and formula and the possibility of higher absorption and lower excretion in young infants’.
The Do Not Consume limit is twice the US EPA 77 1 mg/L 10-day Health Advisory of 1 mg/L for an adult, and almost seven times the Lifetime Health Advisory of 0.3 mg/L recommended for short-term exposures for an infant less than 6 months of age. The Do Not Consume limit is 40-fold higher than the 0.05 mg/L secondary maximum contaminant level, which is a ‘reasonable goal for drinking water quality’ that is intended to control black to brown discoloration, black staining, and bitter metallic taste (40 CFR 143.3). Although people have apparently acclimated to drinking water containing up to 2.16 mg Mn/L 78 water, elevated Mn in water may not be tolerated by those who are not accustomed to its metallic taste.
Conclusion
The Do Not Consume and Do Not Use levels derived above are designed to aid water purveyors and regulatory officials in emergency planning. The Do Not Consume concentration is ~200–300 times less than the KMnO4 concentrations that can cause immediate symptoms in the human gastrointestinal tract. 62 –64 Consumption of 2 L of water at the Do Not Consume concentration is equivalent to 4 mg Mn/day or 36% of the 11 mg tolerable daily upper intake level. 179 As no quantitative ocular toxicity data were located, a Do Not Use level for KMnO4 to prevent ocular injury could be based on pH; solutions with a pH less than 2.0 are considered corrosive in the eye 182 –184 and complaints of ocular irritation tend to increase as solution pH declines. 185 Even at KMnO4 concentrations substantially higher (500 mg KMnO4/L or 174 mg Mn/L) than the limits derived here, the pH of KMnO4 in distilled water (Table 2) is greater than that considered corrosive by regulatory agencies.
The pH of drinking water with elevated KMnO4 can be less (Table 2) than that normally considered acceptable (6.5–8.5) range (40 CFR 143.3). Since water with Mn concentrations greater than 0.05 mg/L (40 CFR 143.3) has a pink to purple tint with a bitter or metallic taste and it stains fabrics and other porous materials, the concentrations derived here are most likely self-limiting. To utilize the values derived here, emergency response plans should account for the KMnO4 exposure scenario and the pH of the water as well as the public health and safety consequences associated with cautions placed on use of municipal water.
Footnotes
Authors’ Note
Mention of trade names, proprietary products, or specific equipment does not constitute an endorsement by NSF International, neither does it imply that other products may not be equally suitable. The drinking water Do Not Consume/Do Not Use limits derived here are based on examination of the toxicity of specific materials in mammalian systems. These limits are designed to protect the public from short-term adverse health effects associated with either ingestion or other direct contact (skin and eye) during bathing and showering. As such, beneficial uses of water intended for other than domestic use (e.g. firefighting, landscape or agricultural irrigation, sanitary or storm sewers, industrial process waters, and hemodialysis) are not considered here and the Do Not Consume/Do Not Use limits should not be applied to exposure situations other than potable water or to species other than mammals.
Funding
NSF International gratefully acknowledges financial support from the State of Pennsylvania Department of Environmental Protection.
Acknowledgment
ANSF International is indebted to the volunteer members of its Health Advisory Board for critical review of the draft manuscript.
