Abstract
Alzheimer’s disease (AD) could result from a multifactorial process involving both genetic predisposition and exposure to environmental factors like pesticides. A case control study of 70 patients of AD and 75 controls was done to examine the association between organochlorine pesticides (OCPs) and risk of AD. OCPs (hexachlorocyclohexane (HCH), aldrin, dieldrin, endosulfan, pp′-dichlorodiphenyldichloroethylene (pp′-DDE), op′-DDE, pp′-dichlorodiphenyltrichloroethane (pp′-DDT), op′-DDT, pp′-dichlorodiphenyldichloroethane (pp′-DDD) and op′-DDD) were extracted from blood and quantitatively estimated using gas chromatography. A Mann–Whitney U test revealed significant difference in β-HCH levels (U = 1237.00, W = 4087.00, z = −6.296, p = 0.000, r = −0.71), dieldrin levels (U = 1449.00, W = 4299.00, z = −5.809, p = 0.000, r = −0.68) and pp′-DDE levels (U = 2062.00, W = 4912.00, z = −2.698, p = 0.007, r = −0.59) between AD patients and controls. In conclusion, this study supports epidemiological studies that associate exposure to pesticides with increased risk of AD, and we identified the specific pesticides β-HCH, dieldrin and pp′-DDE that are associated with the risk of AD in the north Indian population. However, further research is needed to establish the potential role of these OCPs as an etiologic agent for AD case.
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disease defined by specific neuropathological and neurochemical features. 1 –5 The etiology of AD has not been resolved yet. AD has been linked to at least five chromosomes. However, only a portion of AD case account for known genetic components. 6 It has been suggested that AD could result from a multifactorial process involving both a genetic predisposition and an exposure to environmental factors. 7 Pesticides are environmental chemicals used extensively throughout the world including India. Of the 2.5 million tons of pesticides used worldwide each year, less than 0.1% reaches the pest target. 8 Thus, 99% of currently applied pesticides are being released indiscriminately into the environment, many of which will persist for years and travel far from the point of application. 8 Many pesticides exert their killing effects through neurotoxic mechanisms. Historically, most attention was focused on acute effects and relatively large exposures to humans, but in recent years neurological effects from chronic and low-level exposures have been more widely studied in people who apply pesticides and in general public. 9 –11 Although studies are not fully consistent, they demonstrate the neurologic impacts at lower levels of chronic exposure to neurotoxic pesticides. These neurologic impacts include neurobehavioral performance impairments, sensory, motor and nerve dysfunction, memory, attention, visual–spatial processing, pattern memory and others. Pesticides have been implicated as one of the major environmental risk factors for neurodegenerative disease. 12,13 Toxicological studies revealed that some pesticide families (carbamates, organophosphates, organochlorines and bipyridyles) can cause serious damage to the nervous system. 14 The basic mechanism of action for most organochlorine pesticides (OCPs) is an alteration in the transfer of a signal along a nerve fiber and across the synapse from one nerve to another or from a nerve to a muscle fiber. Different classes of pesticides altered this process in different ways, but the end result is an alteration in normal nerve signal propagation. OCPs act primarily by altering the movement of ions across the nerve cell membranes, thus changing the ability of the nerve to fire. Many studies have shown significant association between exposure to pesticides and AD. 7,15 –17 However, most of these studies have not identified specific pesticides that may contribute to AD. In India, of all the pesticides used, 40% belong to the organochlorine class of chemicals. Among OCPs, hexachlorocyclohexane (HCH), dichlorodiphenyltrichloroethane (DDT), dieldrin and endosulfan are persistent environmental pollutants. 18 OCPs are chemically stable, strongly lipophilic as well as very slow in degradation. These compounds are concentrated high up in the food chain and can be detected in the diet including drinking water. OCPs can disturb the function of the mitrochondrial system and produce free radicals that induce oxidative stress. 19,20 Many studies have demonstrated that OCPs induce oxidative stress and are neurotoxic. 21,22
Thus, the combination of their persistence in the environment and their potential to neurotoxicity suggests that the risk of AD may be significantly associated with OCPs. This study was designed to examine OCP levels in the blood without exposing them to the same in the patients of AD and controls.
Material and methods
Study population and design
In this study, subjects (AD and control) were examined by neurologist in out patients department of neurology during February 20, 2010 to December 30, 2011, Institute of Human Behaviour and Allied Science, Delhi. A total of 145 subjects were enrolled for the study, which included 70 patients in the AD group and 75 subjects in the control group.
Subjects were of 50–85 years old with probable AD, defined by the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) 23 and a magnetic resonance imaging (MRI; diffuse cerebral atrophy)/computed tomography (CT; reduction in the size of the brain, cerebral ventricles and cortical atrophy)/positron emission tomography (PET; global decreases in metabolic function, particularly in the temporoparietal regions) brain scans supporting the clinical diagnosis of AD were eligible. Additional inclusion criteria were a score of 0–23 on the Mini-Mental State Examination and a Clinical Dementia Rating score of ≥0.5. Control group comprised age-matched healthy volunteers.
Subjects were excluded in both the cases and control groups, if there was no consent for participation in the study, history of cerebral stroke, epilepsy, a history of head trauma and other concomitant disease potentially associated with dementia, chronic intake of drugs affecting cognitive processes, moderate-to-severe depressive episode and familial history of any kind of cognitive/behavioral abnormality. Nutritional deficiency, metabolic abnormalities and central nervous system infections were ruled out.
Standard protocol approvals, registration and patients consent
The study protocol and the Informed Consent Form were reviewed and approved by Institutional Ethics Committee of our institute. Before any study specific procedures were performed, written consent was obtained from each subject.
Sample collection and estimation of OCP levels
Blood samples were collected using standard venipuncture techniques and stored at −80°C until analysis. The OCPs α-HCH, β-HCH, γ-HCH, aldrin, dieldrin, a-endosulfan, b-endosulfan, pp′-dichlorodiphenyldichloroethylene (pp′-DDE), op′-DDE, pp′-DDT, op′-DDT, pp′-dichlorodiphenyldichloroethane (pp′-DDD) and op′-DDD were assayed. OCPs such as HCH, DDE, DDT and DDD contain various isoforms, which have unique toxicological significance making it necessary to examine various isoforms of OCPs in blood sample. The compounds selected for assay were based on previous findings 24 ,25 that indicated the pesticides most commonly found in human samples and based on the compounds assayed by the Environmental Protection Agency. Samples were analyzed on Perkin Elmer gas chromatograph equipped with 63Ni electron capture detector under standard operating procedure. 25
OCPs were extracted from blood according to the method described by Bush et al. 26 Quantitative analysis of OCPs in each sample was analyzed by comparing the peak area with those obtained from a chromatogram of a mixed organochlorine standard (Supelco, Sigma-Aldrich, New Delhi, India) of known concentration. The detection limit of the detector was <0.05 pg per chloroethylene with nitrogen as a carrier gas. The detection limit of the method was 4 pg/ml for each OCP. For quality control process, five blood samples in triplicate were spiked with a mixed standard of OCPs at 5 and 25 ng/ml. The average recoveries of prepared samples exceeded 95%. The case and control samples were run in the same analytical batches, and moreover, a quality check sample was always run with each set of samples for pesticide analysis to maintain accuracy.
Statistical analysis
The statistical analysis was carried out using SPSS version 14.0. Data are expressed in the form of OCPs detected in the percentage (%) of subjects and mean ± SD. The p < 0.05 was considered statistically significant. Independent t test was used for comparing the difference between detected levels of OCPs in both groups. Mann–Whitney U test was used for comparing the difference between the mean levels of OCPs in AD and control group. Logistic regression was performed, with β-HCH, dieldrin and pp′-DDE as predictor variables for AD versus control status.
Results
Demographic characteristics of the study population are given in Table 1. There was no significant difference between the AD and controls in terms of demographic characteristics and genders. OCPs, op′-DDE, op′-DDT, pp′-DDD and op′-DDD were not detected in the present study. The unpaired sample t test results (Table 2) showed a highly significant difference in the groups in terms of presence of β-HCH (p < 0.001), dieldrin (p < 0.001) and pp′-DDE levels (p < 0.05). Post hoc test done by Mann–Whitney U test (Table 3) revealed highly significant difference in β-HCH levels (p < 0.001), dieldrin levels (p < 0.001) and pp′-DDE levels (p < 0.01) in AD and controls.
Demographic characteristics of AD and controls

AD: Alzheimer’s disease.
Presence of OCPs in AD and control subjects

AD: Alzheimer’s disease; OCP: organochlorine pesticide; HCH: hexachlorocyclohexane; pp′-DDT: pp′-dichlorodiphenyltrichloroethane; pp′-DDE: pp′-dichlorodiphenyldichloroethylene.
a p < 0.001 highly significant.
b p < 0.05 significant.
Distribution of OCP in AD and control subjectsa
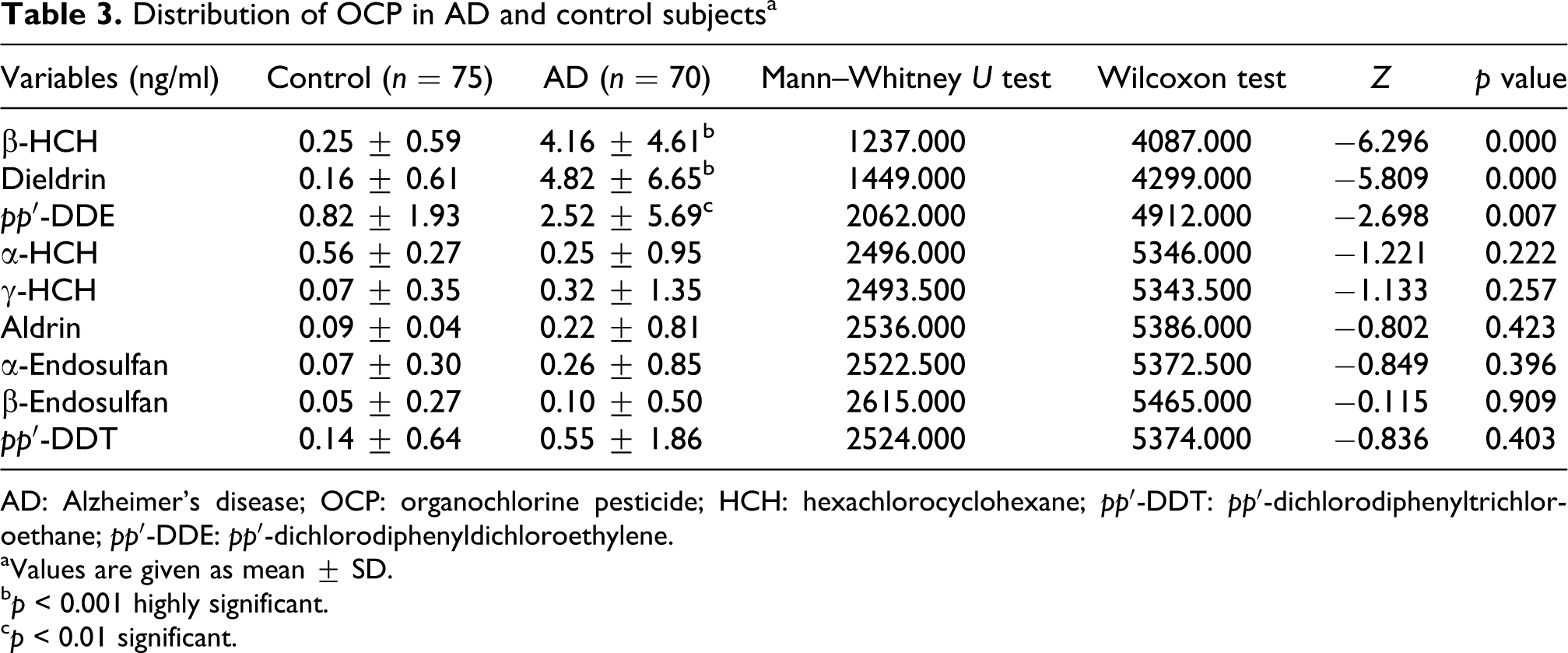
AD: Alzheimer’s disease; OCP: organochlorine pesticide; HCH: hexachlorocyclohexane; pp′-DDT: pp′-dichlorodiphenyltrichloroethane; pp′-DDE: pp′-dichlorodiphenyldichloroethylene.
aValues are given as mean ± SD.
b p < 0.001 highly significant.
c p < 0.01 significant.
Direct logistic regression was performed to assess the impact of number of factors on the likelihood of developing AD. The model contained three independent variables (β-HCH, dieldrin and pp′-DDE levels). The full model containing all 3 predictors (Table 4) was statistically significant (χ 2 = 169.03, p <0.001) in all the subjects (N = 145), indicating that the model was able to distinguish between the patients with AD and controls. As shown in Table 4, all three independent variables made a unique statistically significant contribution to the model.
Variables in the equation

HCH: hexachlorocyclohexane; pp′-DDE: pp′-dichlorodiphenyldichloroethylene; OR: odds ratio; CI: confidence interval; SE: standard error; Sig.: p value; B: beta coefficient; Wald: Wald statistics of the variable in the model.
aVariable/variables entered on step 1: β-HCH, dieldrin and pp′-DDE.
Discussion
OCPs are a large class of multipurpose chlorinated hydrocarbon chemicals. Their extensive use in public health programs and agriculture has resulted in severe environmental pollution leading to potential health hazards. These compounds are an integral part of the ecosystem. These are concentrated up in the food chain. Humans, being at the top of the food chain, are most vulnerable to health effects as ingestion of toxic chemicals is several folds higher through the process of biomagnification. OCPs are highly lipophilic, break down slowly in the environment, accumulate in the blood and fatty tissues and are often associated with neurodegenerative disease. 27 The effect of OCPs is probably initiated due to an imbalance between pro-oxidants and antioxidant defense mechanism in individuals. 28 Oxidative stress, generated by OCPs, causes peroxidation of lipids and formation of other reactive intermediates that may also result in increased oxidative damage of biomolecules as well as formation of protein carbonyl adducts, which lead to the disruption of normal protein metabolism. Thus, exposure to OCPs may contribute to cholinergic system deficiency and the production of free radicals in AD but this remains nonspecific. One theory on the pathogenesis of AD postulates that neurodegeneration is the result of oxidative stress and damage to vulnerable cerebral tissues. The brain’s high lipid content and poor antioxidant defenses render it an ideal target for free radical attack. The transfer of a signal along a nerve occurs by changes in the electrical potential across the nerve cell membrane, which is created by the movement of ions in and out of the cell (ion channel). Different classes of pesticides inhibit this process in different ways, but the end result is an alteration in normal nerve signal propagation. The primary action of chlorinated pesticides is interfering with axonal transmission by disrupting ion flux, leading to overstimulation of the nerves and uncontrolled neuronal discharge.
In this case control study, we found significant increased level of β-HCH, dieldrin and pp′-DDE in patients with AD when compared with controls. Many studies have shown an association between OCP levels and neurodegenerative disease, 29,30 while Richardson et al. 27 showed no association. Li et al. 31 estimated global emissions of β-HCH from the usage of technical HCH to be between 1945 and 2000 at 8,50,000 tons, of which 2,30,000 tons were emitted into the atmosphere over the same period. Its chemical structure seems to confer the greatest physical and metabolic stability. The isomers of HCH (α-HCH, β-HCH and γ-HCH) can enter human body via food or drinking water contaminated with HCH. Among the HCH isomers, β-HCH is the most slowly excreted from the body. We also found β-HCH in some controls, but the mean level was much lower compared with patients with AD. This result suggests that there are other factors that may interact with β-HCH exposure to alter the risk of AD. For patients with AD, it is possible that genetic polymorphism in enzymes that metabolize β-HCH may contribute to their high levels of β-HCH.
Technical grade DDT is a mixture of three forms, pp′-DDT (85%), op′-DDT (15%) and oo′-DDT (trace amounts). In our study, pp′-DDE, a major metabolite of DDT (1,1,1-trichloro-2,2-bis(p-chlorophenyl)ethane), was significantly high (p = 0.015) in AD patients when compared with controls. pp′-DDE is more persistent than DDT. Fleming and coworkers 20 found that DDT and its metabolites were more likely to be found in AD brain tissue than in the brain tissue of those with PD or of control samples. Our data also concurs with the Fleming and coworkers results, although in both the studies, the sample size is small. We also found that dieldrin levels were significantly high in AD patients when compared with controls (p = 0.000). Dieldrin is a major metabolite of aldrin. Point of entry of aldrin into the environment has most typically been the soil because of their strong adsorption to soils and their low aqueous solubilities. Biodegradation of aldrin is generally slow and by hydrolytic process, it is converted into dieldrin. Bioconcentration and bioaccumulation of these compounds and their residues are significant and, in addition to their being constant contaminants of soil, water and air, they are often found in aquatic organisms, wildlife, foods and humans. 32 Aldrin is quickly metabolized to dieldrin, mediated by the mixed-function mono-oxygenases. Liver tissue has the largest activity but several other organs such as the lung and skin also have this enzymatic process. Dieldrin is then metabolized at a much slower rate to hydrophilic metabolites. Because dieldrin is also extremely apolar, it displays a high affinity to fat and is thus retained in animal fats, plant waxes and other similar organic matter in the environment. This fat solubility can lead to a progressive accumulation of dieldrin in the food chain, which theoretically could eventually produce concentrations in organisms that might exceed lethal limits to predators or consumers.
Direct logistic regression results of the present study showed that all the independent variables (β-HCH, dieldrin and pp′-DDE levels) made a unique statistically significant contribution to the model, recording an odds ratio of 7.2, 6.0 and 4.8, respectively. This indicates that for additional 1 unit increment in β-HCH, dieldrin and pp′-DDE levels, chances of developing AD in a population will be 7, 6 and 4 times, respectively. We also found that in some patients with AD, the β-HCH, dieldrin and pp′-DDE were not detected, which suggest the multifactorial etiopathologic nature of AD.
In conclusion, the present study supports epidemiological studies 7 ,15 –17,27 that associate exposure to pesticides with increased risk of AD, and we identified the specific pesticides β-HCH, dieldrin and pp′-DDE that are associated with the risk of AD in the north Indian population.
Footnotes
Funding
The author (N.K.S.) was provided with Senior Research Fellowship (Reference No. 3/1/2/5/Neuro/2004-NCD-I) from Indian Council of Medical Research (ICMR), New Delhi, India.
Conflict of interest
The authors declared no conflicts of interest.
