Abstract
Oxidative stress has been proposed as a possible mechanism involved in manganese (Mn) toxicity. Using natural antioxidants against metal-induced hepatotoxicity is a modern approach. The present study investigated the beneficial role of silymarin, a natural flavonoid, in Mn-induced hepatotoxicity focusing on histopathology and biochemical approaches. Male Wistar rats were exposed orally to manganese chloride (20 mg/mL) for 30 days followed by intraperitoneal cotreatment with silymarin (100 mg/kg). Exposure to Mn resulted in a significant elevation of the plasma marker enzyme activities and bilirubin level related to liver dysfunction of reactive oxygen species (ROS) production and hepatic oxidative stress indices. This metal reduced the activities of superoxide dismutase, catalase and glutathione peroxidase and nonenzymatic antioxidant levels such as reduced glutathione, total sulfhydryl groups and vitamin C. In addition, it caused hepatic hemorrhage, cellular degeneration and necrosis of hepatocytes as indicated by liver histopathology and DNA fragmentation studies. Coadministration of silymarin alleviated Mn oxidative damage effects by inhibiting ROS generation. Histological studies also supported the beneficial role of silymarin against Mn-induced hepatic damages. Combining all, results suggested that silymarin could protect hepatic tissues against Mn-induced oxidative stress probably through its antioxidant activity. Therefore, its supplementation could provide a new approach for the reduction in hepatic complication due to Mn poisoning.
Introduction
Manganese (Mn) is the fourth most widely employed industrial metal worldwide. 1 General population may be exposed to Mn from food, water contamination and air pollution caused by industrial emission and fuel additives, as an antiknock gasoline (methylcyclo-pentadienyl manganese tricarbonyl) and fungicides (e.g. maneb and mancozeb). Other occupational settings including welding and steel or dry battery manufacturing are also implicated. 2,3 The increasing environmental exposure justifies the extensive study performed in the last years for a correct assessment of the potential health risk due to this metal. 4 The latter is an essential trace element and its deficiency leads to several diseases in human. 5 Furthermore, it acts as a catalytic cofactor in a number of redox enzymes required for several metabolic processes. 6 When its intake exceeds the tolerable limit, it exerts toxic effects leading to cell death. 7,8 The toxicity and oxidative stress caused by Mn is well investigated in nervous tissues 9,10 but poorly understood in hepatic cells. The exact cellular mechanisms by which Mn produces hepatotoxicity in vivo are unknown, but the advancement of research demonstrates that the oxidative stress is the key contributor in Mn-induced hepatic injury as it is known to produce reactive oxygen species (ROS). 11 Recently, Huang et al. have indicated that excess Mn induces a set of toxicological activities such as oxidative stress, changes in lipid profile and hepatic dysfunction and more critical consequences. 12 Moreover, Mn has a high affinity for sulfhydryl (thiol) group in proteins, reduced glutathione (GSH) and amino acids, especially cysteine. 13 It may disturb the antioxidant barrier via inhibition of the functional SH groups. Thus, the maintenance of redox metal homeostasis is supposed to be an effective way for preventing tissue damage. Metal-chelation therapy is a modern pharmacotherapy used to chelate redox-active metals and remove toxic metals from living systems to avoid metal poisoning. The chelating agent must possess donor electron groups such as carboxyl, amine, hydroxyl and/or thiols group. 14 Several Mn chelators, such as EDTA or para-aminosalicylic acid, have successfully been tried in removing Mn and have beneficial effects against various neurodegenerative diseases accompanied by Mn accumulation. 15,16 Thus, the recent research deals with the mitigation of Mntoxicity through dietary supplementation of natural antioxidants like ascorbic acid (AA) and vitamin E or herbal antioxidant like Melissa officinalis. 17 –19 Effect of silymarin against Mn-induced hepatic impairment was the first step in this direction. Silymarin, considered as a medicinal plant, is purified from milk thistle plant (Silybum marianum (L.) Gaertn) originating from Mediterranean area and used for centuries in the treatment of liver diseases. Silibinin, the main bioactive reagent of silymarin, has been reported to possess antioxidant, 20,21 anti-inflammatory, 22 antiapoptotic 23 and anticancer properties, 24 and thus, has a potential protective effect against various malignant diseases. 25 In fact, silymarin has been shown to be a potent scavenger of a variety of ROS, including superoxide anion, hydroxyl radical and nitrogen dioxide radical. 26 In addition, silymarin exerts its antioxidant properties by increasing the expression of enzymes, including superoxide dismutase (SOD) and glutathione peroxidase (GPx). 27 The possible hepatoprotective activity of silymarin against Mn-induced hepatotoxicity has not been reported so far. Therefore, in this study, we aimed to evaluate the hepatoprotective effects of the silymarin by using the Mn-induced subchronic oxidative liver injury in adult rats.
Materials and methods
Chemicals and reagents
Silymarin and all other chemicals, required for all biochemical assays, were obtained from Sigma Chemicals Co. (St Louis, France).
Treatment of animals and tissue preparation
A total of 28 male rats of Wistar strain weighing 190 ± 23 g were obtained from Central Pharmacy (SIPHAT, Tunisia). They were fed pellet diet purchased from the Industrial Society of rodents diet (SICO, Sfax, Tunisia). Diet composition was detailed by Fetoui et al. 28 All animal procedures were conducted in strict conformation with the local Institute Ethical Committee Guidelines for the Care and Use of laboratory animals of our institution. Tissue samples for this study were obtained from rats treated similarly as in the animal model described and published elsewhere. 29,30 Briefly, rats were randomly divided into four groups of seven animals each: The first group of rats served as the controls, received ad libitum distilled water and 0.5 mL vehicle solution of silymarin given daily by intraperitoneal (i.p.) injection. The second group (Mn group) received a solution of manganese chloride (MnCl2; 20 mg/mL) through drinking water. Animals in the third group (Mn + SIL group) were given a single i.p. injection of silymarin (100 mg/kg body weight per day), 24 h after the administration of MnCl2 solution. The fourth group (SIL group) was given daily a single dose of silymarin (100 mg/kg body weight). At the end of the experimental period (30 days), the animals of different groups were killed by cervical decapitation to avoid stress conditions. The liver was quickly excised, rinsed in ice-cold physiological saline, weighed and tissue homogenate was prepared in the appropriate buffer at 10% (w/v) as indicated in the procedures measurement of each parameter. The supernatant aliquots were stored at −80°C and used for biochemical assays.
Biochemical assays
Measurement of ROS generation
ROS levels in liver were measured using 2′,7′-dichlorofluorescein diacetate (DCFH-DA).
31,32
Briefly, the liver homogenate was diluted 1:20 times with ice-cold Locke’s buffer (154 mM NaCl, 5.6 mM KCl, 3.6 mM NaHCO3, 2.0 mM CaCl2, 10 mM
Assessment of serum specific markers related to hepatic dysfunction
The activities of serum aspartate transaminase (AST), alanine transaminase (ALT), lactate dehydrogenase (LDH), total bilirubin (TB) and gamma glutamyl transferase (GGT) were assayed spectrophotometrically according to the standard procedures using commercially available diagnostic kits (Biomaghreb, Ariana, Tunisia, Ref. 20043; 20047; 20012; 20102; 200222, respectively).
Determination of liver enzymatic antioxidant activities
Total SOD activity was evaluated by measuring the inhibition of pyrogallol activity as described by Marklund and Marklund. 33 One unit (U) corresponded to the enzyme activity required to inhibit half of the pyrogallol oxidation. SOD activity was expressed as Units per milligram of protein.
Catalase (CAT) activity was determined by the method of Aebi 34 using hydrogen peroxide (H2O2) as substrate. A decrease in absorbance due to H2O2 degradation was monitored spectrophotometrically at 240 nm for 1 min and the enzyme activity was expressed as µmol H2O2 consumed per minute per milligram of protein.
GPx activity was measured according to Flohe and Gunzler. 35 The activity was expressed as nmoles of GSH oxidized per minute per milligram of protein.
Determination of nonenzymatic antioxidants levels in liver tissue
Total cellular GSH levels were determined as described previously by Ellman. 36 Distilled water was used as blank and standards were prepared with a commercial GSH solution (10–100 µmol/L). The slope of the reaction rate was used to calculate the GSH content. Total GSH content was expressed as micromoles per gram of tissue.
Total thiols estimation was determined according to the methods of Ellman. 36 Total thiols content was expressed as nanomoles per milligram of protein.
A content was determined spectrophotometrically by dinitrophenyl-hydrazine method. 37 The calibration curve was prepared using a standard ranging between 0 and 100 µg of AA per milliliter. Results were expressed as microgram per gram of tissue.
Measurement of lipid peroxidation, hydroperoxides, protein carbonyl and nitrite levels in liver tissue
The extent of lipid peroxidation by measuring thiobarbituric acid reactive substances (TBARS) in terms of malondialdehyde (MDA) formation was measured according to the method of Draper and Hadley. 38 The MDA values were calculated using 1,1,3,3-tetraethoxypropane as the standard and expressed as nanomoles of MDA per gram of tissue.
Lipid hydroperoxides (LOOHs) were estimated using the ferrous oxidation in xylenol orange assay (FOX assay) described by Jiang et al. 39 The amount of hydroperoxide produced was calculated using the molar extinction coefficient of 4.59 × 104 M−1 cm−1 and the results were expressed as nanomoles per milligram of protein.
Protein carbonyls were determined using 2,4-dinitrophenylhydrazine and the basis of the assay involved the reaction between protein carbonyl and dinitrophenylhydrazine to form protein hydrazone. 40 The absorbance was measured at 370 nm, using the molar extinction coefficient of DNPH, e = 22,000 M−1 cm−1 and the results were expressed as nanomoles of carbonyl per milligram of protein.
Nitrite levels (NO2 −), served as an indicator of nitric oxide production, were measured by a colorimetric assay using Greiss reagent. 41 Concentrations were determined by using 2.0–80 μmol of sodium nitrite as a standard. The results were expressed as micromoles of nitrite accumulated per milligram of protein.
Quantitative protein determination
Protein concentration in liver homogenates was measured by Bradford method (BCA Protein Assay Kit, Pierce Biotechnology Inc., Rockford, USA) using bovine serum albumin as standard.
DNA fragmentation assay
To verify the extent of DNA damage, we perform the DNA smear technique. This technique consists of electrophoresis of full genomic extracted DNA resolved in agarose gel. The DNA was extracted according to Kanno et al. 42 Briefly, frozen liver samples (50 mg) were homogenized in lysis buffer (10 mM Tris–HCl, 50 mM NaCl, 1 mM EDTA, 0.5% SDS, pH 8). The lysate was incubated for 3 h with proteinase K (final concentration 100 µg/mL) at 56°C. After incubation, the solution was centrifuged at 10,000g for 20 min. DNA was extracted from the supernatant with equal volume of phenol/chloroform mixture. The aqueous layer was taken in a fresh tube. 1/10th volume of 3 M sodium acetate (pH 7.4) and 2.5-fold volume of ethanol were added and centrifuged. The precipitated DNA was washed with 80% ethanol. The pellets were dissolved in Tris–EDTA buffer (10 mM Tris–HCl and 1 mM EDTA), RNase A (DNase-free) was added and the mixture was incubated at 37°C for 1 h and genomic DNA yield was measured spectrophotometrically at 260 nm. DNA samples were carried out at 80 V for 1 h on 0.8% agarose gel in Tris-acetate-EDTA (TAE) buffer, containing 0.5 μg/mL ethidium bromide. The gel was observed under an ultraviolet lamp and photograph has been taken
Histopathological analysis
Portions of the liver were placed in 10% neutral formalin. Fixed tissues were embedded in paraffin, sectioned to a thickness of 5 µm and stained with hematoxylin and eosin, and examined with Leica® microscope fitted with Sony® digital camera to capture images for histological studies.
Statistical analysis
Results were expressed as mean ± SD. All analyses were carried out with GraphPad Prism 4.02 for Windows (GraphPad Software, San Diego, California, USA). Significant differences between treatment effects were determined by one-way ANOVA, followed by Tukey’s post hoc test for multiple comparisons with statistical significance of p < 0.05.
Results
Effects on body weight and liver body weight ratio
Table 1 shows the changes in the body weight and absolute and relative weights of liver in experimental rats at the end of the treatment. No significant changes in final body weights were observed between groups. A significant (p < 0.05) decrease in liver body weight ratio was noted in Mn-treated rats. These changes induced by Mn intoxication were (p < 0.05) alleviated significantly by silymarin coadministration. No significant changes were observed between control and silymarin-treated rats
Effects of different treatments on body weight and absolute (g) and relative weight of liver (absolute liver weight/body weight × 100) in experimental animals: controls (C) and rats treated with Mn, SIL or their combination (Mn + SIL)a

SIL: silymarin; Mn: manganese.
aValues are expressed as mean ± SD of six rats in each group.
b p < 0.01 Mn group versus control group.
c p < 0.05 Mn + SIL group versus Mn group.
Effects on serum biochemical parameters
Table 2 shows the status of enzymes activities and bilirubin level in the serum of control and experimental rats. Oral administration of Mn (p < 0.05) increased the activities of biomarkers like AST, ALT, LDH and GGT and level of TB significantly when compared with those of controls. Coadministration of silymarin (p < 0.05) decreased these biomarkers of hepatotoxicity significantly when compared with Mn-treated rats. Administration of silymarin (100 mg/kg body weight) to normal rats did not show any significant effect on hepatic markers.
Changes in the activities of serum AST, ALT, LDH, GGT and total bilirubin levels in control and experimental ratsa
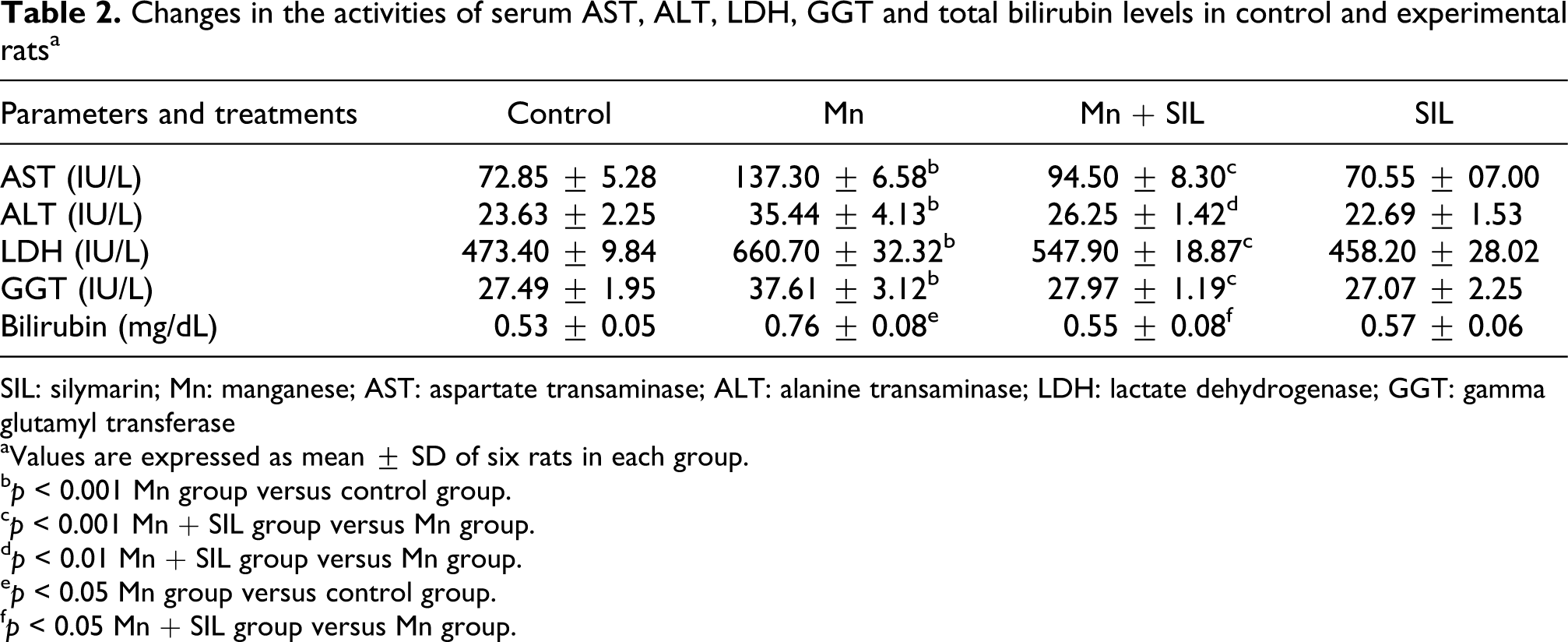
SIL: silymarin; Mn: manganese; AST: aspartate transaminase; ALT: alanine transaminase; LDH: lactate dehydrogenase; GGT: gamma glutamyl transferase
aValues are expressed as mean ± SD of six rats in each group.
b p < 0.001 Mn group versus control group.
c p < 0.001 Mn + SIL group versus Mn group.
d p < 0.01 Mn + SIL group versus Mn group.
e p < 0.05 Mn group versus control group.
f p < 0.05 Mn + SIL group versus Mn group.
Effect on ROS production
To evaluate the effects of silymarin on Mn-induced oxidative stress, we investigated the levels of ROS production. As shown in Figure 1, significant increase in hepatic ROS level was observed in Mn exposed animals when compared with control group; whereas, the cotreatment with silymarin (p < 0.05) reduced ROS generation significantly when compared with Mn-treated rats.

Effects of different treatments on the levels of ROS in the liver of controls (C) and rats treated with Mn, SIL or their combination (Mn + SIL). Data are expressed as fluorescence units per minute per milligram of protein. Values are expressed as mean ± SD of six rats in each group. Mn group versus control group: ***p < 0.001. Mn + SIL group versus Mn group: ¥¥¥ p < 0.001. ROS: reactive oxygen species; SIL: silymarin; Mn: manganese.
Effects on lipid peroxidation, protein oxidation and nitrite
Table 3 shows the changes in the levels of lipid peroxidation product in experimental animals. In Mn-treated rats, the level of thiobarbituric acid reactive substances (MDA), LPO, protein carbonyl content (PCO) and nitrites were (p < 0.05) increased significantly. Coadministration of silymarin (100 mg/kg body weight) along with Mn (p < 0.05) lowered the levels of these products significantly in the liver.
Effects of different treatments on lipid and protein parameters: LOOHs, TBARS in terms of MDA, PCO and NO production in the liver of controls (C) and rats treated with Mn, SIL or their combination (Mn + SIL)a

SIL: silymarin; Mn: manganese; LOOHs: lipid hydroperoxides; TBARS: thiobarbituric acid reactive substances; MDA: malondialdehyde; PCO: protein carbonyl content; NO: nitric oxide.
aValues are expressed as mean ± SD of six rats in each group. Data are expressed as LOOHs (nmoles /mg protein), TBARS (nmoles of MDA/g of tissue), PCO (nmoles/mg protein) and nitrite (NO2 −) levels (µmoles/mg protein).
b p < 0.001 Mn group versus control group.
c p < 0.001 Mn + SIL group versus Mn group.
d p < 0.01 Mn + SIL group versus Mn group.
Effects on nonenzymatic antioxidant levels in liver
Table 4 shows the levels of hepatic nonenzymatic antioxidants namely reduced GSH, total thiol group and vitamin C in the liver of control and experimental rats. A significant (p < 0.05) decrease in the levels of nonenzymatic antioxidants was noticed in rats treated with Mn when compared with control rats. Cotreatment with silymarin (100 mg/kg body weight) along with Mn (p < 0.05) restored the levels of nonenzymatic antioxidants significantly in the liver when compared with Mn-treated rats.
Effects of different treatments on nonenzymatic antioxidant status in the liver of Mn-treated rats, controls (C) and rats treated with Mn, SIL or their combination (Mn + SIL)a

SIL: silymarin; Mn: manganese; GSH: glutathione.
aValues are expressed as mean ± SD of six rats in each group. Data are expressed as GSH (µmoles/g tissue), total thiol (nmoles/ mg protein) and vitamin C (µg/g tissue).
b p < 0.001 Mn group versus control group.
c p < 0.001 Mn + SIL group versus Mn group.
d p < 0.05 Mn + SIL group versus Mn group.
e p < 0.01 Mn + SIL group versus Mn group.
Effects on the activities of antioxidant enzymes on the liver
Figure 2 shows the levels of enzymatic antioxidants namely SOD, CAT and GPx in the liver of control and experimental rats. A significant (p < 0.05) depletion in the activities of enzymatic antioxidants in Mn-treated rats was observed. Cotreatment with silymarin increased (p < 0.05) the levels of antioxidants significantly in the liver of Mn-treated rats.
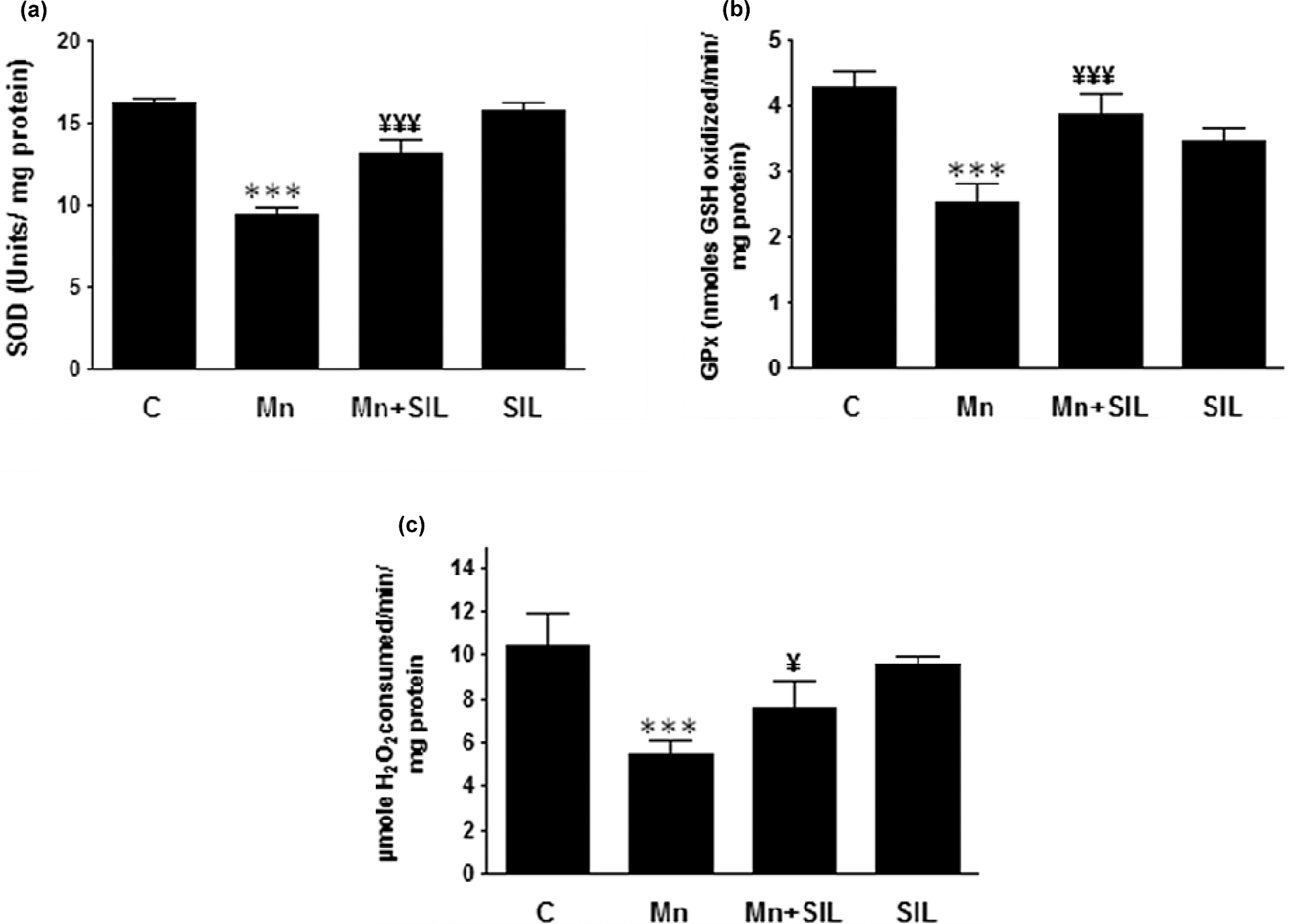
Effects of different treatments on antioxidant enzyme activities (CAT, GPx and SOD) in the liver of controls (C) and rats treated with Mn, SIL or their combination (Mn + SIL). (a) SOD, (b) GPx and (c) CAT. Values are expressed as mean ± SD of six rats in each group. Mn group versus control group: ***p < 0.001. Mn + SIL group versus Mn group: ¥¥¥ p < 0.001 and ¥ p < 0.05. SIL: silymarin; Mn: manganese; SOD: superoxide dismutase; GPx: glutathione peroxidase; CAT: catalase.
Effects on oxidative DNA damage
As shown in Figure 3, a smear (hallmark of necrosis) reflected by a higher Et/Br fluorescence intensity without ladder formation on agarose gel, indicating random DNA degradation, was observed through the entire lane of DNA liver samples of Mn-treated rat. Silymarin cotreatment (SIL + Mn) exerted a protective effect against Mn-induced damages by reducing the smear formation. No difference in appearance could be observed in DNA damage among the control and the silymarin group.

Agarose gel electrophoresis of DNA fragmentation. DNA isolated from experimental liver tissues was loaded into 0.8% (w/v) agarose gels. M: marker (1 kb DNA ladder); Lane 1: DNA isolated from normal liver; Lane 2: DNA isolated from manganese intoxicated liver; Lane 3: DNA isolated from silymarin-cotreated liver samples and Lane 4: DNA isolated from silymarin-treated liver samples.
Histological assessment
As shown in Figure 4, the results of histopathological evaluation showed that silymarin exhibited hepatoprotective effects against Mn-induced liver injury. The liver of control rats showed normal liver architecture, polyhedral hepatocytes and the boundaries of the sinusoids exhibited a single layer of fenestrated endothelial and kupffer cells (Figure 4(A)). Silymarin alone had no significant effects on liver histology (Figure 4(F)). While, Mn treatment caused visible histological damages in hepatocytes. They were characterized by the appearance of hepatocellular necrosis, leukocyte infiltration and activated kupffer cells. In addition, the sinusoids between the plates of hepatocytes were markedly dilatated in the liver of Mn-treated rats (Figure 4(B) to (D)). Coadministration of silymarin in Mn-treated group alleviated partially the liver damages with mild portal inflammation (Figure 4(E)).
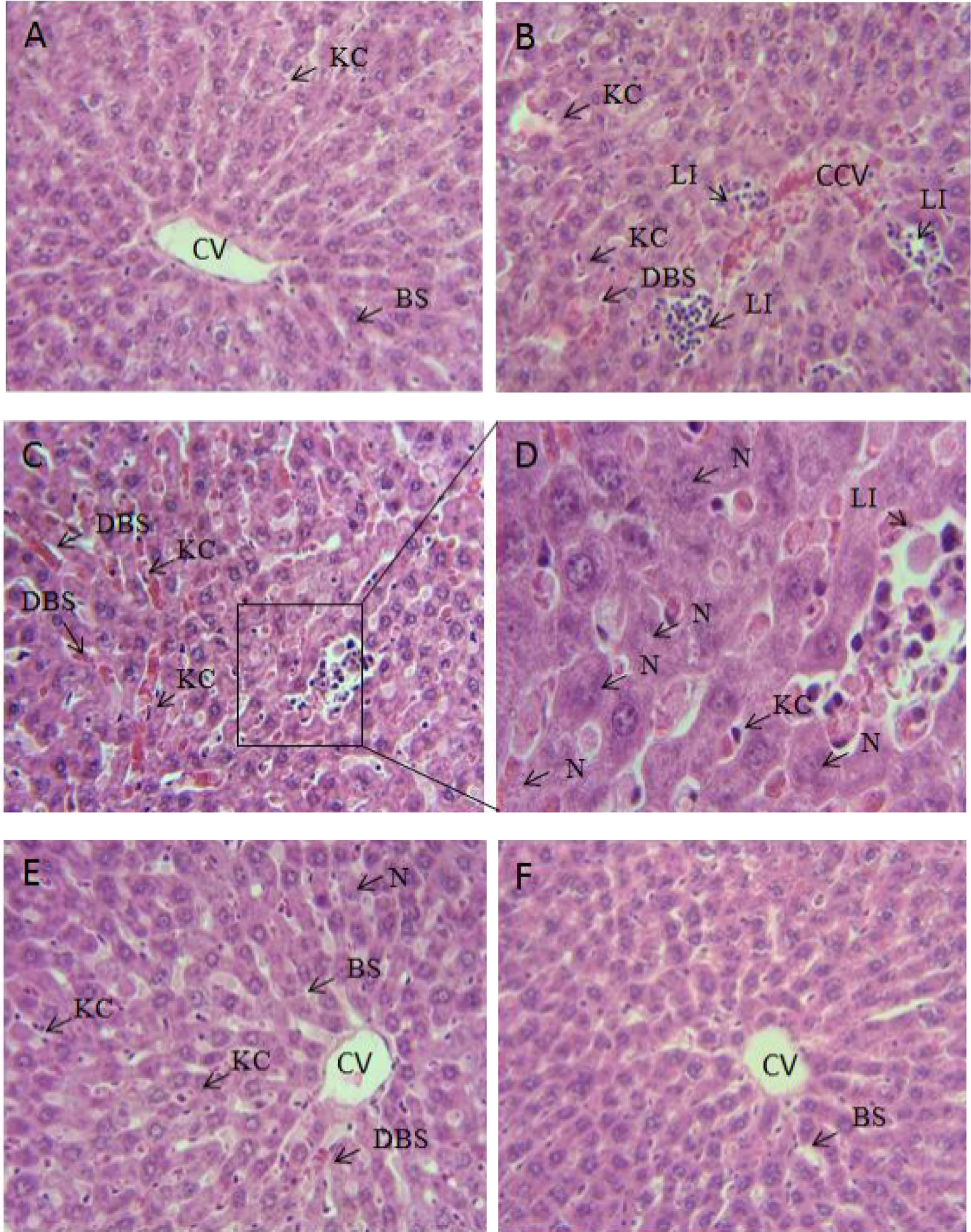
Hematoxylin and eosin stained liver sections of adult rats: controls (A), manganese treated rats (B, C, D), manganese treated rats cotreated with silymarin (E) or treated only with silymarin (F). Each microphotograph represents a section from an individual liver. Magnification: X400 (A, B, C, E, F); X1000 (D). The arrows indicate: BS (blood sinusoids), KC (Kpuffer cells), N (hepatic necrosis), LI (inflammatory leukocyte infiltrations), DBS (dilatation in blood sinusoids), CCV (congested central vein).
Discussion
Exposure to Mn can cause adverse neurological, reproductive and respiratory effects in both animals and humans. 43 Mn toxicity is unusual and is primarily manifested in the liver, a critical organ for Mn homeostasis, due to its primary role in removing Mn from the blood for biliary excretion. 9 So, accumulation of Mn in the liver causes hepatic damages and thereby restricts the rate of its elimination. 44 In the present study, administration of Mn induced the alterations of organ–body weight ratio without mortality. The reduced relative weight of liver in Mn-treated rats is an important indication of the pathological improvement of the liver. Our results were consistent with previous studies of Huang et al., 12 , 45 who have reported that Mn accumulation causes disturbances in the total body weight and absolute and relative weights of liver in rats probably by an increase in lipids and proteins degeneration caused by the high level of Mn in hepatic cells. In our study, animals exposed to Mn showed necrotic changes resulting in the release of liver injury markers (AST, ALT, LDH, GGT and bilirubin). According to previous studies, 46 , 47 the following enzymes (AST, ALT and LDH) were released from cytoplasmic area into circulation in case of cellular damages. GGT has been used as an index of liver dysfunction, its measurement may be useful to study oxidative stress-related tissues. The products of the GGT reaction may themselves lead to the increased free radical production, particularly in the presence of iron. 48 –50 Other clinical studies corroborate with our results showing that the increase in plasma TB level was associated with exposure to Mn. 51,52 Coadministration of silymarin attenuated Mn-induced hepatotoxicity as shown by the morphological changes and by the decreased activities of enzymes and bilirubin level, thus offering protection against Mn toxicity by stabilizing the cell membrane of rat hepatocytes, which may decrease the leakage of the enzymes into blood stream. The current work provided a considerable support for evidencing the protective effects of silymarin on liver damage induced by Mn. Furthermore, oxidative stress may be a result of excessive ROS generation or failure of the cellular antioxidant system. The rate of free radical generation and its elimination by antioxidant defense mechanisms are balanced under normal conditions. However, mitochondria are the major sites of cellular ROS production, which binds with a number of cellular components including lipid, protein, DNA, carbohydrate, thiols and other low-molecular-weight antioxidants causing oxidation of macromolecules leading ultimately to pathogenesis. 53 Our results demonstrated that Mn markedly increased the levels of ROS in liver of Mn-treated rats when compared with controls. Previous in vitro and in vivo studies have indicated that Mn causes oxidative stress, by inducing the generation of ROS, in mitochondria isolated from liver. 54 , 55 Excessive ROS production and/or loss of antioxidant defenses cause an oxidant–antioxidant imbalance making membrane lipids vulnerable and protein to oxidative damage. In the present investigation, there is a significant increase in the levels of TBARS, LPO and PCO in the liver of Mn-treated rats, which confirms the onset of oxidative stress. Accordingly, it could be concluded that Mn-induced oxidative damage in hepatic cells via generation of free radicals like ROS and/or reactive nitrogen species (RNS). Generally, ROS react with the polyunsaturated fatty acids to cause peroxidative changes leading to an enhanced lipid peroxidation levels. However, a concomitant use of silymarin significantly reduced the hepatic levels of lipid peroxidation and protein carbonylation. Nitric oxide (NO), source of RNS, is a highly reactive oxidant that is produced through the action of inducible isoform of NO synthase (iNOS). In pathological stressed conditions, iNOS is an inflammation responsive enzyme expressed de novo by different cell types, including macrophages and hepatocytes, responsible for enhanced tumor growth in hepatocellular carcinoma. 56 In the current study, exposure to Mn induced a significant increase in the level of NO. NO can also exacerbate oxidative stress by reacting with ROS, particularly with the superoxide anion forming peroxynitrite (ONOO−), which affect the redox balance of cells inducing protein, lipid and DNA modifications. 57 In our study, silymarin coadministration could decline the increase in NO level in the liver suggesting its protective effects by attenuating ROS production.
The ability of liver detoxification resides in its rich metabolizing enzyme profile and intracellular antioxidant reserve. The overproduction of free radicals overwhelms the detoxification and scavenges capacity of cellular antioxidant enzymes, namely SOD, CAT and GPx, and nonenzymatic antioxidants, namely vitamin C, total thiol and GSH, resulting in a severe damage to macromolecules and toxic lipid peroxides formation, which ultimately contribute to cellular hepatotoxicity. 58 Mn-treated rats exhibited a decrease in the activities of enzymatic antioxidants in the liver in agreement with our previous report describing its neurotoxicity in adult rats. 29 Coadministration of silymarin markedly increased enzymatic antioxidant activities by upregulating the gene expression of SOD, CAT and GPx as reported previously. 27 The nonenzymatic antioxidant like vitamin C, a primary antioxidant, can directly scavenge singlet oxygen, superoxide and hydroxyl radicals in aqueous medium. 58 Vitamin C can also scavenge peroxynitrite, an awful nitrating and oxidizing molecule, which itself induces lipid peroxidation, nitration of amino acids and apoptosis. Another important nonenzymatic antioxidant, a reduced GSH, plays a crucial role in the detoxification process and neutralizes the electrophilic site by providing an –SH group, renders the metabolite more water solubility and detoxify certain ROS (e.g. hydroxyl radical). The decline in the level of vitamins C, GSH and total thiols in Mn-induced rats might be related to the increased utilization of these antioxidants counteracting the high lipid peroxidation and oxidative stress. Silymarin cotreatment increased the levels of these antioxidants by scavenging free radicals and alleviating oxidative stress.
In addition, free radical generation following xenobiotic exposure may lead to extensive DNA damage giving rise to mutations and/or cell death. 59 In the present study, DNA damage was evaluated by electrophoresis of DNA isolated from liver. In general, DNA intact band appears to be condensed near the application point with no DNA smearing suggesting no DNA fragmentation. Administration of Mn provoked DNA damage resulted by DNA shearing without classical DNA ladder pattern seen in apoptosis. Silymarin cotreatment was found to be effective and prevent the Mn-induced smear formation. It could prevent DNA oxidation mainly by quenching free radicals and modulating enzymes metabolism.
Consequently, the biochemical findings in our study were supported by the liver histopathological examination, which demonstrated changes in liver of Mn-treated groups. The abnormal histology included the presence of pyknotic nuclei (necrosis) and inflammatory consistent with Mn-induced cellular injury, like leukocyte infiltrations, activated kupffer cells, dilatation in blood sinusoids and extensive cytoplasmic vacuolization. This might be attributed to the fact that Mn was primarily metabolized in the liver. So, reactive generated metabolites were thereby known to damage hepatocytes. Our results were in agreement with those of Huang et al. 12 Coadministration of silymarin improved the histological alterations induced by Mn exhibiting less disarrangement and degeneration of hepatocytes. This improvement might be attributed to the efficiency of silymarin by reducing intracellular ROS levels of hepatocytes in order to prevent oxidative stress-induced cellular damage. Furthermore, hepatic cell proliferation was found to be stimulated after silymarin cotreatment, suggesting that the enhanced liver regeneration might help to replace the damaged liver cells.
In conclusion, this investigation showed that silymarin could possess protective effects against Mn-induced oxidative stress in the liver of rats. Its overall preventive role could be due to its free radical scavenging properties, which diminished the oxidative impact by Mn burden in the liver. This finding provided a clue on antioxidant properties of silymarin and its future prospects in combating Mn-mediated hepatic damage.
Footnotes
Acknowledgments
The authors thank Miss Dalenda Kchaou for her assistance in histolological techniques.
Funding
The present work was supported by the grants of DGRST (Appui a la Recherche Universitaire de base, ARUB 99/UR/08-73), Tunisia.
Conflict of Interest
The authors declared no conflicts of interest.
