Abstract
This article offers a broad assessment of the hormetic dose response and its relevance to biomedical researchers, physicians, the pharmaceutical industry, and public health scientists. This article contains a series of 61 questions followed by relatively brief but referenced responses that provides support for the conclusion that hormesis is a reproducible phenomenon, commonly observed, with a frequency far greater than other dose–response models such as the threshold and linear nonthreshold dose–response models. The article provides a detailed background information on the historical foundations of hormesis, its quantitative features, mechanistic foundations, as well as how hormesis is currently being used within medicine and identifying how this concept could be further applied in the development of new therapeutic advances and in improved public health practices.
Part I. Historical foundations and concepts/principles
Introduction
Hormesis has become an important concept in biological and biomedical disciplines that utilize dose response information. This is evident by the fact that the terms hormesis or hormetic have become more common and widely cited in the research literature. In the 1980s, these terms were cited only 10–15 times per year in the Web of Science database. In 2010 and 2011, they were cited over 3200 times in this database. Furthermore, during the past decade, for the first time hormesis has also became fully integrated into leading textbooks in pharmacology 1 and toxicology, 2,3 while five books have focused on different aspects of hormesis over the past several years. 4 –8 Similarly, several professional organizations have had keynoted presentations and special sessions on this topic. High-profile journals such as Science published a four page news story on hormesis, 9 introducing the concept to the broader scientific community, while Nature published a highlighted commentary on the implications of hormesis for the discipline of toxicology. 10 The reasons commonly underlying this interest in the concept of hormesis have been due to the fact that it is a reproducible phenomenon that is very generalizable and has important implications in drug discovery, in the clinical trial, in therapeutics, as well as in toxicological hazard assessment, risk assessment, and risk communication for chemicals, radiation, and pharmaceutical agents.
This article is intended to provide a broad overview of the concept of hormesis for biomedical scientists, pharmacologists, physicians, and public health scientists. The format of this review is intended to be via a large number of relatively short questions and referenced answers. This format has been followed in two articles, one directed toward toxicologists and risk assessors 11 and the other toward biogerontologists. 12
Q/A# 1. What is hormesis?
The term hormesis is derived from the Greek meaning ‘to excite.’ It can be defined at multiple levels: the descriptive (what is it?), the mechanistic (how does it work?), and at the evolutionary levels (what is its adaptive significance?). 13,14 Descriptively, hormesis is a dose–response relationship that is generally characterized as a biphasic dose response with a low-dose stimulation and a high-dose inhibition. These findings are typically plotted on graphs as either inverted U-shaped or as J-shaped dose responses. Of considerable significance to the hormesis concept is that the biphasic dose response has consistent quantitative features including the magnitude of the stimulation, the width of the stimulation, and their relationship to the zero equivalent point (i.e. threshold). With respect to mechanism, there are numerous proximate mechanisms that may account for specific types of biphasic dose responses. The proximate mechanisms may be either ‘receptor- or non-receptor-’ based mechanisms. 11 The highly consistent quantitative features of the hormetic dose responses at the cell, organ, and organismic level may be mediating the effects of upstream and highly conserved allometric gene clusters that control and direct the allocation of biological resources within complex biological systems. 15 From an evolutionary perspective, hormesis may be viewed as an adaptive response that mediates cellular stress involved in a broad range of preventive, reparative, and signaling activities. 5 The hormetic dose response, therefore, is a specific type of biphasic dose response with definable quantitative characteristics relating to the magnitude and width of the low-dose stimulatory response. By lending specificity to the type of biphasic dose response observed, 11 it is a better term than biphasic.
Time can be a crucial component of the hormetic dose response, especially when the hormetic dose response represents an overcompensation to a disruption in homeostasis. 16 The dose–time response, which was initially reported by Schulz, was confirmed later by Branham 17 in an extensive replication of the original findings of Schulz 18,19 concerning the effects of chemical disinfectants on yeast metabolism. This perspective was incorporated into a sophisticated cybernectic regulatory model system by Stebbing 8,20,21 and provided substantial confirmatory validation by Calabrese. 22 Assessing hormesis within a dose–time-framework can be challenging since it requires the use of many doses, with responses evaluated over multiple time points. Approximately 20% of the 9000 dose responses in the hormetic database include a time component. 23
Q/A# 2. Why is the term hormesis poorly known and/or rarely used in the pharmaceutical industry?
The term hormesis was first mentioned in the peer-reviewed scientific literature in 1943 by Southam and Ehrlich, two forestry researchers at the University of Idaho. 24 They observed that low doses of extracts from the Red Cedar tree enhanced the metabolism of multiple fungal species, whereas higher concentrations were inhibitory. Both these authors soon left the University of Idaho, with Ehrlich moving to the University of Minnesota to work on enhancing the production of penicillin in World War II and Southam to Columbia University from which he graduated Medical School. He would go on to have a career in oncology with a focus on tumor immunology. Thus, neither investigator followed-up on their dose–response concept discovery, and their new term. Despite this first mentioning in the scientific literature, the term hormesis was not even used as a key word for this original article for searching purposes, and was therefore not cited in widely used databases such as Pub Med and the Web of Science. In fact, the term hormesis or hormetic was only rarely cited through the 1970s and only about 10–15 times per year in the Web of Science through the entire decade of the 1980s. Therefore, the terms hormesis or hormetic had little intellectual penetration within the standard scientific literature in the 35 years since first mentioned by Southam and Ehrlich. 24
Prior to when Southam and Ehrlich 24 coined the term hormesis, the biphasic dose response was often referred to as the Arndt–Schulz Law. 18,25 Hugo Schultz, a professor at the University of Greiswald, Germany, reported the occurrence of multiple biphasic dose responses in studies on the effects of chemical disinfectants on yeast metabolism (see work by Crump 26 for an English translation of H. Schulz autobiography), presenting these findings in 1884 at a meeting of the Greifswald Medical Association (http://www.Medizin.uni-greifswald.de/medverein/Geschichte.htm). He soon generalized these observations following discussions with Rudolph Arndt, leading to the Arndt–Schulz law. Schulz argued that the biphasic dose response characterized how chemical and physical agents affected biological systems over a broad dose range. The hormesis concept was also widely known during the early decades of the 20th century as Hueppe’s Rule, named after bacteriologist Ferndende Hueppe. 27 In the area of psychological stress biology, a comparable biphasic dose response is known as the Yerkes–Dodson Law. 28 –30 Further complicating the terminology issue, several terms were created in the 1970’s that are scientific discipline-specific that also describe the hormetic dose response. Some of these terms include the following: U-shaped dose response, J-shaped dose response, adaptive response, biphasic dose response, bitonic, inverted U-shaped dose response, preconditioning, postconditioning, paradoxical response, parabolic dose response, and others. More recently, an integrated terminology for biological stress responses has been proposed based on the hormesis concept. 31 Thus, the term hormesis has been poorly known in the pharmaceutical industry because it was introduced about 60 years after creation of the concept and at the time when the concept was severely marginalized within the scientific community. It then had to compete with a plethora of other new terms.
Q/A# 3. Why did the pharmacology field miss the concept of hormesis?
The concept of hormesis was missed by the discipline of pharmacology for a variety of reasons. 15,25,32 –35 On the scientific level, the hormesis concept can be difficult to study and establish. Of importance is that the magnitude of the low-dose stimulation is invariably modest with maximum responses typically (60% of the cases) only 30–60% greater than the control group. Such a modest stimulatory response can be difficult to distinguish from normal control group variation. In order to study possible hormetic effects, study designs generally need to be more rigorous than that which is normally employed, using at least 3–4 properly spaced doses below a previously well-characterized pharmacological or toxicological threshold. There may also be a time requirement, thereby imposing further resource constraints on the experimental studies. The modest stimulatory response would also affect statistical power calculations requiring investigators to use more animals, making the costs of the studies greater. Given the modest nature of the stimulatory response in the hormetic zone, there would be a much greater need than with high dose study responses to validate the experimental findings via more efforts at replication. All of these factors would have a marked impact on whether the hormetic dose response could be observed as well as shown to be a reproducible phenomenon. The above issue of reproducibility would also be further challenged if the control group had normally high variability for the parameters of interest. 36 –38 Finally, even though there were a number of prominent researchers in the early decades of the 20th century that published substantial findings on the hormesis concept, essentially all became redirected to other areas of professional activity including administration and other scientific questions. 39
On the political level, the hormesis dose response (HDR) was put forth as the explanatory principle of homeopathy by Professor Hugo Schulz in the mid 1880s. This linking of the hormesis concept with homeopathy profoundly affected its capacity to be accepted by the fields of pharmacology and toxicology, both mainstream subdivisions of traditional medicine. 40
Despite the scientific, personal, and political impediments affecting the acceptance of the biphasic dose–response model, there were many isolated reports of biphasic dose responses, eventually accumulating to such a substantial number that Szabadi 41 integrated this information within a mechanistic framework. The assessment of Szabadi 41 was the first general and modern pharmacological elaboration on the topic. His article generated considerable follow-up discussion with additional documentation 42 –45 supporting the scientific foundations of the biphasic dose response.
Q/A# 4. Who were the key individuals that affected the acceptance of hormesis within the scientific community?
Despite the prolonged advocacy of the hormesis concept by Professor Hugo Schulz, there was a general absence of significant leadership on this concept within the broader scientific community by others who published scientific findings on it. In tracing the careers of many early hormesis researchers, what seems clear is that much of the early research was of good quality, reproducible, and published in leading journals. 39 In fact, by the early decades of the 20th century, the hormesis concept was incorporated into the textbooks of botany 46,47 and microbiology. 48 –50 A key factor was that the scientific research, especially that US researchers published, did not become embodied in the homeopathy versus medicine dispute, in contrast to that of Hugo Schulz. However, despite the relatively large number of such scientists, these individuals, for the most part, developed other professional interests leaving behind the hormesis concept. Thus, the hormetic concept failed to become a unifying theme for the US researchers. The early researchers on hormesis provided no counter to the influential criticisms offered by Clark. 15,39,51,52 This lack of intellectual leadership at the time of dose–response concept consolidation was an important factor in the failure of the hormetic dose response to become more firmly established within the broader scientific domain. In retrospect, this lack of leadership occurred at the time when the pharmacological community successfully (but incorrectly) linked the hormesis concept to the high dilution elements of homeopathy helping to lead to its scientific demise (see Calabrese 15 for a review).
The most visible subsequent leader of the hormesis concept after Schulz was Professor Thomas Luckey at the University of Missouri. While Luckey first reported on the concept of hormesis in his research on the effects of antibiotics on gnotobiotic animals in the mid 1940s, 53,54 he did not display conceptual leadership on the topic of hormesis for more than 30 years after his initial scientific discoveries relating to the hormesis concept about 45 years after the death of Schulz. In fact, by the time Luckey became involved in studying (rather than providing leadership to) hormesis in the 1940s, the hormetic concept had already become profoundly marginalized. When Luckey did become a leader in this area, hormesis was nearly defunct as a biological concept, being outside the mainstream of the scientific community. Furthermore, once Luckey 55 became involved in debates about hormesis, he did so principally in the area of radiation biology, an area out of his long-standing experience, having little or no standing within the radiation research community. Nonetheless, Luckey’s 1980 book on radiation hormesis provided valuable inspiration for the resurgence of the hormesis concept.
There were many in the biomedical communities that were quick to criticize Schulz but mostly because of how he interpreted his findings, the claim that he had discovered the underlying explanatory principle of homeopathy. That is, low doses of homeopathic drugs induced adaptive responses that enhance the capacity to resist disease. This dispute occurred at a time when there was an intensely hostile competition between what today is called traditional medicine and homeopathy. Over the next 50 years, Schulz would be criticized and even ridiculed by some scientists from traditional medicine while being a heroic figure to most homeopaths. Yet, Schulz was a traditionally trained physician as well as pharmacologist/toxicologist.
The problem that Schulz created for traditional medicine is that he in effect ‘gave’ his biphasic dose response to homeopathy at a time when traditional medicine had yet to even recognize the need for a dose–response model to guide either drug development or therapeutic strategies and practices. 15 However, by 1920s, the need for improved biomedical guidance over therapeutic practices had emerged, making traditional medicine take the dispute over the dose response more seriously. As a result, Schulz and his biphasic dose–response concept came under serious challenge by leading pharmacologists, especially from the UK, lead by the influential University of Edinburgh pharmacology professor, Alfred J. Clark, a leading researcher, textbook author, governmental advisor, and journal editor.
Clark and his colleagues were successful in marginalizing homeopathy, but also in unfairly linking Schulz to the high dilution wing of that medical practice. While the scientific record would show that Clark was incorrect in this regard, 15 his criticism stuck. The concept of hormesis failed to thrive within the scientific community, while also being rejected by nascent federal regulatory agencies focused on drug safety and later with environmental risk assessment. The leaders of traditional medicine were also very successful in denying the hormetic concept acceptance in the leading textbooks, in their professional societies, in professorial hirings, as well as in chemical and drug regulation. This marginalization of the hormesis concept was operational throughout the 20th century.
Q/A# 5. Does the threshold dose–response model have important limitations?
This is a question of historical and practical significance since the threshold model acceptance affects how toxicological and pharmacological studies are designed and how acceptable exposures are derived from clinical testing procedures. Of considerable importance is that the threshold dose response was probably never validated during the entire 20th century even though widely accepted by the scientific and regulatory communities. While it is not possible to prove a negative, substantial efforts have been made by the University of Massachusetts hormesis study team to uncover any study (studies) that were intended to validate the threshold model for below threshold responses. To date, none have been found in the historical toxicological and pharmacological records. The threshold dose–response model was assumed to make accurate predictions of responses in the low-dose zone without having been proven to do so. If true, this would represent a profound error by the pharmacological and toxicological communities and the vast regulatory apparatus in industrial countries, such as the numerous environmental protection agencies and the food and drug administrations. It is also an important failing of the regulated chemical and pharmaceutical industries, many of which possess enormous technical resources. Thus, society is faced with a serious problem since the threshold dose–response model, upon which many health standards were based, was never validated. In addition to its failure to be vetted and validated during the entire 20th century, the threshold dose–response model failed in multiple validation testings when finally assessed. 56 –58 What are the limitations of the threshold model? In simple terms, it fails to do what society requires, that is, to make accurate predictions of chemical and drug effects in the low-dose zone, below the toxicological threshold where society lives.
Q/A# 6. How were biphasic dose responses interpreted by the field of pharmacology?
In 1977, Szabadi 41 indicated that the biphasic dose response was a general one, providing a means by which cells could regulate a broad range of responses to numerous stimuli. He identified a general mechanistic strategy by which biphasic dose responses occurred. Szabadi 41 reported that an agonist often activated two (or more) receptor subtypes. The receptor subtypes displayed differential binding affinity for the agonist. The receptor with the greater binding capacity had far fewer receptors, whereas the receptor with the weaker affinity had greater capacity, that is, more receptors. These receptor subtypes would lead to either stimulatory or inhibitory pathways. When the cells were exposed to a broad range of agonist concentrations, the high-affinity receptors would dominate the response at lower concentrations, whereas at the higher concentrations, the receptor with the greater capacity and lowest affinity would dominate the response. When assessed along a broad concentration response continuum such responses would be biphasic.
This type of response reflects a general response strategy, affecting several dozens of receptor systems, and an extensive array of biological responses (Table 1). There are now numerous complementary mechanisms by which biphasic dose responses occur that have been documented, including receptor linked-signaling pathways. Multiple-signaling pathways mediating hormetic dose responses have been reported. Over the past decade, this area has shown enormous development with several hundred hormetic dose responses having mechanistic explanations down to receptor-specific signaling pathway/pathways. Considerable evidence has demonstrated the involvement of the three major mitogen-activated protein kinases (MAPK) pathways, including the extracellular signal-regulated kinase 1/2 (ERK1/2) pathway, the c-Jun N-terminal kinase (JNK), and the p38 MAPK pathway. For example, ERK1/2 and JNK pathways mediate the biphasic dose response of arsenite 96 and cadmium 97 on cell proliferation, while the PI3K (part of an intracellular signalling pathway)/AKT (protein kinase B, a serine/threonine-specific protein kinase) pathway mediates the biphasic proangiogenic effects of hydrogen sulfide (H2S). 98
Representative receptor systems displaying biphasic dose–response relationships. 59

This type of response has also been reported to occur with a single agonist acting on a single receptor but one in which there were two distinct binding sites. 99,100 Of relevance is that the quantitative features of hormetic dose responses are similar regardless of the number of receptors involved for either the stimulatory or inhibitory pathways or whether there are two receptor subtypes of two different receptors or whether there are two receptor sites on a single receptor or other series of possible permutations. Such findings suggest that there are numerous ways to affect the regulation and distribution of resources within biological systems of profoundly differing levels of complexity (i.e. cell, organ, and whole organism) and function. Despite such avenues of complexity, the quantitative features of the dose response are quite similar and reveal a common underlying dose–response strategy.
Q/A# 7. Can several proximate mechanisms of hormesis be clarified?
Brain-derived neurotrophic factor (BDNF). 101 The facilitating and inhibitory effects of exogenous BDNF on motor axonal regeneration are mediated by trkB and p75 receptors, respectively.
Delta-opioid peptide. 102 (p.86) ‘. . . endogenous opioid peptides may, at low concentrations, promote cell survival via the MEK-ERK pathway through delta2 opioid receptors, whereas they may kill cells at high concentrations via the activation of FasL through an as-yet unknown mechanism involving mu opioid receptors.’
Estrogen. 103 The stimulatory response to low concentrations of estrogen is due to increased phosphorylation of the enzyme (tyrosine hydroxylase; high affinity for the substrate and cofactor). The inhibitory effect of the high concentration of estrogen is caused by a decreased phosphorylation (low affinity for the substrate and cofactor).
Prostaglandin. 104 15d-PGJ2 acts as an antioxidant at low concentrations and pro-oxidant at higher concentrations. This pattern of activity was closely associated with apoptotic response patterns. That is, the low level of 15d-PGJ2 induced anti-apoptotic patterns on gene and protein expressions, while high concentrations induced proapoptotic patterns.
Q/A# 8. Are there different biomedical implications if the mechanism is receptor-based or nonreceptor-based?
This question has not been addressed in the literature. However, it should be noted that the quantitative features of the dose response are similar regardless of whether mechanism underlying the dose response is receptor-based or nonreceptor based. 11
Q/A# 9. How were biphasic dose responses applied to the pharmaceutical field, if they were?
The biphasic dose response has been recognized and extensively used in the pharmaceutical industry in the process of drug development, preclinical investigations, and clinical trials for specific classes of drugs. The low-dose range of a drug, for example, may affect a response that is desirable. This has typically been the case for agents such as anxiolytic 105 and antiseizure drugs. 106 With respect to anxiolytic drugs, low doses often increase the amount of time that a rodent will spend in lighted areas, suggesting a reduction in anxiety. At a higher dose, anxiety typically ensues. Based on such preclinical rodent studies, the doses of these agents will be selected for human clinical trials. The pharmaceutical industry has typically referred to these dose responses as biphasic responses not relating the observations to the possibility that it may be an hormetic dose response.
Q/A# 10. When was the concept of hormesis (using the term) first employed in drug applications?
Food and Drug Administration (FDA) researchers employed the term in the mid 1940s in their assessment of several antibiotics and their capacity to enhance the proliferation of harmful bacteria in mouse models. These investigators highlighted the capacity of commonly employed antibiotics such as penicillin and streptomycin to enhance the risk of death in bacterially exposed mice administered with low doses of antibiotics (i.e. stimulating proliferation of the harmful bacteria), 107,108 while at higher doses, the antibiotic had its expected protective effect (i.e. killing of the harmful bacteria). These striking findings, however, were dominated by concerns associated with the development of resistance to antibiotics and therefore never received a high biomedical priority. The hormesis term was also not assimilated by the pharmaceutical industry for the remainder of the 20th century. While the term hormesis would be initially introduced into environmental toxicity by Stebbing 20,21,109,110 and by Luckey 55,111 into radiation biology, the pharmaceutical industry was adopting the terms such as biphasic for experimental laboratory data whereas the terms U- or J-shaped would tend to be used in human population/epidemiology studies.
Q/A# 11. Why were biphasic dose–responses long-neglected in the pharmaceutical industry?
Even though it might appear as if biphasic dose responses were ignored or neglected by the pharmaceutical industry, the evidence does not support this conclusion. Numerous pharmaceutical agents have been approved for patients in whom the low-dose stimulation as part of the dose response provides the therapeutic response. Entire pharmaceutical areas have been built upon the use of the biphasic dose response. What has been generally missed and/or underappreciated is that these responses appear to fit the quantitative features of the hormetic dose response. Consequently, the industry has been slow to broadly recognize that these hormetic-like biphasic dose responses may represent a general biological dose–response pattern or strategy.
Q/A# 12. How does the hormetic concept relate to homeopathy?
Homeopathy has had a long and complex relationship with the hormesis concept, beginning when Hugo Schulz claimed that he had discovered the underlying biological principle of homeopathy. In 1884, Bloedau 112 claimed that the homeopathic drug called veratrine was effective in the treatment of a type of gastroenteritis. At about the same time, the causative bacteria of this disease was identified and cultured at the laboratory of Robert Koch. Schulz then assessed whether veratrine would kill or prohibit the proliferation of the causative bacteria in a bioassay. To Schulz’s surprise the drug was unable to affect bacterial growth even at high concentrations. Many researchers may have concluded that the drug was not effective in treating this disease and that the initial conclusions of Bloedau 112 were probably wrong. However, Schulz accepted the clinical data as valid and offered an alternative hypothesis. He proposed that the veratrine was therapeutic but that it enhanced the adaptive capacity of the patient to resist infection rather than directly killing the bacteria. While later studying the effects of chemical disinfectants on the metabolism of yeasts, Schulz noted that the agents tested induced a biphasic dose response, enhancing the release of carbon dioxide at low concentrations, while at higher concentrations, this response was decreased in a concentration-dependent manner. 18,19 He was able to replicate these findings, permitting high confidence in the data. While Schulz did not immediately grasp the potential biological significance of these observations, he subsequently formulated a far reaching conceptual dose–response framework. The low-dose stimulatory response in the yeast was believed to be an adaptive response, whereas the responses at higher concentrations reflected toxicity. He then argued that the reason why the veratrine worked in the clinical setting was that it had induced an adaptive response at low concentrations. This adaptive response resulted in the patients being able to resist the infection without directly killing the bacteria. 113 It was this relationship that Schulz proposed, strongly advocated, and popularized and for which he also became highly criticized by the medical community. He believed that he had discovered the underlying principle of how homeopathic drugs act.
Recent assessments of the Schulz 18,19 publications by Calabrese and Jonas 114 argue that these early yeast studies of Schulz were improperly applied to homeopathy. Under most medical situations, the patient would seek treatment for a disease condition, that is, the patient would be treated with a homeopathic preparation after exposure to a disease inducing agent. In case of Schulz’s investigations, there was no prior administered stressor agent or condition. Therefore, the experimental system of Schulz lacked potential exposure and temporal relevance for most clinical evaluative systems. This was noted by Wiegant et al., 115 –117 van Wijk and Wiegant, 118 and Van Wijk et al. 119 who developed a model homeopathic experimental system in which human liver tumor cells were initially administered with stressor agents such as heat or toxic metals and then treated with one of these agents with a dose that was sufficiently low that it failed to induce a response in control cells. This low dose, while having no effect on the control cells, enhanced the adaptive response over what the original stressor exposure did. Their experimental system, therefore, provided a biomedical model to assess the effects of homeopathic drugs within a routine biomedical setting. Calabrese and Jonas 114,120 indicated that this research method represented a vehicle to assess whether such homeopathic drugs may act via a postconditioning hormesis process. 120 This framing of how homeopathic drugs may act could be experimentally tested, creating a legitimate ‘point of contact’ between homeopathy and traditional biomedical research. 121 This perspective could create opportunities in which homeopathic drugs could be assessed within a framework fully consistent with modern molecular biology research, that is, in concentration ranges typical of those routinely employed in traditional pharmacological/biomedical research.
Q/A# 13. How does the concept of hormesis relate to the high-dilutional aspect of homeopathy?
It is clear that, based on his written statements in the historical record, Hugo Schulz rejected the high dilutional aspect of homeopathy. 15,122 His biphasic dose–response phenomenon was seen within the context of a traditional biomedical dose response. Schulz linked the low-dose stimulation to the low-dilutional ‘wing’ of homeopathy. Such a distinction, which is quite clear from the historical literature, was glossed over and/or overlooked by leaders in the traditional medical community during the conflict with homeopathy, thereby misrepresenting the position of Schulz. 39,40,51,52
Q/A# 14. How did the dose–response and threshold dose–response concepts emerge within the field of pharmacology?
The dose–response concept is believed to have been originally developed by the French biologist Claude Bernard (1813–1878) 123 based on studies concerned with the excretion of glucose. As a result of these studies, Bernard first formulated the threshold dose–response concept. Other researchers extended this idea to the excretion of additional pharmacological/physiological agents such as chloride, urea, and other metabolic products. 124 –128 The threshold dose–response concept was subsequently placed within a more general framework by the renal pharmacologist Cushny (1866–1926), 129 who developed a simple formula-based model to describe the threshold dose response. The threshold dose–response model was further supported by Alfred J. Clark (1883–1941), 130 who observed a biological threshold for acetylcholine, which required approximately 20,000 molecules acting via receptors to produce an initial effect on a heart cell (e.g. isotonic contraction). Other research supporting the threshold dose–response concept in pharmacology was reported by Aitken 124 while working in the laboratory of the Nobel Prize winner and British Pharmacologist Professor Charles Scott Sherrington.
While the above-noted research of Clark, 131 Cushny, 129 Aitken, 124 and others was principally pharmacologically oriented, support also emerged in the allied fields of toxicology, 132 –134 radiation occupational health, 39,135,136 and immunology. 137
Despite these converging and supportive dose–response findings from diverse researchers, major credit goes to Alfred J. Clark for his efforts in the area of quantitative pharmacology, placing the dose–response on a solid theoretical foundation, including mechanistic, biomathematical, and population-based perspectives. His highly influential textbooks had a major impact on several generations of pharmacologists and toxicologists during the middle decades of the 20th century, which is the period when these fields were establishing their fundamental scientific principles and research methods, including study designs, statistical methods, and hazard/safety assessment methods that would be applied to the emerging field of risk assessment. 51,52 Clark’s efforts lead to the establishment of the primacy of the threshold dose–response model in pharmacology and toxicology as well as other biological subdisciplines. The textbooks of Clark also profoundly criticized the biphasic dose–response concept of Hugo Schulz, which became known as hormesis, leading to its marginalization throughout the remainder of the 20th century.
Q/A# 15. Do hormetic dose–response relationships have definable quantitative features?
When the criteria for the hormesis database were initially developed in 1996, there was no consensus or much previous debate/discussion given to what were the quantitative features for this type of dose–response relationship. However, following the analysis of about 6000 dose–response relationships satisfying the evaluative criteria for hormesis, hormetic dose responses typically (approximately 60% of cases) displayed a modest stimulatory response, being only about 30–60% greater than the control group at maximum (Figure 1). Approximately 20% of dose responses had their maximum response greater than twice the control value. These findings were unexpected as they implied a possible dose–response maxima that would later provide a framework for integrating the pharmacological ceiling effect and biological plasticity within the hormesis foundation. 11,13,30 This so-called 30–60% ‘rule’ has also raised the possibility of whether dose responses with a maximum response greater than three- to fourfold of the control are still legitimate examples of hormesis despite conforming to a biphasic dose response.
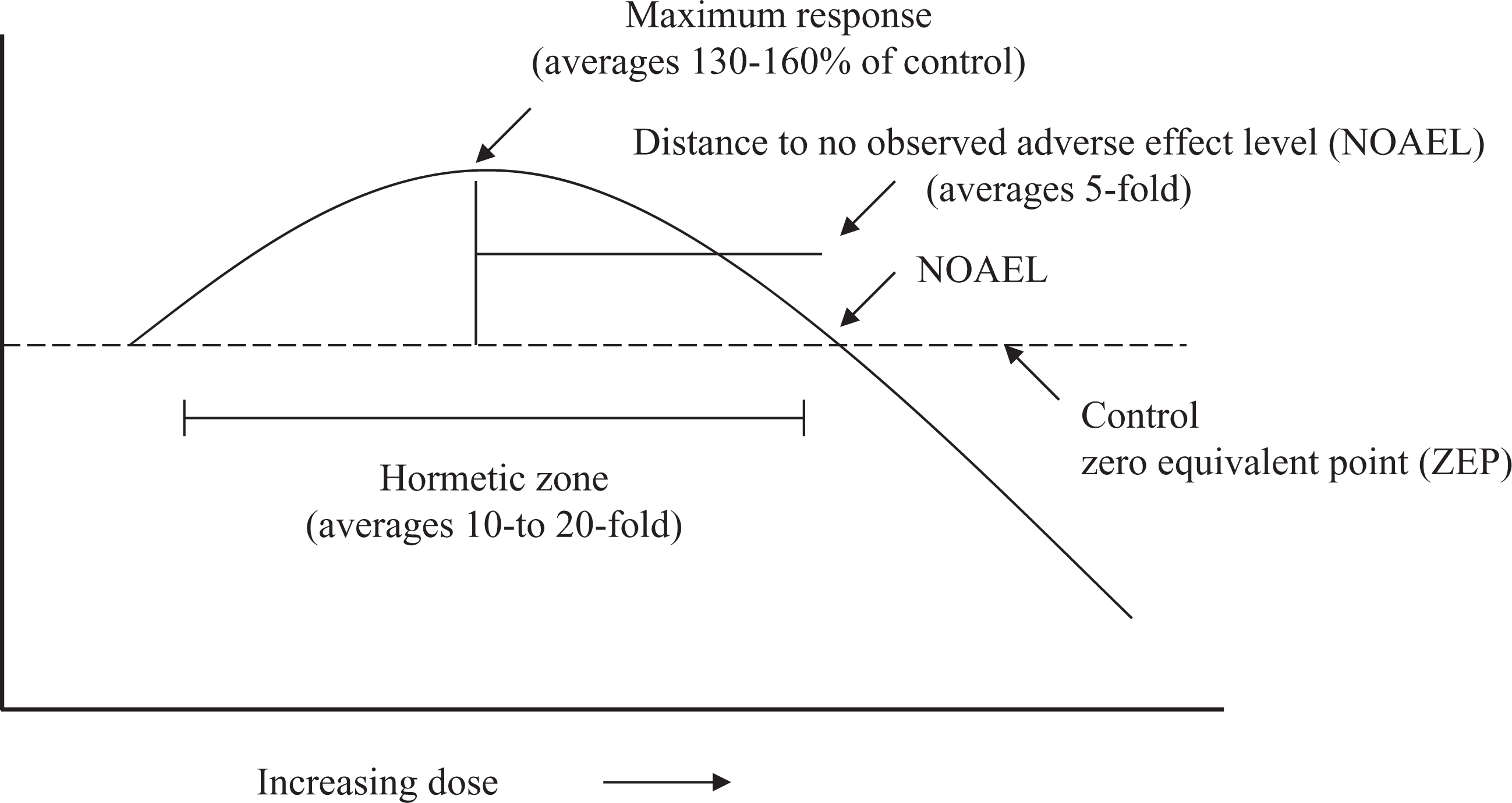
Dose–response curve depicting the quantitative feature of hormesis.
Why is there such a strong general consistency in the magnitude of the hormetic stimulation, independent of biological model, level of biological organization, biological end point, and chemical class/physical stressor? This dose–response property has been highly conserved from bacteria to humans. 138,139 It may be hypothesized that the limiting of the maximum stimulatory response represents a strategy by which biological resources are managed, controlled, and allocated. Given the vast array of regulatory processes that could be activated within any limited time period, having a generalized constraint of resource allocation may offer a survival advantage over the modulation of resource allocation to the specific requirements of each dose response for each specific condition.
The width of the stimulatory response is generally within 10- to 20-fold of the toxicological or pharmacological threshold. 23 Nonetheless, in a low percentage of cases (i.e. 5–10%), the width of the stimulatory response was much broader, exceeding a factor of 1000-fold. The factors contributing to this marked variation in the width of the stimulatory zone are unknown. They may, in part, be related to an unique combination of heterogenicity of sample population and pharmacokinetic characteristics.
Q/A# 16. How does the ceiling effect concept relate to hormesis?
The ceiling effect entered into the pharmacological literature in the early 1990s and has greatly expanded over the subsequent years. 140 This was an activity that independently paralleled the occurrence of hormesis in the biomedical literature. A careful review of the ceiling effect literature reveals that it reflects the dose–response maxima. Since the dose–response maxima is determined by the limits of biological plasticity and is described by the hormetic dose response, it follows that the ceiling effect is in fact the response maxima of the hormetic dose response.
Q/A# 17. Can the hormetic response be optimized?
Optimization of response is not only theoretically possible but also the object of researchers in a range of biological disciplines, dealing with tumor and nontumor cell biology, 141 wound healing, 142 and in plant biology. 143 Information supporting this concept will be presented for the cases of tumor cell biology and wound healing.
During the 1970s, low concentrations of the chemotherapeutic adriamycin (AD) were reported that could stimulate cellular growth parameters. In 1972, Wang et al. 144 noted that AD increased thymidine incorporation into DNA at low doses at short duration times. Roper and Drewinko 145 later reported stimulation of DNA synthesis at low AD doses. Huybrechts et al. 146 subsequently found increased survival of hematopoietic stem cells at subtoxic AD concentrations. For each of these situations, the stimulatory effects were of limited magnitude (10–50%), while at higher concentrations, the drug produced its well-known cytotoxicity. These findings led Vichi and Tritton 141 to conduct a systematic evaluation of how varying the experimental conditions affected the occurrence of stimulatory responses in various tumor and nontumor cells. They reported that AD induced biphasic concentration responses in multiple biological models for a range of end points. These included L1210 cells (survival), S180 cells (survival), HL-60 cells (survival), A431 cells (cell number), 3T3 cells (thymidine incorporation), and mouse fetal heart cells (protein synthesis) at 13–15 days of gestation, a period of nonactively dividing cells. While the above-noted experiments assessed AD-induced cytotoxicity, low-dose growth stimulation was an unexpected but consistent finding. The marked consistency of these so-called ‘accidental’ stimulatory responses lead Vichi and Tritton 141 to assess the broader biological significance of these biphasic dose–response relationships.
The extent of the AD induced enhanced survival depended on the cell density of the suspension culture. Since cells grown in higher densities yielded a greater proliferation response stimulation, this may have resulted from high cell density or because growth at high densities depleted growth factors in the serum containing medium. Subsequent experiments were conducted with low-density cultures with different fetal serum levels (0, 5, and 10%) and exposure to 10−9 M AD. While growth was decreased at 10% fetal serum, it was enhanced by 30–50% at the 0–5% fetal serum levels.
Various experimental conditions were needed for the stimulatory response. These included: a low-AD concentration, just below that which induced a decrease in proliferation, the medium supporting cell growth needed to be incomplete (i.e. suboptimal), and the drug should be able to induce toxicity. This latter condition was based on the observations that all cytotoxic anthracyclines stimulated growth at low doses, while inactive aglycones did not enhance growth. The hormetic response and its optimization were dependent on cell density, medium composition, and the culture’s history. Vichi and Tritton 141 concluded that to most reliably obtain growth stimulation, it was necessary to grow high-density cells in partially exhausted medium.
The concept that hormetic responses are optimized when cells are grown under suboptimal conditions has been more recently addressed by Demidova-Rice et al. 142 based on the experiments on the effects of low-level light on dermal wound healing. Wound healing was most efficiently enhanced in animal models with suboptimal wound healing capacity. It was not possible to enhance the healing capacity of wounds in mouse strains that had a normal high efficiency in wound repair. Such observations lead to the conclusion that these strains are probably responding in an optimized manner.
Such collective studies are of importance since they were well conceived and based on substantial findings. While the hormesis concept has been systematically deconstructed and reconstructed with respect to experimental protocols in so few systems, these findings have important theoretical and practical implications if they could be further generalized.
Q/A# 18. Are there hormetic ‘principles’?
Based on an assessment of the hormetic database, a set of nine hormetic principles has been derived. They are summarized in Table 2.
Hormetic principles.

Q/A# 19. Should hormesis be considered a biological expectation or a methodological flaw?
With the large number of data-based articles published on the reproducible occurrence of hormetic dose responses in numerous biological models, for a broad range of biological end points, for chemicals from numerous chemical classes, for a broad range of physical stressors, and with numerous well characterized mechanisms, this would seem to be an odd question to raise. 72,147 –150 However, a publication on the occurrence of cellphone-induced brain cancers by Interphone Study Group 151 suggests that this question is pertinent. This study 151 indicated a J-shaped dose response. At low doses, there was a decrease in tumor incidence with nonoverlapping confidence intervals with control values, suggesting the likelihood of real decreases in response. However, the authors discounted the low dose ‘protective’ effect as biologically implausible, while being unable to account adequately for these decreases via other potential factors in their methodology. While the point here is not to reassess their data, it is necessary to emphasize that such researchers continue to ascribe to an assumption that the hormetic dose–response model lacks validity and that other models, such as the threshold and linear models, are the vehicles by which scientific thought is to be guided. All this comes after the publication of multiple articles demonstrating that the hormetic dose response has substantially outperformed the capacity of these models to make accurate predictions in the low-dose zone. 56 –58 In fact, the predictive capacity of the threshold and linear low-dose models were consistently poor regardless of biological end point, chemical class, or physical agent.
Q/A# 20. How does the FDA consider the concept of hormesis?
It is uncertain how the FDA considers the concept of hormesis. As previously discussed (see Q/A# 10), FDA researchers published research findings on hormetic dose responses as far back as the mid 1940s. 107,108 The FDA has also approved large numbers of pharmaceutical agents that display hormetic dose responses, having key drug effects in the hormetic zone. This suggests that the FDA observes hormetic-biphasic dose responses on a regular basis, especially in their evaluation of preclinical data. Therefore, the FDA is aware that it is the low-dose stimulatory response component that is the basis of efficacy for numerous drug classes. However, despite this situation, there is no evidence that the FDA has incorporated the term hormesis into their regulatory lexicon as of the present time.
Q/A# 21. Is hormesis a measure of biological performance?
The dose response has two basic components, responses at doses greater than and less than the threshold. The responses at the doses greater than the threshold dose generally define a toxic response, while responses to doses less than the threshold, that is, responses in the hormetic zone of the dose response, represent a novel concept/interpretation called biological performance. These two areas of the biphasic dose–response relationship define fundamentally different phenomena with profoundly different biological implications. Biological performance activities are integrated biological responses that are adaptive in nature and are necessary for survival. The spectrum of end points considered within the context of biological performance is broad, including responses such as growth, memory, bone strength, disease resistance, and longevity. These biological effects are mediated by complex and highly integrated processes that typically display hormetic dose responses. The quantitative features of these dose responses, as affected by either endogenous or exogenous agents, display hormetic characteristics. Such findings have substantial implications for pharmaceutical companies since numerous products are beneficially active in the hormetic zone of the dose response. 152 The magnitude of such responses will be defined by the limitations imposed on maximal hormetic response (30–60% increase) by biological plasticity. There are, therefore, significant biological dose response restrictions based upon the hormesis concept that are imposed on the pharmaceutical industry, affecting drug discovery, development, and regulatory agency approval.
Q/A# 22. Are there drugs where the response maximum exceeds the 30–60% general ‘rule’?
In the hormesis database, there are examples in which the responses are greater than twofold that of the control group. These represent about 15–20% of the database. It is likely that some proportion of these responses may reflect high variability in a particular experiment; other elevated responses may be an indication of biological dysregulation. Furthermore, the high response may also represent an unexpectedly low control group. This general area requires considerably more research attention.
Q/A# 23. Is hormesis likely to occur only for drugs that act via specific types of mechanisms?
This is not likely the case as hormetic responses have been shown to occur in a broad range of biological models and in different cell types using a plethora of different proximate mechanisms. Therefore, the hormetic dose response occurs independent of the specific mechanism. 23
Q/A# 24. Are hormetic effects trivial with little practical application?
A large body of evidence indicates that hormetic effects are modest, being in the percentage rather than the fold zone. In about 75–90% of the cases, the maximum stimulatory responses are less than twice that of the control group, while about 60% of the cases have a maximum response of about 30–60% greater than the control group. 153 In fact, for the development of pharmaceutical agents that relate to the concept of enhancing biological performance, this is the range of possible user improvement. Since the increase is modest, it is more difficult to establish the occurrence of these effects in clinical trials without the expenditure of greater resources. It is also likely that physicians may wish that a drug would increase biological performance by more than a modest extent as demonstrated with the hormesis perspective. Nonetheless, improvement of a modest degree (30–60%) has the potential to profoundly affect patient health and the overall patterns of health seen in populations. The hormetic concept may also have important implications for the area of environmental regulations, relating to both hormesis induced benefits and harmful effects. In addition, the hormetic dose response also represents a fundamental strategy for biological processes, including developmental morphogenesis that guide the formation of biological curvatures such as capillary formation, eye shape, and a plethora of other similar biological patterns that employ concentration gradient signaling that follow the hormetic dose/concentration response. 154 Thus, the hormetic concept, while biologically subtle, is far from trivial.
In case of environmental regulation, the issue of whether responses at low levels of exposure are ‘real’ or not in human populations is problematic because chemicals are rarely tested at low doses. Regulatory agency testing strategy typically involves exposing animal models to high doses of chemicals and extrapolating via mathematical models to human populations under the assumption that the animal is an appropriate model both qualitatively and quantitatively. The principal human validation of such assumptions involves epidemiological investigations. However, these are often out of temporal sequence from the animal studies by some 20–50 years. Epidemiology also has a weak capacity to detect biological effects, rarely detecting population responses below a risk of about 2–3 times greater than the background. Biological changes in the 30–60% range as is the case for hormesis are inherently difficult for epidemiology to detect. Therefore, epidemiology is far less likely than toxicological studies to have a major impact on the assessment of possible hormetic dose responses.
Q/A# 25. Can toxicological/pharmacological mechanisms account for the quantitative dose response features of hormesis?
There are a large number of studies that have assessed the occurrence of hormetic-like biphasic dose responses. Many of these have provided mechanisms that have been summarized and compared for common biological and signaling features. Despite this array of advances in the assessment of biological systems affecting the occurrence of biphasic dose–response relationships, no mechanistic research has yet accounted for the quantitative features of the hormetic dose response. These include assessing why the ceiling effect may be at 25, 50, 75, 100, or 150% greater than the control value or why the width of the stimulatory response may be fivefold in one case while being over 1000-fold in another instance. Answers to these questions would have important implications for the process of environmental risk assessment as well as in the development of therapeutic strategies for patient treatment.
Part II. Biomedical applications
Introduction
This section represents the second part of a comprehensive assessment on how the concept of hormesis may affect the biomedical sciences, ranging from the basic experimental sciences to therapeutic applications. Part II represents the applications of hormesis for the biomedical sciences, while Part I provides the scientific foundation of hormesis.
Q/A# 26. How can the concept of hormesis help the pharmaceutical industry?
The hormesis concept has important implications for the pharmaceutical industry. It can assist in the process of drug discovery, drug development, design and interpretation of preclinical studies, and in the design of the clinical trial. The hormesis concept can also affect sample size and statistical power considerations. Hormesis will also provide critical insight into estimating a maximum treatment effect that can be obtained from drug treatments. 152 Each of these areas will be specifically discussed in subsequent questions in this article.
Q/A# 27. How does hormesis affect the therapeutic assessment of drugs?
The concept of hormesis can have an important impact on patient treatment strategies, especially in targeting of the therapeutic zone. The ideal therapeutic situation would involve having a broad hormetic zone. If this range were to extend over several orders of magnitude, it would minimize the likelihood of experiencing adverse responses due to an overdose, while also minimizing risks associated with interindividual variation (Figure 2).
Q/A# 28. How does hormesis affect the safety assessment of drugs?
While there has been no formal inclusion of the hormesis concept in the safety assessment of drugs, it could be of considerable value. For example, for some drug classes, the response in the hormetic zone may have negative public health and clinical consequences. Many antitumor drugs have the potential to enhance the proliferation of the human tumor cells within the hormetic (i.e. stimulatory) component of the concentration response relationship. 72 This has also shown to occur for antibiotics, 48 –50,155 –158 anti-yeast 56 –58 and antiviral agents. 159 –161 In a similar fashion, undesirable effects can be seen in areas in which low doses of drug may enhance smooth muscle proliferation resulting in the restriction of blood or urine flow. 67,162,163 Many other such types of potential adverse effects can occur due to stimulatory effects in the hormetic zone.
Q/A# 29. What is the role of hormesis in drug discovery?
Hormesis can play a constructive role in drug discovery. For example, in the case of antitumor drugs, numerous agents are screened in multiple biological model systems (e.g. numerous types of human tumor cell lines) with the initial intention of ‘discovering’ agents that are effective in killing tumor cells. Based on these findings, the most effective drugs are selected for further evaluation. One factor that has not been considered in this process is whether the effective compounds also demonstrate evidence of an hormetic response (i.e. enhancing cell proliferation) at low concentrations. If this is the case then the agent may have both attractive and unattractive properties. The more attractive agent would be one that both effectively killed the tumor cells, while at the same time not showing the hormetic response at the lower concentrations. A further permutation to the negative scenario is the one in which an agent that kills the tumor cells effectively but also displays hormesis and a long biological half life. A similar agent but with a shorter biological half life might prove to be more acceptable since it would not remain as long in the patient due to pharmacokinetic factors. This general type of perspective could also be applied to other drugs that are designed to kill harmful organisms or cells, such as antibiotics, antifungal, anti-yeast, and antiviral agents. Furthermore, there will also be numerous situations in which cellular proliferation is an undesirable feature of a drug’s action. This could occur in treatment related to vascular and ocular surgery. 67 Drugs targeted for their capacity to inhibit cell proliferation at high doses may in fact be hormetic at lower doses, leading to reduced patient success. In these cases as well, the hormetic dose–response concept could play a constructive role in the process of drug discovery.
Q/A# 30. What is the role of hormesis in preclinical testing?
Hormesis has the potential to play a significant role in preclinical testing, by affecting the range of scientific questions that are proposed and tested, the types of animal models that could be used to test such hypotheses, the nature of the study design, including the number and spacing of doses, sample size, statistical power, need for study replication, and in the interpretation of data. For example, selection of an animal model with a very low disease incidence in the control group will preclude being able to assess which treatment can affect a decreased incidence of disease. 164
Q/A# 31. How would hormesis affect the design and conduct of the clinical trial?
Many agents have passed preclinical testing only to fail at the clinical trial. 165 The transition from the animal model evaluation to human testing is a critical step in the drug development process. Since animal models that are used in most testing schemes are far more homogeneous than human populations, special challenges are faced in the clinical trial, making it a much harder test to pass. Human variability can exceed over two orders of magnitude in response to many agents whereas inbred animal strains may have group variation only 1/20th as large. 166 Since the hormetic zone may be generally in the 10- to 20-fold range below the threshold dose, some human subjects administered a drug in clinical trials may experience toxicity at the same dose of the drug that another individual may not have any treatment effect whereas a third person may be intermediary, experiencing a drug benefit, and no undesirable side effects. While this spectrum of interindividual response may be not unexpected, the problem comes when the data are analyzed and averaged together. The net result may be that a drug fails to show adequate drug efficacy due to human variability, failing the test. Yet, in the animal model, the drug may have been a striking success due to its normal effectiveness and low interindividual variation.
The hormetic dose response can assist the pharmaceutical industry in confronting these types of research obstacles. For example, given the nature of human interindividual variation, each individual could have their own dose response profile. In this way, clinical researchers may be able to tailor a dose response that takes into account human variability. If such variability could be identified and predicted by biomarkers, it would be of considerable value. This could also affect the process by which the study is conducted, affecting sample size and the number of subgroups to be evaluated.
Q/A# 32. Is hormesis likely to occur for all types of drugs?
In large scale tests of hormetic frequency in the pharmacological and toxicological literature, it was shown that the hormetic effects occurred at about a 40% frequency using very strict a priori entry and evaluative criteria. 167 If the criteria were made only modestly less strict, there would have been a notable increase in the estimated hormesis frequency. Thus, while it is not possible to have a definitive answer to this question, there are sufficient data to conclude that the hormetic dose response is common, reproducible, and a biological expectation in the vast majority of biological systems, end points measured, and chemical classes tested.
Hormetic dose responses have been reported for a very broad range of drugs. This would include drugs that affect responses via receptors that act via opposing receptor subtypes. Similarly, a single endogenous agonist can affect an increased or decreased rate of response when the concentration increases or decreases. Since this a very basic type of biological strategy that affects many receptors, it follows that numerous types of drugs will have the capacity to display hormetic dose responses.
Q/A# 33. Are there drugs based on the hormesis concept being used by the pharmaceutical industry without the industry being aware that this is the case?
The answer to this question is broadly affirmative as there are a plethora of drugs both on and off the market that are based on hormesis without there being any recognition of such as association by the pharmaceutical industry. 31,67,104 This is principally because of the use of different terms for the same concept as mentioned above. The failure to recognize that this is occurring has significantly contributed to the fact that the hormesis concept is not recognized as a basic biological principle by the biomedical community.
Q/A# 34. How does hormesis deal with the issue of interindividual variation?
The hormesis concept deals with interindividual variation in response to drugs, chemicals, and physical stressor agents as would other dose–response models. This question, which was explicitly addressed by Calabrese and Baldwin, 14 demonstrated that individuals that widely differ in their susceptibility to toxic agents also display hormetic dose responses to those agents. The quantitative features of the hormetic dose response in highly resistant and highly susceptible subjects were similar. The major difference in response is that the more susceptible individuals have their dose responses shifted to the left on the dose response continuum. In about 20% of the cases, the reason for the enhanced susceptibility was largely related to the absence of the hormetically based adaptive response (see the Calabrese and Baldwin 14 article for examples of these situations). Thus, the concept of hormesis can be useful to the pharmaceutical industry and regulatory agencies such as the FDA in the assessment of drug effects on heterogeneous populations. The hormesis concept could also be instrumental in guiding risk assessment activities and in the establishment of acceptable exposure levels based on insights provided on potential high-risk groups.
Q/A# 35. How would the activities of the pharmaceutical industry change if it believed that many of their drugs were based on hormesis?
It is likely that the industry would explore the possibility that the hormesis concept is a general one with broad applications, and one that could enhance the more efficient use of existing resources. 168 –172 This concept would quickly infiltrate the educational system affecting graduate education, the type of research funded, the content and the language of professional journals, the programs of annual meetings, and corporate scientific goals and strategies. With respect to the later, the HDR would provide guidance on the magnitude of potential drug effects and this would drive whether a therapeutic agent may have clinical utility. The width of the hormetic effect would facilitate estimating the therapeutic window or ratio. Not only would this be helpful in the comparative assessment of drugs but also in the type of guidance provided to clinicians. Adopting a hormetic dose response perspective may lead the industry to review many of their agents, especially those with encouraging preclinical data that failed the clinical trials, and determine whether their overall assessment of these drugs could use revision, whether the marketing of approved drugs could be altered or improved and whether there could be new applications for these agents (Table 3).
Implications of hormesis for clinical practices/pharmaceutical companies.

Q/A# 36. Does the hormesis effect vary with age from the young to the elderly?
The question has not been systematically studied. However, there are numerous examples in the peer-reviewed literature demonstrating differential susceptibility by age 14 and with age–gender interactions. In many of these studies, hormetic responses have been demonstrated.
Q/A# 37. What are the limitations of epidemiology in the study of hormesis?
If a population comprised multiple subgroups with differential susceptibility to toxic substances as well as having different capacities to display hormesis, these factors will affect the capacity to detect hormetic dose responses. Assume further that these different population subgroups comprise varying percentages of the entire population. These three conditions are likely to be common within humans. Using these three factors, we have conducted numerous simulations designed to estimate overall population-based dose responses across the dose–response continuum. Depending upon the number of subgroups, their specific dose–response susceptibilities and the varying proportions in the population, it is possible to derive a broad range of population-based dose–response relationships, including linear, threshold, biphasic, and polyphasic dose–response relationships. 1
Such simulation exercises are significant in several ways. First, they demonstrate that the shape of the dose response within a heterogeneous population is the summation of the dose responses of all contributing subgroups. Change the population subgroup characteristics and one could profoundly alter the population-based dose response. Second, it illustrates the difficulties that can be involved in assessing the hormesis concept within the context of a population with either a large number of population subgroups or high interindividual variation. Third, these findings suggest that the use of a default dose–response curve of possibly any type in the risk assessment process may be a questionable concept. Fourth, the complexity of population subgroup variables and how this affects the population structure would likely be an important factor affecting epidemiological study replication especially for relatively minor risk factors or for agents that act hormetically. It was the recognition of such population subgroup variability and their differential contribution to the total population that supported a decision to principally assess the hormesis concept using homogenous animal models within an experimental context.
Q/A# 38. Can hormesis contribute to the healthy worker effect?
The concept of the healthy worker effect is well known in occupational epidemiology. Workers at chemical, nuclear, and other facilities typically show lower rates of various types of diseases than the general public. 173 The typical explanation of this so-called healthy worker effect has related to the initial selection of a healthy individual for entry into the work force. Such workers might be required to pass entry physical and mental examinations as well as having past success in other positions. These activities would tend to result in the exclusion of some proportion of individuals potentially at risk to a wide range of chronic diseases. These individuals may be excluded from the workforce via company selection practices as well as via self selection. Nonetheless, these individuals who have been selected out of the workforce will still have their health responses recorded in county and state disease related data bases that serve as a control group in occupational epidemiology investigations. The workers and the general public to which they are compared are not necessarily ideal comparisons. It is not surprising for workers, even those in occupations involving potential exposures to hazardous substances, to display disease rates lower than the general public or control group. Researchers have generally concluded that the lower disease incidence in such workers is not the result of any beneficial effects due to exposure to toxic substances in the workforce but rather due to the so-called healthy worker effect. However, while the healthy worker effect concept offers explanatory value and face validity, it may not be the full explanation as to why workers at chemical and nuclear facilities typically show less disease incidence than the general public. One potential additional explanation is that occupational exposures to low levels of toxic substances and ionizing radiation may be inducing hormetic-adaptive responses that result in overall health benefits. The hormetic dose–response hypothesis, therefore, is a factor that should be considered by occupational epidemiologists in designing of epidemiological studies. The hormesis phenomenon has long been ignored by the epidemiological community in such occupational settings. However, in a world of low–dose exposures, it is expected that hormetic effects may be operational, making it a concept that epidemiologists must address.
Q/A# 39. How can statistical simulations improve the assessment of hormesis?
All biological responses express some degree of normal variation, ranging from relatively small to rather large. The variability may be due to genetic, environmental, or experimental conditions. Let us assume that the variability of your test system is such that the control shows a mean value that is normalized to 100% with one standard deviation being ±20%. Thus, 99% (i.e. 3 SDs) of the population responses would be within a range of ±60% of the control mean. Let us now assume that we are conducting a dose–response study with a control and five treatments and further assume that there is no treatment effect and the control and five treatments all display a mean of 100% and the same variability. Let us now conduct a computer simulation of 10,000 dose responses assuming that the control and treatments have their responses occurring in a random manner according to the variability description given above. What would emerge is that a certain percentage of the 10,000 dose responses would display excellent examples of hormesis, yet they would all be false positives, due to normal variation. One could change the variability assumption to 10 or 50%, and the number of false positive hormetic responses would decrease or increase, respectively. Such computer simulations are important as they indicate how susceptible one’s experimental system is to false positive values. Simulation is an important statistical tool that has broad application for biomedical researchers but especially for those interested in assessing possible hormesis effects. See Calabrese et al. 174 for an example of the value of statistical simulations in the assessment of hormesis for mutations.
Q/A# 40. How do triphasic and other polyphasic dose responses relate to hormesis?
Polyphasic dose–response relationships have been reported in the biomedical literature. In general, these dose responses have not been broadly investigated, possibly due to the heightened difficulties of using study designs that are even more extensive than those needed to assess the hormetic dose response. The Web of Science database lists over 20 times more citations for biphasic than for triphasic dose responses.
There are at least two general ways by which triphasic and polyphasic dose responses may occur and be related to the concept of hormetic. Triphasic dose response: some types of hormetic dose responses occur as a result of an initial disruption of homeostasis followed by a modest overcompensation stimulatory response. This type of hormetic dose response has been widely reported, requiring a time component for detection. 175 –179 Within this context, it is not unreasonable to assume that many, if not most biological systems, have evolved the capacity to detect damage before adaptive repair processes are induced. It is also not unreasonable to assume that there may be a ‘damage detection threshold’ that must be exceeded in order to induce reparative responses. Furthermore, it would be important that biological systems not engage in a biological version of ‘false alarm’ signaling, or at least not on numerous occasions and/or at inappropriate times. This would suggest that the threshold of detection would likely be set sufficiently high to avoid the so-called biological false alarm yet low enough to affect the initiation of repair responses in order to prevent damage of a potentially serious nature. Where to set the threshold would be important and must have been a factor that was broadly tested and highly selected for within an evolutionary context. Furthermore, given human heterogeneity, it is possible that there is a distribution of damage detection thresholds. Those individuals with a very low-damage detection threshold would be expected to be in a frequent state of biological over-reactivity. Individuals with a very high-damage detection threshold would be at the opposite end of that spectrum, being too slow to respond to injury or damage. For those individuals in the normal part of the population distribution one could expect to observe that some damage that exceeded ‘normal’ background but not enough damage to be ‘detected’ for repair purposes. In order for a new and more aggressive repair process to be induced sufficiently more damage than background would need to occur. If a low dose of a toxic substance induced such a modest increase in damage, but not enough to be detected, this damage may be readily measured. At higher doses, more damage may occur – perhaps exceeding the detection threshold. This process would eventually result in a triphasic dose response. There would be undetected and unrepaired damage (i.e. phase 1 of the dose–response curve, i.e. an upward occurrence of damage). At a higher dose, damage level would become detected. This would lead to the initiation of a repair process and the occurrence of a modest overcompensation response and less damage than seen in the unexposed controls (i.e. phase 2 of the dose–response curve, that is, the occurrence of the hormetic reduction in damage). At still higher doses, more damage occurs, overwhelming the repair process and leading to massive damage (phase 3 of the dose–response curve). Figure 3 provides a description of the triphasic dose response.

Triphasic dose response.

Hormesis dose response and the therapeutic zone.
The triphasic dose response as described above may be seen in the research of Sykes et al. 176 that demonstrated a radiation-induced triphasic dose response in the PKZ1 animal model. This research indicates that biological systems required some level of damage to occur above background before the system detected the newly induced changes (i.e. mutations). Once detected, hormetic-adaptive procedures became activated and the biphasic part of the dose–response emerged. With the addition of a higher exposure, the triphasic dose response was displayed. Much remains to be clarified for triphasic dose responses. Perhaps the reason that Sykes demonstrated the triphasic dose response was that the model has a high detection threshold. If other models had lower detection thresholds, it would be harder to discern a triphasic dose response. Thus, while it is likely that the triphasic response may commonly occur, it is also a phenomenon that would be difficult to discern unless attempts were made to detect relatively faint signals at low dose in a dose response context that was resource intensive.
It should be noted that models of multiple phase dose responses have been reported. For example, substance P enhances neutrophil adhesion with two distinct peaks. The initial peak between 10−17 and 10−13 M was mediated by the NK1 (neurokinin-1) receptor, while the second peak (10−11 to 10−7) was mediated by both NK1 and NK2 (neurokinin-2) receptors. 177
Q/A# 41. What are the biomedical implications of a triphasic dose response?
The triphasic dose response suggests that at exposures below the hormetic adaptive response that risks would theoretically increase. 176,178,179 The triphasic dose response concept has a reasonable theoretical foundation but requires better documentation across model and biological end point.
Q/A# 42. How will the trend toward high throughput assays affect observations of hormesis?
The growing use of high throughput assays (i.e. an experimental method that allows quick evaluations of large numbers of test compounds) will provide an experimental framework to identify potential hormetic dose responses. The use of a large number of concentrations, especially below the toxic threshold, increases the likelihood of observing hormetic responses. 180 A major limitation with high throughput assays is that they are often employed as a screening technique. In case of hormetic dose responses, a strong effort to replicate findings is required. If researchers are primarily concerned with high concentration effects in a high throughput assay, it is likely that possible evidence of hormetic dose responses would be missed. Investigators need to evaluate the entire concentration response continuum, including the possibility of hormetic responses in the low concentration zone and then be willing to validate the hormetic effects with further testing.
Q/A# 43. May drugs be acting hormetically even though the experimental data appear inconsistent with this interpretation?
This sounds like an odd question but in fact it is not and the answer is possibly. Calabrese et al. 57 demonstrated that many chemicals tested in the NCI (National Cancer Institute) antitumor agent database displayed hormetic dose–response relationships. In a subsequent analysis, a distribution of chemical responses was made assuming a threshold model, with random responses below the estimated threshold dose. The 50th percentile chemical was constrained to have a calculated value of 100% with the remaining chemicals being symmetrically distributed above and below the median response (100%) value. The actual data showed a distribution of treatment responses that was skewed to the right across the entire distribution of chemicals assuming a threshold for treatment responses. That is, the chemicals showed higher response values than predicted by the threshold model for the entire population of chemicals tested consistent with the hormetic dose–response model. The findings were interpreted as indicating that all chemicals demonstrated evidence supportive of an hormetic dose response, even when the average response was less than 100%. However, is this possible? In all studies, random variation occurs. It may be estimated and then used to simulate responses of all chemicals assuming no treatment effect using a threshold model. For example, the lowest response predicted as a chemical consistent with the threshold model within this distribution of chemicals was about 90%. Yet the lowest response of a chemical based on the real data was about 95%. This response exceeds that predicted by the threshold response by 5%, a response consistent with an hormesis interpretation. Many investigators may consider such responding agents as not showing evidence consistent with an hormetic interpretation. However, within the context of a large chemical screening assessment study that interpretation would be incorrect. Thus, these data should be interpreted as indicating that all the assessed chemicals satisfying the a priori entry criteria provided evidence consistent with an hormetic interpretation. 58
Q/A# 44. How should hormesis affect the education of pharmacologists, pharmacists, and government regulators?
The concept of hormesis should be incorporated into introductory courses on pharmacology and toxicology. In these academic courses, the history of the dose response should be presented 39,40,181 –185 along with the reasons why the hormetic dose response was marginalized within the biomedical community and the reasons for its scientific resurgence. Emphasis should be placed on the development of a common dose–response terminology, following the suggestions of a large grouping of interdisciplinary biomedical scientists. 31 Educational practices should also address how hormetic dose responses are evaluated, and applied within medical and pharmaceutical settings. Furthermore, the concept of hormesis should be actively integrated into graduate student research activities.
Q/A# 45. How does hormesis affect the occurrence of drug–induced side effects?
Hormesis is a biphasic dose response that often results from the actions of partial agonists and partial antagonists. Partial agonists/antagonists are extremely common, affecting most, if not all, receptor systems. The use of partial agonists/antagonists diminishes the likelihood of adverse effects while creating a broader dose range over which the response would occur. 186 –189 These two features are extremely important for health and survival. One can imagine the survival implications of individuals affected by adverse side effects, ranging from headaches to dizziness, to seeing double, among others. Therefore, a major factor in evolutionary success is to minimize undesirable side effects of endogenous agonists. As one can see with the modern pharmaceutical world this is not an easy task. However, this could be another critical dimension of hormesis within an evolutionary context.
Q/A# 46. How does hormesis deal with the issue of chemical/drug interactions
Hormesis has important implications for the general area of chemical interactions. In case of the dose response, interactions may occur with exposures above or below the threshold. Typically, what is presented in textbooks on toxicology are those interactions occurring above the toxic threshold. 190 Nonetheless, some studies have explored whether and to what extent chemical interactions occur when both agents are in the hormetic zone. 191 Of particular interest to the pharmaceutical industry is that these studies have focused on memory in rodents. It was shown that numerous agents could increase memory via hormetic processes. 192 –194 These initial studies lead to the development of chemical interaction experiments using a broad range of possible experimental drug dose combinations. The maximum increase in memory performance did not exceed that induced by a single agent. Nonetheless, while at very low doses, there was evidence of chemical additivity and synergy; however, the maximum response never was greater than any of these agents acting individually at their optimal dose. Thus, there was evidence of interaction but it did not exceed the hormetic maxima. The capacity for interactions occurred in a response range far below the maximal hormetic response level. This suggests that the response maximum of the interaction was also constrained by the limits of biological plasticity, which are revealed in the hormetic maxima. These observations have important implications for the pharmaceutical industry as they suggest that under normal biological conditions, the response maxima will conform to that described by the hormetic dose response. This effectively places a cap on the maximum biological performance of a drug. This also has implications for the study design of preclinical and clinical trial investigations for biological end points that are concerned with biological performance. 11,67,104
Q/A# 47. How does hormesis relate to quantitative structure activity relationships (QSAR)?
The role of chemical structure in affecting the hormetic dose response is far from clear. There is strong evidence that hormesis can occur from a variety of unrelated chemical and physical stressor agents. On the other hand, there are a large number of examples in which a chemical-induced hormetic dose response has an unmistakable chemical structural relationship. It is important to better clarify the range of nonspecific hormetic stress responses and the domain of the highly specified chemical structural determinants of hormetic responses. 20,187 –189,195,196 Regardless of which of these categories a dose response resides the available data indicate that the quantitative features of the hormetic dose responses for either group are similar. Such observations are commonly reported and highly reproducible. Therefore, it appears that both are constrained by the limits imposed by biological plasticity. 197
Q/A# 48. Is there such a phenomenon as ‘aberrant’ hormesis?
There are cases in which the hormetic dose response can lead to undesirable and harmful effects in humans. This was first recognized in the time of Hugo Schulz as seen in the publication of Martius-Rostock 198 who noted that at low doses ionizing radiation could enhance the growth of tumors while being inhibitory at high doses. This concept was independently recognized some 20 years later by researchers in the FDA who reported that low doses of several antibiotics could stimulate the proliferation of harmful bacteria resulting in the death of the experimental animals. 107,108 Similar types of potentially harmful effects to humans have been proposed for antitumor agents as well 40 and that this situation may be particularly common for drugs with long biological half-lives. It is also likely that drugs or environmental chemicals that enhance the proliferation of the prostate could contribute to the development of an enlarged prostate, having clinical implications. 67 There are many other examples that have been discussed in the literature in which an hormetic dose response could yield an undesirable effect. These would be examples that are referred to here as ‘aberrant’ hormesis since this dose response reflects the same pattern of response, the same quantitative features of the dose response and same mechanistic strategies that are seen when positive adaptive responses are observed.
Q/A# 49. Can hormesis enhance the occurrence of cancer?
There is the likelihood that an hormetic dose response can contribute to the development of cancer. As noted above, this may occur via the capacity of low doses of endogenous or exogenous agents to enhance the proliferation of tumor cells as well as affecting their capacity to be transformed into more aggressive tumors. 199,200 In this later respect, hormetic dose responses have been reported to occur with p-glyco-protein-ATPases and can increase the capacity of tumor cells to prevent the entry of antitumor agents. 201 Based on this concept, it would be predicted that the hormesis concept could play a role in tumor recurrence. 199
Q/A# 50. Are preconditioning/postconditioning responses examples of hormesis?
Preconditioning is a term that was introduced into medicine by researchers 202 who found that a prior brief hypoxic stress to dogs markedly reduced their risk of cardiac damage following a subsequent myocardial infarct within the next 24–48 hours. The prior stress induced an adaptive response that lead to the observed protection; the investigators labeled this phenomenon as ‘preconditioning’. Despite its novelty in the medical domain, the preconditioning concept was not new to the field of toxicology, where a prior low dose exposure to a toxic substance could also significantly protect against the occurrence of toxicity from a subsequent and more massive exposure. This phenomenon had been reported for chloroform, carbon tetrachloride, and numerous other agents starting in the 1960s, 203 –205 and for chemical mutagens, starting with Samson and Cairns in 1977. 206 By 1984, this phenomenon was reported for radiation induced mutations. 207 Since researchers in these other biological fields used different terms such as autoprotection, heteroprotection, and adaptive response, these earlier articles were missed by Murray et al., 202 thus leading to their use of ‘preconditioning’ for the same general biological concept. Of particular relevance to the hormesis concept is that the preconditioning dose may be optimized to affect the most protective response to the subsequent massive stress. Responses of a wide range of preconditioning doses reveal an underlying hormetic dose response. 168 Thus, the preconditioning phenomenon is a specific type of hormetic dose response, demonstrating the same quantitative features of the hormetic dose response.
With respect to postconditioning, this phenomenon occurs when a low dose of the stressor agent/condition is administered following a massive exposure/trauma. The same type of protective response has been reported in animal models and patients as occurs within preconditioning procedures. The dose response for postconditioning also follows the hormesis model. Based on such observations pre- and postconditioning concepts may be incorporated into an integrative and consistent biomedical terminology framed within an hormetic context. 30,31
Q/A# 51. Is angina pain prior to a heart attack an example of an hormetic preconditioning effect?
In the aftermath of the experimental studies in which preconditioning was demonstrated to reduce cardiac damage in dogs, which subsequently experienced a massive myocardial infarction, epidemiologists determined that heart attack patients who had experienced angina pain due to mild hypoxia in the days just prior to their heart attack had less cardiac damage than those patients that did not experience the prior angina pain. 208,209 These observations lead to the hypothesis that prior angina pain due to hypoxia is a manifestation of preconditioning similar to that which was studied in the canine model.
Q/A# 52. Is there an hormetic relationship between antigen exposure and antibody production?
In 1981, Volkman et al. 210 indicated that exposure to the antigen keyhole limpet hemocyan (KLH) in human peripheral blood mononuclear cells induced a biphasic dose response for the production of a specific antibody to the KLH antigen. Low doses of KLH resulted in a several fold increase in antibody production, whereas at higher doses, the production of the specific antibody was profoundly decreased. In this study, the subjects first received a primary subcutaneous immunization from exposure to KLH followed by booster immunization. An in vitro dose–response study was then conducted 2 weeks following the booster exposure. Of interest was a progressive increase in serum immunoglobulin M levels, reflective of an expansion of polyclonal antibody production and a decoupling from the specific antibody response. A similar type of response was also reported for pokeweed mitogen-derived anti-KLH. Such investigations indicate that the potential for antigens to induce specific antibody production can be highly dose dependent and biphasic in nature. There was also sufficient interindividual variation in these investigations to suggest that the optimal dose of antigen for antibody production could markedly vary between individuals. These findings raise important questions concerning the dosing strategy for specific antibody production in the execution of mass vaccine distribution programs.
Q/A# 53. Can low levels of fluroquinolone antibiotics protect tendons at low doses but enhance tendon rupture at higher doses?
Numerous studies have been published indicating that the general class of fluoroquinolone antibiotics can pose a risk of tendon rupture, especially the Achilles tendon, and particularly in elderly patients. 211,212 The average duration of treatment prior to tendon injury is relatively short being about 2 weeks, with some patients showing extreme susceptibility with rupture responses after only a few doses. There are a number of other contributing factors such as renal disease, cotreatment with glucocorticoids, and conditions created by the bacterial infectious disease process itself such as increases in collagenase activity in circulating macrophages. In studies designed to clarify the role of oxidative stress induced in rabbit tendon cells by various fluoroquinolones, Pouzaud et al. 213 reported that each of the four drugs tested displayed a biphasic dose response for cellular viability, with there being a low-dose stimulation and a high dose inhibition. The low stimulatory concentrations were in the serum concentration zone for human patients. These findings raised the question whether fluoroquinolones may not only enhance the occurrence of tendon rupture but also whether at lower doses may also be protective of tendon tissue. While the predominant research focus has been on the capacity of these agents to cause tendinitis and tendon rupture, the findings of Pouzaud et al. 213 indicate the occurrence of an hormetic dose response. Research as to whether fluoroquinolones may help prevent tendon tears at low doses is worthy of further investigation.
Q/A# 54. Does H2S act via hormetic processes?
The simple answer is that it does so in a wide variety of ways, as amply demonstrated in numerous animal models for a broad range of end points. Most striking are reports that H2S displays hormesis via both pre- and postconditioning in rodents with the response being both time and dose dependent. The optimal time for the administration of the H2S donor was 1 day prior to the induction of the ischemia-reperfusion process. If H2S was administered 5 days prior to the ischemia-reperfusion, no protection occurred. Intermediate protection was observed at 3 days prior to the ischemia-reperfusion process. The dose response was U-shaped with the optimal dose decreasing damage by approximately 60%. A similar, but less dramatic, decrease in damage occurred using a postconditioning protocol, with a maximal decrease in damage of approximately 30%. 214 H2S also protected the liver against damage due to ischemia reperfusion when the H2S was administered at 55 min into a 1-hour ischemia (i.e. just before the onset of reperfusion). While the optimal H2S treatment reduced the damage by approximately 60%, the relationship between the optimal dose and the dose causing the onset of liver damage was very close, being within a factor of two. The chemoprotective effects of H2S have also been reported in wound healing experiments 215 using multiple endpoints, all displaying clear evidence of hormesis. H2S also modulated vascular tension in the aorta 216 and immunological functions 217 in a manner consistent with the hormetic dose response. There is also evidence that H2S enhances the proliferation of human colonic tumor cell lines in an hormetic dose response fashion. These observations suggest that H2S might have the potential to act as a promoter of colonic cancer. 218 These findings are generally consistent with the capacity of H2S to enhance wound healing, in part, via the capacity to biphasically increase the proliferation of endothelial cells, providing a proangiogenic function. In conclusion, the recognition of hormetic dose responses with H2S is broadly developed and well established.
Q/A# 55. Can hormesis be incorporated into and enhance personalized medicine?
Pharmaceutical companies are challenged by the frequent failure of candidate drugs to successfully pass the clinical trial. The reasons for such failures can be varied and complex as well as agent specific. However, important general reasons for failure are related to the occurrence of human response heterogeneity among the test population to the candidate drug. This heterogeneity of response will relate to both the potential for an adverse response as well as the optimal dosage to achieve the intended therapeutic response. Averaging responses across a group of subjects with wide interindividual variability is problematic since it will flatten out the potential beneficial response, perhaps leading to a conclusion that the drug lacks sufficient efficacy. The issue may well be that most individuals may respond in a positive way to the candidate drug but that each population subgroup or for that matter, individual, may display their own specific optimal dose. One dose fits all protocols, therefore, are likely to provide misleadingly low efficacy potential results and lead to the failure of drugs in the clinical trial.
The concept of hormesis incorporates interindividual variation within its testing and evaluation, providing a dose response foundation upon which the personalized medicine framework can be based. This would be the case for not only avoiding drug-induced injury but also for the achieving dose optimization for intended drug benefits. The continued failure to properly incorporate the HDR concept in the development of clinical protocols for the assessment of both potentially undesirable and beneficial effects is therefore a possible explanatory factor for why excellent candidate drugs often fail clinical trials.
Q/A# 56. Does failure to incorporate hormetic behaviors underlie the metabolic syndrome?
The metabolic syndrome, a condition that precedes the onset of type 2 diabetes, is characterized by obesity along with elevated triglycerides, reduced high-density lipoprotein, hypertension, and evidence of pathological insulin resistance based on high plasma glucose concentrations or prior diabetes. The evolutionary foundation of the metabolic syndrome as well as its heightened occurrence in modern industrialized societies and its effective treatment has been placed within an hormetic context by Nunn et al. 219,220 These authors have proposed that the metabolic syndrome represents part of a biological continuum, sitting at the opposite end of the oxidative stress spectrum to the caloric restriction phenotype (i.e. propensity to minimize energy expenditure, i.e. biological ‘thriftiness’) with its enhanced longevity. Both these opposing biological phenotypes are mediated by the involvement of the insulin/insulin-like growth factor axis. In the case of the caloric restriction phenotype, there is an increased activity of DAF16/ FOXO (forkhead) stress transcription factors that inhibit insulin signaling. Insulin resistance leads to fat deposition, while diminishing oxidative redox signaling induced stress. Insulin resistance is determined by the capacity to resist oxidative stress, which itself is mediated by preconditioning hormesis stimuli (e.g. fasting, physical activity, plant polyphenols, unsaturated fats, alcohol, a variety of pharmacological agents, as well as a broad spectrum of environmental stressor agents including low doses of ionizing radiation). According to Nunn et al., 219,220 the metabolic syndrome may therefore represent a physiological tipping point (i.e. their thrifty-inflammatory tipping point) where the insulin resistance transitions from being restricted to muscle to affecting the onset of inflammatory responses in a broad range of tissues, especially adipose. Temporal and tissue-specific insulin resistance would be beneficial if one stays within the so-called hormetic exposure zone while becoming unhealthy, if placed within the framework of the so-called modern sedentary lifestyle. Thus, this perspective provides a further generalizing of the hormetic dose–response concept into that of physiological homeostasis.
Q/A# 57. Is radiation therapy an example of hormesis?
The first half of the 20th century witnessed the widespread use of x-rays to treat a broad range of inflammatory and infection diseases. 221,222 This literature is extensive and generally consistent in the claim that low doses (i.e. fractions of the erythema dose) could enhance the healing process while higher doses were inhibitory. Research was published in leading journals, such as New England Journal of Medicine, Journal of the American Medical Association, and Radiology, with investigators from the leading academic institutions. The primary publication limitations were that the data are generally from case studies although this was not always the case. 223,224 When controls were used studies similarly supported a low-dose beneficial treatment response. The use of x-rays for inflammation and infectious disease was, for the most part, eliminated from practice as a result of the introduction of antibiotics form the early 1940s onward. Nonetheless, the biphasic nature of these x-ray induced beneficial effects of the early 20th century is both an important historical medical legacy but perhaps an area that warrants reexamination in light of current concerns over antibiotic resistance.
Q/A# 58. Could wound healing be enhanced via hormesis?
The idea that wound healing could be accelerated originated with Alexis Carrel in approximately 1907 while working at the Rockefeller Institute. Carrel’s research evaluated the wound healing acceleration concept in both in vivo animal research and via the use of tissue culture. Efforts arising from this research were pivotal in the development and maturation of tissue culture. In fact, one of Carrel’s young students, Raymond C. Parker, would come to play a significant role in the production of the polio virus for the Salk vaccine based upon an optimization procedure intended to enhance virus production using the hormetic concept (see Q/A# 59).
In the first half of the 20th century, wound healing acceleration was centered on several areas of research: tissue extracts (i.e. skin, heart, liver, and brain) from adults and embryos using tissue culture, whole animals, and in clinical settings, typically with application to indolent wounds, numerous sulfhydryl compounds, wound hormones, cartilage extract, x-rays, chemicals that induced hyperplasia, vitamins, and a broad mix of pharmaceutical-like agents. 225 –227
The results of such testing generally failed to establish a major supported area of research in which wound healing showed convincing enhancements. However, research with cartilage showed generally consistent healing in the range of about 20–30% increase in wound tensile strength. 225 In case of tissue extracts, some notable articles indicate that indoledent wound healing could markedly improve. 226,227 However, this area failed to proceed as a result of the profound success of antibiotic treatments in the second half of the 20th century.
With the 1970s came the discovery of growth factor families renewing interest in the possibility of wound healing acceleration. In addition, the process of wound healing has been distilled into numerous discrete steps. 228 Numerous examples have now been published showing that both individual steps and the overall process of wound healing could be accelerated by specific treatments. Thus, over the past several decades, there has been enormous progress into the area of wound healing and its possible acceleration. There are now several hundred articles that have been published which have demonstrated that numerous chemicals (e.g. growth factors and numerous other agents), laser, ultrasound, and shock waves accelerate wound healing in in vitro and whole animal systems. Some of the findings have been incorporated into clinical settings. A large number of studies have explored wound healing within extensive dose response frameworks. In many of these instances, the findings have revealed a hormetic dose response. These findings generally indicate that the magnitude of the accelerated wound healing is bounded by the limits imposed by the quantitative features of the hormetic dose response.
Q/A# 59. Did the hormesis concept affect the Salk vaccine?
The Salk vaccine was derived from the capacity of monkey kidney cells to synthesize the polio virus. The synthesis occurred in a synthetic nutrient media called Mixture 199. During the development of nutrient media, it was necessary to add several fat soluble vitamins (vitamins A and D) and cholesterol. A small amount of ethanol was added in order to place these agents into solution. Further testing was then undertaken to determine what ethanol concentrations would adversely affect cell function using a broad concentration range (0.001–5.0%). It was determined that concentrations of >1.0% ethanol were toxic. However, concentrations of ethanol in the range of 0.2–0.5% were stimulated over controls by 30–40%. 229 Therefore, the concentration response was hormetic, showing the characteristic low-dose stimulation, high dose inhibition. Using these experiments, the concentration of ethanol in the mixture was increased to 0.21% based on its low concentration stimulatory action. This concentration was used in Mixture 199 and later Mixture 597 for polio virus production for the next two decades. 230 Thus, the hormesis concept was incorporated into the Salk vaccine, with the goal of enhancing the more efficient production of the virus.
Q/A# 60. How can hormesis affect risk assessment?
There are fundamental ways in which hormesis can affect risk assessment. These are summarized in Table 4.
Implications of hormesis for toxicological risk assessment.

Q/A# 61. Does EPA include the concept of hormesis in risk assessment policy and practices?
The US Environmental Protection Agency (EPA) 231 risk assessment process explicitly states that its goal is to prevent pollutant-induced harm and not to consider possible health benefits (i.e. ‘as the purpose of a risk assessment is to identify risk (harm, adverse effects etc), effects that appear to be adaptive, non-adverse or beneficial may not be mentioned’). This EPA risk assessment goal establishes a framework in which the hormetic dose response could be accepted or ignored. For example, this risk assessment framework indicates that EPA would consider estimated harm related to an hormetic/biphasic dose response, but not if benefits occurred. In case of potential harmful effects, the EPA could accept data showing a low-dose hormetic stimulation leading to adverse health effects (e.g. increased prostate gland size 30 or an acceleration in a developmental process such as the onset of puberty). 232,233 In contrast, the EPA would disregard the hormetic response when there was a reduction in population-based risk. For example, in case of carcinogens, EPA could recommend an exposure standard at a very low dose, essentially a de minimus risk, using a linear at low-dose model instead of following hormetic dose–response data, which demonstrated a significant health benefit (i.e. reduction in tumor incidence) within the observable experimental data. This example demonstrates how the EPA concept of risk is too limited. For example, in the carcinogen case, there is an assumed risk of harm, and there is the risk of losing a demonstrated benefit. Both types of risk need to be integrated within a public health assessment. This perspective was supported in a recent survey of the membership of the US Society of Toxicology and the Society of Risk Analysis, where 68% supported the incorporation of health benefits into the risk assessment process. 234 The data indicate that the risk assessment policy position of EPA is inconsistent with the strong majority of toxicologists and risk assessors.
The EPA risk assessment policy statement is also inconsistent with long standing community based fluoridation programs. The fluoride risk assessments have historically focused on preventing harm at high doses, while ensuring the existence of community-based drinking water fluoridation programs at lower doses, that is, a beneficial dental response. 15
Another conflict of this risk assessment policy occurs when an agent displays a beneficial effect for one population subgroup at a dose that would be harmful to another (e.g. high-risk group). In similar situations, the same agent may affect a beneficial response with the high-risk group at a low dose, while having no measurable biological response on the normal population subgroup at this dose. This general population-based dose–response possibility would likely be a common occurrence.
These policy based inconsistencies reveal that the EPA risk assessment policy guidance document fails to consider interindividual variability in a hormesis-based risk assessment process. If hormesis is not incorporated into the risk assessment definition, the EPA policy would lead to greater overall population risks of adverse health effects.
If the hormesis concept were to become formally incorporated into the risk assessment process, it would create a series of new challenges and opportunities to improve the public health 235,236 . With the incorporation of the hormesis concept into the risk assessment process, there would be different population-based ‘stake-holders’, with each displaying a different group dose response. That is, the hormetic benefits or adverse effects will occur at different dose regions for the various population subgroups. Therefore, the benefits and risks to each of population subgroup would now need to be considered, quantified, and made explicit in the risk assessment process and then be integrated within a comprehensive negotiation or risk management decision making process. While this presents a new challenge for agencies like EPA, it also reflects emerging toxicological developments that need to be properly managed, with the goal of optimizing the population-based response.
The 2004 EPA risk assessment guidance document fails to integrate the health assessment needs of society. Its limited definition of a risk assessment creates a significant gap in the assessment of human health, leaving an institutional blind spot. This EPA strategy will lead to inadequate population-based health assessments and wasteful allocation of resources. The above assessment demonstrates that the EPA risk assessment policy is insufficiently flexible to address the above real life examples of hormesis and interindividual variability, thereby suggesting a revision to the EPA definition of a risk assessment to one that estimates the net population-based disease incidence at each level of incremental exposure. 15
Final perspectives
The hormesis concept is a fundamental dose response, highly conserved, and set in an evolutionary framework. The hormesis concept establishes a biological context for some of the key ‘rules’ of pharmacology and toxicology within which pharmaceutical companies compete and where physicians work. 168 –173 Hormesis also offers the pharmaceutical industry dose response quantitative parameters that describe and constrain the maximum potential of a drug that is designed to enhance biological performance. Thus, the hormetic dose response should play a significant role affecting the study design, dose number, dose range, and statistical power, all factors that enhance the capacity to address key biological questions more incisively. Knowledge of hormesis is also significant since it helps to clarify the health implications where agents induce harmful responses below the threshold. Given its potential significance, it is important to find practical ways to incorporate it within the educational framework of physicians, pharmacologists, and biomedical scientists. Since hormesis is a basic biological concept leaders of these fields need to ‘institutionalize’ the teaching of this concept and its applications. Considerable progress has been achieved along these lines within the field of toxicology where hormesis is prominent in leading textbooks. It is expected that this concept will become more generally integrated within the formal educational process of these important biomedical, therapeutic, and public health disciplines over the next few years. Such an educational transformation will provide the foundation for integrating hormetic principles into modern medicine and biomedical research and their widespread applications.
Footnotes
Authors’ Note
The US Government is authorized to reproduce and distribute for governmental purposes notwithstanding any copyright notation thereon. The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsement, either expressed or implied, of the Air Force Office of Scientific Research or the US Government.The sponsors had no role in study design, collection, analysis and interpretation of data, writing of the report, and decision to submit the manuscript for publication. This document contains 9 similar questions/answers (Q/A#: 1, 5, 6, 9, 12, 15, 17, 18, and 50) as previously published in Biogerontology 12 ; since they relate to core biological concepts for hormesis. However, each of the answers to these questions has been restructured and edited for the present manuscript. In general, the degree of overlap between the two articles was carefully evaluated and determined to be appropriate. The lead author (EJC) has consulted ExxonMobil on the topic of soil ingestion.
Funding
This work was sponsored by the Air Force Office of Scientific Research, Air Force Material Command, USAF, under grant number FA9550-07-1-0248. The lead author (EJC) has received unrestricted research grants from ExxonMobil Foundation concerning hormesis. However, this funding was not applied to the present manuscript.
Conflict of Interest
The authors declared no conflicts of interest.
