Abstract
Lead (Pb) is a developmental neurotoxicant found in industrial activities, many of them already prohibited worldwide. This study aimed to evaluate current blood Pb (PbB) levels in children in Cordoba, Argentina, and to compare these with similar studies performed before Pb was banned in gasoline in 1996. We also sought to identify mechanistically relevant biomarkers by measuring δ-aminolevulinic acid dehydratase (δ-ALAD), superoxide dismutase (SOD), and catalase (CAT) activities. We finally aimed to determine whether sociodemographic characteristics are associated with Pb toxicity. Blood samples collected from 161 healthy children between September 2009 and February 2010 revealed mean PbB levels of 2.58 ± 0.30 µg/dl. Enzymatic δ-ALAD, CAT, and SOD activities showed no significant variations when plotted against PbB levels. Finally, children living in the suburbs have higher PbB levels than their city counterparts, while low socioeconomic status increased δ-ALAD inhibition compared with that of middle-income children. Overall, these results evidenced a substantial reduction in exposure to Pb in this pediatric population over a decade after Pb was restricted in gasoline and reveal the importance of pursuing novel biomarkers of toxicity along with the sociodemographic profile to complement Pb diagnosis.
Introduction
Lead (Pb) is a highly toxic metal widely distributed in the environment, largely as a consequence of indiscriminate use in leaded gasoline, but also related to Pb smelting and mining activities, its presence in paints, tubing systems, batteries, prints, and other poorly controlled items, for example, toys, school supplies, folk medicines, vitreous clay, and so on. 1,2 The decision to ban the major Pb applications in the United States over 30 years ago (gasoline in 1973; paints in 1978) resulted in a substantial decrease in children’s mean blood Pb (PbB) levels, as evidenced in numerous reports. 3 –5 Yet Pb exposure still represents a significant public health concern for the pediatric population.
It has been reported that coexposure to neurotoxicants (e.g. metals and pesticides) or the experience of stressful situations, such as alcoholism, delinquency, poor medical attention, and family violence, may potentiate deleterious effects of Pb. 6 –9 Many Pb-exposed populations are also at risk due to concurrent hematological disorders 10 or to nutritional deficiencies (i.e. a diet low in nutrients and essential metals such as iron and calcium) that may result in a diminished capacity to respond to the toxic effects of Pb. 10 –13 These findings are also supported by experimental data in animals, 14 –17 suggesting the possibility of a close relationship between high Pb levels, poor nutritional status, and high stress levels. 8,18
The regulation and prohibition of Pb applications in Argentina are relatively recent (leaded gasoline was phased out in 1996), and the benefits gained from such controls have not yet been adequately evaluated. 19 The majority of the local reports focus on children living near industrial and mining areas, and therefore exposed to dangerously high Pb levels. 20 –23 Few studies 13,24 have explored the effects of the metal in child populations living in areas devoid of a nearby industrial source of Pb, leaving unanswered the question of the prevalent factors that contribute to the subclinical adverse effects associated with chronic low-level Pb exposure in the Argentinean pediatric population.
It is well known that young children are the most vulnerable segment of the population, in part because of the immaturity of their developing nervous system and mouth–hand activities, but also due to differential toxicokinetic parameters compared with adults. These include an increased respiratory rate, higher gastrointestinal absorption (over 50% of ingested Pb as against 10–15% in adults), a diminished proportion of metal storage in hard tissue, and lower excretion. 25 –28
Although Pb induces alterations in multiple organs, its effects on the central nervous system are crucial for human health, particularly for the pediatric population. Low-level Pb exposure is associated with diminished intelligence quotient, impaired neurological development, and behavioral problems such as attention deficit and hyperactivity. 29 –31 These effects are evident even at PbB concentrations below 10.0 mg/dl, the level of concern established until recently by the Centers for Disease Control and Prevention (US CDC). 32 However, at the recommendation of its Advisory Committee on Childhood Lead Poisoning Prevention, CDC sets up now a new reference value of 5 μg/dL which is based on the 97.5th percentile of blood lead levels in U.S. children aged 1-5 years, as measured by the agency's National Health and Nutrition Examination Survey (NHANES). 33
Pb also affects the gastrointestinal, renal, and hematopoietic systems, and the latter provides useful biomarkers for diagnostic purposes. 34 In this regard, Pb inhibits δ-aminolevulinic acid dehydratase (δ-ALAD), an enzyme involved in heme biosynthesis by catalyzing the condensation of two δ-aminolevulinic acid (δ-ALA) molecules to generate porphobilinogen. 35,36 This step results in an increase in δ-ALA blood levels and urinary excretion. δ-ALA suffers self-oxidation, promoting the generation of reactive oxygen species (ROS) such as superoxide, hydroxyl radical, and hydrogen peroxide (H2O2). 37,38 In turn, ROS-induced toxicity is counteracted by antioxidant compounds, including enzymes such as superoxide dismutase (SOD), which dismutes the superoxide ion generating H2O2, and catalase (CAT), an enzyme that, along with glutathione peroxidase (GPx), decomposes the H2O2. 37,39 It is noteworthy that several studies postulate that evaluating the enzyme δ-ALAD may be indicative of both Pb-induced hematological effects and Pb-induced oxidative stress. 40,41
With this background, the aim of this study was to establish current Pb exposure in the pediatric population of Cordoba city and its vicinity, by measuring traditional biochemical indicators (PbB levels and δ-ALAD enzymatic activity). In addition, to explore molecular biomarkers of Pb effects on redox status, the activity of two antioxidant enzymes, SOD and CAT, was evaluated. Finally, the aim was to identify whether differential vulnerability to Pb toxicity is manifested in child subpopulations, to try to unravel the interaction between high PbB levels and adverse sociodemographic status.
Materials and methods
Subjects
The protocol employed was approved by the Institutional Ethics Committee for Research in Child and Adult Health of the Hospital de Niños de la Santísima Trinidad, Cordoba, Argentina. Blood samples from 161 children (84 girls and 77 boys), aged between 1 month and 14 years were collected during a 6-month period, between September 2009 and February 2010, 13 years after Pb was banned in gasoline in Argentina. The children were randomly chosen among those who attended the hospital due to minor health problems such as headaches, colds, abdominal pain, and domestic accidents or simply to perform routine medical checks. Patients with a chronic pathology (such as renal failure, cancer, neurological, or metabolic diseases) or under medication for prolonged treatment were excluded.
Sample collection and treatment
After both the children and their parents were informed about the study, written parental consent along with a written assent from children aged 7 years and older was obtained before sample collection. An aliquot (3–4 ml) of whole blood drawn by venipuncture was placed in 1.5 ml microcentrifuge tubes using sodium heparin as an anticoagulant. It was separated in two aliquots; one was used for the hematocrit, hemoglobin (Hb), PbB, and δ-ALAD activity determinations described below. The remaining aliquot was spun at 825g for 10 min to separate the plasma from the blood cells. Plasma was stored at −20°C for SOD determination, while the erythrocytes were washed three times with isotonic saline and stored at −20°C for CAT activity measurement.
Survey for the identification of potential Pb sources
Immediately after blood sample collection, parents or guardians were given a questionnaire that evaluated the environmental, sociodemographic, and economic profile of the child and family members. This six-part survey was structured as follows: part 1: personal data (gender, age, education, and number of family members); part 2: occupation; part 3: medical record (previous diseases and disorders, use of prescribed medications, and ‘pica’ behavior); part 4: food and nutritional status (common foods in the diet and vitamin intake); part 5: home condition (location—urban vs. suburbs, time living in that house, type of building materials, wall deterioration, closeness to Pb sources, etc.); and part 6: child and family recreation and cleaning habits (daily time spent in the house, frequency and type of cleaning, playtime, and gardening).
Classification of neighborhoods by family income was based on the report ‘Guía Estadística de Córdoba’ (Cordoba Statistics Guide) published in 2008 by Cordoba Town Hall: http://www.cordoba.gov.ar/cordobaciudad/principal2/imagenes/Diseno/estadisticas.html.
Determination of PbB
To avoid contamination, all glass material was washed three times with a nonionic detergent, rinsed with distilled water, and treated for 24 h with 30% nitric acid, rinsed again with milli-Q purified water, and dried. PbB determination was performed using an atomic absorption spectrophotometer equipped with graphite furnace (Buck Scientific 200gf). The instrument was calibrated using aqueous standards of 0.5, 1.0, 5.0, 10.0, 20.0, and 50.0 μg/dl Pb as lead acetate. To determine PbB levels, 100 μl of whole blood was placed in microcentrifuge tubes, mixed with 100 μl of 0.1% Triton X-100 and 2% ammonium pyrrolidine dithiocarbamate, with the subsequent addition of 100 μl of methyl isobutyl ketone. All samples were centrifuged at 573g for 5 min and the supernatant measured at 283.2 nm according to Hessel. 42 Each sample was determined in triplicate with a detection limit of 0.2 µg/dl.
Biochemical assays
Blood Hb content was measured according to the Drabkin and Austin method 43 and the hematocrit level by the microcentrifugation method. 43 Enzymatic activity of δ-ALAD was calculated within 6 h of the sample collection by the standardized European method. 45 Briefly, 200 μl of whole blood was mixed with distilled water and phosphate buffer pH 6.4 containing δ-ALA (substrate) and incubated at 38°C for 1 h. Subsequently, 10% trichloroacetic was added and the tubes centrifuged at 825g for 10 min. The supernatant was collected and mixed with the Ehrlich reagent for subsequent spectrophotometric determination at 555 nm. Plasma SOD activity was determined by the method described by Beauchamp and Fridovich 46 with slight modifications. In brief, 200 μl of plasma was placed in tubes containing 75 µM nitro blue tetrazolium (NBT), 13 mM methionine, 100 nM EDTA and 2 µM riboflavin, and illuminated with white light for 90 min. The photoreduction of NBT to blue formazan was measured at 560 nm. One unit of SOD was considered as the amount of the enzyme that produced a 50% inhibition of photoreduction of NBT under these conditions. To determine erythrocyte CAT activity, the red cells were first hemolyzed with distilled water (1/20 dilution); the resulting hemolysate was further diluted 1/100 with phosphate buffer pH 7.0. Blood CAT activity was assayed by measuring the decrease in absorbance of the substrate (H2O2) at 240 nm (∊ 240 = 0.0394 per mmol/cm) according to Aebi. 46 The resultant activity was expressed as micromoles of H2O2 decomposed per minute per gram of Hb. All the spectrophotometric determinations were performed in a ultraviolet–visible spectrophotometer Varian Cary 50 scan.
Statistical analysis
Data were analyzed by a Shapiro-Wilk normality test that evidenced nonparametric distribution for PbB levels and a normal distribution for hematocrit, Hb, CAT, SOD, and δ-ALAD activity values. The study population was divided in groups by PbB levels and by age (≤1-year-old, 2–3, 4–5, 6–7, 8–9, 10–11, and 12–14 years old), gender, and socioeconomic and demographic characteristics. We used ln-transformed PbB values to normalize PbB data distribution. An unpaired t test was used to compare PbB levels, hematocrit, Hb, δ-ALAD, CAT, and SOD activities by gender and different socioeconomic and demographic status. A one-way analysis of variance (ANOVA) and a linear regression analysis were used to compare these parameters between age groups. For further analysis, the samples were split into four groups according to PbB levels (geometric mean values in parenthesis): PbB levels <5.0 µg/dl (1.99 ± 0.11), between 5.0 and 10.0 µg/dl (7.03 ± 0.25), between 10.1 and 20.0 µg/dl (14.11 ± 2.09), and a last group comprised children with PbB levels above 20.0 µg/dl (24.40 ± 2.12).
To reduce the influence of extreme values on the statistical analysis, we analyzed PbB levels in quartiles (lowest: 0.20–1.60 µg/dl; second: 1.61–2.86; third: 2.87–4.26; highest: >4.26), and a one-way ANOVA was used to compare the hematocrit, Hb, CAT, SOD, and δ-ALAD activity values in these groups. Finally, a linear regression analysis of variance was used to evaluate the relationship between PbB levels and δ-ALAD, CAT, or SOD activities.
Results
Biological indicators in the population under study
The geometric mean PbB levels obtained for the pediatric populations under study were 2.58 ± 0.30 µg/dl (range: 0.20–28.71 µg/dl). As mentioned in the statistics section, the PbB concentrations showed a nonparametric distribution, with most of the values shifted to the left end of the tail (Figure 1, insert), which led to a ln transformation to normalize the data for further comparisons. A more detailed distribution is plotted in Figure 1.

Percentage of children by PbB levels (n = 161) and frequency of distribution of PbB levels (insert).
Further analysis evaluated the relationship between enzymatic and hematological indicators in relation to the age of the population. A significant interaction was observed between PbB concentrations and age (p = 0.0314), but no age differences were found for δ-ALAD, CAT, and SOD activities (p = 0.8622, p = 0.2549, and p = 0.3380, respectively). On the other hand, significant differences were observed in the hematological parameters given that, as expected, the 1 month to 1-year-old group evidenced a decrease in hematocrit and Hb levels: (p < 0.001 for both indicators; Table 1). However, the results demonstrated a positive correlation between hematocrit, Hb, and SOD activity and age, and a negative correlation between PbB and CAT and age, but no correlation was evident between δ-ALAD and age (Table 2). In contrast, when all biochemical parameters were compared by gender, no differences were seen between boys and girls (Table 3).
PbB concentrations, δ-ALAD, CAT, and SOD activities, Hb and hematocrit levels expressed across the different age groups.

SE: standard error; hematocrit: %; Hb: hemoglobin (g/dl whole blood); δ-ALAD: δ-aminolevulinic acid dehydratase (U/L blood); CAT: catalase (x104 μmol H2O2 decomposed/min/g Hb); PbB: blood lead (μg/dl); SOD: superoxide dismutase (U/ml plasma). a,cDifferent letters in PbB values indicate a significant difference at p < 0.05. bStatistically significant (p < 0.001).
Correlation analysis between PbB concentration, enzymatic activities, Hb, and hematocrit levels and age.

δ-ALAD: δ-aminolevulinic acid dehydratase; SOD: superoxide dismutase; CAT: catalase; Hb: hemoglobin; PbB: blood lead.
aStatistically significant.
PbB concentrations, δ-ALAD, CAT, and SOD activities, and hematocrit and Hb levels across genders.

SE: standard error; δ-ALAD: δ-aminolevulinic acid dehydratase (U/L blood); SOD: superoxide dismutase (U/ml plasma); CAT: catalase (×104 μmol H2O2 decomposed/min/g Hb); Hb: hemoglobin (g/dl whole blood); PbB: blood lead (µg/dl), hematocrit: (%).
Biological indicators in relation to the socioeconomic and demographic profile of the children
Regarding the relationship between PbB levels and sociodemographic parameters (Table 4), the variables analyzed in the survey revealed only a significant difference between PbB levels in children living in urban versus suburban environments (p = 0.0455), with higher PbB levels in the latter. Results obtained from comparisons regarding socioeconomic status did not reveal a significant difference between PbB levels in children of low versus middle socioeconomic status, although borderline significance was obtained (p = 0.0579). Individual treatment of the data revealed that the six children with PbB levels >10.0 µg/dl reside in different neighborhoods of Cordoba, four of them in homes considered as within the low socioeconomic stratum (Table 5). Coincidentally, two of them have parents that perform plumbing-related activities, while another two children live in an area with high industrial activity. There is no identified Pb contamination source for the remaining two children.
PbB levels and socioeconomic and demographic characteristics.a

SE: standard error; PbB: blood lead.
aThe numbers in parentheses represent the percentage of the total children included in each sociodemographic classification. bStatistically significant (p < 0.05).
Detail of children with PbB levels over 10.0 µg/dl.
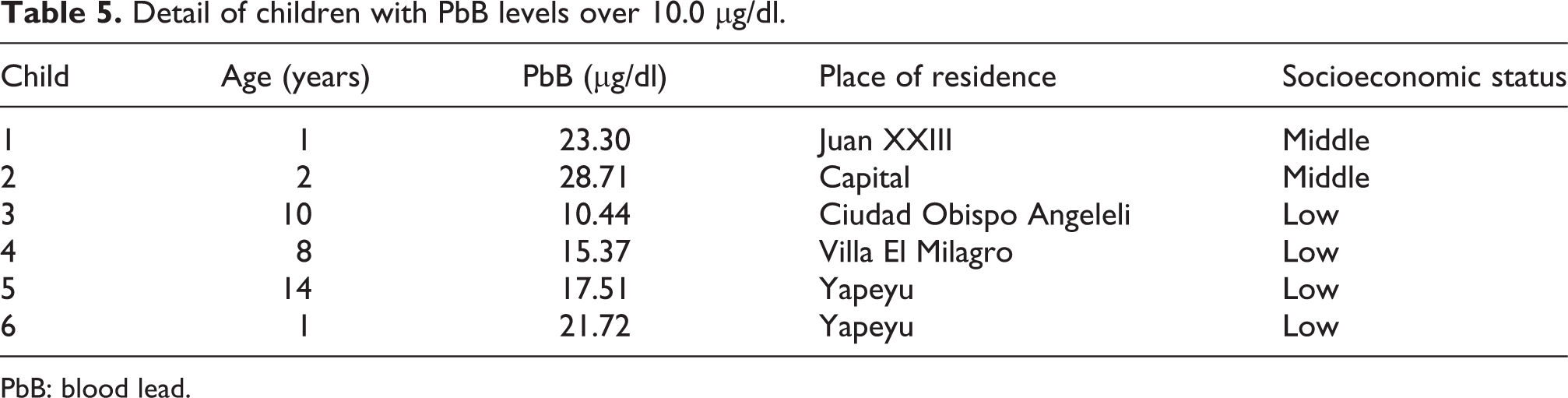
PbB: blood lead.
Comparative analysis of δ-ALAD, SOD, and CAT activities in relation to the socioeconomic and demographic profile (Table 6) evidenced a significant reduction in δ-ALAD activity only in low versus middle socioeconomic status children (p = 0.0034).
δ-ALAD, CAT and SOD activities, and socioeconomic and demografic caracteristics.a

SE: standard error; δ-ALAD: δ-aminolevulinic acid dehydratase (U/L blood); SOD: superoxide dismutase (U/ml plasma); CAT: catalase (×104 μmol H2O2 decomposed/min/g Hb).
aThe numbers in parentheses represent the percentage of the total children included in each sociodemographic classification.
bStatistically significant (p < 0.005).
PbB levels, hematological indices, and enzymatic activities
The statistical analysis of the hematocrit and Hb levels indicates that there were no significant differences in these parameters when analyzed across the different PbB range levels (p = 0.1331 and p = 0.2452, respectively; Table 7).
Hematocrit and Hb levels in relation to PbB levels.

SE: standard error; Hematocrit: %; Hb: hemoglobin (g/dl whole blood); PbB: blood lead.
The data represented in Figure 2 were plotted to show a correlation (if any) among PbB levels and all three enzymatic activities. The results demonstrated a negative correlation between δ-ALAD activity and PbB levels, while no correlation was evident between PbB levels and either CAT or SOD activity.

Correlation between δ-ALAD (a), CAT (b), and SOD (c) enzymatic activities and PbB concentrations. The linear equation, correlation coefficient, and p value are shown in the plot. Blood δ-ALAD activity showed a negative correlation with PbB levels (*p < 0.05) (a). Blood CAT (b) and SOD (c) activities failed to show a statistical correlation with PbB levels (*p ≥ 0.05). δ-ALAD: δ-aminolevulinic acid dehydratase; CAT: catalase; PbB: blood lead; SOD: superoxide dismutase.
Enzymatic δ-ALAD, CAT, and SOD activities were plotted against PbB levels and represented in Figure 3(a) to (c), respectively, showing no significant variations (p = 0.1500, p = 0.2913, and p = 0.5265, respectively).

Blood δ-ALAD (a), CAT (b), and SOD (c) activities in function of PbB levels. There were no changes in any of the enzymatic activities across the different PbB levels. δ-ALAD: δ-aminolevulinic acid dehydratase; CAT: catalase; PbB: blood lead; SOD: superoxide dismutase.
Discussion
To our knowledge, the present study is the first to evaluate PbB levels in a pediatric population in Cordoba, Argentina, since Pb was phased out in gasoline and banned in paints in 1996 and 2004, respectively. We report here a mean PbB level of 2.58 ± 0.30 µg/dl in children living in Cordoba and surrounding areas, a value significantly lower than the 7.70 ± 1.10 µg/dl reported by Hansen et al. (in samples taken between 1995 and 1996) in a similar Cordoba population. 24 Moreover, in their study, the percentage of children with PbB levels above 10.0 μg/dl was 26.7%, whereas our data showed a significant reduction to 3.72%, reflecting current low exposure to this metal. Indeed, these results seem to be indicative of the positive impact of Pb restrictions among the Argentinean pediatric population, a finding that is highly relevant for public health policies. This parallel reduction in PbB levels along with the restrictions in leaded gasoline has been profusely described worldwide, 3 –5,48,49 but in Argentina few reports are available. In this regard, Disalvo et al. 13 recently analyzed PbB levels in children from the general population of La Plata (Argentina), with mean PbB concentrations of 5.4 μg/dl, and with 10.9% of the total children presenting PbB values above the level considered safe by the US CDC. They also reported a clear association between Fe deficiencies and Pb exposure, concluding that nutritional and occupational variables represent a higher risk for Pb contamination. In addition, and although including different pediatric populations, our results can be contrasted with a study performed by De Oto et al. 21 in newborns from an industrial setting in the Buenos Aires (Argentina) suburbs. They reported that, although the median PbB value of 3.7 µg/dl was relatively low, as many as 37% of newborn PbB levels were between 5 and 10 µg/dl, and 2.25% were above 10 µg/dl, reflecting the influence of industrial activity in PbB levels in children living in that setting. Finally, Lopez et al. 50 observed a time-dependent decrease in PbB levels in the adult population living in the Buenos Aires metropolitan area, ascribing this to the restriction of leaded gasoline and Pb welding from canned food. Thus, on the basis of these reports, the present results are expected to have a public health impact, since this is the only study that compares current PbB levels in similar pediatric populations over a 10-year-period.
Although the US CDC defined 10.0 µg/dl as the PbB level of concern, 32 further recent studies have demonstrated that the metal affects the physical and mental development of young children at much lower levels 31,51 –56 encouraging a reduction in the PbB levels considered safe. 57,58 In our study, 13.04% of the pediatric population evidenced PbB levels between 5.0 and 10.0 µg/dl (Figure 1) in the absence of an identified source of exposure or a known risk factor. Furthermore, analysis of the questionnaire failed to reveal a generalized source of exposure for the entire population under study; still, according to PbB levels, we were able to identify higher vulnerability among the suburban than the urban subpopulation. Indeed, there is evidence of high Pb levels resulting from urbanization 4,59,60 in developing countries. 61,62 However, in our study mean PbB levels were higher in children in suburban areas than in those that live in urban areas (p = 0.0455; Table 4). This can be explained, in the first place, by taking into consideration the reduction in environmental contamination in large cities due to the phasing out of leaded gasoline, and second as a result of the high number of children that reside in suburban areas associated with high industrial activity. However, our data did not provide evidence of a statistically significant relationship of Pb levels according to industrial versus nonindustrial settings (p = 0.3245).
In addition to the occupational exposure of some family members, 63 a large percentage of children may be exposed to less-controlled sources of Pb, such as soil and dust contaminated with high Pb concentrations both inside and around the home, 64 –68 drinking water, containers and kitchen utensils, food and toys, among others. 69,70 Our data indicate that the source of exposure for two of the six children presenting high PbB levels was directly related to welding activities performed by their parents at their homes. Two other children with high PbB levels live in an industrial area, a finding that matches reports from the US Environmental Protection Agency 71,72 indicating the importance of industrial development (Pb smelting, batteries, and paint factories) in Pb environmental contamination. Analysis of the remaining two children with high PbB levels did not identify a Pb source, due either to an incomplete parents’ report, exposure to a less common source of this metal, or the coexistence of other neurotoxicants that may potentiate Pb toxicity such as mercury (Hg). It has been described that exposure to Hg and risk of toxicity during early human development are modulated by the vaccination schedule, diet, and maternal amalgams. 73 Pb interaction with other neurotoxicants should also be considered in future studies.
In contrast to several reports that indicate a higher risk of intoxication in US children living in deteriorated houses or those that have been recently remodeled, 28,74,75 our results showed no significant difference in PbB levels between children that reside in deteriorated or recently remodeled houses and those that do not (p = 0.5868 and p = 0.9483, respectively; Table 4). Thus, in spite of the fact that Pb was eliminated from paints in 2004 in Argentina, this restriction does not seem to have had the high impact in PbB levels that was evident after the ban in gasoline. Despite its major limitations, the present study, in agreement with Disalvo et al., 13 suggests that Pb-based paints may not be an important risk factor for Pb exposure nowadays, given that the exposure factors identified in this pediatric population were ascribed to Pb-related activities performed at home or in the surroundings (welding, industries, etc.).
Additionally, our studies failed to demonstrate a significant difference between PbB levels in children from low versus middle socioeconomic status (p = 0.0579; Table 4). However, this difference became evident when δ-ALAD enzymatic activity was correlated with the survey, revealing lower enzymatic activity in low-income children (p = 0.0034; Table 6). Studies published in Argentina and several other countries 13,57,76 –79 suggest that children of low socioeconomic level are those that present higher PbB levels, with deteriorated homes, inadequate nutrition, contaminated water, the absence of good cleaning habits and education, predisposing these children to a higher exposure and contamination with Pb.
Finally, regarding differences in PbB levels by age group (Table 1), we observed a significant age-related interaction, matching data reported by Disalvo et al., 13 which could be ascribed to the higher vulnerability of smaller children, probably resulting from their hand-mouth activities 25 as well as the immaturity of the organs involved in Pb toxicokinetics. 80 In addition, our results showed a correlation between PbB levels and age as reported by Dascanio and Valle 81 and Brody et al. 82 However, the survey-related and sample size limitations must also be considered when any of these factors (industrial setting, age, socioeconomic status, etc.) are analyzed. As regards the hematological parameters, Pb effects on the hematopoietic system comprise anemia, manifested as basophile puncture in red blood cells, and alterations in the mediators involved in heme biosynthesis, such as the reduction in δ-ALAD activity, elevation of δ-ALA, and protoporphyrin levels, among others. 7,35,83 The Hb and hematocrit values obtained in this study indicate that there were no alterations in these indices at any PbB values (p = 0.2452 and p = 0.1331, respectively; Table 7) in agreement with results obtained by Ahamed et al. 40 This is expected considering the low PbB levels obtained for this pediatric population, since it has been demonstrated that only Pb exposure that raises PbB levels to 25.0 µg/dl and above is capable of reducing heme formation sufficiently to be seen in these hematological indicators, 84 which indicates that they can be useful to evaluate poisoning only at moderate to high Pb exposure. 39 In contrast, Sakai and Morita 36 reported that inhibition of δ-ALAD activity was already present at very low PbB concentrations (around 5.0 µg/dl), and suggested that this inhibition could be an early biomarker of biological damage. However, when geometric mean PbB levels were analyzed, we, as did others, 4,40,41,85,86 observed a significant reduction in the activity of this enzyme at PbB levels higher than 20.0 µg/dl (p = 0.0031; data not shown), with a negative correlation between the enzyme and Pb levels, as shown in Figure 2(a). These results also agree with previous studies that reported that PbB levels higher than 25.0 µg/dl affected δ-ALAD activity. 87,88 Similar results were obtained in studies correlating an increase in plasmatic and urinary δ-ALA with PbB levels, 89 –91 suggesting that δ-ALAD activity is not a sensitive indicator of low Pb exposure. One important aspect of the δ-ALAD biomarker that arises from the present study is the reduction in δ-ALAD activity inhibition (61.59 ± 1.15 U/L) from the value obtained by Hansen et al. 24 of 38.80 ± 1.10 U/L, a finding that is closely related to the decrease in PbB levels reported here compared to those obtained in that study.
Regarding the mechanism of action of Pb, it has been proposed that it is able to induce oxidative stress, with considerable evidence indicating the relevance of this imbalance in its toxicity. Glutathione (GSH) and glutathione disulfide (GSSG), as well as the activity of antioxidant enzymes such as CAT, SOD, GPx, and glutathione reductase (GR), have been utilized as biomarkers of the oxidative damage induced by Pb. 37,39 Some authors demonstrated an increase in CAT activity at elevated PbB levels, and a positive correlation between these parameters in both children 4,40 and Pb-exposed workers. 41,85 Gurer et al. 92 have tried to characterize this increase as a compensation mechanism against the increment in H2O2 resulting from the oxidative stress generated by Pb. Our results did not show a statistically significant correlation between these two variables (Figure 2(b)) nor a modification in CAT activity at any PbB levels (p = 0.2913; Figure 3(b)). These findings agree with those of Oktem et al. 93 who failed to observe changes in CAT activity in relation to PbB levels in adolescents exposed to Pb at work. However, when mean SOD activity was compared to PbB levels, there was evidence of an increase in SOD activity selectively at PbB concentrations between 10.1 and 20.0 µg/dl (p = 0.0032; difference not evidenced when PbB levels were analyzed in quartiles; Figure 3(c)), indicating that the correlation between PbB levels and the activity of this enzyme is not linear (Figure 2(c)). Although several reports indicate a positive correlation between these two variables, 94,95 others show no clear association between them, 85,93,96,97 with some authors even reporting a decrease in the activity of this enzyme in plasma and red blood cells at high PbB levels. 98 –100 These differences could be explained considering that, although Pb induces an increase in ROS that leads to an increase in SOD activity and other antioxidant enzymes such as CAT and GPx, it is also able to inhibit these enzymes, increasing their activity at relatively low levels in blood, but reducing them at higher levels. 39 In addition, our study failed to reveal a correlation between δ-ALAD and CAT and SOD activities (data not shown), although some authors found evidence of a relationship between antioxidant enzymes and δ-ALAD activity. 4,40,41,101 Similarly, it has been suggested that there is a correlation between these and increased δ-ALA levels in blood and urine, 38,93 suggesting that δ-ALAD activity and δ-ALA levels can also be considered indicative of Pb-induced oxidative stress. On the other hand, we did find a positive correlation between SOD and a negative correlation between CAT and the age of the children from this study (Table 2), an effect that may be dependent of Pb exposure or interpreted in the context of developmental changes in the antioxidant defenses of the living organisms. 102 –104
In summary, our study reveals a notable reduction in PbB levels in the pediatric population of Cordoba and its vicinity, probably due to the phasing out of Pb in gasoline. Importantly, these results also show that a small but significant percentage of children are still exposed to toxic concentrations of this metal, with identifiable subpopulations at a greater risk. They also emphasize the importance of pursuing novel, mechanistically relevant biomarkers of toxicity. It is thus imperative to identify and reduce the primary sources of exposure for these children either at home or in its surroundings. It is hoped that these exposure indices obtained in otherwise ‘healthy’ children will be taken into consideration in the development of prevention politics and follow-up strategies. Such actions should be aimed at (1) identifying patients at risk with the use of both early biomarkers and a suitable survey and (2) performing follow-up monitoring of children with slightly higher PbB levels, which could account for behavioral and intellectual impairment that may arise in these children not ascribed to other causes.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
The authors wish to thank Maria Eugenia Marull and Marisol Ujaldón Trevisson for their help with the questionnaire analysis, Dr Diego Gaiero for his collaboration in PbB level determinations, and the professionals at the hospital laboratory, particularly Andres Suarez, for their cooperation with sample collection.
Conflict of Interest
The authors declared no conflicts of interest.
