Abstract
A low cytotoxic elastic niosomal formulation loaded with salmon calcitonin was developed. The elastic niosomes were prepared from Tween 61 mixed with cholesterol at various concentrations of the edge activators (sodium cholate (NaC) and sodium deoxycholate (NaDC); 0.25, 0.5, 2.5, 5 and 10% mole) or ethanol (10–30% v/v). The effects of the niosomal concentrations (5, 10 and 20 mM) and phosphate buffer at pH 7.0 (5, 10, 20 and 30 mM) on the physical characteristics of niosomes were investigated. The 5 mM elastic niosomes in 5 mM phosphate buffer containing calcitonin 0.22 mg/mL gave the highest elasticity (deformability index (DI)) at 6.79 ± 2.03 determined by the extrusion method. The blank elastic niosomes comprised 2.5% mole NaDC, 5% mole NaC or 20% v/v ethanol showed the highest elasticity. The 5% mole NaC elastic niosomes loaded with calcitonin gave the highest DI (21.59 ± 0.91) and percentages of calcitonin entrapment efficiency (60.11 ± 4.98). This study has demonstrated that this NaC elastic niosome did not only reduce the cytotoxicity of the loaded calcitonin but also gave superior cell viability to the ethanolic elastic niosome as well.
Introduction
Calcitonin, a polypeptide hormone with 32 amino acids synthesized in mammals, is a major regulatory factor in calcium and skeletal metabolism. Calcitonin belongs to amyloid family, which is toxic via oxidative mechanism. The fibrils of human or bovine calcitonin were toxic to cell culture. 1 Salmon calcitonin has been found to have as much neurotoxicity as calcitonin from other species. 2 Clinically, calcitonin can be administered via intramuscular, subcutaneous, intravenous injection (100 IU), intranasal inhalation (200 IU) and oral administration (2500 IU). The transdermal route has many advantages including the avoidance of hepatic first pass effects, delivering the drugs continuously, having fewer side effects and improving patient compliance. Effects of calcitonin on fibroblasts and collagen formation in rabbits have also been reported. 3 Long-term administration of calcitonin has been shown to enhance fibroblast growth and collagen synthesis. These fibroblasts exhibited hypertrophy cells, highly developed rough endoplasmic reticulum, several polyribosomes, large nuclei, hypertrophic Golgi complex and many dense granules and lysosomes. Many transdermal deliveries of calcitonin have been developed, for example, ViaDor-Calcitonin (ViaDerm-Calcitoin) from TransPharma Medical Ltd. 4 The major obstacles of transdermal drug delivery are the low percutaneous penetration and drug stability. Liposomes have been proposed as efficient drug carriers for topical delivery, since they have the potential to enhance drug penetration into the skin, improve therapeutic effectiveness and increase the stability of the drug. Law and Shih 5 have demonstrated that calcitonin entrapped in liposomes was stable after storing at various conditions for 1 month and had a sustained release profile by intravenous injection, while calcitonin solution did not show this effect. Calcitonin entrapped in liposomal polymer–protease inhibitor conjugate (chitosan–aprotinin) complexes demonstrated not only a significant decrease in calcium plasma level of about 15-fold compared with calcitonin solution but also the prolonged pharmacological effects of calcitonin after intragastric administration. 6 Recently, elastic nanovesicles have been developed to facilitate drug transport across the skin since they can pass through the skin that has the pore sizes smaller than their diameters. 7 Several substances have been added to prepare elastic nanovesicles, including ethanol, sodium cholate (NaC) and sodium deoxycholate (NaDC). The examples of elastic vesicles are transfersomes (vesicles containing phospholipid and edge activator), bilosomes (vesicles containing bile salt) and ethosomes (vesicles containing ethanol and polyols). However, several literatures have reported that ethanol is toxic to human dermal fibroblast cells. 8,9 The less toxic edge activators including NaC, NaDC, Span 80, Tween 80 and dipotassium glycyrrhizinate have been introduced to develop less cytotoxic elastic nanovesicles. Both NaC and NaDC are biodegradable, biocompatible and nonimmunogenic bile salts, which have been found to be effective to form a new type of elastic vesicle called bilosomes for entrapping many peptide drugs, such as interleukin 2, interferon α 10 and insulin. 11 In this present study, the edge activators including NaC and NaDC were used to prepare elastic vesicles or bilosomes loaded with calcitonin compared with elastic vesicles prepared from ethanol. The physical characteristics including vesicular sizes, zeta potential values, deformability index (DI), entrapment efficiency and the cytotoxicity to normal human fibroblasts were investigated in order to develop the low cytotoxic elastic niosomes loaded with salmon calcitonin.
Materials and methods
Materials
Tween 61 (polyoxyethylene sorbitan monostearate), NaDC, acrylamide and glycerol were purchased from Sigma Chemicals Company (Missouri, USA). Cholesterol was purchased from Serva Co. (New York, USA). NaC was obtained from Himedia (India). Ammonium molybdate and tris(hydroxymethyl) methylamine from Fisher Scientific UK Limited (UK), were used. Dulbecco’s modified Eagle’s medium (DMEM) and penicillin–streptomycin were purchased from Gibco BRL (New York, USA). Fetal bovine serum (FBS) was purchased from PAA Laboratories GmbH (Austria). Trichloroacetic acid and disodium hydrogen phosphate from Merck (Germany) and sulforhodamine B (SRB) monosodium salt, N,N′-methylenebisacrylamide and N,N,N′,N′-tetramethyl ethylenediamine from Fluka (USA), were used. Trypsin was purchased from Gibco Invitrogen Corp. (New York, USA). Sodium dodecyl sulfate and coomassie brilliant blue R-250 from Bio-Rad Laboratories (UK) and bromophenol blue dye and ammonium persulfate from Amresco Inc. (USA) were used. Sodium dihydrogen phosphate was purchased from BDH Chemicals Ltd (England). Salmon calcitonin (molecular weight = 3432 Da, isoelectric point (PI) = 9.47) was purchased from Kunming Jida Pharmaceutical Co. (China). Ethanol, methanol, chloroform and glacial acetic acid were analytical grade solvents. Ethanol was purchased from the Liquor Distillery Organization Excise Department (Thailand). Methanol, chloroform and acetic acid were obtained from ACI Labscan Limited (Thailand).
Effects of niosomal concentrations and various dispersants on physical characteristics of elastic niosomes loaded with calcitonin
Preparation of blank niosomes (the niosomes without the calcitonin) and niosomes loaded with calcitonin
The nonelastic and elastic niosomes (Table 1) were prepared by the thin film hydration method with sonication. 12 Briefly, cholesterol was first mixed with Tween 61 at 1:1 molar ratio of various total mixture concentrations (5, 10 and 20 mM). Then, the mixture was dissolved in chloroform/methanol (1:1) and evaporated under vacuum to get a thin film. For nonelastic niosomes, the film was hydrated with various dispersants (distilled water or 20 mM phosphate buffer, pH 7.0). For ethanol elastic niosomes, the dispersants with 20% v/v ethanol was used. The resulting dispersion was sonicated by a microtip probe sonicator for 10 min at 4 ± 2°C by placing the dispersion in an ice bath.
Descriptions of various niosomal systems for the investigation of physical characteristica
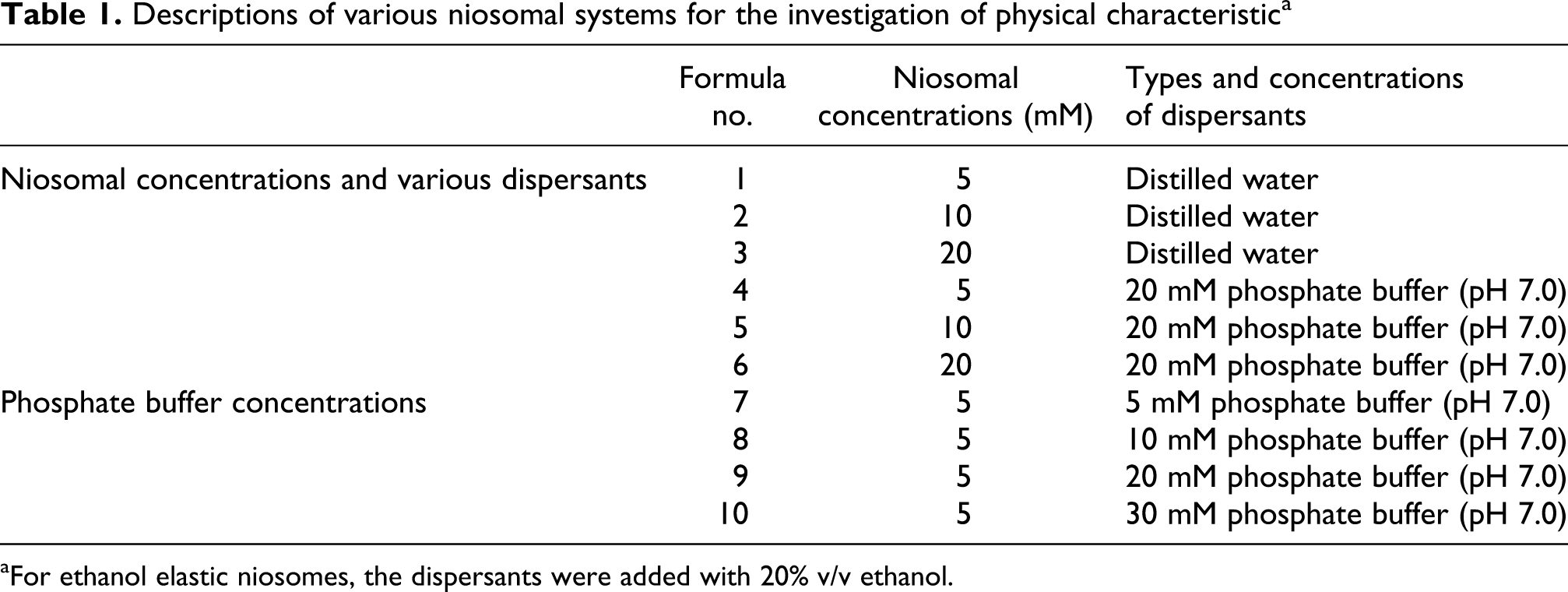
aFor ethanol elastic niosomes, the dispersants were added with 20% v/v ethanol.
Niosomes loaded with calcitonin were prepared with the same above method, except that calcitonin at 0.22 mg/mL (equivalent to the recommended dose of calcitonin nasal spray in postmenopausal osteoporosis treatment) in distilled water or 20 mM phosphate buffer pH 7.0 was used to hydrate the film instead of distilled water or buffer alone.
Physical characteristics of niosomes
The appearance, color and sedimentation of the niosomal dispersion were optically observed, while the size and zeta potential values of the niosomal dispersions were determined by the dynamic light scattering (Zetasizer Nano ZS British, Malvern Instruments, UK) with an angle of detection at 173°.
Measurement of DI
Elasticity of the vesicular membrane is an important and unique parameter for deformable vesicles. Elasticity of niosomes was determined by the extrusion method as described previously.
12,13
Briefly, the niosomal dispersion was extruded through a polycarbonate membrane filter with the pore size of 50 nm (Millipore, USA) and at the constant pressure (2.5 bar). After the 10-min extrusion, the vesicular sizes and the weight of the niosomal dispersion were measured. The elasticity of the vesicles was expressed in terms of DI, according to the following equation
14

Where, j is the weight of the niosomal dispersion, which is extruded in 10 min through a polycarbonate filter (50 nm pore size). rv is the size of the vesicle after extrusion, and rp is the pore size of the filter membrane.
Effects of dispersants on the maximum loading and entrapment efficiency of calcitonin in elastic niosomes
Calcitonin at various concentrations (0.1, 0.2, 0.5, 1.0, 1.5 and 2.0 mg/mL) was loaded in nonelastic and elastic (20% v/v ethanol) niosomes. The maximum loading of calcitonin in niosomes was determined using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and gel documentation.
15
Briefly, niosomes dispersed in electrophoresis loading buffer were loaded on the gel consisting of 20% w/v separating gel and 4% w/v stacking gel. Electrophoresis was performed at the constant voltage of 80 mV until the bromophenol dye front reached the bottom of the gel. The gel was stained with coomassie blue G-250 solution for 30 min and put in the destaining solution to visualize the calcitonin bands. Electrophoretic data were documented by a gel documentation system (Bio-Rad Laboratories, UK) and analyzed by Quantity 1-D analysis software. The maximum loading of calcitonin in niosomes was determined from the amount of the loaded calcitonin that the free calcitonin band was first seen in the gel. The area and intensity of the free calcitonin bands were determined and the calcitonin concentrations were calculated from the standard curve.
16
The entrapment efficiency was expressed as the percentages of the loaded calcitonin when compared with the total drug according to the following equation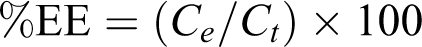
EE is the entrapment efficiency. C e is the concentration of calcitonin loaded in niosomes and C t is the total concentration of calcitonin initially loaded in the niosomal dispersion.
Effects of phosphate buffer concentrations on physical characteristics of elastic niosomes
The nonelastic and elastic niosomes were prepared with the same method as described in the Preparation of Blank Niosomes and Niosomes Loaded With Calcitonin section. Various concentrations (5, 10, 20 and 30 mM) of phosphate buffer solution (pH 7.0) were used to hydrate the film. The phosphate buffer solution was prepared from sodium dihydrogen phosphate and disodium hydrogen phosphate. The pH value was measured by a pH meter (Laboratory Benchtop 86502, AZ Instrument Corp., Taiwan). Physical characteristics and DI of niosomes were investigated by the same methods as described in the Physical Characteristics of Niosomes and Measurement of DI sections, respectively.
Comparison of physical characteristics and cytotoxicity of elastic niosomes
Preparation of elastic niosomes
Niosomes were prepared with the same method as described in the Preparation of Blank Niosomes and Niosomes Loaded With Calcitonin section. For elastic niosomes containing ethanol, the 5 mM phosphate buffer solution at pH 7.0 containing various amount of ethanol (10, 15, 20 and 25% v/v) was used to hydrate the film. For elastic niosomes containing NaC and NaDC, various amounts of NaC and NaDC at 0.25, 0.5, 2.5, 5 and 10% mole were mixed with Tween 61 and cholesterol before hydration and the film was dispersed by 5 mM phosphate buffer solution (pH 7.0). (The %mole means the mole ratio in the niosomal formulation. The 5% mole NaC means 5% of the total moles of the other niosomal compositions besides NaC (Tween 61/cholesterol/NaC = 1:1:0.1).) Physical characteristics and DI of niosomes were investigated by the same methods as described in the Physical Characteristics of Niosomes and Measurement of DI sections, respectively.
Morphology of elastic niosomes
The shape and morphological features of niosomes were investigated by staining the niosomal dispersions with 1% methylene blue on the slides under an optical microscope (Olympus CHS, Olympus optical Co., Ltd, Japan). For confirmation of the lamellar structures, a drop of niosomal dispersion was applied on a 300-mesh formvar copper grid on paraffin and allowed the sample to adhere for 10 min. The remaining dispersion was removed and a drop of 2% aqueous solution of ammonium molybdate was applied for 5 min. The remaining solution was then removed, air dried and examined by a transmission electron microscope (TEM; TEM 1200S JEOL, JEOL Ltd, Tokyo, Japan) operated at 80 kV. The morphology and lamellarity of the niosomes were observed.
Entrapment efficiency of calcitonin loaded in elastic niosomes
The entrapment efficiency of salmon calcitonin loaded in elastic niosomes was determined by the same method as mentioned in the Effects of Dispersants on the Maximum Loading and Entrapment Efficiency of Calcitonin in Elastic Niosomes section. Calcitonin loaded in each elastic niosomal group (i.e. ethanol, NaC and NaDC) from the Preparation of Elastic Niosomes section, which gave the highest DI, were used to determine the entrapment efficiency.
Cytotoxicity of elastic niosomes
Cell cultures
The human skin fibroblast was obtained from Faculty of Tropical Medicine, Mahidol University in Bangkok, Thailand. The cells were maintained as adherent cells in T75 culture flasks at 37°C in a humidified air incubator containing 5% CO2. DMEM were supplemented with 10% FBS, 100 U/mL of penicillin and 100 mg/mL of streptomycin and used as growth medium. For subculture, the cells were rinsed with phosphate buffer saline at pH 7.4 and finally detached with trypsin-EDTA solution (0.25%). The cells at the 25th passage were used.
Cytotoxicity assay
Cells were seeded in 96-well plates at an amount of 10,000 cells/well and allowed to attach overnight. Then the cells were exposed to various concentrations of the niosomal dispersions (0.05, 0.125, 0.25, 0.375 and 0.5 mM) and calcitonin solution (2.2, 5.5, 11, 16.5 and 22 mg/mL, which were equivalent to those loaded in the niosomal dispersions) for 24 h. After incubation, the adherent cells were fixed by adding 50% w/v cold trichloroacetic acid and further incubated for 1 h at 4°C. Then the cells were rinsed with distilled water, air dried and stained with 0.4% SRB in 1% glacial acetic acid for 30 min at room temperature (27 ± 2°C). The unbound SRB was removed by washing with 1% glacial acetic acid solution for four times. After air drying, 100 µL per well of 10 mM Tris base were added to dissolve the bound stain.
17
After mixing, the absorbance was measured at 540 nm with a microplate reader (Biorad, Milan, Italy). The untreated cells were used as a negative control. Cell viability (%) was calculated using the following equation
18,19

Statistical analysis
Data were expressed as mean ± SD. Statistical analysis was carried out using the analysis of variance using the software SPSS 13.0 for Windows and p < 0.05 was considered statistically significant.
Results and discussion
Effects of niosomal concentrations and various dispersants on physical characteristics of elastic niosomes
All niosomal dispersions were in translucent colloidal appearances with no sedimentation or layer separation. The vesicular sizes of calcitonin loaded niosomes were in the range of 138–292 nm. Niosomes loaded with calcitonin and dispersed in distilled water showed larger vesicular size and less negative zeta potential values than the blank (unloaded) niosomes. This may be due to the positive charge of calcitonin at pH 7.0 that can interact with the negative polarity of the niosomal membrane (from the hydroxyl group of cholesterol in the niosomal composition) resulting in less net negative of the system. However, the zeta potential values of niosomes loaded with calcitonin and dispersed in phosphate buffer (pH 7.0) remained unchanged (no significant difference at p < 0.05). The phosphate buffer may contribute to the counter ions that balance the charges of the system. The elasticity (DI values) of various niosomal formulations was summarized in Tables 2 and 3. The DI values of the elastic niosomal membranes were higher than the nonelastic niosomes of 1.04- to 4.19-fold. Nonelastic niosomes also showed some deformability owing to the influence of the surfactant (Tween 61) compositions. Tween 61 may accommodate at the high stress sites within the vesicles resulting in forming a highly curved area of the vesicles. 20 The high curvature of the membrane surface gave high elastic bending energy according to the Hooke’s Law, 21 thereby a more deformability of the vesicular membrane was obtained.
Effects of various niosomal concentrations on vesicular deformability indexa

aFormula nos. 1–3, please see Table 1.
Effects of dispersant types on vesicular deformability indexa
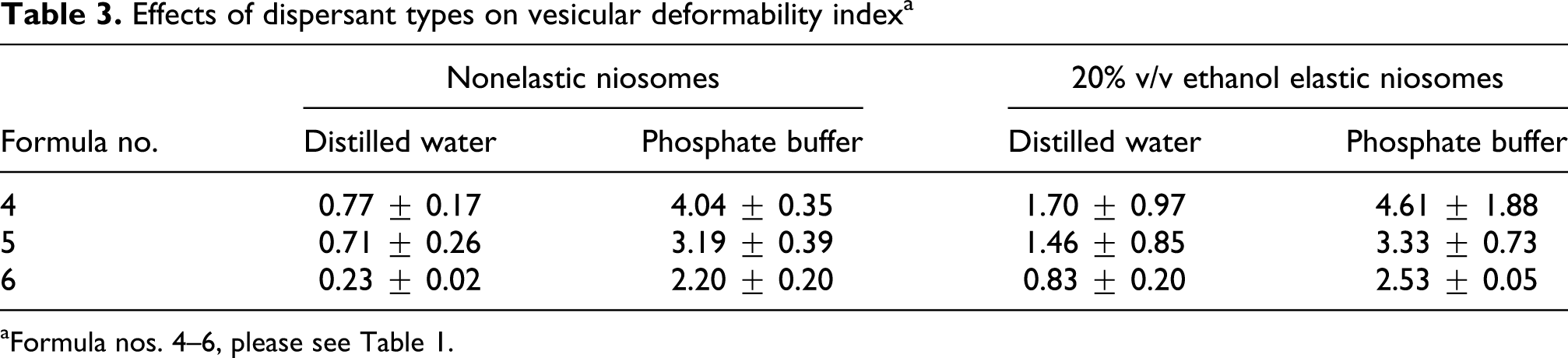
aFormula nos. 4–6, please see Table 1.
For the effects of various concentrations of niosomes (Tween 61 mixed with cholesterol) on vesicle elasticity in distilled water and phosphate buffer, the decrease in concentrations (from 20 and 10 mM to 5 mM) demonstrated the increase in vesicular elasticity. At the lowest concentration of the niosomes (5 mM), the packing properties of the surfactant (Tween 61) depend on the balance between the optimal cross-sectional surface area of the head groups (a 0), the volume of the hydrocarbon chains (v) and the maximum hydrocarbon chain length (l c). According to the geometrical rule, the value of V/a 0 l c at more than 0.5 gives the possibility to form bilayer vesicles. 22 So, the lowest concentration at 5 mM of the niosomes was used. The presence of the bilayer vesicle at this concentration was confirmed by the TEM images. Moreover, the formulation of bilayer vesicles at this low concentration of the total lipid was also reported. 23 Vesicles formed from low concentration of Tween 61 mixed with cholesterol have a decreased packing density of the surfactant molecules. This will lead to a decrease in the aggregation numbers and an increase in flexible spacer length. 24 The formation of smaller vesicles with thinner membrane assembly is obtained. 25 Therefore, vesicles formed from low concentration of Tween 61 mixed with cholesterol can certainly have larger degree of motion and give the more flexible vesicles.
Effects of dispersants on the maximum loading and entrapment efficiency of calcitonin in elastic niosomes
The maximum loading and entrapment efficiencies of calcitonin in niosomes by SDS-PAGE were shown in Figure 1. All niosomal formulations that dispersed in phosphate buffer (at pH 7.0) gave the maximum loading of calcitonin at 0.5 mg/mL, whereas those dispersed in distilled water was at 0.2 mg/mL. Entrapment efficiency of calcitonin loaded in elastic (containing ethanol) and nonelastic niosomes was summarized in Table 4. The entrapment efficiencies of calcitonin in niosomes decreased as the concentration of calcitonin increased. When the amounts of the vesicles were fixed, the osmotic pressure difference between the outer and inner vesicular membranes depended only on the calcitonin concentrations. The high calcitonin concentration might cause some damage to the formed vesicles and make it easier for the drug to escape from the inner phase. 26 The higher entrapment efficiency of calcitonin was observed with 20 mM phosphate buffer (pH 7.0) than with distilled water. The ionic strength in the buffer system may increase the ionic interaction between calcitonin and the vesicular membranes. 27 Calcitonin in the buffer system can interact with the vesicular membranes more than in the distilled water system. The higher entrapment efficiency of calcitonin in elastic than nonelastic niosomal formulation was also observed owing to the aqueous solubility enhancement of calcitonin by ethanol that may facilitate the entrapment of calcitonin in the aqueous phase of the elastic niosomes.

The maximum loading and entrapment efficiencies of calcitonin in niosomes by sodium dodecyl sulfate polyacrylamide gel electrophoresis. (a) Elastic niosomes loaded with various calcitonin concentrations (0.1–2.0 mg/mL) dispersed in distilled water, (b) nonelastic niosomes loaded with various calcitonin concentrations (0.1–2.0 mg/mL) dispersed in distilled water, (c) elastic niosomes loaded with various calcitonin concentrations (0.1–2.0 mg/mL) dispersed in 20 mM phosphate buffer (pH 7.0), (d) nonelastic niosomes loaded with various calcitonin concentrations (0.1–2.0 mg/mL) dispersed in 20 mM phosphate buffer (pH 7.0); The arrow indicates the maximum loading of calcitonin loaded in niosomes in which the small amount of the free calcitonin band was first seen in the gel.
Entrapment efficiency of calcitonin loaded in ethanol elastic and nonelastic niosomes

Effects of phosphate buffer concentrations on physical characteristics of elastic niosomes
The vesicular sizes did not depend on the phosphate buffer concentrations. When the phosphate buffer concentrations increased (5, 10, 20 and 30 mM), the negative zeta potential values were decreased (data not shown). Buffer at high concentration can result in a strong compression of the electric diffusion layer. The elasticity of the elastic niosomes loaded with calcitonin dispersed in 20 mM phosphate buffer (pH 7.0) was higher than those prepared in distilled water of 2.71-, 2.28- and 3.05-fold for the niosomal concentration at 5, 10 and 20 mM, respectively (Table 5). The ionic strength of the buffer system may affect the vesicle elasticity, as the phosphate buffer (pH 7.0) concentrations decrease from 30, 20, 10 mM to 5 mM, the increase in vesicle elasticity was observed. Buffer at higher concentration may have more influence on the charge interactions between the vesicular membranes than at lower concentration. Claessen et al. have indicated that at low ionic strength, the Debye length is large and the electrostatic repulsion between the membranes becomes effective. 28 This will lead to a decrease in the packing density of the surfactant molecules in the vesicles and the molecular assemblies with a large curvature according to the Hooke’s Law as mentioned previously. Thus, niosomes at 5 mM dispersed in 5 mM phosphate buffer was selected for further study because of its high entrapment efficiency and vesicular elasticity.
Effects of various concentrations of phosphate buffer on vesicular deformability indexa

aFormula nos. 7–10, please see Table 1.
Comparison of physical characteristics and cytotoxicity of elastic niosomes containing ethanol and elastic niosomes containing the edge activators (NaC and NaDC)
Physical characteristics of elastic niosomes
Table 6 showed the vesicular sizes, zeta potential values and DI of various blank nonelastic and elastic niosomal formulations. The vesicular sizes of all ethanol, NaC and NaDC elastic niosomes were in the range of 108.30 nm to 126.33 µm and zeta potential values at −17.88 to −45.10 mV. Ethanol elastic niosomes showed larger vesicle sizes, but lower zeta potential values than the NaC and NaDC niosomes. Higher ethanol concentrations gave larger vesicular sizes. This was in concordance with the previous investigation. 29 The average size was decreased at low ethanol concentration (0–3.3%), whereas an increase in vesicular sizes was observed at high ethanol concentrations (3.3–30%). The decreased niosomal sizes may be due to the effect of the edge activator at high concentration on the reduction of the interfacial tension between the vesicular membrane leading to the decrease in the bilayer thickness. 30 However, when the concentrations of NaC and NaDC increased, not much influence on the zeta potential values was seen. Most NaC and NaDC elastic niosomes gave more negative zeta potential values (−39.92 to −45.10 mV) than the nonelastic niosomes (−38.62 mV) and ethanol elastic niosomes (−17.88 to −27.40 mV). This may be due to the anionic property of NaC and NaDC. The slight differences of the zeta potential values between NaC and NaDC niosomes might be from the different functional groups in their structures (Figure 2). NaC has one hydroxyl group more than NaDC, which may affect the zeta potential values. 31 Niosomes containing ethanol gave the zeta potential values less than those without ethanol. The zeta potential values decreased when the ethanol contents were increased. This may imply the effect of ethanol on the reduction of the electrostatic repulsive energy of the vesicular surface due. 32 In general, the system that shows zeta potential values of more than ±30 mV is normally considered stable. The ethanol elastic niosomes gave the zeta potential values of less negative than −30 mV. So, this system was probably not stable.
Physical characteristics and DI of various blank nonelastic and elastic niosomal formulations

DI: deformability index; NaC: sodium cholate; NaDC: sodium deoxycholate.

Chemical structures of sodium cholate (NaC) and sodium deoxycholate (NaDC).
The vesicular sizes and zeta potential values of niosomes loaded with calcitonin were demonstrated in Table 7. The size of niosomes increased when loaded with calcitonin. The vesicular sizes of niosomes loaded with calcitonin were 150.98, 143.01 and 138.53 nm for nonelastic, NaC and NaDC elastic niosomes, respectively. Ethanol elastic niosomes loaded with calcitonin gave smaller size (194.69 nm) than the blank ethanol elastic niosomes but larger than the NaC and NaDC elastic niosomes loaded with calcitonin. Both NaC and NaDC might be suitable to prepare elastic niosomes loaded with calcitonin because they gave the smaller niosomes than ethanol.
Physical characteristics and DI of the selected nonelastic and elastic niosomes loaded with calcitonin when compared with their corresponding blank niosomes
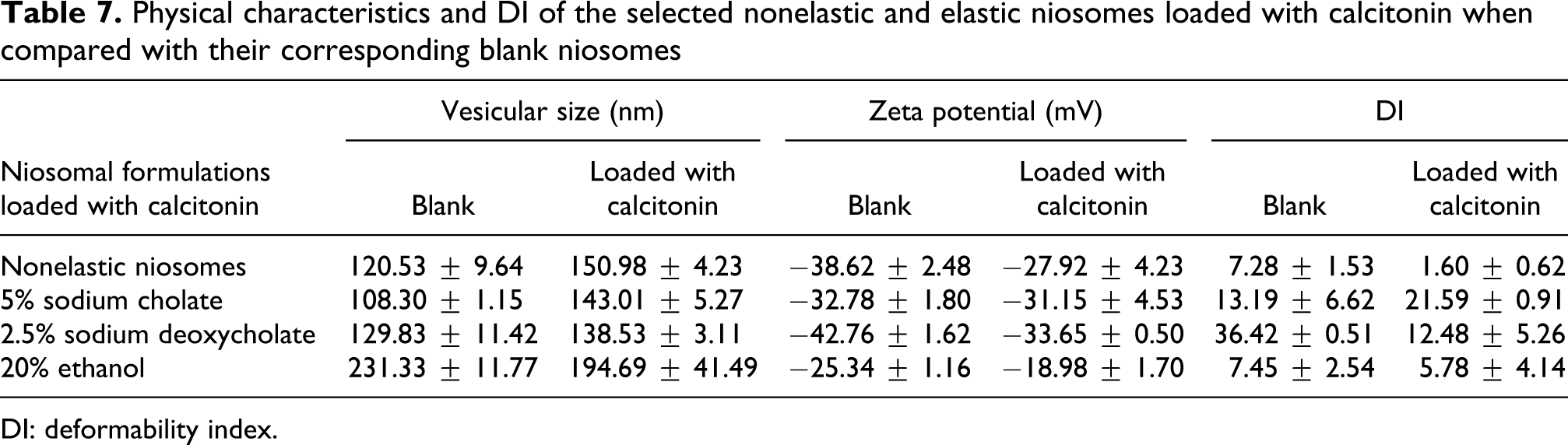
DI: deformability index.
The morphology of the niosomes was in the spherical shape when observed under optical microscope. From negative-staining electron micrographs (Figure 3), both nonelastic and elastic niosomes were in unilamellar structure with the vesicular size of less than 300 nm. All elastic niosomes appeared to have higher deformability structure than the nonelastic niosomes. The TEM of some elastic niosomal vesicles that had the elongated and self-adaptable shape may be from the conformation change of the niosomal membrane affected by the neighbor niosomal vesicles. 33 However, the nonelastic niosomes exhibited spherical shape. Perhaps, ethanol in elastic niosomes provided elasticity to the vesicular membrane by reducing the interfacial tension. Thurmond et al. have demonstrated that cholate molecules have a very strong tendency to increase the curvature of the niosomal membranes. 34 Thus, these two cholates (NaC and NaDC) may have the elasticity effect on the spontaneous curvature and bending rigidity of the niosomal membranes.

Negative-staining TEM images of niosomal formulations (×20,000) comprised Tween 61/cholesterol (1:1) dispersed in 5 mM phosphate buffer pH 7.0; (a) to (d) were blank nonelastic, 20% v/v ethanol elastic, 5% mole NaC elastic and 2.5% mole NaDC elastic niosomes, respectively, while (e) to (h) were nonelastic, 20% v/v ethanol elastic, 5% mole NaC elastic and 2.5% mole NaDC elastic niosomes loaded with calcitonin (0.22 mg/mL), respectively. TEM: transmission electron microscopic; NaC: sodium cholate; NaDC: sodium deoxycholate.
DI of elastic niosomes
The elasticity of NaC and NaDC niosomal membranes did not depend on the concentrations of the edge activators. However, the elasticity of these bilosomes was more than the ethanol elastic niosomes. Besides the interfacial tension reduction effects, the head groups of these two bile salts also intercalated between the surfactant molecules, producing the less packing density with more flexibility of the surfactant molecules. Thus, NaC and NaDC provided both interfacial and head group region mechanisms, whereas ethanol gave only the elasticity to the vesicular membrane by reducing the interfacial tension. 13 In fact, Maghraby et al. have also found that surfactants such as NaC and NaDC may interact with the lipid vesicles and change the conformation by fitting their lipophilic parts between the hydrocarbon chains, compensating the bulkiness of the head groups. 35
The blank elastic niosomes comprised 2.5% NaDC (36.42 ± 0.51), 5% NaC (13.19 ± 6.62) and 20% ethanol (7.45 ± 2.54) which gave the highest elasticity (DI) in each elastic niosomal group of 5-, 1.8- and 1.02-fold higher than their corresponding nonelastic niosomes (7.28 ± 1.53), were selected to loaded with calcitonin. The NaC elastic niosome loaded with calcitonin gave the elasticity of 1.64-fold higher than its blank niosome. This can be explained that the hydrophilic nature of the loaded calcitonin may enhance the elasticity of the vesicle. 36 On the contrary, the vesicle elasticity of the blank NaDC, ethanol elastic and nonelastic niosomes was more than their corresponding niosomes loaded with calcitonin of 2.92-, 1.29- and 4.55-fold, respectively. The positive charge of calcitonin at pH 7.0 may interact with the negative charge of the niosomes (from the cholesterol compositions) resulting in the decrease in the electrostatic repulsion between the vesicular membranes. This also led to the increase in packing density of the surfactant molecules in the vesicles and the molecular assemblies with a small curvature according to the Hooke’s Law. In DI determination, the large vesicle sizes and the rigid property of the nonelastic niosomes loaded with calcitonin (the vesicle sizes ranging from 120.53 ± 9.64 nm to 150.98 ± 4.23 nm) can affect the DI value during the extrusion process through the polycarbonate membrane filter. According to the DI equation (Measurement of DI section), the pore size and the weight of the extruded niosomal dispersion were fixed. The DI values were thus only affected by the vesicular sizes after extrusion (rv). Since the nonelastic niosome loaded with calcitonin, which was in large sizes with rigid structures, only its small vesicle sizes could be extruded through the membrane resulting the low rv and DI values.
Entrapment efficiency (%EE) of elastic niosomes
The nonelastic, ethanol (20% v/v) elastic, NaC (5% mole) elastic and NaDC (2.5% mole) elastic niosomes gave the entrapment efficiencies of calcitonin (0.22 mg/mL) at 50.97 ± 3.91%, 51.95 ± 9.47%, 60.11 ± 4.98% and 56.51 ± 7.47%, respectively. The NaC and NaDC elastic niosomes indicated higher entrapment efficiency of calcitonin than ethanol elastic niosomes. The more negative charge from the hydroxy group of the two bile salts can interact with calcitonin that has the positive charge (PI = 9.47) in phosphate buffer (pH 7.0) better than ethanol. Hence, calcitonin can be both adsorbed on the niosomal membrane by the electrostatic interaction and loaded in the aqueous layers in the vesicles from its hydrophilic property. Both NaC and NaDC elastic niosomes showed no significant difference of calcitonin entrapment efficiency despite their different structures. This may be due to their same hydrophilic-lipophilic balance (HLB) values of 16.7, which have the same affinity for calcitonin. 37
Cytotoxicity of elastic niosomes
The percentages of human skin fibroblast viability treated with blank and loaded nonelastic and elastic (ethanol, NaC and NaDC) niosomes with calcitonin were shown in Figure 4. The cells incubated with the buffer only gave more than 80% viability, even the concentration of the buffer was more than 0.5 mM. However, when the cells were incubated with calcitonin in buffer solution, a significant decrease in cell viability of only 20% was observed (p < 0.05). The positive charge (PI = 9.47) of calcitonin in phosphate buffer (pH 7.0) may be toxic to cells. 38 At 0.05 mM, blank nonelastic and elastic niosomes showed no cytotoxicity compared with buffer (no significant difference p < 0.05). However, when the concentrations of the blank nonelastic and elastic niosomes were increased from 0.05 to 0.5 mM, cell viability was decreased. This may be affected by increased permeability, lysis and solubilization of the lipid–protein–detergent association from the exposure of the cells to the surfactants (Tween 61, NaC and NaDC). 39 Cell viability when treated with NaDC niosomes was higher than that treated with NaC niosomes, but no significant difference (p < 0.05) was observed. Since high charge density is also related to cytotoxicity, 40 the cell viability of NaDC elastic niosomes that had high charge density based on the zeta potential values was expected to be lower than that of the NaC elastic niosomes. However, no significant cell viability between NaDC and NaC elastic niosomes was observed. This may be due to the more amount of NaC (5% mole) in the NaC elastic vesicles than NaDC (2.5% mole) in the NaDC elastic vesicles. At 0.125 mM of niosomes, the blank NaC and NaDC niosomes showed significant higher cell viability than the blank ethanol elastic niosomes. For the blank nonelastic niosomes, the 20% v/v ethanol, 5% mole NaC and 2.5% mole NaDC niosomes gave cell viability of 58.96 ± 0.91%, 48.71 ± 7.26%, 67.35 ± 5.43% and 71.45 ± 7.50%, respectively. The lowest cell viability was found in ethanol elastic niosomes, since ethanol can damage the cells by inducing oxidative stress in mitochondria DNA and leads to an impaired function of mitochondria and inhibits the synthesis of protein encoded by mitochondrial DNA in cells. 41
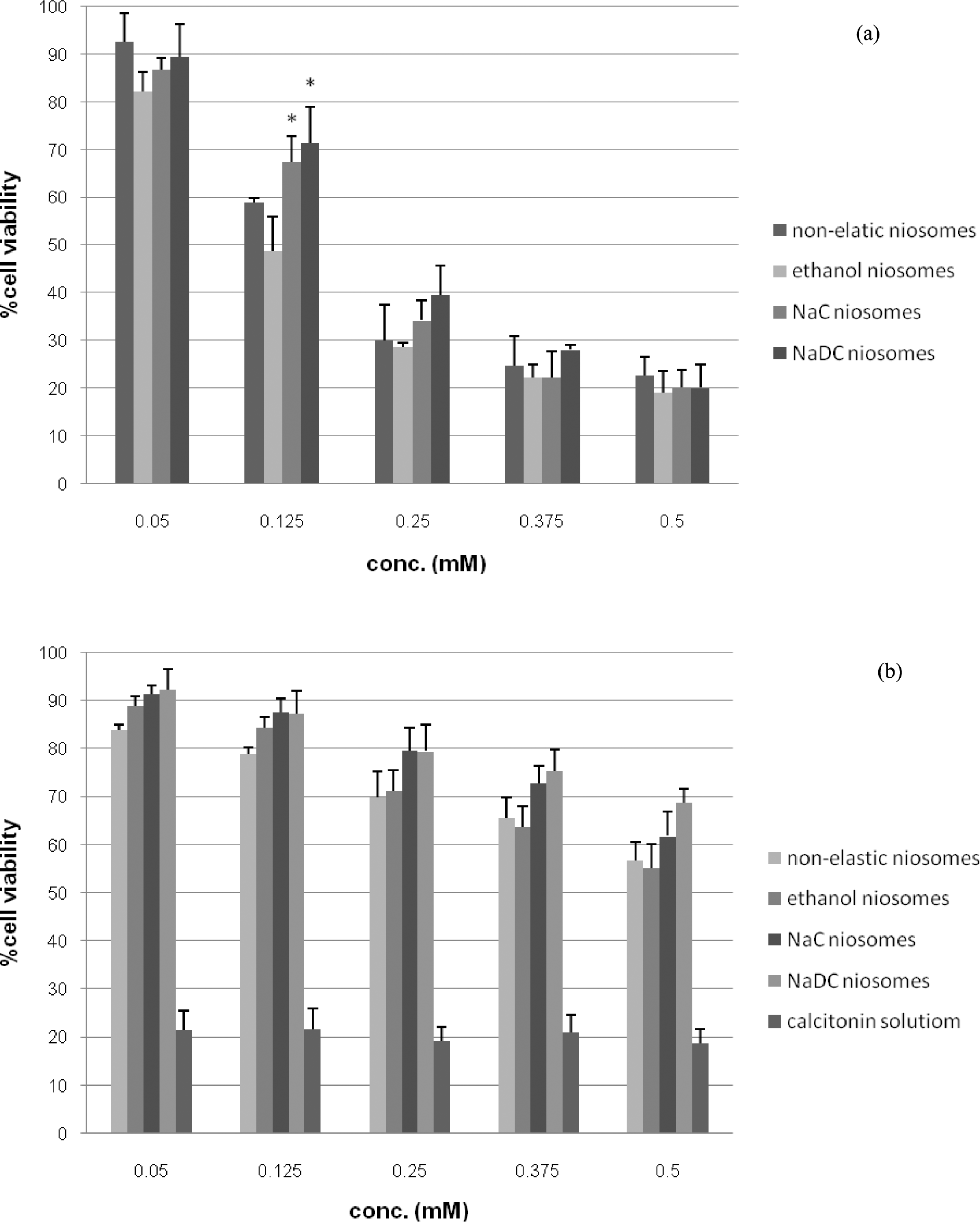
The percentages of human skin fibroblast viability by SRB assay of the blank niosomes (a) and calcitonin loaded in niosomes (b) (each value represents mean ± SD, n = 3). *Significant difference (p < 0.05) compared with the blank ethanol niosomes. SRB: sulforhodamine B.
In addition, nonelastic and elastic niosomes loaded with calcitonin gave higher cell viability than their corresponding blank nonelastic and elastic niosomes and the calcitonin solution. At 0.5 mM, niosomes loaded with calcitonin gave more than 55% of cell viability. When calcitonin at 0.22 mg/mL was loaded in nonelastic, ethanol elastic, NaC elastic and NaDC elastic niosomes, cell viability was increased to 3.02-, 2.93-, 3.29- and 3.65-fold, respectively, when compared with their corresponding blank niosomes. The neutralization of the positive charge of calcitonin at pH 7.0 by the negative charge of niosomes may result in charge density reduction, thereby decreasing the cytotoxicity. Moreover, when loaded with calcitonin, the negative zeta potential values and the cytotoxicity of the nonelastic and elastic niosomes decreased more than their blank niosomes. Thus, zeta potential values may relate to cell viability. The higher the zeta potential values of the niosomes, the stronger the interaction of the niosomal membrane with calcitonin can be expected. Furthermore, the decrease in cytotoxicity was also correlated with the binding process of cellular protein and the surfactant. The protein may act as a detoxifying agent on surfactant cytotoxicity. 42 In fact, the toxicity of niosomes is due to not just one, but many characteristics of the niosomal systems, such as surface charges, zeta potential values and protein binding process. The results from this study have suggested that the cytotoxicity of calcitonin can be reduced when loaded in elastic niosomes.
Conclusion
This study has indicated that the concentrations of niosomes (Tween 61 mixed with cholesterol), types of dispersants and the phosphate buffer concentrations (pH 7.0) can affect the physical characteristics (vesicular sizes and zeta potential values), vesicular elasticity and entrapment efficiency of calcitonin. Among various concentrations of different edge activators containing in elastic niosomes, the NaC (5% mole) elastic niosomes showed the highest entrapment efficiency of calcitonin. Calcitonin loaded in NaDC (2.5% mole) and NaC (5% mole) elastic niosomes gave higher cell viability in human skin fibroblast than the unloaded calcitonin. In summary, the 5% mole NaC elastic niosome appeared to be the most appropriate niosomal system to load calcitonin because of its high elasticity and low cytotoxicity on human skin fibroblast.
Footnotes
Funding
This work was supported by Natural Products Research and Development Center (NPRDC); Science Technology Research Institute (STRI); Faculty of Pharmacy, Chiang Mai University, Thailand; Biotechnology Department, Iranian Research Organization for Science and Technology (IROST), Tehran, Iran; the Thailand Research Fund (TRF) under the Royal Golden Jubilee (RGJ) PhD program; and Nanoscience and Nanotechnology Research Center Project, Faculty of Science, Chiang Mai University, Thailand.
