Abstract
Study objective:
Naltrexone is a competitive opioid receptor antagonist acting at the µ- and k-opioid receptors that blocks the euphoric effects of exogenous administered opioids. When used in opioid-dependent patients, naltrexone can cause acute and severe withdrawal symptoms.
Methods:
This was a cross-sectional study conducted from December 2007 to March 2008 and consisted of patients who had used naltrexone accidentally or deliberately and were referred to Loghman-Hakim Poison Hospital, Tehran, Iran. All symptoms and signs were assessed and the relationship between the dose of naltrexone, opioid dependence, and outcome was evaluated.
Results:
In 132 patients referred to our hospital, the most frequently reported symptoms and signs occurring in more than 10% of patients were agitation (96.2%), altered level of consciousness (38.6%), nausea (28%), vomiting (27.3%), abdominal pain (24.2%), diarrhea (16.7%), bone and muscle pain (15.9%), tachycardia (12.9%), and dilated pupils (11.4%). Being the most prominent symptom, the agitation was the most difficult aspect of withdrawal to manage. Except for agitation, no relationship was found between the presence of these symptoms and the dose of naltrexone used. Outcome of the patients (classified as complete recovery, partial recovery, death, and no follow-up) was related to the substance of addiction (p < 0.05) but not to the naltrexone dose.
Conclusion:
Emergency physicians should be aware of the potential for severe agitation from naltrexone-precipitated hyperacute withdrawal and its appropriate management. Opioid-dependent patients who wish to continue withdrawal and abstinence must be encouraged to visit trained physicians and be warned about misuse of naltrexone.
Introduction
Background
Drug abuse and dependence are the increasing problems among young people, and the adverse effects of many drugs abuse are well recognized. Our previous study that was carried out in the largest clinical toxicology center of Iran showed that most deaths in poisoned patients (42%) were caused by opioids, which may indicate a relatively larger population of opioid users in our community. 1 A similar study, also in Iran, showed that opioids are the single most common cause of overdose in all age groups. 2
Naltrexone is a long-acting competitive opioid antagonist, acting at the µ- and k-opioid receptors, which blocks the euphoric effects of exogenous-administered opioids that has been used to treat opioid dependence. This comparatively new medication used in drug rehabilitation programs to maintain abstinence from heroin and methadone and prevent relapse in former opioid-dependent patients. 3 –7 Before being given naltrexone, it is recommended that the patients should be opioid-free for a period of 7–10 days and undergo a supervised naloxone challenge test, before being accepted into a controlled detoxification program. Also, inquiry should be made about recent administration of methadone or any other opioids before administration of naltrexone. Absorption of naltrexone following oral administration is rapid, with peak plasma concentrations within 3 h. The oral availability has been estimated to be between 20% and 60%. Plasma concentration falls gradually in the first 24 h, followed by a subsequent very slow decline. 4 Accidental or intentional ingestion of naltrexone in opioid-dependent patients will result in an acute blockade of opioid receptors and precipitate a rapid onset of severe opioid withdrawal reaction within 5 min, which may last up to 48 h. Theoretically, when the µ-opioid receptors are sufficiently occupied by naltrexone, no additional antagonist can stimulate them, while it has an affinity at least 20 times that of morphine. 8
According to Iranian Pharmacopoeias, naltrexone is recommended after finishing abstinence therapy to prevent relapse. Naltrexone should be started at least 5 days after stopping opioid use; if used earlier, it can precipitate withdrawal signs and symptoms. 9
The opioid withdrawal syndrome due to abstinence is rarely life threatening or associated with significant aberrations of mental state. 10,11 Clinical manifestation and management of precipitated withdrawal are shown in Figure 1. Naltrexone when used in opioid-dependent patients can cause acute, severe withdrawal symptoms by a much more frequent and severe agitation than described in withdrawal caused by abstinence. 3 ,5,6,10 –13
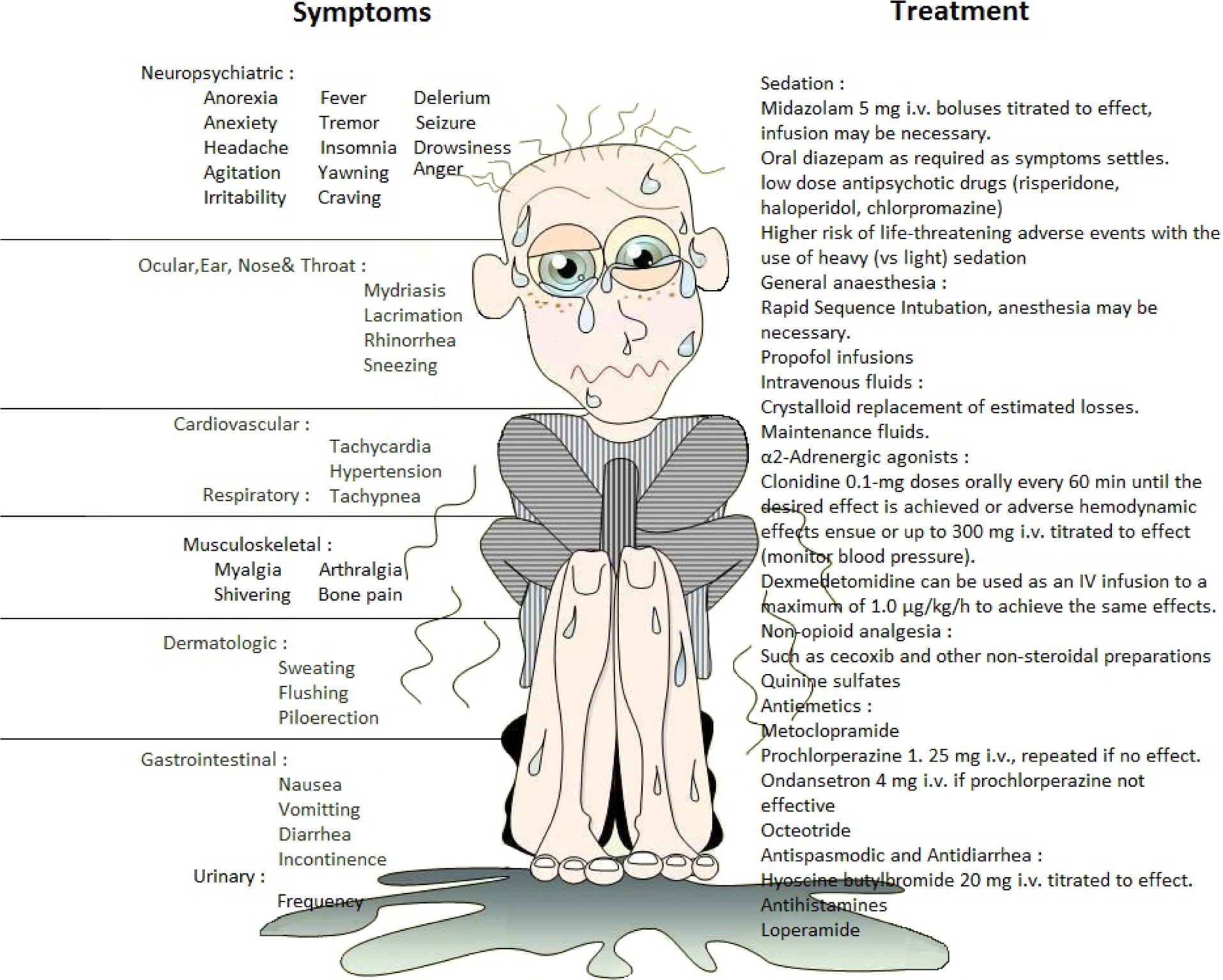
Clinical manifestation and brief management of acute opiate withdrawal syndrome.
Objectives
This study aimed to evaluate the clinical findings of naltrexone interaction in opioid abusers. We assessed symptoms and signs of opioid withdrawal in a large group of patients and tested for the presence of a relationship between dose of naltrexone, opioid dependence, and outcome.
Materials and methods
Study design
This was a single-center cross-sectional study and focused on the signs and symptoms in a large group of hospitalized patients who had recently used naltrexone alone or in combination with other drugs, deliberately or accidentally. The study period was from December 2007 to March 2008 and for a period of 121 consecutive days. To achieve a 95% confidence level, ± 5% precision and agitation prevalence of 91% (as the most common variable which was found in the first 50 cases), Cochran's sample size calculation for categorical data was 125 patients. Similar study in the same population had approximate rate of agitation. 9 Finally, 132 patients were enrolled in this study.
Setting
The study was carried out at Loghman-Hakim Poison Hospital in Tehran, Iran. The Loghman-Hakim toxicology unit serves a population in excess of 12.5 million and normally has 30,000 cases presenting to the emergency ward due to poisoning each year, of which 14,000 are hospitalized. This is the only tertiary hospital for the poisoned patients in the capital city and is the largest in the country. To the best of our knowledge, our inpatient complex seems to be the largest clinical toxicology department in the world. 1,14
Selection of participants
The criteria for the diagnosis of naltrexone consumption and the circumstances were defined based on the history when arriving at the emergency department (ED) and/or withdrawal syndrome and was confirmed through interviewing the patients and their friends or relatives in psychiatric in-person interviews. Those who denied naltrexone consumption were excluded.
Data collection and processing
In all the patients, epidemiological variables, symptoms, and signs were assessed, and the presence of a relationship between dose of naltrexone and type of substance used (classified as opium, heroin, none, and unknown) was evaluated. Heroin with relatively high purity is the main constituent of the illicit drugs obtained under the street name “crack” in Iran. 15
Outcome of the patients, classified as complete recovery, partial recovery (where the patients needed outpatient medical care due to naltrexone physical complications), death, and no follow-up, and biochemical changes (sodium, potassium, and creatinine concentrations) were assessed in the studied patients. Residents conduct the original history and physical examinations for clinical purposes, and then the data is subsequently extracted from the information of medical records using a questionnaire designed by toxicology experts. This questionnaire was specifically designed for the purpose of performing research on this problem. Accuracy of data is reassessed in psychiatric interview. Altered level of consciousness was defined as any measure of arousal other than normal (Table 1). 16 Agitation was defined as a state of restlessness and increased arousal, which is experienced by the patient as inability to relax and was seen by resident physicians as restless activities. Treatment was given according to the available conservative protocols including clonidine and benzodiazepines but without the use of opioids. This work conducted in accordance with the Declaration of Helsinki (1975) revised Hong Kong (1989).
Altered level of consciousness

Source: reproduced with permission from Dr Mojgan Karbakhsh, 2007. 16
Primary data analysis
To start the analysis process, data were coded and checked for errors. Descriptive statistics were arranged according to sex, age, and marital status distribution.
Naltrexone capsules are available in two generic forms in Iran; 25 and 50 mg with different colors. Naltrexone dose was defined in six categories: below 25 mg, 25–50 mg, 51–100 mg, 101–150 mg, more than151 mg, and unknown, based on colors and numbers they took. The relationship between dose and the presence of each complaint was assessed using χ2 test using SPSS software v 13.
Similar method was applied to find out whether the outcome was related to the dose of naltrexone. Meanwhile, one-way analysis of variance (ANOVA) was performed to assess whether there is any significant difference between the mean of sodium, potassium, blood urea nitrogen, and creatinine concentration in different naltrexone dose groups (defined above). All these analyses were done at the 0.05 level of significance.
Results
Overall, in 132 studied patients, 122 (92.4%) were male. The patients’ ages ranged from 15–72 years, with a median of 38 years (interquartile range 30–46.5). Regarding marital status, 72 (54.5%) patients were married, 15 (11.4%) were single, and 45 (34.1%) had unknown marital status. None of the patients were reported as divorced. The demographic data, variety of the opioid derivatives, their route of use by the patients, reason for use of naltrexone, and final outcome are presented in Table 2.
Demographic characteristics of 132 hospitalized patients with naltrexone consumption

aCessation of opium abuse.
bUse of opium on regular naltrexone intake.
Naltrexone is often given to the victim by a friend or relative in a misguided attempt to withdraw the victim from opioids and not to precipitate an acute life-threatening withdrawal syndrome (73.4%). Clinical features of acute naltrexone-accelerated withdrawal syndrome are presented in Table 3.
Clinical features of acute naltrexone-accelerated withdrawal syndrome in 132 hospitalized patients

LOC: level of consciousness.
Agitation, which is often very rapid in onset (sometimes as little as 5 min after ingestion of naltrexone), is the most frequent symptom (96.2%) and also the most difficult to manage. One case of seizure was encountered, and another (0.8%) of our patients died of a myocardial infarction during the period of withdrawal, in which stress in combination with preexisting factors might result in death. Unexpected sudden death did not let us perform echocardiography but electrocardiography showed diffused ST-segment changes.
No relationship was found between the presence of these symptoms and signs and the dose of naltrexone used (dose groups), except for agitation in which the relationship was significant by χ2 test (p < 0.05). In addition, except for agitation and yawning (p < 0.05), there was no significant relationship between the type of substance used and the presence of the other symptoms and signs using χ2 test. In an analysis carried out by χ2 test, outcome of the patients was related to the substance of addiction used; opium and heroin had more physical complications than the other opioids (p < 0.05). Outcome was not related to the naltrexone dose (p = ns).
Sodium, potassium, blood urea nitrogen, and creatinine concentrations were assessed in the studied patients. None of these were related to the dose of naltrexone used, but potassium concentration was related to the type of substance used by the patients, using ANOVA test (p < 0.05). Compared with others, heroin users showed a higher potassium concentration (Duncan’s and Tukey’s tests).
Of all our patients, 103 (78%) had consumed naltrexone only and most of them (74.4%) had used 25–50 mg. The substances used concomitantly by the patients are shown in Table 4.
Concomitant substances used in the studied patients

BZD: benzodiazepines; TCA: tricyclic antidepressants; AH: antihistamine.
Discussion
In this study, 132 naltrexone consumers were evaluated during a 4-month study period. The short time period needed to recruit this many patients is a reflection of a significant problem of illicit drug abuse and dependence. The present study showed that adult males were exposed much more frequently than females, most probably due to a much higher rate of opioid dependence among males.
In most studies of naltrexone-induced withdrawal presenting to ED, naltrexone was used accidentally; similarly, in this study, most of the patients used naltrexone accidentally nand were not aware of the interactions and only a few patients used it deliberately. 3 –5 The only route of naltrexone consumption in our patients was oral, because it is the most available form of the drug in pharmacies in Iran. In one other study, it was therapeutic and by implantation. 6
Regardless of the intent of consumption of naltrexone, all patients (97%) with a recent history of opioid addiction were referred to the ED in severe opioid withdrawal. As shown in Table 3, agitation, decreased level of consciousness, nausea, and vomiting were the main clinical findings. These features are a significant risk of aspiration pneumonia, a major complication that may explain the high morbidity (32% partial recovery) of patients, which is shown in Table 2.
Our results showed that the most frequent clinical features on presentation were agitation, nausea, and vomiting, while in another study vomiting, abdominal pain, and drowsiness were the most significant findings. 6
The opioid withdrawal syndrome is rarely life-threatening or associated with significant aberrations of mental state. 10 ,11 In our study, we encountered one seizure and a case of death due to myocardial infarction in a 52-year-old male patient; in addition, the vast majority of our patients had significant agitation.
Most of the patients used naltrexone alone and most used it (mistakenly) to reduce their withdrawal syndrome, because their friends offered it as a drug that is applied for being abstinent. Only nine patients used it deliberately in a suicidal attempt and in combination with other drugs. Two patients had used naltrexone mistakenly for naproxen and mefenamic acid to reduce toothache. Also, naltrexone was inadvertently prescribed in clinic to one opioid-dependent patient.
Limitations
This was a cross-sectional single-center study in which resident physicians were the primary providers of care, and caution should be used in attempting to generalize the results to facilities involving attending physicians or to nonacademic settings. As with any interviewer-administered survey, interviewer bias may have been present. The potential for interviewer bias was minimized using a standardized script for interviewers to read.
As the present survey was a single-center study, the most fundamental problem with the quality of the data was the lack of explicit definitions for documentation on arrival time. The incomplete documentation of some diagnoses and apparent lack of uniform methods for different resident physicians who were recording the patients’ data made it sometimes impossible to define the cases.
Also, we were not being able to detect the real value of the drug and opioids in body fluids. So the process of within-interview cross validation was not crucial to confirm the clinical manifestations are only due to interaction between naltrexone and opioids. The impurity of illegal substances may differ substantially and most patients are not aware of it.
Conclusion
The nature, severity, and duration of naltrexone-induced acute opioid withdrawal vary greatly between the individuals, and the clinical course of events is unpredictable. With the increasing trend for treating more opioid-dependent patients with naltrexone and the fact that opioid-dependent patients may consider naltrexone as a misguided means to break the cycle of drug dependence, the potential exists for increasing numbers of similar presentations. Therefore, physicians involved in the emergency care of these patients must be aware of the dramatic clinical course of the ingestion of naltrexone in opioid dependence and be prepared to manage the complications.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgments
The authors thank Professor Ian M. Whyte for his great comments and Davood Hassanian-Moghaddam for schematic presentation of withdrawal syndrome in Figure 1 by revising it through ![]() .
.
Declaration of Conflict of Interest
The authors declared no conflicts of interest.
