Abstract
Objective:
We investigated both the effects of the endothelin type B (ETB) receptor antagonist, BQ-788, on amitriptyline-induced cardiotoxicity and the role of ETB receptors on amitriptyline-induced cardiovascular depression.
Methods:
Male Wistar rats were anaesthetized with urethane/chloralose. Mean arterial pressure (MAP), heart rate (HR) and QRS duration were recorded. Toxicity was induced by amitriptyline infusion (0.94 mg/kg per min) until the 50% inhibition of MAP. In the first protocol, 5% dextrose or BQ-788 bolus was administered to control or experimental group animals, respectively. In the second protocol, after incubation with BQ-788 or 5% dextrose, amitriptyline was infused.
Results:
Amitriptyline caused a significant decrease in MAP, prolonged QRS duration and decreased HR for both the groups. BQ-788 administration improved MAP (5, 10 and 15 min), shortened the prolonged QRS (5 and 10 minutes) and increased HR (5, 10 and 15 min) compared with dextrose group. While all the amitriptyline-infused rats survived in BQ-788 group, all the amitriptyline-infused rats died within 20 min in dextrose group. In the second protocol, BQ-788 incubation did not cause any statistically significant change in amitriptyline-induced cardiovascular depression.
Conclusion:
BQ-788 may have beneficial effects in amitriptyline-induced cardiovascular changes via a physiologic antagonism. ETB receptor antagonists may be the promising antidotes for the cardiovascular toxicity with hypotension and bradycardia.
Introduction
Tricyclic antidepressant (TCA)-induced cardiotoxicity contributes to mortality and morbidity of patients in overdose. Hypotension and QRS prolongation, which are the consequences of alpha-1 adrenergic receptor blockade, noradrenaline reuptake inhibition and fast sodium channel blockade in the heart, are the major cardiovascular findings of TCA overdose. 1,2
Treatment of cardiovascular toxicity in TCA overdose mainly consists of the sodium bicarbonate, norepinephrine, phenylephrine or vasopressor doses of dopamine with supportive treatment. 3 Although some investigational antidotes such as NG-nitro-L-arginine methyl ester (L-NAME), adenosine receptor antagonists, theophylline or glucagon were found to be effective in reversing amitriptyline-induced cardiovascular toxicity in experimental animal models, there is not any effective antidote for amitriptyline poisoning.
Endothelin (ET) is a peptide produced by endothelial cells and a powerful vasoconstrictor agent. It has a role in some cardiovascular events such as hypertension, myocardial infarction, heart failure and ischemia–reperfusion injury. In different in vivo studies, it was demonstrated that ET caused an increase in cardiac output and systemic vascular resistance. 4 ET exerts its cardiovascular effects by binding to two distinct types of receptors, entitled ET type A and type B receptors (ETA and ETB, respectively), which are distributed to cardiac cells and vascular system. 5 –7 While vasoconstriction and an increase in blood pressure occur via ETA receptor stimulation, ETB receptors induce vasodilatation by releasing nitric oxide (NO) from the endothelial cells. The other important role of ETB receptors is the clearance of ET-1 from the circulation. As a result, ETB receptors clear the ET from plasma and reduce the ET concentration available to the ETA receptors. 7
In our study, we aimed to investigate the effects of BQ-788, an ETB receptor antagonist, on amitriptyline-induced decrease in mean arterial pressure (MAP) and heart rate (HR), QRS prolongation and the role of ETB receptors in a rat amitriptyline cardiotoxicity model.
Methods
The experimental protocol was approved by the Dokuz Eylul University, School of Medicine Animal Use Committee with Guide for the Care and Use of Laboratory Animals (NIH: Publication no.85-23, revised 1996). Adult male Wistar rats (n = 30), weighing 250–280 g, were fasted overnight with free access to water before the experiments.
Rats were anaesthetized with urethane/chloralose (500/50 mg/kg intraperitoneally, respectively). The trachea was cannulated using a 8-Gauge nasogastric cannula for spontaneous breathing. The right common carotid artery was cannulated with polyethylene tubing (50 mm OD (in.) 0.97 mm (0.038) ID (in.) 0.58 mm (0.023)) containing heparinized saline (100 U/mL) for blood pressure measurements. The left external jugular vein and left femoral vein were cannulated with polyethylene tubing for amitriptyline and dextrose or BQ-788 administrations, respectively. MAP, HR and electrocardiogram were recorded via a pressure transducer (BPT 300, Commat Ltd, Ankara, Turkey) on a data acquisition system (BIOPAC, MP35B-CE, 206B1564; USA). An infusion pump (Braun, Perfusor Compact S, Germany) was used for continuous administration of amitriptyline. The body temperature of rats was kept at 37°C with a desk lamp during the experiment. After the completion of the cannulation procedure, animals were allowed to stabilize for 15 min.
Experimental protocol
In a preliminary study, a 10 nmol/kg bolus dose of BQ-788 was administered to the rats via femoral vein to test the effects on the baseline values of MAP, HR and QRS duration during the whole experiment (n = 3, 10 nmol/kg). BQ-788 did not cause any significant change in baseline levels of MAP, HR and QRS duration within 30 min (Table 1).
The effects of BQ-788 bolus (10 nmol/kg) on MAP, HRs and QRS duration.
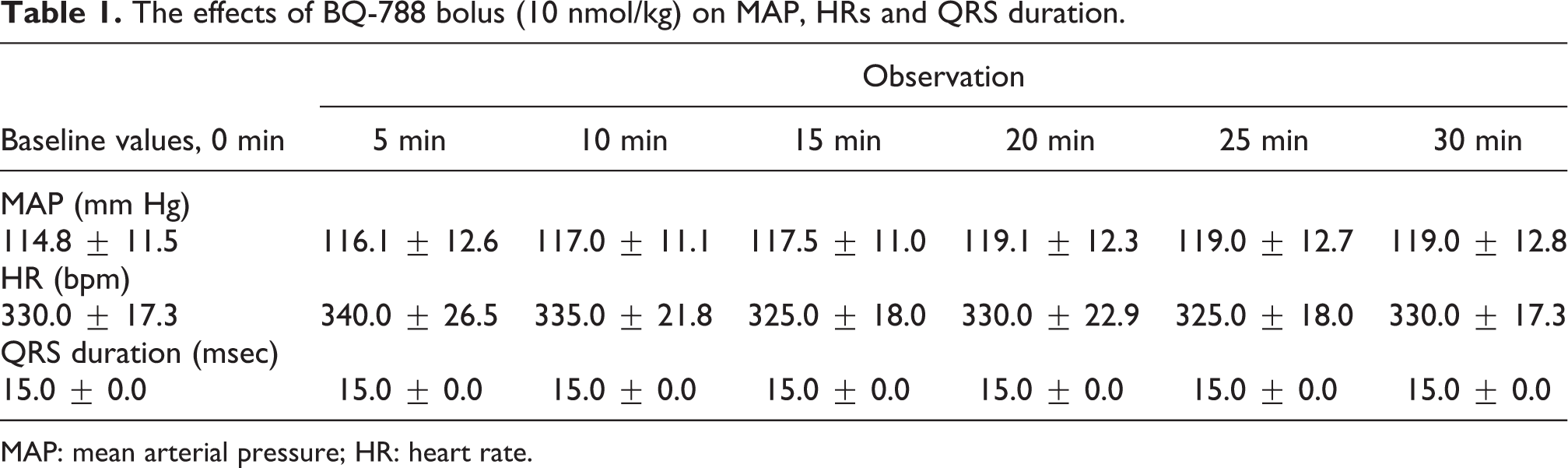
MAP: mean arterial pressure; HR: heart rate.
There were two study protocols. While the effects of an ETB receptor antagonist, BQ-788, on amitriptyline-induced MAP and HR decrease and QRS prolongation was investigated in protocol 1, the role of ETB receptors in amitriptyline-induced cardiovascular depression was investigated in protocol 2.
Protocol 1
At the end of the stabilization period, toxicity was induced by amitriptyline infusion of 0.94 mg/kg per min. After the 50% inhibition of MAP, amitriptyline infusion was instantly stopped and 5% dextrose (n = 8) or a 10 nmol/kg bolus dose of BQ-788 (n = 6) was given to the control or the experimental group of rats, respectively, to evaluate the effects of BQ-788 on amitriptyline-induced MAP, HR and QRS duration.
Protocol 2
At the end of the stabilization period, 5% dextrose or 10 nmol/kg BQ-788 bolus were injected via left femoral vein. Amitriptyline of 0.94 mg/kg per min infusion was started 15 min after dextrose or BQ-788 administration. Rats were monitored for the changes in the MAP, HR and QRS duration in control (n = 7) and experimental (n = 6) groups.
Drugs
Urethane and α-chloralose were prepared as 300 and 40 mg/mL stock solutions in distilled water, respectively (Sigma Chemical Company, St Louis, Missouri, USA). Amitriptyline (Sigma-Aldrich Company, Steinheim, Germany) was dissolved in distilled water at a concentration of 30 mg/mL. BQ-788 (Sigma Chemical Company, St Louis, Missouri, USA) was dissolved in (1.2 mg/mL) dimethyl sulfoxide (DMSO). The concentration of DMSO was 10% in the BQ-788 group. In our previous studies, 10% concentration of DMSO did not cause any effect on vascular system. 8 –10 Therefore, we did not include a control group for DMSO in our study.
Statistical analysis
Parametric tests were used for the statistical analyses with percentage changes in MAP, HR and QRS duration. Data within groups were evaluated by Student’s t test for paired data. To analyze the differences between the groups, Student’s t test for unpaired data was performed. All data were presented as mean ± SEM (GraphPad Instat™, 1990–1994, GraphPad Software V2.05a 9342, USA). A p < 0.05 was considered to be statistically significant.
Results
Protocol 1
There was no significant difference between the baseline measurements of the control and experimental groups in MAP, HR and QRS duration (Table 2). Amitriptyline infusion (0.94 mg/kg per min) was stopped at the time of nearly 50% inhibition of MAP compared with baseline. Amitriptyline infusion caused a significant decrease in MAP (48.7 ± 1.1% and 50.3 ± 1.8%) and HR (73.3 ± 4.5% and 69.5 ± 8.8%) and a prolongation in QRS duration (183.4 ± 6.3% and 166.7 ± 8.6%; for control and BQ-788 groups, respectively) within 15 min. There was no significant difference between the experimental and the control groups in MAP at the end of the amitriptyline infusion (p > 0.05). Although all the amitriptyline-infused rats survived in experimental group, all the amitriptyline-infused rats died within 20 min in the control group. Statistical analysis was restricted to an interval of 0–15 min because of the missing values due to death in the control group.
The effects of dextrose (n = 8) or BQ-788 (n = 6) bolus after amitriptyline infusions on MAP, HR and QRS duration (protocol 1).

MAP: mean arterial pressure; HR: heart rate.
a p < 0.001 versus baseline.
b p < 0.001 versus end of amitriptyline infusion.
c p < 0.05 versus end of amitriptyline infusion.
d p < 0.05 versus baseline.
e p < 0.01 versus baseline.
f p < 0.01 versus end of amitriptyline infusion.
In the control group (n = 8), 5% dextrose bolus, after amitriptyline infusion, decreased MAP at 5 and 10 min (22.6 ± 4.2%, p < 0.001; 23.5 ± 5.4%, p < 0.05, respectively), decreased HR at 5 min (31.4 ± 6.7%, p < 0.01) and prolonged QRS duration at 5 min (279.2 ± 29.5%, p < 0.01) of the observation period compared with the end of the amitriptyline infusion.
In the experimental group (n = 6), BQ-788 bolus, after amitriptyline infusion, did not change MAP and HR and improved QRS duration at 15, 20, 25 and 30 min (116.7 ± 11.4.%, p < 0.05; 103.3 ± 3.3%, p < 0.01; 111.1 ± 11.1.%, p < 0.05; 100.0 ± 0.0%, p < 0.001, respectively) of the observation period compared with the end of the amitriptyline infusion.
When compared with the control group, a BQ-788 bolus improved MAP at 5, 10 and 15 min (61.2 ± 5.4%, 22.6 ± 4.1% at 5 min; 70.5 ± 9.1%, 23.6 ± 5.3% at 10 min; 69.6 ± 10.2%, 19.3 ± 8.5% at 15 min; p < 0.001, p < 0.01 and p < 0.05, respectively), increased HR at 5, 10 and 15 min (72.3 ± 9.9%, 31.4 ± 6.7% at 5 min, 74.6 ± 7.5%, 35.8 ± 12.8% at 10 min and 83.9 ± 3.8%, 40.4 ± 9.6% at 15 min, p < 0.05, respectively) and shortened the prolonged QRS at 5 and 10 min (150 ± 11.4%, 254 ± 25.2% at 5 min; 144.4 ± 7.0%, 225.0 ± 25% at 10 min; p < 0.01, respectively, Figure 1).

The effects of 5% dextrose or BQ-788 on amitriptyline induced (a) MAP, (b) QRS duration and (c) HR. *p < 0.05, **p < 0.01, ***p < 0.001 versus control. MAP: mean arterial pressure; HR: heart rate.
Protocol 2
There was no significant difference between the baseline measurements of control (n = 7) and experimental groups (n = 6) in MAP, HR and QRS duration (Table 3). Bolus of 5% dextrose or BQ-788 did not change MAP (95.3 ± 4.2% and 102.3 ± 1.9%), HR (102.8 ± 1.4% and 98.7 ± 1.3%) and QRS duration (100.0 ± 0% and 103.3 ± 3.3%) at the 15th minute compared with baseline for 5% dextrose and BQ-788 groups, respectively (p > 0.05). In both the groups, amitriptyline infusion reduced MAP and HR and prolonged QRS duration significantly when compared with the end of the 15th minute of the 5% dextrose and BQ-788 bolus (Table 2). There was no statistically significant difference in MAP, HR and QRS duration between the control and BQ-788 groups in any time (p > 0.05).
The effects of dextrose (n = 7) or BQ-788 (n = 6) bolus on MAP, HR and QRS duration differences induced amitriptyline infusion (protocol 2).

MAP: mean arterial pressure; HR: heart rate.
a p < 0.05 versus baseline.
b p < 0.01 versus BQ-788 or 5% dextrose bolus.
c p < 0.05 versus BQ-788 or 5% dextrose bolus.
d p < 0.001 versus baseline.
e p < 0.01 versus baseline.
f p < 0.001 versus BQ-788 or 5% dextrose bolus.
Discussion
In the present study, we have demonstrated that an ETB receptor antagonist, BQ-788, improved amitriptyline-induced hypotension, HR reduction and QRS prolongation in a rat model of amitriptyline toxicity.
One of the well-known mechanisms of amitriptyline-induced hypotension is alpha-1 adrenergic receptor blockade and noradrenaline reuptake inhibition. Additionally, concentration-dependent L-type calcium channel blocking effect of amitriptyline is suggested to contribute to the mechanism of hypotension. 11 Treatment attempts for amitriptyline-induced hypotension are sodium bicarbonate, norepinephrine, phenylephrine or vasopressor doses of dopamine. 3 Some investigational treatment modalities such as L-NAME, a NO synthase inhibitor, selective adenosine A1 and A2a receptor antagonists, glucagon or theophylline were reported to be effective in reversing amitriptyline-induced hypotension in animal models. 8,12 –14
ETs, which are powerful vasoconstrictor peptides, show their effects via their receptors entitled ETA and ETB. While ETA receptors are responsible for vasoconstriction and increase in blood pressure, ETB receptors cause vasodilatation by the secretion of NO and prostacycline. ETB receptors are also present on the smooth muscles of human arteries and mediate vasoconstriction. 15,16 ETs vasoconstructor tonus is dominated in the circulation. ETB mediated vasoconstriction may negligible. 17 Verhaar et al. demonstrated that ETB receptor antagonism either alone or with ETA receptor antagonism causes vasoconstriction. 18 In our study, an ETB receptor antagonist, BQ-788, improved amitriptyline-induced hypotension. We might suggest several mechanisms of improvement in amitriptyline-induced hypotension using BQ-788 as given below.
First, the beneficial effect of BQ-788 on amitriptyline-induced hypotension might result from increased ET-1 levels in circulation. Systemic blockade of ETB receptors by BQ-788 results in a significant rise in circulating ET-1, which cause vasoconstriction. In a study by Brunner et al., blockade of ETB receptors by BQ-788 caused a 20-fold increase in ET-1 levels in nonhuman primates. 19 –21 Increased ET-1 levels might improve hypotension by enhancing Ca2+ influx through the L-type calcium channels. Pollock et al. showed that antidepressants block cardiac calcium currents concentration dependently. Therefore, ETB receptor blockade and increased ET-1 levels in circulation might enhance contractility of heart by increasing calcium influx that resulted in improvement of hypotension. 11,22,23 Although we could not measure the ET-1 levels in circulation, we believe that ETB receptor blockade with BQ-788 might increase ET-1 levels and this increase might contribute to the improvement of amitriptyline-induced hypotension.
Second, increased endogen catecholamine releasing activity by ET might also contribute to the improvement of hypotension. Li et al. showed that ET caused endogen release of catecholamines in rat and rabbit papillary muscles. 4
Third, BQ-788 might improve amitriptyline-induced hypotension via inhibition of NO secretion from endothelium. Because stimulation of ETB receptors causes vasodilatation by secreting NO from the endothelial cells, ETB receptor blockade by BQ-788 might block NO release from endothelium that contribute to the improvement of hypotension.
BQ-788 improved the amitriptyline-induced bradycardia in our study. As it is known, ET receptor antagonists have no effect on cardiac chronotropy or positive chronotropic effect mediated by ETB receptors, 24,19 but ETs have sympathoexcitatory effects and these mediate through ETA receptors. Therefore, administration of BQ-788 and blockade of ETB receptors might have beneficial effect on HR through sympathoexcitatory effect of endogen ET. 25
Although an ETB receptor antagonist, BQ-788, alone did not change QRS duration during the experimental period of our study, it surprisingly improved amitriptyline-induced QRS prolongation in rats. The principal mechanism of QRS complex prolongation induced by amitriptyline is voltage-gated sodium channel blockade, which increases the duration of the cardiac action potential and refractory period and delays atrioventricular conduction. However, ETs or ET receptor antagonists have no described sodium channel opener activity. 3 In a study by Cheng et al., a tetradoxin-resistant voltage-gated Na+ current in human cardiac tissue (hH1, Nav 1.5) was demonstrated to be enhanced by ET-1. Therefore, blocking ETB receptors by BQ-788, an increase in circulating ET-1 might shorten QRS duration by increasing Na+ current in the heart. 26
In the second part of our study, lack of beneficial effect on amitriptyline-induced cardiovascular changes with pretreatment by BQ-788 indicates that there might not be any role of ETB receptors in the mechanism of amitriptyline-induced cardiovascular depression. Improvement in amitriptyline-induced hypotension by BQ-788 might be the consequence of the physiological antagonism. These findings suggest that ETB receptor antagonist, BQ-788, may be effective in reversing hypotension and bradycardia induced by other toxicities such as calcium channel blocker or beta blocker. Therefore, in different toxicity models, ETB receptor antagonist, BQ-788, may be tried.
Limitation of our study was lack of ET-1 level measurements in circulation. In some experimental studies, blockade of ETB receptors by selective ETB receptor antagonists resulted in an increase in ET-1 levels in circulation.
Conclusion
An ETB receptor antagonist, BQ-788, improved cardiovascular toxicity is induced by amitriptyline. As a pretreatment with an ETB receptor antagonist, BQ-788 has no effect on amitriptyline-induced hypotension, bradycardia and QRS prolongation; we concluded that ETB receptors have no role in amitriptyline-induced cardiovascular toxicity. Therefore, it is suggested that BQ-788, an ETB receptor antagonist, may have beneficial effects in cardiovascular changes induced by amitriptyline via a physiologic antagonism rather than a pharmacologic antagonism. Also in future studies, ETB receptor antagonist may be tried in some toxicities such as calcium channel blocker and beta blocker.
Footnotes
Authors’ Note
This study was presented at the EAPCCT XXXI International Congress, Dubrovnik, Croatia and was short listed at candidate-young investigator award.
Funding
The present research study was supported by Dokuz Eylul University Research Foundation (2010.KB. SAG. 004).
