Abstract
The comet assay is a well-established, simple and sensitive method to measure DNA damage in single cell and is commonly used in human trials to investigate the effects of pollution, occupational hazards and potential genoprotective agents. Peripheral blood lymphocytes are most commonly used in human biomonitoring studies, but lymphocytes collected from the mouth offer a potentially attractive, noninvasive alternative. The aim of the current study was to develop a buccal cell lymphocyte comet assay procedure. Cells were collected from mouthwash of three healthy volunteers and tested individually. The comet assay was performed under different pH and times of alkaline treatment, electrophoresis run times and hydrogen peroxide concentrations. Optimal conditions for buccal lymphocytes in comet assay were found to be pH >13 for unwinding and electrophoresis buffers, 10-min alkaline unwinding treatment and 20-min electrophoresis run time. We successfully utilized our optimized assay conditions to demonstrate the genoprotective activity of quercetin. This newly established procedure offers an alternative noninvasive sampling method for the investigation of DNA protection and/or damaging effect.
Introduction
Reactive oxygen species (ROS) are highly reactive radical and nonradical oxygen-containing molecules, which are formed as the by-products during normal cellular processes. ROS play a crucial physiological role in cell signalling and homeostasis. Yet, in higher concentrations, ROS can attack vital biomolecules, namely, nucleic acids, lipids and proteins causing cellular damage. Hence, vigorous means are required to eliminate excess ROS instantaneously. 1 Our cells are endowed with effective defence and repair system against oxidative damage. Several enzymatic and nonenzymatic systems function to inactivate, reduce and scavenge ROS. 2 However, the production of ROS sometimes can offset the endogenous antioxidant defence system resulting in oxidative stress. Oxidative stress has been implicated in the aging process, inflammation and the pathogenesis of over 100 ailments including cancers, atherosclerosis, neurodegenerative diseases, acquired immunodeficiency syndrome (AIDS) and metabolic syndrome. 3
Hitherto, the exact pathogenic mechanism of ROS is not well known, but three reactions are recognized to cause DNA damage and cellular injury: lipid peroxidation of membranes, DNA fragmentation and cross-linking of proteins. 2,4 DNA damage can lead to mutagenesis, cellular metabolism defects or even cell necrosis. 5 To assess genotoxicity of ROS and other hazardous substances, in 1984, Östling and Johanson developed the first microelectrophoretic method known as single-cell gel electrophoresis (SCGE) or comet assay, in which mammalian cells were embedded in melted agarose gel and coated onto microscope slide to run electrophoresis under neutral buffer, in order to investigate the genotoxicity of radiation. 6 A few years later, Singh and coworkers modified the procedure to study radiation-induced DNA damage in human cells and to analyze the repair kinetics of DNA in single cells. 7 They modified the microelectrophoretic procedure by changed the pH from neutral to highly alkaline (pH >13), in order to increase the sensitivity of comet assay to investigate the recovery of human lymphocytes after exposure to low dose radiation. This methodology formed the basis of the standard comet assay. 8
The comet assay is fast, cost-effective and small number of cells is required. 9,10 Other modified comet assay versions are also available to achieve different objectives such as neutral pH, enzyme-assisted, proteinase K-assisted, fluorescent in situ hybridization and lysed cell comet assays. 11 –13 Comet assay is also commonly employed in nutraceutical and natural product research, particularly in traditional Chinese medicines. 12 Other novel applications of comet assay include detection of apoptosis and assessment of absorbed dose of diagnostic x-ray. 14,15
The types of cells used in SCGE are variable, ranging from human blood cells to culture cells to bacteria. The criteria of selecting cell type depend on the aim of the experiment, ethical considerations, budget and convenience. Human cells utilized include lymphocytes, fibroblasts, spermatocytes, epithelial cells and bladder cells. 12,16 Previously, buccal epithelia cells were used in genotoxicity studies 17 ; it has been suggested that buccal lymphocytes provided better result in terms of purity and viability, and it was a potential substitute for peripheral blood cells in comet assay. 18 However, we have found that the background score of buccal lymphocytes was very high (data not shown). It is important to optimize experimental conditions for different types of cells 19 and optimization of experimental parameters for buccal lymphocytes in comet assay deemed to be necessary. The aim of this study is to optimize comet assay conditions, such as, pH, duration of alkaline treatment and length of electrophoresis, for buccal lymphocytes.
Materials and methods
Chemicals and reagents
Histopaque 1077, type VII low melting point agarose, disodium EDTA (for both lysis solution and electrophoresis buffer), Triton X-100, hydrogen peroxide (H2O2) solution (30% v/v), quercetin dihydrate and ethidium bromide were purchased from Sigma-Aldrich Chemicals (St Louis, Missouri, USA). Sodium chloride and disodium hydrogen phosphate anhydrous were purchased from Panreac Quimica (Barcelona, Spain), while sodium dihydrogen phosphate dihydrate and sodium hydroxide (for both lysis solution and electrophoresis buffer) were obtained from (BDH Prolabo, Leicestershire, England). Standard agarose was obtained from Amresco (Solon, Ohio, USA).
Sample collection
Three healthy volunteers (one male and two females; age between 37 and 43 years) were recruited for providing buccal lymphocytes. Written informed consent was obtained from each subject, and Ethics in Human Research Committee of Charles Sturt University has approved all experimental procedures. Method for collecting buccal lymphocytes followed previous study with modifications. 20 Volunteers were asked to rinse their mouth once with room temperature normal saline solution that was then discarded; then they were asked to rinse with cold (4°C) normal saline solution for 1 min. The cold saline rinse solution containing cells from the buccal cavity was collected into a 50-mL centrifuge tube. The tubes were centrifuged at a relative centrifugal force of 400g for 10 min at 4°C. The solution was decanted and 25 mL phosphate-buffered saline (PBS) was added for cell washing. Centrifugation step was repeated and PBS was decanted. The cell pellet was collected and transferred into a microtube.
Buccal lymphocyte isolation
After dispersing into 1 mL cold PBS, cells were underlaid with 100 µL of Histopaque 1077 and centrifuged at 400g for 7 min at 4°C. The layer of Histopaque 1077 with buccal lymphocytes was then collected into a microtube. PBS (1 mL) was added to the microtube and centrifugation was repeated to collect buccal lymphocyte.
Cell treatment
The buccal lymphocyte pellet was stressed with freshly prepared H2O2 in PBS. Cells were resuspended and incubated with 1 mL H2O2 (final concentrations of 100 and 200 μM) for 5 min in ice. This was used to investigate the dose response of comet assay score in the procedure.
Quercetin pretreatment
In another set of experiments, cells from same volunteers were pretreated with quercetin before oxidative challenge with H2O2. Briefly, 10 mM of stock quercetin suspension was prepared by mixing and ultrasonicating quercetin dihydrate in deionised water. The stock quercetin suspension was further diluted in PBS as working solution of 10, 20 and 50 µM. Buccal lymphocytes were incubated with 1 mL of different quercetin concentrations at 37°C for 30 min. Afterwards, cells were subjected to optimized comet assay as described below.
Comet assay optimization
Microscopic slides were precoated with 1% standard agarose. Standard agarose (85 µL, 1% w/v in PBS) was added onto precoated slides on the day of experiment. Low melting point (LMP) agarose (85 µL, 1% w/v in PBS), kept at 40°C, was gently mixed with the buccal lymphocytes in the microtube. The mixture was then transferred on the top of the standard agarose layer. These sandwich slides were kept at 4°C until the gel is solidified, before removing the cover slip. Triton X-100 (400 µL) was mixed with 40 mL cold lysis solution in a staining jar. The slides were put into the jar and kept at 4°C for 60 min for lysis. These slides were transferred into another staining jar containing 40 mL cold electrophoresis buffer (pH ~12 or >13) for 10, 20 or 30 min, in order to unwind DNA and remove the residual salt from lysis solution. The slides were then placed evenly on electrophoresis tank platform and covered with electrophoresis buffer. The electrophoresis tank was surrounded by icepacks to keep it cold. Electrophoresis was run for 10, 20 or 30 min at constant voltage (25 V) at 0.04 A and pH 12 or at 0.3 A and pH >13. After electrophoresis, slides were placed in a staining jar with deionised water for 5 min. This step was repeated twice to remove electrophoresis solution. 9
Assay scoring
Slides were air dried and stained with 50 µL ethidium bromide. Approximately 100 cells per slide were scored under a Nikon Microphot-Fx Epifluorescence microscope, with excitation filter at 510–560 nm, (Tokyo, Japan). Different levels of DNA damage were graded from 0 to 4 (Figure 1), where 4 was the most severe damage for each comet image according to the comet tail intensity and length visualized. The score of each gel can be calculated with the following equation

Score (0–4) of comet assay showing different levels of DNA damage.
Statistical procedures
Statistical analysis was carried out using Prism program version 5.0 for Windows (GraphPad software Inc., La Jolla, California, USA). Linear regression analysis was used to evaluate the dose-response of H2O2 treatment. The protective effect of quercetin pretreatment against H2O2 was investigated by one-way analysis of variance (ANOVA) followed by Dunnett’s t test. Results were considered as significantly different at p < 0.05.
Results
The comet scores obtained with electrophoresis conducted at pH >13 were generally higher than that from less alkaline conditions (pH ~ 12) under corresponding alkaline unwinding treatment and electrophoresis time (Figures 2 and 3), which suggests that higher sensitivity can be achieved with pH >13. The second consideration of the parameters used was the dose-response behaviour. Alkaline treatment of 10 min with 20 min electrophoresis at pH >13 showed good linearity across working range of 0–200 μM H2O2 treatment (Figure 3(a)) with the best goodness-of-fit (r 2 = 0.998). Hence, 10-min alkaline unwinding treatment and 20-min electrophoresis time were chosen in the subsequent quercetin pretreatment study.
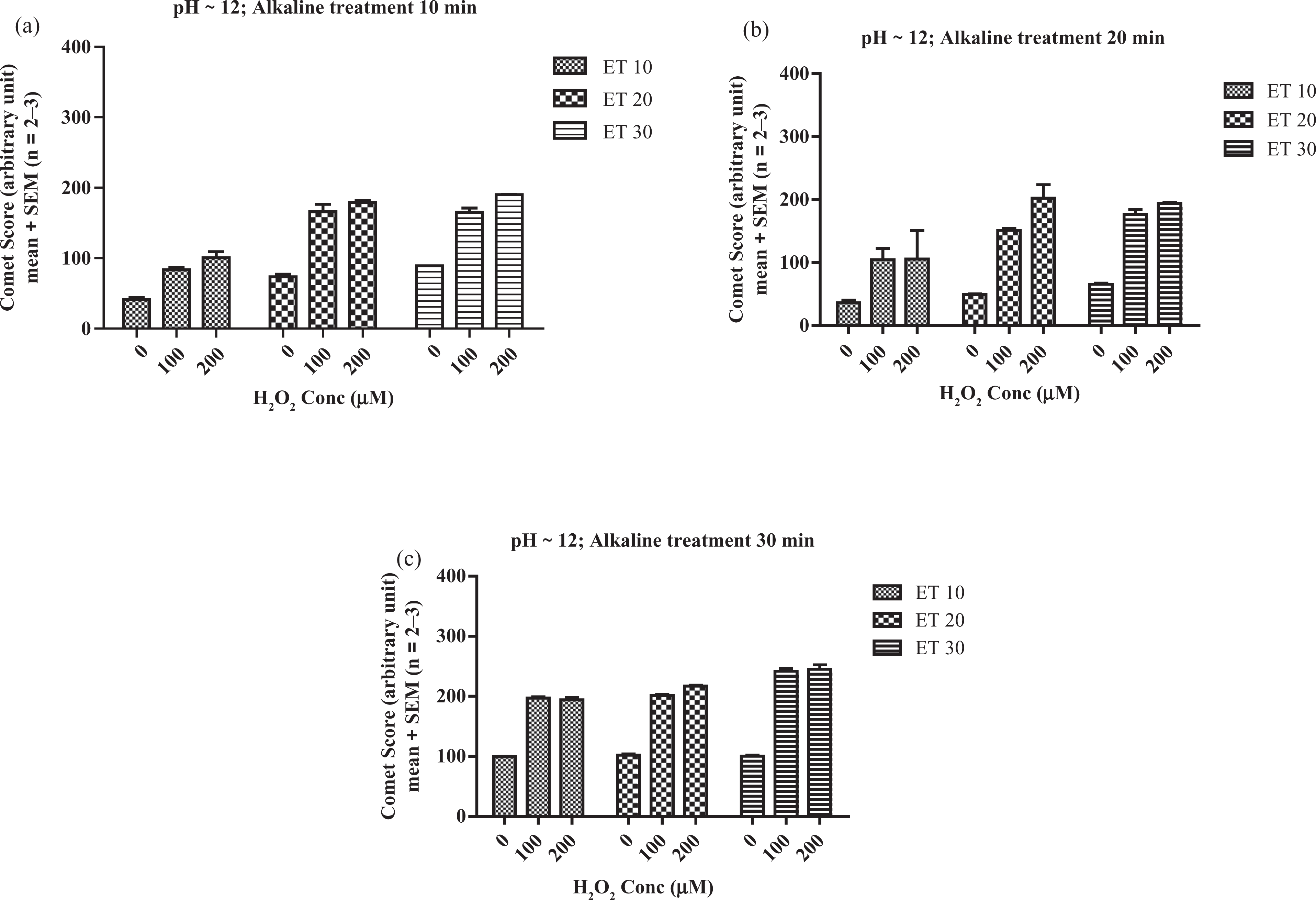
Mild pH (~ 12) comet assay procedure with various electrophoresis times (ET) and different lengths of alkaline treatment: (a) 10 min, (b) 20 min and (c) 30 min.

Strong pH (>13) comet assay procedure with various electrophoresis times (ET) and different lengths of alkaline treatment: (a) 10 mins, (b) 20 mins and (c) 30 mins.
Results showed that 30 min quercetin pretreatment at all tested concentrations (10, 20 and 50 µM) protected DNA from H2O2 (100 µM)-induced oxidative stress (Figure 4; one-way ANOVA followed by Dunnett’s t test, p < 0.0001).

Effect of quercetin on DNA damage (mean +1 SEM) of buccal lymphocytes from three healthy subjects tested. Results of oxidative challenge with 100 μM H2O2, preincubated with quercetin at the concentrations of 0, 10, 20 and 50 μM. One-way analysis of variance, *p < 0.0001.
Discussion
The aim of the current study was to optimize the comet assay parameters for buccal lymphocytes. Results showed that high pH (>13) version of comet assay with relatively short alkaline unwinding treatment (10 min) and electrophoresis run time (2 min) provided highest linearity of the dose-response curve against oxidative challenge, in addition to achieving low-baseline comet score. The span of comet score for pH ~ 12 was generally lower than that of pH >13, which comes in agreement with earlier findings. 21 It has been suggested that high pH conditions can induce disruption of DNA–nuclear matrix interactions and produce more single stranded DNA fragments that forms the comet tail, whereas under lower pH conditions, only relaxed loop domains are formed and thereby ‘shorter’ comet tails. 22 Lower alkaline conditions for unwinding and electrophoresis keep DNA in the comet head and tail in the form of double-stranded DNA, while single-stranded DNA is observed under strong alkaline conditions. Thus, higher pHs yield a maximized score after unwinding and electrophoresis. Both neutral and alkaline comet assay conditions are used based on cell type and the aim of investigation. 8,23,24
Besides the pHs of unwinding and electrophoresis buffers, the duration of unwinding treatment and electrophoresis run time are critical variables and sometimes dependent on cell type. Vijayalaxmi et al. suggested that alkaline unwinding between 20 and 60 min is suitable for most cell types under strong alkaline electrophoresis condition. 25 In our study, comet assay with 30-min alkaline unwinding showed the highest score. However, dose-response curves to H2O2 oxidative challenge for 30-min treatment was not any better than the 10-min treatment. Electrophoresis run time between 5 and 40 min has been recommended. 26 In the current investigation, 30-min electrophoresis time did not increase the sensitivity of the test compared with 20-min runs, that is, both 100 and 200 μM of H2O2 treatment demonstrated similar comet scores.
Quercetin is a potent antioxidant bioflavonoid with well-established protective ability against oxidative stress. 27 The genoprotective activity of quercetin against H2O2-mediated DNA damage has been demonstrated in comet assay previously. 28 In the current study, quercetin was used as a positive control to demonstrate the effectiveness of the selected comet assay parameters for detecting genoprotection. 29 Quercetin showed protective effect on buccal lymphocyte DNA in the range 10–50 µM.
To conclude, pH >13, 10-min alkaline unwinding treatment and 20-min electrophoresis run time provided optimal conditions for buccal lymphocytes in comet assay. This newly established procedure offers an alternative noninvasive sampling method for studying human cells in comet assay.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgment
The authors thank Hong Kong Institute of Vocational Education (Shatin), the Hong Kong Polytechnic University and Kwong Wah Hospital for supporting this work.

