Abstract
The present study aimed at determining the differences in plasma concentrations of traditional antiepileptics such as phenytoin, carbamazepine, and valproic acid in patients receiving monotherapy and combination therapy. In addition, the effect of gender and age on plasma drug concentration was evaluated in these patients. For this purpose, plasma trough concentrations obtained during routine therapeutic monitoring of these drugs were assessed retrospectively. The average plasma concentrations reached the apparent therapeutic ranges, except for the average plasma concentration of phenytoin, which was below the therapeutic range in patients who received only phenytoin or in combination with the other agents. Phenytoin when combined with carbamazepine or valproic acid significantly decreased the average plasma concentrations of these drugs to subtherapeutic concentrations. The results showed that plasma carbamazepine concentrations were higher in men than in women, whereas plasma concentrations of valproic acid and phenytoin were higher in women than in men. The difference in this regard between men and women was found to be statistically significant for phenytoin. The difference between the average plasma concentrations of carbamazepine, phenytoin, and valproic acid among age groups was not significant. In conclusion, our study measured the average plasma antiepileptic drug concentrations in patients with epilepsy who were receiving monotherapy and combination therapy and were routinely monitored, and has thus shown the importance of drug monitoring in the evaluation of the effectiveness of these drugs.
Introduction
Antiepileptics are a group of drugs that are very sensitive to drug interactions, and they are used for long periods for neurological disorder such as epilepsy, neuropathic pain, and migraine. 1 –3 In the early stage of epilepsy therapy, seizures can be brought under control in 60–70% of patients by monodrug therapy. The treatment strategy for epilepsy patients who cannot be treated by monotherapy is to continue monotherapy with an alternative antiepileptic drug or to start polytherapy with two different drugs. In patients who show resistance to this treatment strategy, a combination of three drugs is used in most cases. 1,2,4 It has been shown that polytherapy with antiepileptic drugs is more effective than monotherapy for the control of seizures. However, a combination of two or more antiepileptic drugs can be risky because of the increased possibility of adverse effects and pharmacokinetic drug interactions. 1,2,5 Most of these interactions are based on metabolic features and can be predicted by identifying the isoenzymes responsible for drug metabolism and determining the effects of these drugs on similar isoenzymes. 1,2
Antiepileptic drugs can produce an inducing or inhibiting effect on the metabolism of other drugs, including antiepileptics; thus, these drugs can increase or decrease the effect of other drugs. 6 Phenytoin, carbamazepine, phenobarbitol, and primidone, which are known as classical antiepileptic drugs, reduce plasma concentrations and pharmacologic effects of drugs by inducing enzyme activity. 1,2 The plasma concentration of antiepileptic drugs such as valproic acid tends to increase plasma concentrations because such drugs inhibit enzymes responsible for their metabolism and thus increase the risk of drug toxicity. 2
It is very difficult to evaluate the success of antiepileptic drugs during the treatment. Therapeutic drug monitoring, that is measurement of drug concentrations in biological fluids using clinical pharmacology, is the most popular means for the optimization of antiepileptic therapy. Appropriate and rational use of therapeutic drug monitoring increases the success of drug therapy, achieving maximum seizure control and producing minimum adverse drug reaction. 7
In this study, we investigated the plasma drug concentration in patients with epilepsy who routinely received monotherapy or combination drug therapy with phenytoin, carbamazepine, and valproic acid, and analysed the interactions between these drugs. In addition, the impact of kinetic differences between men and women and different age groups with regard to plasma drug concentration was evaluated by investigating the influence of gender and age on monotherapy and combination drug therapy.
Materials and methods
Within the scope of this study, plasma concentrations of patients with epilepsy aged between 18 and 65 years who underwent routine analysis at Eskisehir Osmangazi University Medical Faculty Department of Pharmacology between 2007 and 2010, and were treated with oral phenytoin, carbamazepine, and valproic acid were evaluated retrospectively. Comorbidities (hypertension, diabetes, and other chronic diseases), chronic drug use (except antiepileptic drugs), hepatic, and renal failure patients were excluded from the study. Patients treated with phenytoin, carbamazepine, and valproic acid for more than 3 months were included in this research. The records of patients whose blood samples were obtained before the morning drug dose (trough level) after the last dose were evaluated. The daily doses of phenytoin, carbamazepine, and valproic acid administered to the patients included in this study were similar to the therapeutic doses. Patients who used carbamazepine, phenytoin, and valproic acid out of daily standard therapeutic dosage were excluded from the study.
The therapeutic index is accepted as 10–20 μg/ml for phenytoin, 4–10 μg/ml for carbamazepine, and 50–100 μg/ml for valproic acid. Patients were classified as monoantiepileptic drug users (monotherapy) and combination drug users (combination therapy). These groups were then divided according to gender and age. Ages were classified as decades as follows: 18–28, 29–38, 39–48, 49–58, and 59–65.
The blood concentrations of phenytoin, carbamazepine, and valproic acid were measured in the aforementioned laboratory with a Boehringer Mannheim Hitachi 912 autoanalyser by using Cedia® EIA (Microgenic Corporation) kits through standardized methods. The assay is for the quantitative measurement of the unbound drug fraction (e.g. free phenytoin) in serum or plasma. This assay is a unique homogeneous enzyme immunoassay system that uses recombinant DNA technology.
The statistical analyses of plasma concentrations of drugs were performed using one-way analysis of variance (ANOVA) with Tukey’s post hoc test, by GraphPad Prism 4 version. Other statistical analyses were performed using one-way ANOVA with Dunnett’s post hoc test, GraphPad Prism 4 version. Plasma concentrations of antiepileptic drugs are shown in tables as arithmetic mean ± standard error of the mean. The p value <0.05 was considered as statistically significant.
Results
In this research, 1741 patients with epilepsy received phenytoin monotherapy; 1598, carbamazepine monotherapy; and 4534, valproic acid monotherapy. There were 304 patients receiving valproic acid–carbamazepine combination therapy, 159 patients receiving valproic acid–phenytoin combination therapy, and 159 patients receiving phenytoin-carbamazepine combination therapy. The classification of evaluated cases is shown in Table 1.
The number of patients treated with monotherapy and combination therapy according to gender

W: woman; M: male; +CARB: in addition to carbamazepine group; +PHEN: in addition to phenytoin group; +VA: in addition to valproic acid group.
Among the female patients, 667 were treated only with phenytoin; 855, only with carbamazepine; and 2418, only with valproic acid. Among the male patients, 1074 were treated only with phenytoin; 743, only with carbamazepine; and 2116, only with valproic acid. There were 73 female patients and 86 male patients treated with phenytoin-carbamazepine, 143 female patients and 161 male patients treated with carbamazepine–valproic acid, and 68 female patients and 91 male patients treated with phenytoin–carbamazepine. Classification of patients receiving monotherapy and combination drug therapy according to gender is shown in Table 2.
The number of patients treated with monotherapy and combination therapy according to age
+CARB: in addition to carbamazepine group; +PHEN: in addition to phenytoin group; +VA: in addition to valproic acid group.
Among the 18–29 patients, 337 were treated only with phenytoin; 446 only with carbamazepine; and 1299 only with valproic acid. Among the 29–38 patients, 327 were treated only with phenytoin; 433 only with carbamazepine; and 1311 only with valproic acid. Among the 39–48 patients, 496 were treated only with phenytoin; 368 only with carbamazepine; and 1050 only with valproic acid. Among the 49–58 patients, 386 were treated only with phenytoin; 278 only with carbamazepine; and 513 only with valproic acid. Among the 59–65 patients, 155 were treated only with phenytoin; 73, only with carbamazepine; and 361, only with valproic acid. Classification of patients receiving monotherapy and combination drug therapy according to age is shown in Table 3.
The mean plasma carbamazepine, phenytoin, and valproic acid concentrations of patients treated with mono- and combination therapy according to gendera

+CARB: in addition to carbamazepine group; +PHEN: in addition to phenytoin group; +VA: in addition to valproic acid group.
a Results are expressed as average ± standard error.
The average plasma carbamazepine concentration in patients who received only carbamazepine therapy was 5.37 µg/ml. This value was significantly reduced to 3.38 μg/ml in patients who used phenytoin combination therapy, and was found to be 5.53 μg/ml in patients who received valproic acid combination therapy (Figure 1). In 67.92% of patients treated with phenytoin combination therapy, the average plasma carbamazepine concentration was below the therapeutic index.

The mean plasma concentration of carbamazepine in patients treated with carbamazepine monotherapy and combination therapy. CARB: patients treated with carbamazepine alone; CARB + PHEN: patients treated with carbamazepine–phenytoin combination therapy; CARB + VA: patients treated with carbamazepine–valproic acid combination therapy. ***Different from CARB (p < 0.01).
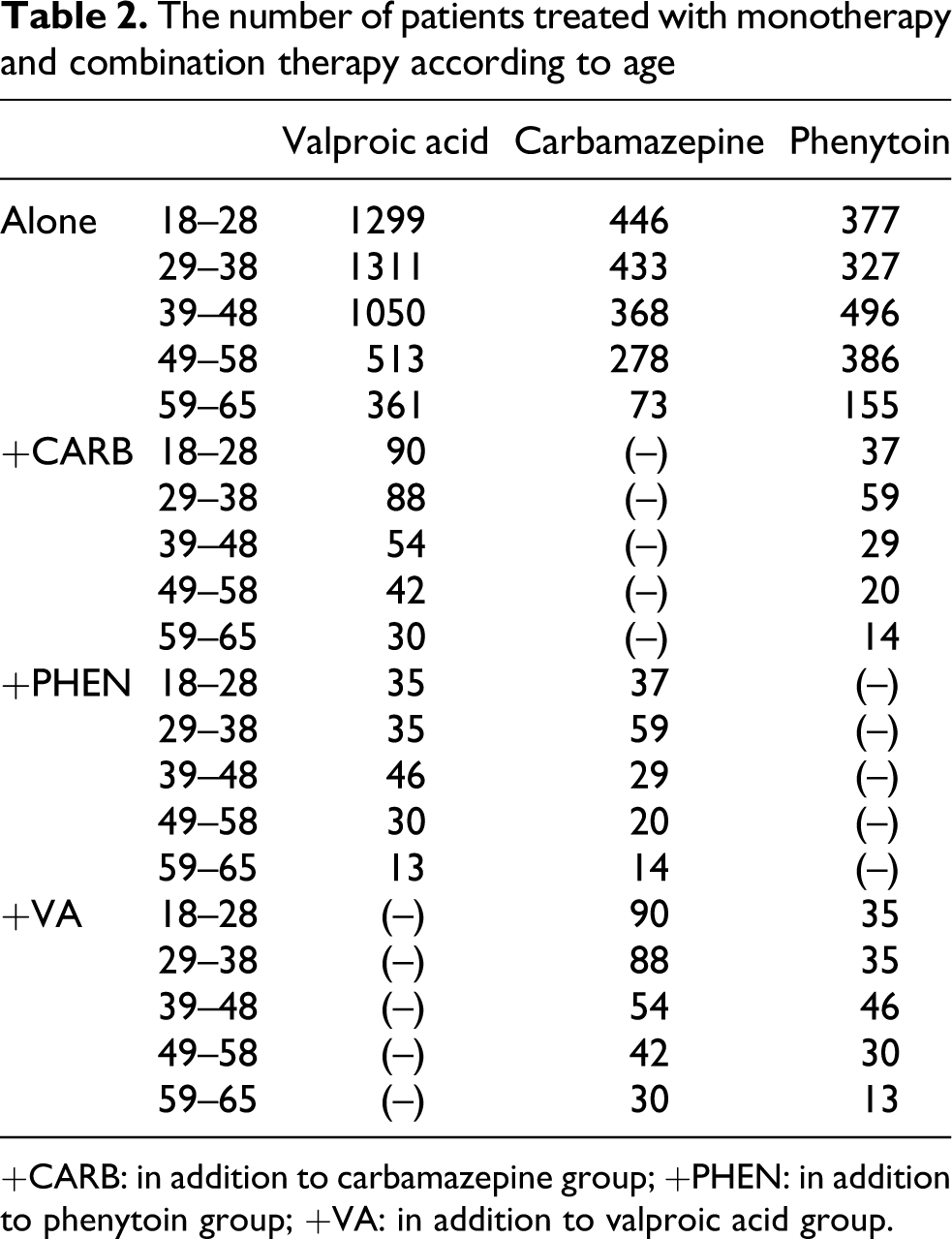
The plasma phenytoin concentration was found to be 6.42 μg/ml in patients treated only with phenytoin; this value is below the therapeutic index. The average plasma phenytoin concentration was 6.24 μg/ml when carbamazepine was included in the therapy, and the value was found to be 6.62 μg/ml when valproic acid was included in the therapy; these values are below the therapeutic index. There were no statistically significant differences between patients receiving monotherapy and combination therapy (Figure 2). It was found that the average plasma phenytoin concentration was below the therapeutic index in 1369 of 1741 patients with epilepsy.
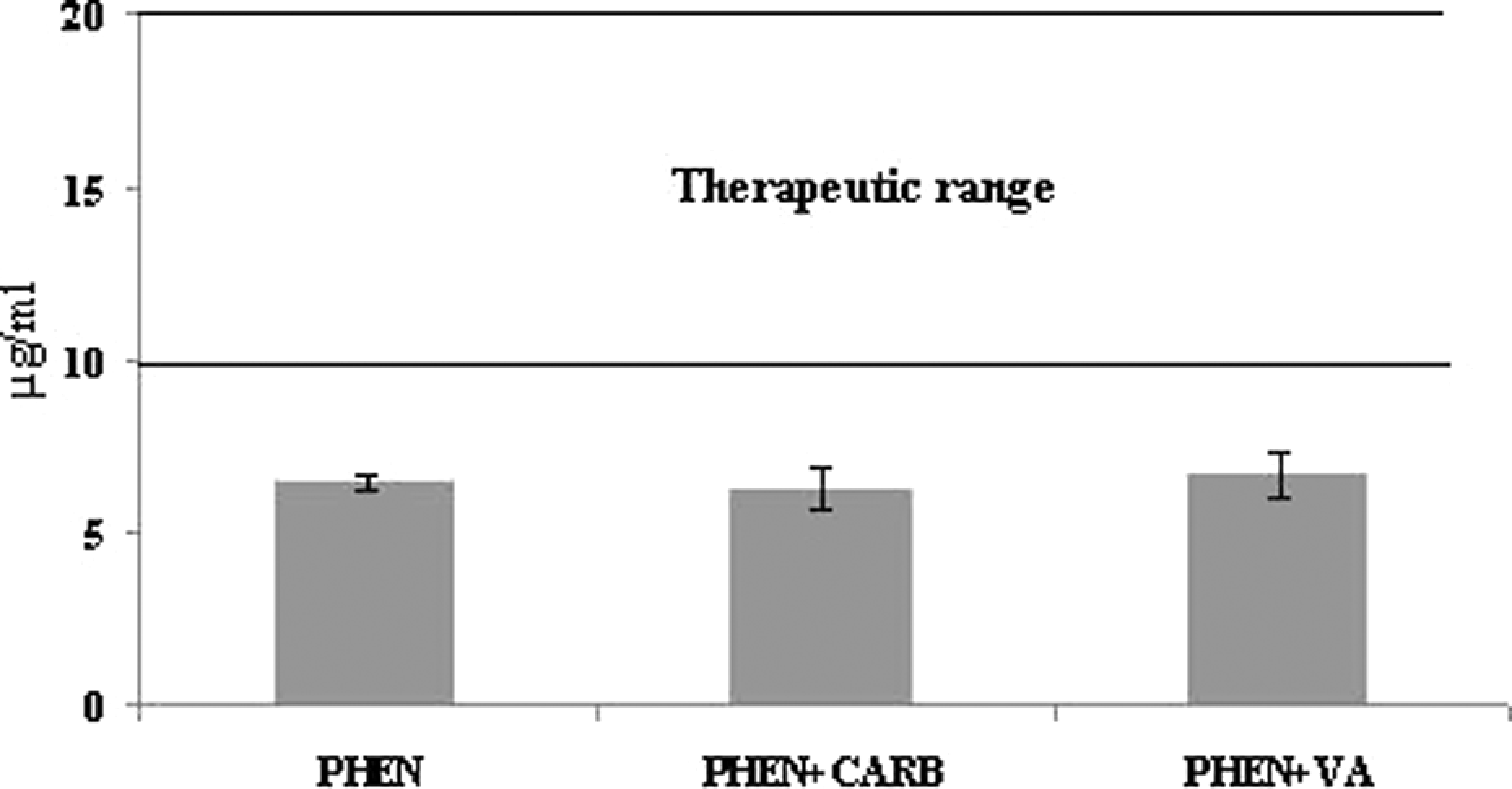
The mean plasma concentration of phenytoin in patients treated with phenytoin monotherapy and combination therapy. PHEN: patients treated with phenytoin alone; PHEN + CARB: patients treated with phenytoin–carbamazepine combination therapy; PHEN + VA: patients treated with phenytoin–valproic acid combination therapy.
The average plasma valproic acid concentration was found to be 69.42 μg/ml in patients receiving only valproic acid therapy, whereas it was 44.44 μg/ml in patients receiving phenytoin combination therapy and 60.96 μg/ml in patients receiving carbamazepine combination therapy. In this study, it was interesting to note that the average plasma valproic acid concentration was found to decline significantly and reached values below the therapeutic index (Figure 3). It was stated that during valproic acid–phenytoin combination therapy, the plasma valproic acid level in 62.89% of the patients was below the therapeutic index.

The mean plasma concentration of valproic acid in patients treated with valproic acid monotherapy and combination therapy. VA: patients treated with alone valproic acid; VA + PHEN: patients treated with valproic acid–phenytoin combination therapy; VA + CARB: patients treated with valproic acid–carbamazepine combination therapy. ***Different from VA (p < 0.01).
The average plasma concentration of carbamazepine was 5.31 μg/ml in female patients and 5.43 μg/ml in male patients who received only carbamazepine therapy; however, the difference between men and women in this regard was not significant. The average plasma concentration of carbamazepine was 3.29 μg/ml in women and 3.46 µg/ml in men after phenytoin was included in the therapy. It was stated that in men and women, the average plasma concentration of carbamazepine declined significantly and was below the therapeutic index in patients receiving combination treatment as compared with those receiving carbamazepine only. The average plasma concentration of carbamazepine was 5.36 μg/ml in women and 5.67 μg/ml in men after the inclusion of valproic acid in the treatment. There were no significant differences between men and women in terms of the average plasma carbamazepine concentration between patients receiving valproic acid combination therapy and monotherapy (Figure 4).
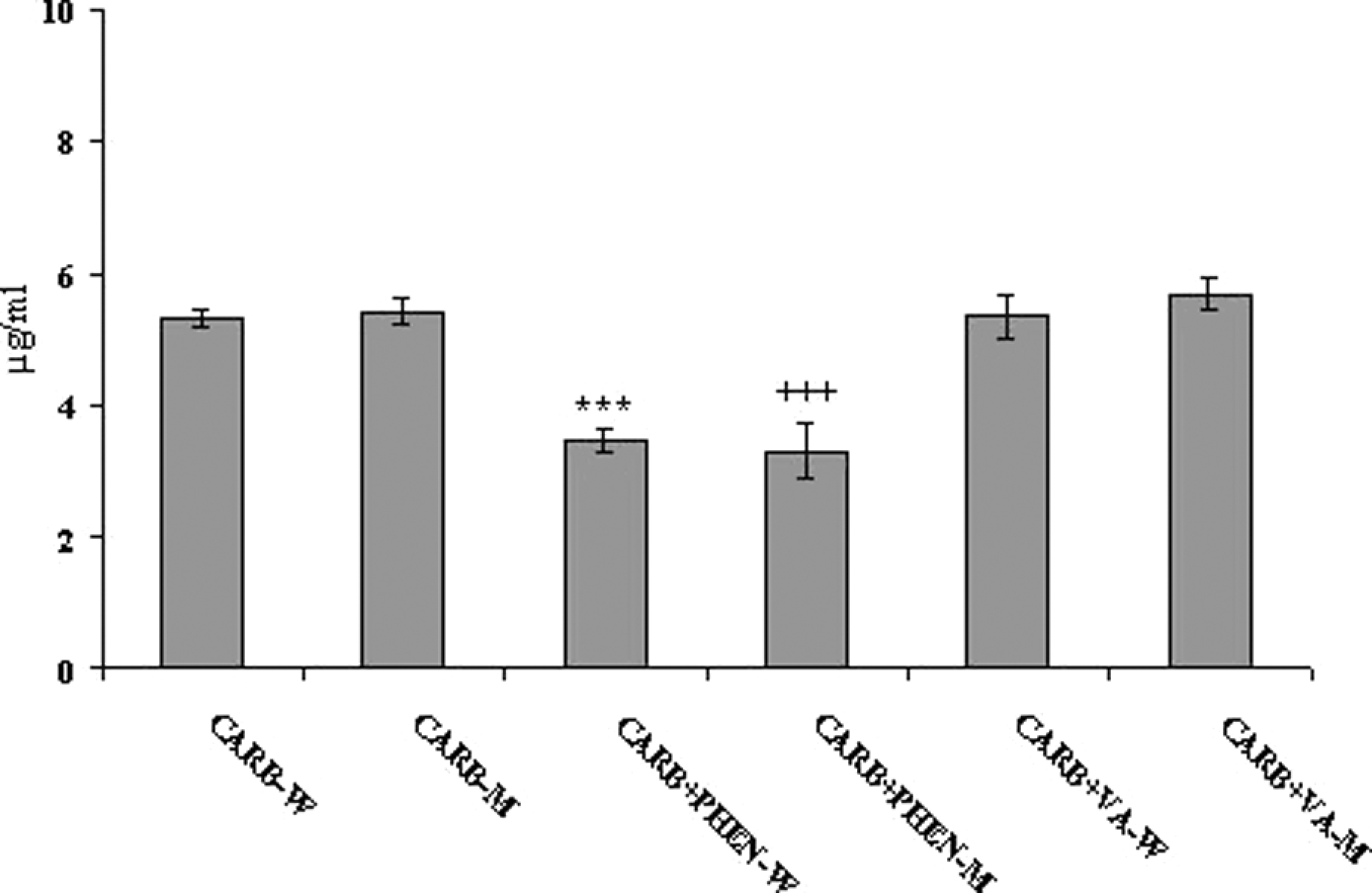
The mean plasma concentration of carbamazepine in female and male patients treated with carbamazepine monotherapy and combination therapy. CARB-W: female patients treated with carbamazepine alone; CARB + PHEN-W: female patients treated with carbamazepine–phenytoin combination therapy; CARB + VA-W: female patients treated with carbamazepine–valproic acid combination therapy; CARB-M: male patients treated with carbamazepine alone; CARB + PHEN-M: male patients treated with carbamazepine–phenytoin combination therapy; CARB + VA-M: male patients treated with carbamazepine–valproic acid combination therapy. ***Different from CARB-W (p < 0.01); +++Different from CARB-M (p < 0.01).
The average plasma phenytoin concentration in patients receiving only phenytoin was 6.96 μg/ml in women and 6.09 μg/ml in men; the difference between men and women in this regard was statistically significant. When carbamazepine was included in the therapy, the average plasma phenytoin concentration decreased to 6.56 μg/ml in women and 5.96 μg/ml in men. However, decrease in the average plasma phenytoin concentration was not significant in men or women. When valproic acid was included in phenytoin therapy, the average phenytoin plasma concentration was 6.93 μg/ml in women and 6.42 μg/ml in men. Similar to previous results, the average phenytoin plasma concentration did not change significantly with the inclusion of valproic acid in the therapy, in men or women (Figure 5).

The mean plasma concentration of phenytoin in female and male patients treated with phenytoin monotherapy and combination therapy. PHEN: patients treated with phenytoin alone; PHEN + CARB: patients treated with phenytoin–carbamazepine combination therapy; PHEN + VA: patients treated with phenytoin–valproic acid combination therapy; PHEN: patients treated with phenytoin alone; PHEN + CARB: patients treated with phenytoin–carbamazepine combination therapy; PHEN + VA: patients treated with phenytoin–valproic acid combination therapy. *Different from PHEN-W (p < 0.05).
The average plasma valproic acid concentration in patients receiving only valproic acid was 71.03 μg/ml in women and 68.73 μg/ml in men, and the difference between men and women in this regard was not found to be statistically significant. The average plasma valproic acid concentration was 38.91 μg/ml in women and 48.57 μg/ml in men when phenytoin was included in the therapy. It was detected that phenytoin combination therapy decreased the plasma valproic acid concentration significantly below the therapeutic index. When carbamazepine was included in the therapy, the average plasma valproic acid concentration was 59.21 μg/ml in women and 62.51 μg/ml in men. In women, the average plasma valproic acid concentration was found to decrease significantly with carbamazepine–valproic acid combination therapy. In men, the decrease in the average plasma valproic acid concentration was not statistically significant (Figure 6).

The mean plasma concentration of valproic acid in female and male patients treated with valproic acid monotherapy and combination therapy. VA: patients treated with valproic acid alone; VA + PHEN: patients treated with valproic acid–phenytoin combination therapy; VA + CARB: patients treated with valproic acid–carbamazepine combination therapy; VA: patients treated with valproic acid alone; VA + PHEN: patients treated with valproic acid–phenytoin combination therapy; VA + CARB: patients treated with valproic acid–carbamazepine combination therapy. ***Different from VA-W (p < 0.01); +++Different from VA-M (p < 0.01).
The average plasma concentrations of the patients receiving carbamazepine monotherapy were 5.33 μg/ml in 18–28 age group of patients, 5.45 μg/ml in 29–38 age group, 5.53 μg/ml in 39–48 age group, 5.02 μg/ml in 49–58 age group, and 5.50 μg/ml in 59–65 age group. No significant difference was observed among the decades. The average plasma concentrations of the patients receiving phenytoin monotherapy were 6.67 μg/ml in 18–28 age group, 6.79 μg/ml in 29–38 age group, 6.04 μg/ml in 39–48 age group, 6.34 μg/ml in 49–58 age group, and 6.50 μg/ml in 59–65 age group. The average phenytoin plasma concentration did not show any significant differences among age groups. The average plasma concentrations of the patients receiving valproic acid monotherapy were 71.84 μg/ml in 18–28 age group, 70.95 μg/ml in 29–38 age group, 67.70 μg/ml in 39–48 age group, 66.02 μg/ml in 49–58 age group, and 64.94 μg/ml in 59–65 age group. In all the age groups, these differences were not found to be statistically significant. Average plasma concentration of patients receiving monotherapy and combination drug therapy according to age is shown in Table 4.
The mean plasma carbamazepine, phenytoin, and valproic acid concentrations of patients treated with monotherapy and combination therapy according to agea

+CARB: in addition to carbamazepine group; +PHEN: in addition to phenytoin group; +VA: in addition to valproic acid group; NS: no significant difference was observed among groups.
a Results are expressed as average ± standard error.
Discussion
Monotherapy is preferred to combination therapy because of the increased possibility of drug–drug interactions and the strong side effects associated with combination drug therapy at the onset of medication in patients with epilepsy. In addition, it has been pointed out in previous studies that monotherapy is sufficient in many patients. Combination therapy has been provided to patients with chronic epilepsy who do not respond to monotherapy. 6 In the previous studies, it was shown that 19–24% of epilepsy patients received two or more antiepileptic drugs simultaneously. 1
Therapeutic drug monitoring enables the regulation of drug dosage and serum drug concentration depending on the clinical effects 1,8,9 and allows for continuity of appropriate drug blood concentration. 1,8 Complex kinetic characteristics of antiepileptic drugs lead to interactions between drugs; these interactions can reduce the effect of the drug in therapy and/or produce toxic effects. Hence, the monitoring of drug plasma concentrations in patients with epilepsy has considerable advantages in therapy. 10 Plasma concentrations of drugs are monitored at regular intervals to evaluate therapy efficiency in patients with epilepsy, especially in patients who receive two or more antiepileptic drugs simultaneously. 1,11
According to our study results, it was found that the average plasma carbamazepine (3.39 ± 0.27 μg/ml; Figure 1) and valproic acid (44.44 ± 2.51 μg/ml; Figure 3) concentrations decreased below the therapeutic index, and the decrease was significant when administered in combination with phenytoin. The average plasma carbamazepine concentration decreased when administered in combination with phenytoin; this observation was similar to that of the other studies. 12,13 The average plasma concentration of both phenytoin and valproic acid was decreased when administered in combination with carbamazepine in patients with epilepsy. However, this decrease was not statistically significant for phenytoin. The decrease in the plasma valproic acid concentration when administered in combination with carbamazepine in patients with epilepsy was also observed in another study. 13 Combination therapy with valproic acid had no significant difference in the average plasma phenytoin and carbamazepine concentrations. Similarly, in another study, the average plasma carbamazepine concentration was not affected when administered in combination with valproic acid. 12
It is known that carbamazepine decomposes into its metabolites in the presence of cyctochrome P 450 (CYP), epoxide hydrolase, and uridine diphosphate (UDP) glucuronide transferase enzymes. 14 CYP3A4, 15 CYP1A2, and CYP2C8 are the cytochrome enzymes responsible for carbamazepine metabolism. 14,16 CYP2C9 17 and CYP2C19 14,5,18 enzymes have a role in phenytoin metabolism. UDP glucuronidation followed by β oxidation and cytochrome-mediated oxidation reactions results in the decomposition of valproic acid into its metabolites. CYP2B, CYP2C9, and CYP2A6 are the cytochrome enzymes responsible for the oxidation of valproic acid. 14,19,20
Phenytoin can cause a decrease in the plasma concentration and therapy effectiveness of carbamazepine and valproic acid by inducing CYP3A4, CYP2C9, CYP2C19, and CYP1A2 enzyme activities and by accelerating carbamazepine and valproic acid metabolism. Furthermore, because of an increase in the level of hepatotoxic metabolites due to the metabolism of antiepileptics, hepatic toxicity induced by carbamazepine and valproic acid can increase by enzyme induction. 2 In our study, it was shown that the decrease in the average plasma carbamazepine and valproic acid levels below the therapeutic index was likely to be related to the induction of enzymes responsible for carbamazepine and valproic acid metabolism, as shown in other studies.
Carbamazepine accelerates the metabolism of other drugs by inducing CYP3A4, CYP2C9, and CYP2C19 enzyme activities; moreover it induces its own metabolism (auto-induction) by inducing CYP3A4 enzyme activity. 1,2 Similarly, we observed a decrease in phenytoin and valproic acid concentrations in patients with epilepsy, in our study. However, this value was close to the sublimit of the therapeutic index for valproic acid (60.96 μg/ml) and below the therapeutic index for phenytoin. The observed decrease in the average plasma phenytoin and valproic acid concentrations when administered in combination with carbamazepine was not clear. In previous studies in which patients received combination therapy of phenytoin and carbamazepine, both of which cause enzyme induction, the decrease in the carbamazepine plasma concentration was maintained. 1,2 In our study, this evidence was supported by a significant decrease in plasma carbamazepine concentration. Furthermore, hepatic toxicity induced by valproic acid can increase by enzyme induction.
Valproic acid may also inhibit CYP2C9 activity in vivo especially in patients requiring high doses of valproic acid. Although valproic acid is a weak inhibitor of CYP3A4, the possibility of inhibition of the intestinal enzyme cannot be completely excluded. 1,2,21,22 With the absence of an inhibitory effect on CYP1A2, CYP2D6, and CYP2E1 activities, and weak inhibition of CYP2A6 and CYP2C19 23 activities, valproic acid is unlikely to produce clinically relevant interactions by inhibiting these CYP isoforms. 21 The use of drugs metabolized by these enzymes and valproic acid would lead to the deceleration of drug metabolism and increase in plasma concentration. 1,2 On the other hand, from our results, we cannot suggest that valproic acid combination therapy has a distinct effect on the plasma concentration of drugs used with valproic acid. Such evidence for carbamazepine was supported by a study by Fukuoka et al. 12
A review of the comparative inducing potency between carbamazepine and phenytoin determined that these agents exhibit approximately the same degree of enzyme induction. Also other studies are suggesting that phenytoin seems to be a more potent inducer than carbamazepine. 22 In our patients with carbamazepine phenytoin combination therapy, carbamazepine plasma concentration was decreased significantly according to carbamazepine monotherapy. However phenytoin plasma concentration was not decreased significantly in patients with carbamazepine phenytoin combination therapy according to monotherapy. Our result showed that phenytoin is a more potent inducer than carbamazepine. Clinicians should closely monitor the efficacy and toxicity of drugs considered to be sensitive to induction or to have a narrow therapeutic ratio when given concomitantly with enzyme inducers, especially during long-term or chronic therapy. 22
Our study results pointed out that the average plasma concentration decreased below the therapeutic index in patients with epilepsy who received monotherapy and combination therapy with phenytoin (Figure 2). This finding shows the need for routine observation of patients receiving phenytoin in order to evaluate the success and continuity of the therapy. In another study, an interesting result was that the plasma phenytoin concentration shows a wide range of scale. 24 In light of this, we can say that it is quite important to monitor the progress of patients, especially of those patients receiving phenytoin medication, so that the phenytoin dosage can be regulated and it can be ensured that the dose administered is appropriate and sufficient.
When we evaluated the influence of gender on the plasma antiepileptic concentration, we found that the average plasma carbamazepine concentration (Figure 4) was higher in men and the average plasma phenytoin (Figure 5) and valproic acid concentrations (Figure 6) were higher in women. The difference between men and women in this regard was statistically significant for phenytoin but was not significant for valproic acid and carbamazepine. Studies on molecular basis draw attention to the fact that gender is an important parameter to be considered in drug pharmacokinetics as it can influence drug efficiency, just as genetic and environmental factors. 14,25 The determining parameters of drug pharmacokinetics, such as body weight, plasma volume, gastric emptying, cytochrome P450 enzyme activity, and the amount of carrier protein, differ between men and women. 25 –27
Differences in the average plasma concentration of valproic acid, carbamazepine, and phenytoin between men and women can be explained by considering different enzyme activities responsible for the metabolism of these drugs. The enzyme activity of CYP2C9 and CYP2C19, responsible for the metabolism of phenytoin, is higher in men than in women. 17,28 Similarly, the UDP glucuronidation pathway, which is responsible for 50% of valproic acid metabolism, is faster in men than in women. 14,17,26 In conclusion, the activity of enzymes responsible for the metabolism of phenytoin and valproic acid is higher in men than in women, because of which the metabolism of these two drugs is slower in women; hence, it can be thought that the plasma concentration of these two drugs is higher in women. The activity of the CYP3A4 enzyme, which is responsible for carbamazepine metabolism, is higher in women than in men. 17,26,28
For this reason, carbamazepine is cleaved into its metabolites at a faster rate, and because of this the plasma carbamazepine concentration is lower in women than in men. However, we should bear in mind that the difference in enzyme activity between men and women is not the only marker of difference in average plasma antiepileptic drug concentrations between men and women. All parameters affecting pharmacokinetic drug features such as plasma volume and quantity of plasma proteins also lead to this difference. In addition, age, cigarette smoking, alcohol abuse, and features specific to a particular sex (menopause, menstruation, and pregnancy) can also affect the drug metabolism. 17,28 Furthermore, individual enzyme polymorphisms can be another parameter influencing the drug metabolism and plasma concentrations. 14,18
The significant decrease in the average plasma carbamazepine and valproic acid concentrations during phenytoin combination therapy was thought to be a noteworthy result in the evaluation of the influence of gender on combination therapy. During carbamazepine combination therapy, the average plasma phenytoin and valproic acid concentrations significantly decreased in female patients. It was observed that valproic acid combination therapy had no effect on plasma phenytoin and carbamazepine concentrations in men or women. On the basis of the significant decrease in the average plasma carbamazepine and valproic acid concentrations in phenytoin combination therapy and the decrease in the average plasma valproic acid concentration in carbamazepine combination therapy in women, we can interpret that women show high sensitivity against enzyme induction.
Age-related changes may affect the pharmacokinetic parameters, efficacy and tolerability of drugs. 29 Particularly, differences may also occur in hepatic enzymes and metabolizing capacity during life span. Dosage adjustments in order to prevent poisonings and adverse reactions can be necessary in some cases because of the variation in these parameters. 30 In our study, when the effects of age on the plasma concentrations of drugs were evaluated, no significant difference was observed. This observation may arise from excluding pediatric and geriatric groups from our study. The patients in our study were only the members of the adult group. The metabolization of these drugs showed differences in the elderly (age 65 and above) 31 –34 or in childhood (age 18 and below), in some studies. 30,35,36 However, there is likely to be no difference between different age groups in other studies. 29,37 –41 In addition, activity or the content of specific enzymes responsible for the metabolism of these drugs may not be influenced by age. 30,42,43 As a result, comparing the plasma antiepileptic drug levels in the age group between 18 and 65 (in decades) we concluded that metabolism of these drugs showed similar rate and quantity in this age range.
In conclusion, our study measured the average plasma antiepileptic drug concentrations in patients with epilepsy who were receiving monotherapy and combination therapy and were routinely monitored, and has thus shown the importance of drug monitoring in the evaluation of the effectiveness of these drugs. In epilepsy treatment, when drugs with drug–drug interaction potentials are combined with each other, therapeutic monitoring should be routinely performed in order to evaluate the possible toxic effects of drugs and their treatment impact. It should always be remembered that a combination of carbamazepine and valproic acid produce toxic metabolites with enzyme-inducing drugs such as phenytoin, which in turn produce serious toxic effects. In addition, the effective phenytoin dosage for therapy should be regulated by routine monitoring of the plasma levels.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
