Abstract
The aim of this study was to investigate the possible effects of ivabradine against doxorubicin (DOX)-induced cardiotoxicity in rats using hemodynamic parameters (electrocardiogram, heart rate (HR), and blood pressure), biochemical markers of oxidative stress, lactate dehydrogenase, aspartate transaminase, creatine kinase-MB, and histopathological analyses both in serum and tissue specimens. A total of 28 female rats were randomly assigned to 4 groups: (a) control (n = 6 rats), (b) DOX group (n = 7 rats), (c) DOX + ivabradine–treated group (n = 8 rats), and (d) ivabradine group (n = 7 rats). When the means of the four groups were compared, there was only a significant difference in the level of HR (p < 0.05). DOX treatment caused more HR elevation when compared to the control group, whereas ivabradine application after DOX treatment significantly reduced HR levels. Cardiomyocytes were revealed as normal histology in the light of both hematoxylin and eosin staining and immunostaining methods (caspase-3 and bcl-2) in all groups. The present study reported the therapeutic effects of ivabradine against DOX-induced cardiotoxicity accompanied by the hemodynamic and biochemical parameters.
Introduction
Doxorubicin (DOX), a member of the quinone-containing anthracycline class of cytostatic antibiotics, is a potent and broad-spectrum chemotherapeutic agent. 1 Its clinical use is seriously limited by its cardiotoxicity. 2 The drug’s toxicity is known to be closely related to the generation of reactive oxygen free radicals (reactive oxygen species), lipid peroxidation, and decreased glutathione (GSH) levels. 3 Increased oxidative stress and antioxidant deficits have been suggested to play a major role in DOX-induced cardiotoxicity. 4 Rossi et al. 5 showed that DOX treatment caused an increase in mean blood pressure (BP) in an experimental model. This important finding may bear relevance to the pathogenesis and therapy of DOX-induced cardiotoxicity.
This hypothesis is also consistent with the findings of the studies, suggesting that elevated heart rate (HR) represents a risk factor for cardiovascular morbidity and mortality both in primary prevention and in patients with hypertension, coronary artery disease, and myocardial infarction. 6 HR variability has been shown to be associated with coronary plaque ruptures and subclinical inflammation in healthy middle-aged and elderly patients. 7,8 Ivabradine, a HR-lowering agent, acts in the sinus node by selective and specific inhibition of the cardiac pacemaker If current that controls the spontaneous diastolic depolarization in the sinus node with no effect on myocardial contractility. 9 Custodis et al. 6 reported that HR reduction by ivabradine reduced oxidative damage and thereby improved endothelial dysfunction. Furthermore, Heusch 10 declared that ivabradine exerted pleitrophic action beyond HR reduction. Many studies and clinical applications of ivabradine belong to the cardiology area and are particularly related to the symptomatic treatment of stable angina pectoris in patients with normal sinus rhythm who have a contraindication to or intolerance to beta blockers and heart failure due to left ventricular systolic dysfunction for its pure pharmacodynamic properties.
All of the investigations mentioned above motivated us to plan this experimental design for the first time. The aim of the present study was to investigate the possible effects of ivabradine against DOX-induced cardiotoxicity in rats using biochemical markers of oxidative stress, lactate dehydrogenase (LDH), aspartate transaminase (AST), creatine kinase (CK), and histopathological analyses both in the serum and in tissue specimens.
Material and methods
Animals and groups
For this study, a total of 28 female Wistar Albino rats of 10–12 weeks of age and weighing 250–300 g were obtained from Inonu University Laboratory Animals Research Center and placed in a temperature (21 ± 2°C) and humidity (60 ± 5%) controlled room in which a 12:12-h light:dark cycle was maintained. The rats were randomly assigned to four groups as follows: (a) control (n = 6 rats), (b) DOX group; injected intraperitoneally (i.p.) with DOX (Doxo-Teva®, Med-Ilac, Istanbul, Turkey) 20 mg/kg in a single dose (n = 7 rats), (c) DOX + ivabradine-treated group; injected i.p. in a single dose of 20 mg/kg DOX plus 10 mg/kg Ivabradine 1 h before the DOX treatment and continued for 10 days by oro-gastric canule (n = 8 rats), (d) ivabradine group; applied by oro-gastric canule for 10 days (n = 7 rats). Ivabradine (Coralan®, Servier Corp, Istanbul, Turkey) was dissolved in 0.5 cm3 of saline (0.09% NaCl) solution and the control group received the same amount of saline by the same route.
The applied dosage of DOX was chosen according to the related literature from our research team’s previous research. 11 In these studies, ivabradine was dissolved in saline to obtain a final concentration of 10 mg/ml. Because of the highly variable ivabradine dosage schemes reported in the literature, we administrated ivabradine at a dose of 10 mg/kg/day po, which has been reported to cause marked antioxidative and endothelial improving effects. 6,7
The rats in all the groups were fed with a standard chow pellet diet with tap water ad libitum. All experiments in this study were performed in accordance with the Guidelines for Animal Research from the National Institutes of Health and were approved by the Committee on Animal Research at Inonu University, Malatya, Turkey.
Blood pressure, HR, and electrocardiographic records
Twenty-four hours after the last injection, which was chosen according to our previous DOX-related study, 1 all rats were first anesthetized with a reversible ketamine hydrochloride (75 mg/kg) and xylazine (8 mg/kg) combination i.p. for determining the hemodynamic parameters. Systemic BP (systolic and diastolic) and HR were monitored and recorded during a period of 5 min from the cannulated-carotid artery by a BIOPAC MP-100 A-CE data acquisition system (Goleta, California), as shown in Figure 1. In addition, electrocardiographic (ECG) signal activity was recorded for at least 5 min with a sampling frequency of 500 Hz under anesthesia using disposable electrodes attached to the rat’s thorax. Next, the ECG traces were visually analyzed by two clinician experts for assessing HR, ventricular ectopic beats (VEB), ventricular tachycardia (VT), ventricular fibrillation, ST depression, QRS enlargement, and major ECG anomalies such as atrioventricular blocks or branch blocks.
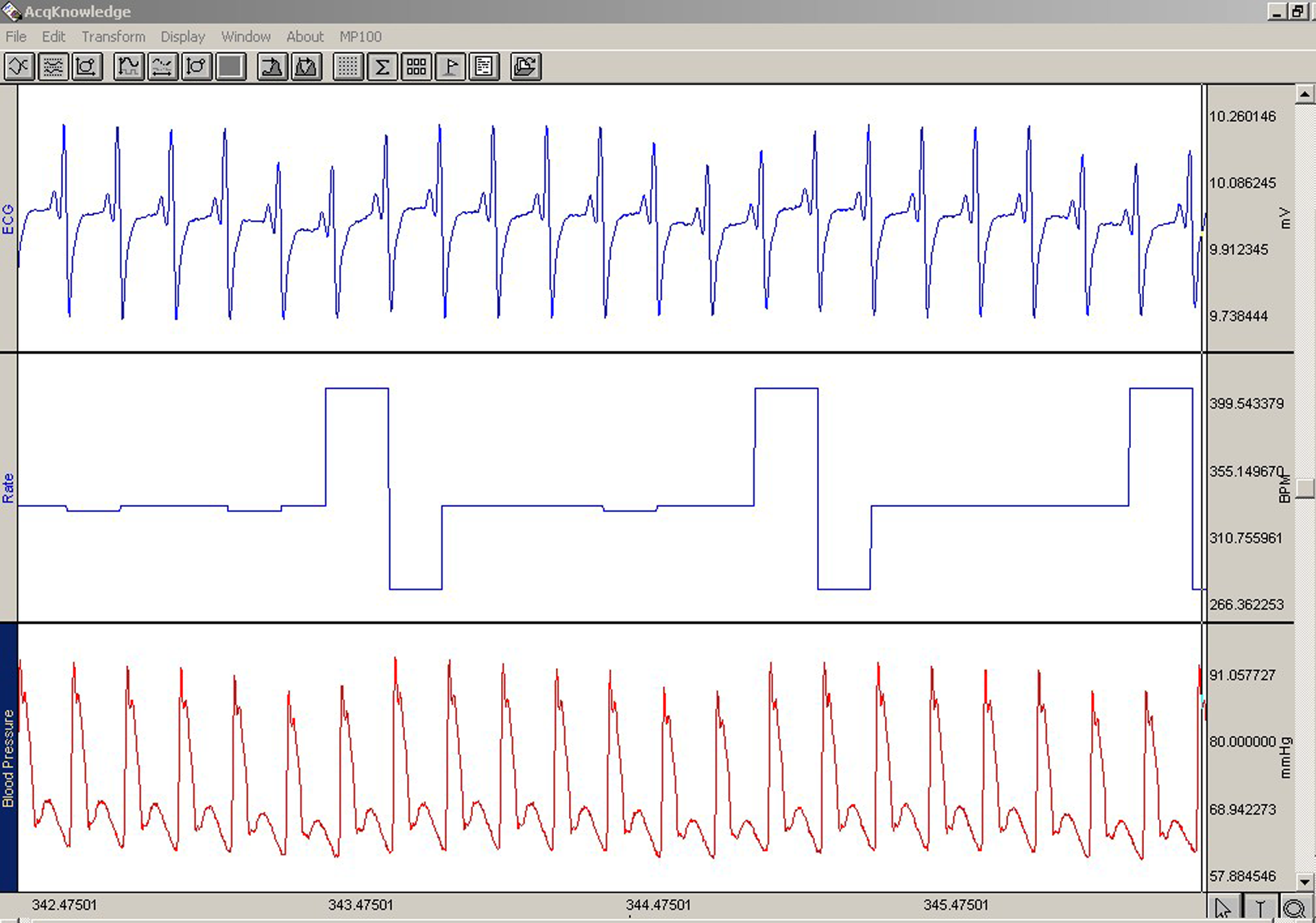
Example of measured electrocardiography (ECG), heart rate, and blood pressure signals through BIOPAC MP-100 A-CE data acquisition system.
Biochemical analyses
After the records were successfully completed in all 28 rats, they were weighed and then killed by an overdose of anesthesia. The hearts were quickly removed and divided equally into two longitudinal sections. One of these parts was placed in formaldehyde solution for a routine histopathological examination by the light microscopy. The other half was frozen in the liquid nitrogen and then stored at −70°C until assayed for thiobarbituric acid reactive substances (TBARS), a lipid peroxidation product, superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) contents. Trunk blood was collected from the inferior vena cava to determine the serum levels of LDH, AST, and CK enzymes-MB enzymes, which were determined using an Olympus Autoanalyser (Olympus Instruments, Tokyo, Japan).
Determination of serum TBARS
The TBARS contents of homogenates were determined by thiobarbituric acid reaction spectrophotometrically. 12 Three milliliters of 1% phosphoric acid and 1 ml 0.6% thiobarbituric acid solution were added to 0.5 ml of plasma pipetted into a tube. The mixture was heated in boiling water for 45 min. After the mixture had cooled, the color was extracted into 4 ml of n-butanol. The absorbance was measured by a spectrophotometer (UV-1601; Shimadzu, Kyoto, Japan) at 532 nm. The amount of lipid peroxides was calculated as TBARS of lipid peroxidation. The results were expressed in nanomoles per gram (nmol/g tissue) according to a standard graph that was prepared from the measurements of standard solutions (1,1,3,3-tetramethoxypropane).
Determination of SOD
Total (Cu–Zn and Mn) SOD (EC 1.15.1.1) activity was determined based on the method of Sun et al. 13 The principle of the method is the inhibition of nitroblue tetrazolium (NBT) reduction by the xanthine–xanthine oxidase system as a superoxide (O2 •−) generator. One unit of SOD was defined as the enzyme amount causing 50% inhibition in the NBT reduction rate. SOD activity was expressed as units per milligram protein (U/mg protein).
Determination of CAT
CAT (EC 1.11.1.6) activity was determined with respect to Aebi’s method. 14 The principle of the assay is based on the determination of the rate constant (k, s−1) or the H2O2 decomposition rate at 240 nm. Results are expressed as k per gram protein (k/g protein).
Determination of GSH-Px
Determination of GSH-Px activity (EC 1.6.4.2) was measured by the method of Paglia and Valentine. 15 An enzymatic reaction in a tube containing nicotinamide adenine dinucleotide phosphate (NADPH), reduced GSH, sodium azide, and glutathione reductase was initiated with the addition of H2O2, and the change in absorbance at 340 nm was monitored by a spectrophotometer. Activity was given in units per gram protein (U/g protein).
Histological examination
For the light microscopic evaluation, the portions of each heart were fixed in 10% of a neutral phosphate buffered formalin solution. Following dehydration in an ascending series of ethanol solutions (70, 80, 96, and 100%), the tissue samples were cleared in xylene and embedded in paraffin. The tissue sections of 6 μm were stained with hematoxylin–eosin (H&E) and examined by an observer blind to the treatment. A pathologist reviewed the H&E stained sections from each specimen to confirm the initial tissue diagnoses. For immunohistochemistry, following steps were applied to the slides: same sequence was followed until microwave treatment. The thick sections were taken on to the polylysine-coated slides. After rehydrating, the samples were transferred to citrate buffer (pH 7.6) and heated in a microwave oven for 20 min. After cooling for 20 min at room temperature, the sections were washed with phosphate-buffered saline (PBS). Then, the sections were kept in 0.3% H2O2 for 7 min, and subsequently washed with PBS. The sections were incubated with primary rabbit-polyclonal cysteine aspartate specific proteinase (caspase-3: Labvision), a key effector protein of the apoptotic machinery. Caspase-3 kit was used according to the manufacturer’s instructions with minor revisions. For the other immunohistochemical staining method, bcl-2 monoclonal antibody (a marker protein of apoptosis) was used according to the manufacturer’s instructions. Antigen retrieval was by steam treatment in a citrate buffer and the sections were quenched for 10 min in hydrogen peroxide. The slides were coated with primary antibody, incubated at room temperature for 30 min. The antibody was detected with a biotin–streptavidin detection system (Bio-Genex, San Ramon California, USA). The sections were examined by a Leica DFC 280 light microscope by a histopathologist unaware of the status of animals. Amino-etilkarbazol (AEC) was used for kromogen system (LabVision Corporation, Union City, CA, ABD).
Statistics
The normal distribution was explored with Shapiro Wilk test. Homogeneity of variances was tested by the Levene’s method. In the analyses, one-way analysis of variance and Kruskal-Wallis H tests were used to compare the four groups. Multiple comparisons were made by Tukey and Tamhane T2 tests and Bonferroni correction with Mann Whitney U test where appropriate. p < 0.05 was considered as significant.
Results
Body and heart weights
There was no difference between the body weights before and after the experiments among the groups. The heart weights of the DOX-treated rats were significantly higher when compared to the control group, whereas the DOX + ivabradine application reduced this finding to the control levels (data not shown).
Hemodynamic parameters
Table 1 indicates the mean arterial BP (mm Hg) and HR (beats min−1) values, which were measured at the end of the experiment. Based on these points, significant differences among the groups in the BP values were not detected. When the means of the four groups were compared, there was only a significant difference in the level of HR (p < 0.05). DOX treatment caused more HR elevation when compared to the control group, whereas ivabradine application after DOX treatment significantly reduced HR levels (p < 0.05).
Hemodynamic parameters

HR: heart rate; DOX: doxorubicin; SBP: systolic blood pressure; DSB: diastolic blood pressure.
a Significant difference as compared with DOX group.
b Significant difference as compared with control group.
c Significant difference as compared with DOX + ivabradine group.
d One-way analysis of variance test.
ECG analysis
The ECG analysis was performed in the computerized assessment (Figure 1). At the end of the experiment day, ECG data were visually evaluated by two specialist clinicians who were blind to the status of the animals. Although they have observed minor ECG changes such as ST depression, QRS enlargement, VEB, and VT at some periods of ECG traces in some subjects, they did not consider these arrhythmias as significant and specific to any group to warrant the claim that the DOX treatment might cause a heart disorder.
Effect of ivabradine on DOX-induced changes in the heart tissue enzymes and lipid peroxides
The results are summarized in Table 2. In brief, a single dose of DOX treatment caused a significant increase in the TBARS levels (37.62 ± 6.25 nmol/g tissue) compared with the control group (27.12 ± 5.58 nmol/g tissue) in the heart tissue. However, ivabradine administration could not reduce this lipid peroxidation (33.49 ± 3.77 nmol/g tissue). Further, DOX treatment caused a decrease in the SOD (0.14 ± 0.03 U/mg prot) and CAT (0.18 ± 0.02 K/g protein) enzyme activities, whereas ivabradine therapy tended to ameliorate them, nearly to the level of the controls (0.23 ± 0.06 U/mg prot, 0.20 ± 0.06 K/g prot vs. 0.22 ± 0.05 U/mg prot and 0.23 ± 0.03 K/g prot, respectively). In addition, GSH-Px levels were found to be significantly increased in the DOX-treated group (3.23 ± 0.85 U/mg prot) when compared to the control group (1.82 ± 0.2 U/mg prot). Ivabradine administration significantly decreased this elevated antioxidant enzyme activity (2.07 ± 0.53 U/mg prot).
Effects of doxorubicin (DOX) and/or ivabradine administration on the heart tissue enzymes and lipid peroxides

CAT: catalase; GSH: glutathione; GSH-Px: glutathione peroxidase; MDA: malondialdehyde; SOD: superoxide dismutase. * One way analysis of variance test
a Significant difference as compared with control group.
b Significant difference as compared with DOX group.
c Significant difference as compared with DOX + ivabradine group.
Serum LDH, AST, and CK levels
Table 3 presents the results of LDH, AST, and CK levels. In sum, DOX treatment caused a significant increase in the LDH (1910 ± 225.9 IU/L) and CK (1515.28 ± 793.1 IU/L) levels when compared to the control group (807 ± 308.54 IU/L and 805.5 ± 314.9 IU/L, respectively) in the heart tissue. Ivabradine therapy statistically decreased these elevations (849.87 ± 154 IU/L and 813.87 ± 248.68 IU/L, respectively). Moreover, DOX application significantly elevated the AST levels when compared to the control groups, whereas ivabradine treatment cold not reduce AST levels.
Effects of doxorubicin (DOX) and/or ivabradine administration on serum enzymes

AST: aspartate transaminase; CK: creatine kinase; LDH: lactate dehydrogenase.
a Significant difference as compared with DOX group.
b Significant difference as compared with DOX + ivabradine group.
c Significant difference as compared with control group.
d One-way analysis of variance test.
e Kruskal-Wallis H test.
Histological results
The cardiomyocytes were revealed as normal histology in the light photographs of both H&E staining (Figure 2) and immunostaining methods (caspase-3 (Figures 3 and 4) and bcl-2 (Figure 5)) in the all groups.
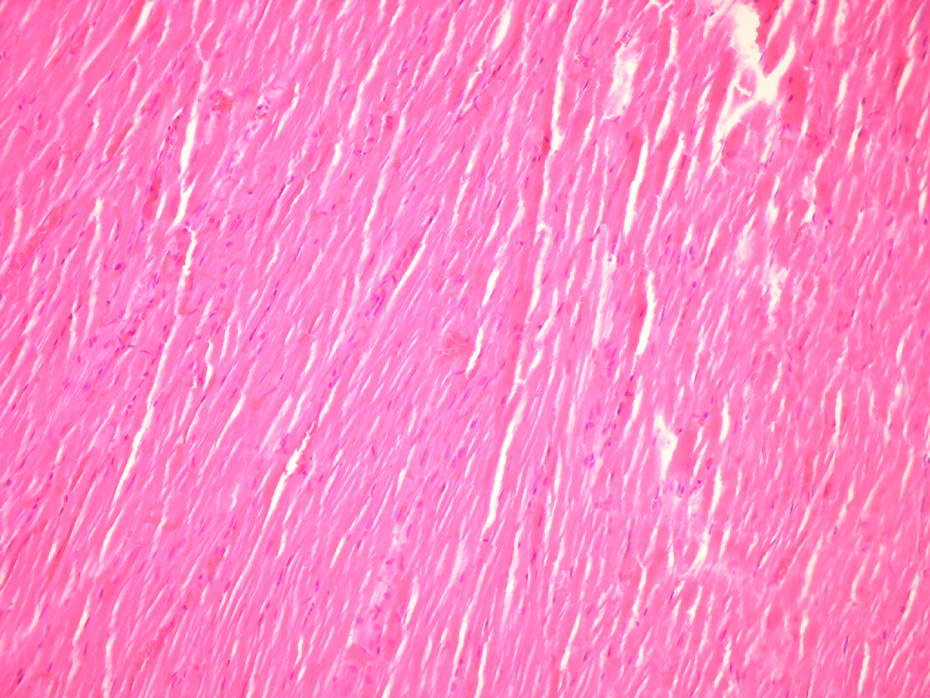
Cardiomyocytes revealed as normal in the all groups (hematoxylin–eosin (H&E) ×200).

Doxorubicin (DOX)-treated rats: immunohistochemical staining of the rat heart. Cardiomyocytes show negative caspase-3 staining in all the groups (Amino-etilkar bazol ×400).

Control group: the view of apoptotic cells that belong to the germinal center of lymph node which was stained with caspase-3 (Amino-etilkarbazol × 400) as negative control.

Light micrograph of the doxorubicin (DOX)-treated group; cardiomyocytes show negative bcl-2 staining in all the groups (Amino-etilkarbazol ×400).
Discussion
DOX is one of the most effective chemotherapeutic agents in a variety of cancer types; however, its usefulness is seriously limited by the risk of developing heart failure. Available laboratory evidence suggests that an increase in the oxidative stress caused by elevated free radical formation and decreased myocardial endogenous antioxidant enzymes play an important role in heart failure. 16 –18 Two different mechanisms of free radical formation by DOX have been described. The first mechanism implicates the formation of a semiquinone free radical by the action of several NADPH-dependent reductases. In the presence of oxygen, redox cycling of DOX-derived quinine-semiquinone yields O2 •− radicals. In the second mechanism, DOX-related free radicals are produced by a nonenzymatic mechanism that involves a reaction with iron. Iron-DOX complex can reduce oxygen to H2O2 and other active oxygen forms. 19,20
Apart from these mechanisms, another explanation for DOX-induced cardiomyopathy is a dose-dependent process with cumulative myocyte damage. During myocyte injury development, there is an overall increase in the adrenergic nervous system activity. 21 Therefore, Rabelo et al. 22 reported that basal tachycardia was observed in DOX-treated rats in relation to control rats. In addition, Venkatesan 23 declared that animals receiving DOX had an increase in HR compared to control groups. In the literature, there have been no reports regarding ivabradine, a specific inhibitor of the cardiac sinoatrial node, against DOX-induced cardiotoxicity. For this reason, the current study was undertaken to address the question as to whether inhibition of cardiac sinoatrial node by ivabradine exerted the reduced susceptibility to DOX-induced heart injury at least, in an experimental study.
The main findings of the current study were as follows: (1) we demonstrated in accordance with the above-mentioned literature that DOX treatment resulted in more significant HR elevation when compared to the control group, whereas ivabradine application after DOX treatment significantly reduced HR levels. (2) Among all groups, there were no differences in BP values. It is well established that elevated HR increases myocardial oxygen consumption 24 in normal myocardium, which is matched by increased coronary blood flow 25 through metabolic coronary vasodilatation. 26 Therefore, a slight increase in myocardial oxygen extraction helps to match oxygen supply with oxygen demand during tachycardia. 27 Therefore, ivabradine has an important protective role against HR elevation by If-channel inhibition. For this reason, many studies and clinical applications of ivabradine have relevance to the field of cardiology. This is particularly true for the symptomatic treatment of stable angina pectoris in patients with normal sinus rhythm who have a contraindication to or intolerance to beta blockers and heart failure due to left ventricular systolic dysfunction for its pure pharmacodynamic properties, which is a specific inhibitor of the cardiac sinoatrial node. 28 –31 Recently, Zhang et al. 32 declared that HR reduction therapy following myocardial infarction, using ivabradine, was shown to preserve maximal coronary perfusion via the reduction in perivascular collagen and caused a decrease in renin–angiotensin system activation. In addition, they proposed that HR reduction by ivabradine facilitated a more favorable O2 microenvironment via improved venous flow and decreased O2 demand.
Many studies and clinical applications of ivabradine belong to the cardiology field, especially related to the symptomatic treatment of stable angina pectoris in patients with normal sinus rhythm who have a contraindication or intolerance to beta blockers and heart failure due to left ventricular systolic dysfunction for its pure pharmacodynamic properties, which is a specific inhibitor of the cardiac sinoatrial node.
Additionally, it is well established that ivabradine exerts anti-anginal effects in preclinical and clinical trials. 6 The If-channel inhibitor ivabradine was studied exclusively due to its specific effects on the sinus node with no influence on myocardial contractility and relaxation. 25 HR reduction has been demonstrated to ameliorate atherosclerosis and vascular inflammation, with an improvement in cardiovascular outcome. 9 Recently, ivabradine treatment was shown to improve endothelial function, to benefit ischemic myocardial blood flow and contractile function, as well as to reduce infarct size and post-infarct remode. 10 In accordance with our hemodynamic results, in this study, male apolipoprotein E-deficient mice were fed a high-cholesterol diet and were treated with ivabradine (10 mg/kg/d) or vehicle for 6 weeks (n = 10 per group). As a result, ivabradine reduced HR by 13.4% (472 ± 9 bpm versus 545 ± 11 bpm; p = 0.01) but did not alter the BP or lipid levels. There are many articles related to ivabradine’s protective effects against experimental cardiac ischemia. Based on this relationship, Drouin et al. 33 demonstrated that chronic HR reduction by ivabradine significantly improved endothelium-dependent vasodilatation to acetylcholine, and they suggested that these beneficial effects of ivabradine result secondarily in the lowering of HR. Parallel to these results, more recently, Custodis et al. 6 have reported that ivabradine reduces oxidative stress, improves endothelial function, and prevents atherosclerosis. Moreover, Heusch 10 declared that ivabradine exerted pleitrophic actions beyond HR reduction. In accordance with this notion, more recently, Walcher et al. 34 have reported that ivabradine reduces chemokine-induced CD4-positive lymphocyte migration. Also, Baumhakel et al. 35 showed that ivabradine given to mice via chow at an oral dose of 10 mg/kg/d improves penile endothelial function by reducing oxidative stress and penile fibrosis. They also concluded that these beneficial effects resulted in the prevention of endothelial dysfunction. More recently, our another investigator team concluded that ivabradine exerts a dose-dependent response beyond heart reduction against renal ischemia/reperfusion (I/R) injury. Also they demonstrated that prophylactic administration of 0.6 mg/kg ivabradine protected the kidneys from I/R injury, whereas 6 mg/kg ivabradine did not show any beneficial effects on either antioxidative enzymes or serum blood urea nitrogen and creatinine levels. In explaining this surprising result, they suggested one possibility is that the higher dose of ivabradine (6 mg/kg) reduces HR in the rat, thereby causing a reduction in blood flow, including renal vessels. 36
The other main finding of the current study was as follows: ivabradine, a selective and specific inhibitor of the cardiac pacemaker If current, could have more therapeutic effects on the enzymatic defense system when compared to the DOX alone treated group in a rat model. In the current study, a single dose of DOX treatment caused a significant increase in the TBARS levels and caused a decrease in the SOD and CAT enzyme activities. Ivabradine administration could not reduce elevation of lipid peroxidation, whereas ivabradine therapy tended to ameliorate SOD and CAT enzymes to nearly the same level as controls. Oxygen radical-induced injury of membrane lipids is considered the most important factor responsible for the development of DOX-induced cardiotoxicity. 37 Lipid peroxides, which are the breakdown products of lipids, are measured in the tissues as an indictor of the lipid peroxidation. Ivabradine reduces the vascular NADPH oxidase activity to 48.6% and decreases markers of O2 •− production and lipid peroxidation in the aortic wall. 6 O2 •− generated by DOX is transformed to H2O2 by SOD and further detoxified by CAT or GSH-Px. 38 In the present study, GSH-Px levels were significantly increased in the DOX-treated group when compared to the control group. Ivabradine administration significantly decreased this elevated antioxidant enzyme activity. One of the possible explanations for these results is that GSH is the major intracellular antioxidant, and GSH redox cycle is a major defense system in cardiomyocytes against oxidative stress because the CAT level is low. Since DOX causes the depletion of cardiac GSH, 39 this may reflect the consistent formation of oxygen free radicals. 40 Therefore, GSH-Px enzyme activities may be increased in the acute stage using GSH substrate, which can be detected as reduced in the serum for suppressing the overexpression of oxygen radicals induced by DOX. Moreover, when the other antioxidant enzymes (SOD and CAT) are insufficient to protect the cell against free radical attacks, GSH-Px enzyme expression is stimulated for further defense systems. Ivabradine supplementation with DOX application enhanced the antioxidant status; therefore, GSH-Px levels were reduced in this group.
The last main finding of the current study was as follows: DOX treatment caused a significant increase in the LDH and CK levels when compared to the control group in the heart tissue. Ivabradine therapy statistically decreased these elevations. In accordance with our results, Iqbal et al. 41 reported that single-dose DOX (20 mg/kg) caused cardiotoxicity, as evidenced by the elevation of serum LDH activity apart from other parameters in rats. Similarly, Osman et al. 42 showed that DOX treatment showed a significant 2.6- and 3-fold increases in the cardiac CK and LDH enzyme activities in the serum of the animals, respectively.
DOX-induced morphological changes in the cardiac tissues were observed by light microscopy. Several studies have reported that DOX treatment induces significant morphological changes, while others have reported that there are no significant morphological changes until 5 days after acute treatment 43 or until 8 weeks after the beginning of long-term treatment. 41 According to our histopathological observation, the cardiomyocytes were revealed as having normal histology (Figure 2), whereas both staining methods (H&E and caspase-3/bcl-2) worked flawlessly (Figures 3 –5) in the all groups. In the literature, histological findings in DOX-induced cardiomyopathy include myofibrillar loss, myocyte vacuolization, apoptosis, and eventually the death of the myocyte. 14 However, we did not observe any differences among the groups including the immunohistochemical caspase-3 and bcl-2 examinations for apoptotic cell death.
In sum, this study is the first report regarding the therapeutic effects of ivabradine against DOX-induced cardiotoxicity accompanied by the hemodynamic, biochemical parameters, and histopathological observations. However, further experimental and clinical studies are required to confirm these findings before applying ivabradine clinically to treat DOX-related cardiac injuries.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflict of Interest
All authors declare that they have no conflicts of interest.
