Abstract
The aim of this study was to assess the influence of soy isoflavones, administered pre- and later postnatally, on the estrogen receptor α (ERα) and β (ERβ) expression in bones and to examine the mineral metabolism of the skeletal system in male rats. In bones, ERs were examined with an immunohistochemical method; in blood, estradiol with chemiluminescence immunoassay and in blood and bones, calcium and magnesium with atomic absorption spectrometry and fluorides with a potentiometric method were examined. Decreased immunoexpression of ERα and the increased intensity of immunofluorescence of ERβ in osteocytes in the femur of experimental rats were observed. In the serum of treated rats, a significantly higher concentration of estradiol and lower calcium were observed. The content of magnesium and fluoride were significantly higher in the bones of the examined animals. The data presented show that pre- and postnatal supplementation of male rats with soy isoflavones may considerably increase the concentration of estrogens in serum, with a concurrent effect on the mineral composition of bones.
Introduction
Phytoestrogens, present in the environment, may significantly influence the osseous system through mimicking, antagonizing and masking estrogen action. 1 They bind to estrogen receptors (ERs) due to the similarity of their chemical structure to endogenous estrogens. 2 The estrogenic activity of phytoestrogens is dependent on their interaction with ERα or ERβ. At nutritionally relevant doses, phytoestrogens may selectively interact with ERβ and thus affect only the expression of a subset of estrogen-responsive genes. 3,4 The differences between the respective N-terminal regions of the ERs have been suggested as a possible explanation for their diversity of responsiveness to several ligands. 4
Recent observations made of rare defects in ER sensitivity or estrogen synthesis have called attention to the importance of estrogens in the developing male skeleton. The affected men have demonstrated continuous linear skeletal growth, lack of pubertal growth spurt and reduced bone mass. 5 –7 The pubertal growth spurt coincides with the rise in the concentration of estradiol (E2). 8 E2, while inducing the pubertal spurt in linear growth, leads concurrently to the exhaustion of proliferation and to replicative senescence. This concept is consistent with the age dependency of the duration of estrogen exposure to induce epiphyseal fusion. The younger the child when exposed to estrogen, the longer the duration in years before disappearance of the growth plate. 9
Otherwise people exposed to phytoestrogens exhibit phytoestrogen concentrations that may be 20 times higher than that of endogenous estrogens. 2 The resultant elevated level of estrogens in blood serum may lead to a disturbed distribution of elements in the body. 10
Concentration of sex hormones is low in the blood serum of children. There are a number of studies demonstrating the placental transfer of phytoestrogens from mother to the fetus. 11 –13 It is suggested that the rates of transfer of phytoestrogens from the mother to the fetus and from the fetus to the mother are different. 12 ,14 Therefore, once phytoestrogens are transferred to the fetus, they tend to stay in the fetal side longer than in the maternal side. 12
Large amounts of potentially estrogenic substances are consumed by children during the period of their highest susceptibility, mainly in baby food. 11 –13,15 It has been reported that children feeding on isoflavone-based supplements have 1000 times greater estrogen concentration than in their breast-fed peers with mothers on soy diet. 16 Irvine et al. 14 have shown that children from 4 months are capable of digesting and assimilating isoflavones levels like adults. The hypothesis by Messina 17 has been that isoflavones exert antiestrogenic effects in high-estrogen conditions, such as in premenopausal women, and estrogenic effects when in low-estrogen conditions, such as in men and postmenopausal women. 18 Although there is no convincing proof that early exposure affects the later development of children, especially boys, the fact that infancy is a time of elevated sensitivity to estrogens 19 which may make the changes during sexual maturation more likely.
Phytoestrogens are the largest group of dietary xenoestrogens and are naturally present in plants. 20 ,21 Studies show that their estrogenic potential is lower than that of 17β-estradiol (E2) but, at a constant or long-term exposure, the effect of these compounds on health should not be underestimated. 2 Bearing this in mind, the aim of this article is to examine the effect of soy isoflavones with a proven estrogenic potential, administered pre- and postnatally, on the expression of ERs α and β, and on the mineral metabolism of the osseous system in mature male rats.
Materials and methods
Animals
Three-month-old female Wistar rats were bred with the male of the same strain. Pregnant rats (n = 12) were divided into two groups: control (n = 6) and experimental group (n = 6). They were housed separately in a room with controlled temperature (21–23°C) and lighting (12 h/12 h light/dark) and were allowed free access to food and water. During pregnancy and lactation, mothers in the experimental group received individually, per os, soy isoflavones (mixture of daidzein and genistein, at a dosage of 200 mg/kg body weight (b.w.)/day; Meno Stop—HASCO Lek, Poland, 75 mg of isoflavones sum or SoyaMeno—Terpol, Poland, 60 mg of isoflavones sum) from the first day of experiment until weaning and then during lactation. Soy isoflavones were given once a day (in the morning) individually to each rat in the form of pellet composed of isoflavones preparation mixed with humid regular rat chow. Each pellet contained the dose of isoflavones, accurately to the b.w. of rat because before preparing the pellet, the powder was weighed and then mixed with the humid chow. The females of the control group (n = 6) received regular rat chow. After separating pups from their mothers, young males (n = 6) of experimental group received soy isoflavones individually at the same daily dose of 200 mg/kg b.w. until reaching the sexual maturity (3 months). The young male of mothers unexposed to soy isoflavones (n = 6) were fed without soy isoflavones until reaching sexual maturity. After the treatment period, animals (males) from both groups were killed under thiopental anesthesia (120 mg/kg b.w., intraperitoneally (i.p.), Biochemie GmbH, Austria). Then the blood and femurs were immediately taken. The experiment was conducted in full adherence to Polish Law and the ethics committee of the Pomeranian Medical University gave its approval for the study protocol (approval No BN-060/76/06).
Chemicals
The chemicals used for Mg and Ca determination in serum and bones—element standards and HNO3 (ultra pure)—were obtained from Merck (Poland). The chemicals used for F determination in serum and bones—element standards and total ionic strength adjustment buffer (TISAB)— were obtained from Thermo Orion (Boston, USA); perchloric acid and sodium citrate were obtained from Chempur (Poland).
The chemicals used for microscopy—paraffin, ethyl alcohol and xylene—were purchased from POCh (Poland).
The immunohistochemistry-specific primary antibodies—ERα (F-10) mouse IgG antibody and ERβ (H-150) rabbit IgG antibody—were obtained from Santa Cruz Biotechnology, Inc (Germany). The secondary antibodies: anti-mouse IgG-fluorescein isothiocyanate (FITC) for ERα from Sigma-Aldrich (Poland) and anti-rabbit IgG-TexasRed for ERβ from Vector Laboratories (USA). Water was filtered through a Milli-Q Millipore purification system.
Analytical procedures
Sample preparation
Blood samples obtained from control and experimental animals were centrifuged for 30 min after collection and serum was separated. Serum was kept frozen at −80°C until analysis. Serum E2, calcium, magnesium and fluoride concentrations were determined.
Femurs of control and experimental animals were cleaned of soft tissue and in each case the left femur was decalcified after fixation and embedded in paraffin. Paraffin-embedded specimens were cut into 5 μm-thick sections for immunolocalization of ERα and ERβ. Right femurs were boiled three times in distilled water, dried for 24 h at 60°C and pulverized in an agate mortar.
Determination of estradiol in serum
In the blood serum of control and soy isoflavone-treated rats, concentrations of E2 were determined by electroluminescence (ECLIA—enhanced chemiluminescence immunoassay) method (Cobas 6000 analyzer, Roche, Switzerland). The range of determination was 18.4–15 781 pM/L. The apparatus was calibrated using standard solutions. The calibration curve was drawn automatically by the computer.
Immunolocalization of ERα and ERβ in bones
Paraffin sections of the femur tissue of control and treated rats were deparaffinized, hydrated and heated in microwave oven for antigen retrieval using citrate buffer (pH = 6). The immunohistochemistry was performed using specific primary antibodies: ERα (F-10) mouse IgG antibody (diluted 1:50) and ERβ (H-150) rabbit IgG antibody (diluted 1:50). The sections were incubated overnight at 4°C with primary antibodies. Sections were then washed twice in phosphate-buffered saline (PBS) and incubated for 45 min at room temperature with the secondary antibodies conjugated with flouorochrome. The secondary antibodies were anti-mouse IgG-FITC for ERα and anti-rabbit IgG-TexasRed for ERβ, diluted 1:64 and 1:100, respectively. The sections were then washed twice in PBS. Image analysis was performed with a fluorescent microscope (AxioImager M2, Zeiss) integrated with monochromatic camera (AxioCamera MRm, Zeiss), using filters GFP for green fluorescence and CY3 for red fluorescence. Images were taken from both channels simultaneously. Images are presented as fixed gallery images from both channels and as co-localization of both.
Determination of Ca, Mg and F in serum and bones
Bone
For calcium and magnesium determination, the bone powder (10 mg) was dissolved in 1 ml of concentrated 65% HNO3 (24 h at room temperature). From this basic solution, 10 μl was taken to plastic test tubes and added 5 ml H2O. Calcium and magnesium concentrations were determined using atomic absorption spectrophotometer (AAS) 9100X PHILIPS. The lower limit of detection was 0.1 mM for Ca and Mg, and the response was linear, up to at least 30 mM. The AAS was calibrated using standard solutions. The calibration curve was drawn automatically by the computer coupled with the apparatus. The determinations were performed in an air–acetylene flame for the relevant lamps at the following wavelengths: Ca—422.7 nm (with an added 0.5% lanthanum buffer solution) and Mg—285.2 nm (with an added 0.1% lanthanum or strontium as a buffer solution). The coefficient of variation was 9% for Mg.
For fluoride determination 10 mg of bone powder was dissolved in 1 ml of 2 M perchloric acid (1 h/95°C). From the resultant solution, 0.5 ml was poured into polyethylene cups, to which 2.5 ml of TISAB II solution and 2 ml of 1 M sodium citrate were added. Fluoride contents were determined with a potentiometric method, using Thermo Orion ion-selective electrode (The Thermo Scientific, USA). The electrode was calibrated using standard solutions. The lower limit of detection was 10−6 M for fluoride content. Reproducibility of the method was ±2%.
Serum
The serum calcium and magnesium concentrations were determined using an AAS technique. The determinations were performed in air–acetylene flame using lamps at the following wavelengths: Ca—422.7 nm and Mg—285.2 nm.
A 0.5 ml of TISAB III solution was added to 0.5 ml serum sample and the fluoride content was determined with a potentiometric method.
Statistical analysis
The results were analyzed statistically using the software package Statistica 6.1. Arithmetical mean and the standard deviation (SD) were found for each of the studied parameters. The distribution of results for individual variables was obtained with the Shapiro-Wilk W test. As most of the distributions deviated from the normal Gaussian distribution, nonparametric tests were used for further analyses. Correlations between the changes in the parameters were examined with the Spearman’s rank correlation coefficient. To assess the differences between the studied groups, the nonparametric Mann-Whitney test was used. The level of significance was p ≤ 0.05.
Results
E2 level in blood
The level of E2 in the blood serum of rats receiving phytoestrogens (27.62 ± 3.52 pg/ml) was significantly higher (p = 0.026) than in the control (21.40 ± 2.01 pg/ml; Figure 1).
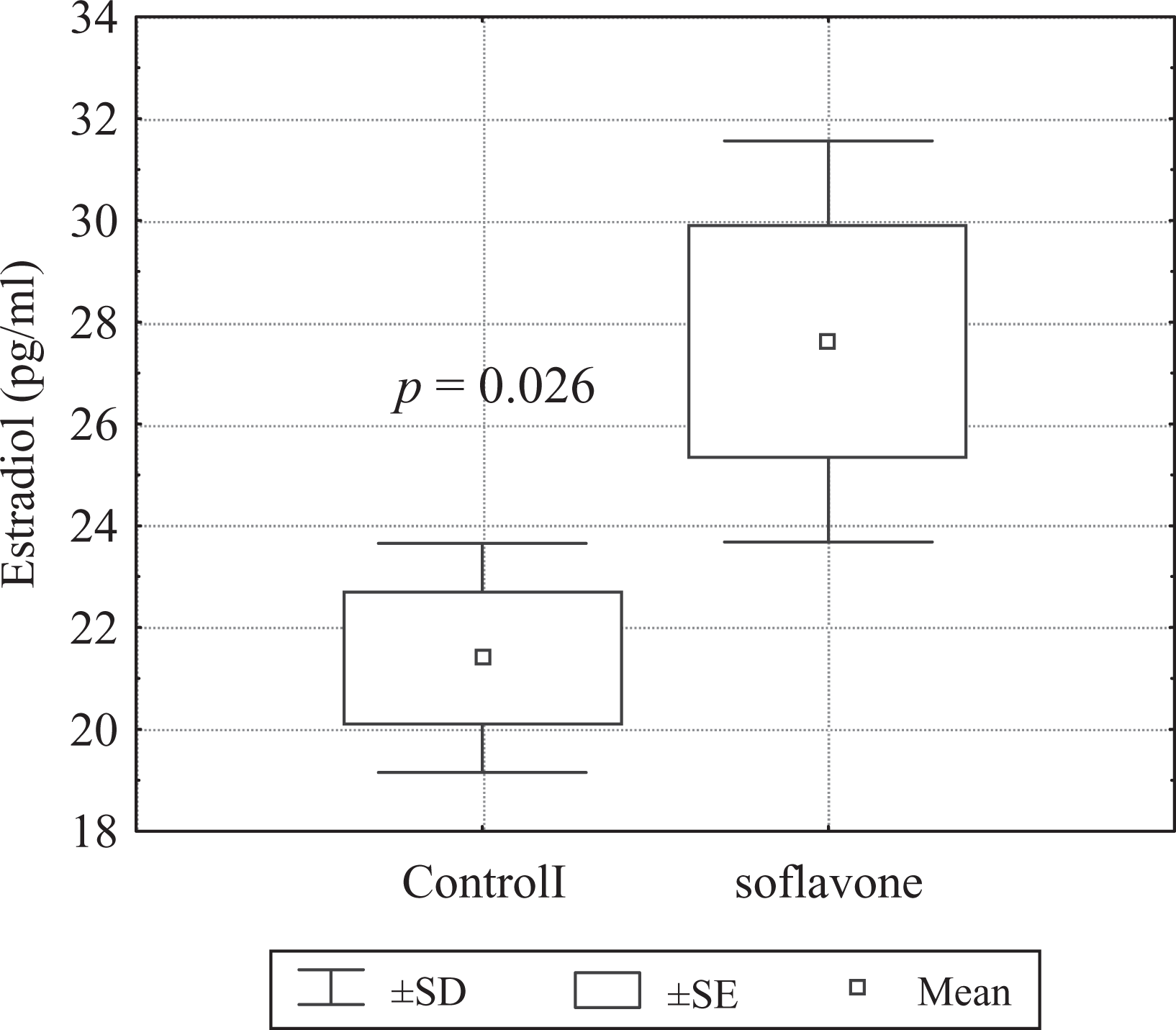
Estradiol concentration in serum of control and soy isoflavone-fed rats. During pregnancy and lactation, female rats received per os soy isoflavones at a daily dose of 200 mg/kg body weight. After that, young males until reaching the sexual maturity received soy isoflavones at a daily dose of 200 mg/kg body weight. The control group was male pups of mothers unexposed to soy isoflavones. In the blood serum of control and soy isoflavone-treated rats, estradiol concentration was determined using the ‘enhanced chemiluminescence immunoassay’ (ECLIA) electroluminescence technique.
Expression of ERα and ERβ in bone
The expression of ERα and ERβ was noted in the osteocytes of femurs of both control and experimental rats. In bone tissue of control rats, immunofluorescence indicating ERα and ERβ expression was detected in osteocytes located between lamellae and osteoblsts in non-haversian canal (Figure 2).
Expression of estrogen receptor α (ERα) and ERβ in the femur of control rats. (a) Immunofluorescence of ERα in the osteocytes located between lamellae (arrow) and osteoblasts in non-haversian canal (arrow head). (b) Immunofluorescence of ERβ in the osteocytes (arrow) and osteoblasts (arrow head). (c) Colocalization of ERα and ERβ.
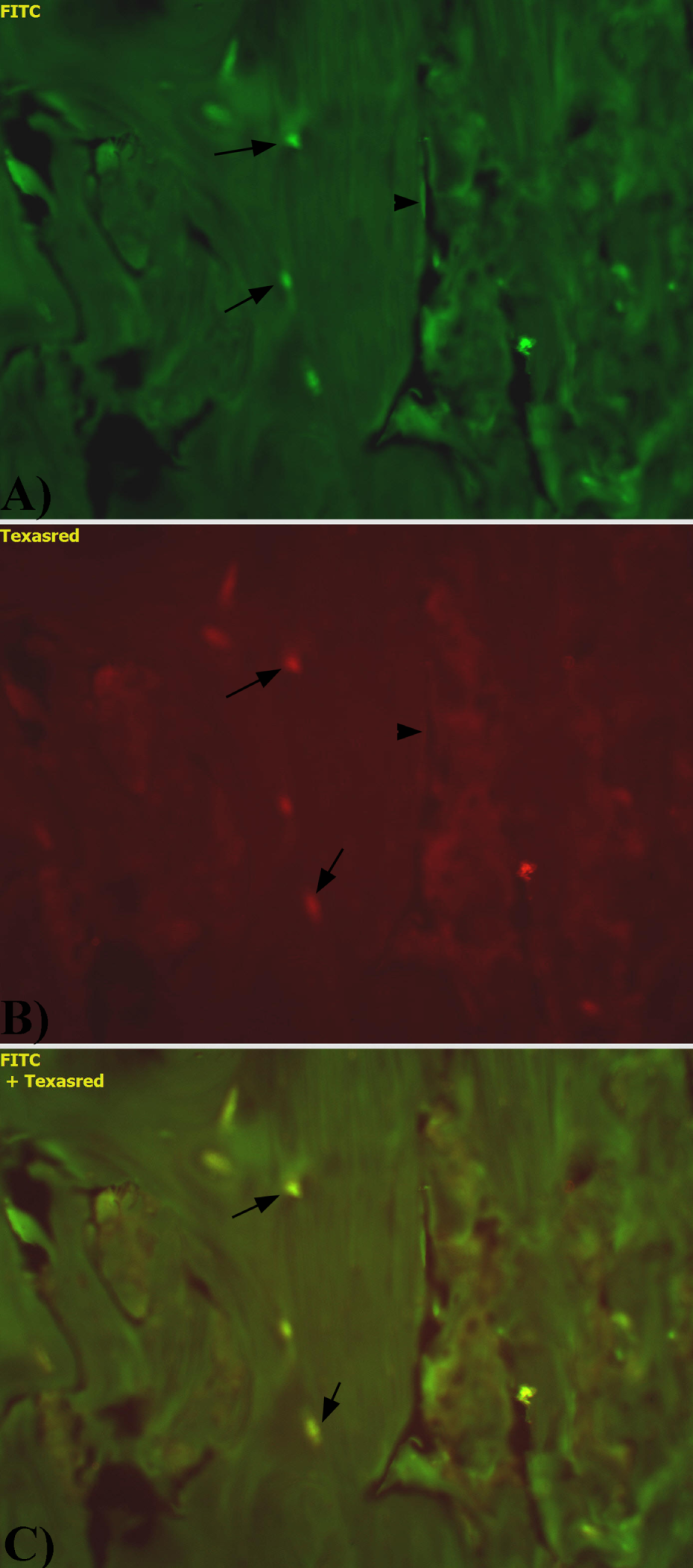
In the bone tissue of rats exposed to isoflavones, the distribution of ERα and ERβ was the same as in the bone of control rats. However, significantly decreased ERα immunofluorescence intensity and increased ERβ immunofluorescence intensities were observed in osteocytes (Figure 3).

Expression of estrogen receptor α (ERα) (a) and ERβ (b) in the femur of experimental rats. (a) The decreased immunoexpression of ERα in osteocytes (arrow). (b) The increased intensity of immunofluorescence of ERβ in osteocytes (arrow). (c) Colocalization of ERα and ERβ.
Ca in serum and bones
We observed a statistically significant lower calcium concentration in the blood serum (p = 0.0003) of animals treated with soy isoflavones (64.26 ± 6.14 mg/l) compared with the control group (92.36 ± 7.48 mg/l). Calcium concentration in bones slightly increased in the group of experimental rats (236.51 ± 12.01 g/kg dry mass (d.m.)) compared to the control group (229.82 ± 19.81 g/kg d.m.), but the observed differences were not statistically significant (Figure 4(a)).

Concentration of calcium (a), magnesium (b) and fluoride (c) in the femur bone and serum of control and experimental rats.
Mg in serum and bones
We observed a statistically significant higher magnesium concentration in the bones (p = 0.0015) of rats treated with soy isoflavones (4.78 ± 0.29 g/kg d.m.) compared with the control group (4.08 ± 0.45 g/kg d.m.). In the blood serum, we found no significant differences in magnesium concentration between the groups (p = 0.097; control group: 19.46 ± 3.92 mg/l, examined group: 16.29 ± 2.72 mg/l; Figure 4(b)).
Fluoride in serum and bones
Fluoride concentration in the bones of rats treated with soy isoflavones (544.82 ± 147.97 mg/kg d.m.) was significantly higher (p = 0.039) than in the control group (430.38 ± 63.69 mg/kg d.m.; Figure 4(c)). At the same time, the blood serum of examined rats (0.068 ± 0.031 mg/l) had a lower fluoride concentration than the control group (0.075 ± 0.024 mg/l) but the difference was not statistically significant.
Correlations between examined elements and estradiol in both groups
We observed a statistically significant correlation between E2 and fluoride (p = 0.022, R = −0.51) and between E2 and calcium concentrations (p = 0.004, R = −0.61) in the blood serum and between E2 and magnesium (p = 0068, R = 0.54) in bones.
Discussion
In this experiment, we have shown the changes in the expression of ERα and ERβ in the osteocytes of femurs and the content of some mineral elements in rats treated with isoflavones during prenatal and postnatal life until achieving the sexual maturity. The noted alterations probably can be a consequence of hormonal unbalance observed in soy isoflavones-treated rats.
In our study, the analysis of the E2 concentration in the serum showed that soy isoflavones-treated rats were characterized by higher concentration of this hormone compared with the control group (Figure 1). Simultaneous results were obtained by Richter et al. (2009) in the research provided on human term trophoblast cells. The authors also observed a significant stimulating effect on estrogen production by genistein and daidzein. 22 Because the phytoestrogens have higher binding affinities for the ERs but lower than E2 18 probably the increased concentration of phytoestrogens in rats’ serum and then its increased binding to ERs caused the compensated estrogen production noted in our research. 22 Estrogen has the greatest effect in the osseous tissue 3 through the decreasing osteoclast formation and activity and increasing osteoclast apoptosis. 18,23 Both genistein and estrogen increased the endogenous gene expression of the alkaline phosphatase, but inhibited osteopontin gene expression and interleukin-6 (IL-6) protein levels. 24 Studies in cell culture have shown that genistein, similarly to E2 may act on osteoblasts to reduce osteoclastic resorption by directly inhibiting IL-6 synthesis. In addition to IL-6, the osteoblastic synthesis of other osteoclast-stimulating cytokines may also be inhibited, presumably by influencing the ER-regulated pathways. 18
For many years it was thought that sex hormones in men are only testosterone and dihydrotestosterone, but the importance of estrogens in the male body is demonstrated by the distribution of ERs present in many tissues and organs, including bones. The ERs, ERα and ERβ, are expressed in all types of bone cells, osteoblasts, osteocytes and osteoclasts 18,20 and play a central role in mediating the biological effects of estrogen. 25 The ERs regulate gene expression by binding to specific response element sequences in the promoters of estrogen target genes or modulate transcription indirectly, via protein–protein interactions. 26,27 Both ERs upregulate the bone morphogenetic protein 6 (BMP6), a potent osteogenic factor played an important role in the bone-protective actions of estrogens and parathyroid hormone (PTH)-like hormone (PTHLH/parathyroid hormone-related protein (PTHrP)), a homolog of PTH, that functions as an autocrine growth inhibitor for osteoblast-like cells and as an anabolic agent in osteoporosis. Neurotensin was another growth factor/hormone found to be upregulated by estrogen via ERα and ERβ, but cystatin D, gene encodes an inhibitor of cysteine peptidases (cathepsins S and H), was stimulated very preferentially via ERβ and the increased expression of ERβ noted in our study might function in several roles in bone physiology. 25
In the present study, we observed coexpression of ERs in osteocytes, which can influence bone tissue metabolism. 4,18,20 Both ERs upregulate the BMP6, a potent osteogenic factor played an important role in the bone-protective actions of estrogens and PTHLH/PTHrP, a homolog of PTH, that functions as an autocrine growth inhibitor for osteoblast-like cells and as an anabolic agent in osteoporosis. Neurotensin was another growth factor/hormone found to be upregulated by estrogen via ERα and ERβ, but cystatin D, gene encodes an inhibitor of cysteine peptidases (cathepsins S and H), was stimulated very preferentially via ERβ and the increased expression noted in our study might function in several roles in bone physiology. 25 At nutritionally relevant doses, phytoestrogens may selectively interact with ERβ 4 and thus affect only the expression of subset of estrogen-responsive genes. 3 Genistein may differentially bind to ERα and ERβ receptors, acting as a partial agonist with ERβ receptors. 18 Daidzein and genistein potently trigger transactivation with ERβ from estrogen response element-reporter genes. 3
Both ERα and ERβ are present in developing human bone and the expression of ERβ increases most significantly during bone mineralization 3 causing increased accumulation, needed in this process, of elements, which was confirmed in our study (Figure 4). However, in the bone tissue of rats exposed to soy isoflavones showed reduced level of ERα expression and increased expression of ERβ (Figures 2 and 3). The results obtained by Zhao et al. (2007) indicated that ERβ induces proteasome-dependent degradation of ERα, presumably through the formation of ERβ/ERα heterodimers. The authors suggest that ERβ-mediated degradation of ERα is at least one mechanism whereby expression of ERβ inhibits recruitment of ERα to the estrogen-responsive promoters, leading to the suppression of ERα-regulated genes. 28
Increased levels of estrogens during sexual maturation result in increased growth hormone concentration, leading to the pubertal growth spurt. 23 Additionally, it stimulates the formation of blood vessels penetrating the epiphyseal gristles, delivering inorganic substances (including fluorides, calcium and magnesium). It causes the mineralization of the chondral matrix and consequently an increase in the concentration in osseous tissue of the elements analyzed in this study. This effect may cause a high level of fluoride and magnesium, resulting in increased levels of amorphous minerals in bone. 29,30
An elevated level of estrogens in the blood serum may result in the disrupted distribution of magnesium in the body due to increased ER sensitivity on the surface of estrogen-sensitive cells. It facilitates the uptake of elements by their cytoplasm; thus decreasing their availability to other tissues. Moreover, high concentration of estrogen in the serum could additionally cause a decrease in the reabsorption of fluoride, calcium and magnesium from bone, by the inhibition of parathormone secretion. 10 This may explain a higher concentration of these elements (especially magnesium) in the osseous tissue of soy isoflavones-treated rats in the present study.
The role of magnesium in the bone metabolism is mainly to activate enzymes taking part in the synthesis, resorption and remodeling of the osseous tissue. 10 In this study, we observed significantly higher magnesium content in the bones of the experimental group. This may have been caused by the increased estrogen concentration in the serum. However, the inflow of magnesium into the osseous tissue may not be advantageous, since the element stabilizes an amorphic (immature) form of hydroxyapatites.
In our study, we observed an increase in bone fluoride concentration, with a simultaneous decrease in fluoride concentration in the serum. Fluorine ions are capable of stabilizing the hydroxyapatite structure, causing an increase in its hardness. There are reports suggesting that ovariectomized rats combined therapy (estrogen and fluoride) can result in better bone strength than the single administration of either alone. 31,32 On the other hand, it has been shown that fluoride intake in the bone could interrupt the formation of the crystal nucleation process. 29 Crystallization would occur continuously toward c-axis at the peripheral area, whereas the central area would remain amorphous, resulting in crystal defects with a fuzzy structure appearing in the bone. 29
In our study, we did not observe any differences in the calcium content in the bone of the experimental and control rats. However, there are reports suggesting that isoflavones, combined with high Ca, are more protective against the loss of femur and vertebra bone mineral density than isoflavones or high Ca diets alone. 33 Other studies have found that in comparison with animal protein, soy protein decreases calcium excretion, a result of the lower sulfur amino acid content of soy protein. 34
A higher E2 concentration in serum may be an explanation for a decrease in the levels of calcium, magnesium and fluoride in the serum, and the simultaneous increase in the concentration of these elements in the bones of rats treated with phytoestrogens. E2 affects receptors and probably inhibits the release of IL-6 from osteoblasts, stimulating them to secrete transforming growth factor β, resulting in enhanced osteogenesis and increased absorption of magnesium, fluoride and calcium from the serum. 3
Conclusion
Pre- and postnatal diet supplementation of the male rats with soy isoflavones may considerably increase the concentration of estrogens in blood serum, affect ERα and ERβ receptors expression and influence on the mineral composition of bones.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declared no conflicts of interest.
