Abstract
Sertraline (SER), a tricyclic antidepressant, is considered to belong to the group of selective amine reuptake inhibitors. Its ability to cross the blood–brain barrier and transplacental transport has been reported previously. It is widely distributed in the brain and is bound to human glutathione S-transferase-π (GST-π). If SER is taken during pregnancy, it gets accumulated in the embryo and fetus, and some studies have suggested it may cause congenital malformations, thus the study of the interaction of GST-π with antidepressants is crucial. In this study, the interaction of human placental GST-π with SER in the presence of the natural ligand, reduced glutathione (GSH) and a xenobiotic ligand, 1-chloro-2,4-dinitrobenzene (CDNB) was investigated. The V m values obtained at variable [CDNB] and variable [GSH] were 61.3 ± 2.3 and 46.4 ± 1.7 U/mg protein, respectively. The k cat and k cat/K m values for GSH and CDNB were 3.63 × 106 s−1, 2.59 × 1010 M−1 s−1 and 4.79 × 106 s−1, 1.29 × 1010 M−1 s−1, respectively. The half maximal inhibitory concentration value for SER was 4.60 mM. At constant [CDNB] and variable [GSH] the inhibition type was linear mixed-type, with K s, α, and K i values of 0.14 ± 0.02, 2.90 ± 1.64, and 2.18 ± 0.80 mM, respectively. On the other hand, at fixed [GSH] and at variable [CDNB], the inhibition type was competitive, with K i value of 0.96 ± 0.10 mM. Thus, these findings weaken the importance of the protective role of GST against toxic electrophiles in vivo in adults, but due to its immature enterohepatic system SER may accumulate in the fetus and cause congenital malformations.
Introduction
Psychiatric illnesses such as mood and anxiety disorders and especially depression are commonly observed in pregnant women and in women at reproductive age and are pharmacologically managed by the use of antidepressants. 1,2 The biogenic amine hypothesis suggests that the functional increase in the activity of serotonin, norepinephrine, or dopamine will result in mood elevation, whereas their decrease will result in depression. 3 Nowadays the three modern classes of antidepressants are considered as the ‘first-line’: (i) selective serotonin reuptake inhibitors (SSRIs), (ii) serotonin–norepinephrine reuptake inhibitors, and (iii) norepinephrine–dopamine reuptake inhibitors. 4 Sertraline (SER) is one of the most potent SSRIs, and it has high affinity for serotonin uptake sites, low affinity for norepinephrine uptake sites, and very low affinity for neurotransmitter receptors. 5 –7 Thus, increased amine concentrations in the synaptic cleft will cause the speed up of synaptic transmission which will result in mood elevation.
Although, the side effects of SER is less hazardous than those tricyclic antidepressants, 8 several adverse side effects for SER have also been reported, such as sexual dysfunction 9,10 and congenital malformations. 11 –15 However, there are not enough firm data to demonstrate the actual contribution of prenatal exposure to SER on the neurophysiological development of the embryo and fetus; it is likely that the underlying pathology of the mother explains part of the anomalies. 8 SER is still primarily prescribed and widely used in severe endogenous depression therapy of the pregnant women. 5,6
Although SER is almost completely absorbed from small intestine following its oral administration, its absorption is rather slow, with a time to reach the maximum plasma concentrations of about 6–8 h and its distribution in the body is uneven; its concentration in brain is 40 times higher than plasma. 7,16 There are contradictory results about the contribution of cytochrome P450 2D6 (CYP2D6) to SER metabolism. SER is metabolized mainly by N-demethylation, and demethylated SER (N-desmethylsertraline, NSER) has longer half-life than SER: 20–30 h versus 60–100 h. 16 SER is a substrate for CYP3A4, suggesting the potential for drug interactions at this isozyme. 17 Although SER and/or NSER show high inhibitory potency on CYP2D6 in vitro with a K i of 0.7 µM, 18 studies on patients, however, have failed to show clinical relevance, 7 and only a modest inhibition was found for individuals with high CYP2D6 activity. 19 SER due to its lipophilic structure can easily cross the blood–brain barrier 7,14 and transplacental transfer of SER and its derivative NSER from mother to fetus has also been shown in both rat and human. 14,20 The accumulation of SER and NSER in the neonate resulted in congenital heart 11 and birth defects 12 ; congenital malformations 13 and toxicity in newborn was also reported. 21,22
Glutathione-S-transferases (GSTs, E.C.2.5.1.18) are the large family of enzymes involved in the chemical defense in cells by the detoxification of xeno- and endobiotics as a part of phase II detoxification system. They also serve as a ligand for various molecules such as steroid hormones, bilirubin, and heme and play a role in signaling cascades as a regulator via protein–protein interaction, synthesis and degradation of eicosanoids, synthesis of steroids, and degradation of aromatic amino acids. 23 –25
GSTs comprise various enzymes located in the cytosol, mitochondria, or microsomes. 26 It was shown that GST-π, one of the cytosolic GSTs, regulates apoptosis and cellular survival by inhibiting c-Jun-N terminal kinase 1 (JNK-1), a protein which has a role in apoptosis and cellular survival. GST-π, under stress-induced conditions (such as ultraviolet-irradiation or H2O2), oligomerizes and causes the dissociation of GST-π-JNK-1 complex leading to apoptosis. 27,28 Placental GST-π may have an important role in protecting the fetus against harmful nucleophiles produced from maternally ingested antidepressants. 12 –15,29,30 The effect of antidepressants on the fetus might be more severe due to immature hepatic clearance. 31,32 Interaction of antidepressants with human placental glutathione S-transferase-π (GST-π) and fetal brain GST-π may reduce their availability and therapeutic activity. On the other hand, they may decrease the efficiency of the enzymatic barrier formed by GST-π, thus increasing the exposure time of the fetus and the mother to toxic reactive electrophiles which may contribute to the adverse effects of antidepressant drugs. 30 Thus, elucidation of the inhibitory behavior of SER on GST-π is important. This study was conducted to investigate the possible effects of SER on human placental GST-π purified from term placenta.
Materials and methods
Materials
Ethylenediamine tetraacetic acid (EDTA), reduced glutathione (GSH), 1-chloro-2,4-dinitrobenzene (CDNB), SER, Trizma base, and all other chemicals used were of analytical purity and were purchased from Sigma, St. Louis, USA.
Methods
Purification of GST-π from human placenta
GST-π was purified from the placenta of a healthy woman who was admitted to the Department of Obstetrics and Gynecology for normal delivery. This study was approved by the Hacettepe University Ethical Committee (HEK 07/22-35) and ‘Patient Consent Form’ was completed. Following delivery, the placenta was immediately placed into the ice-cold physiological serum solution and transferred to the laboratory. Human placental GST-π was purified and characterized as previously described. 30 Briefly after homogenization, the placenta was centrifuged at 10,000 rpm for 1 h using GSA rotor (Du Pont Instruments, USA) on Sorvall RC-5B. The supernatant was collected and 35–90% ammonium sulfate fractionation was performed. After dialysis, the sample was applied to the S-hexylglutathione Sepharose 6B affinity column. Human placental GST was eluted with S-hexylglutathione. Activity containing fractions were collected and after dialysis it was applied to the polybuffer exchanger 94 chromatofocusing column. The enzyme was eluted using Polybuffer 74 (PB74). The activity containing fractions were combined, divided into 1.3 ml aliquots, and were stored at −80°C. This enzyme was used as the enzyme source.
Determination of protein concentration
Protein concentration was determined according to the method described by Bradford and bovine serum albumin was used as the standard. 33
Activity measurement during concentration-dependent inhibition of GST-π by SER
GST-π activity was determined according to the method of Habig and Jakoby. 34 The reaction mixture consisted of 100 mM potassium phosphate buffer, pH 6.5, 4 mM EDTA, 1 mM GSH, 1 mM CDNB, and appropriate amount of enzyme. All the measurements were performed at 37°C with a Perkin Elmer λ25 spectrophotometer (Perkin Elmer Instruments, CT, USA). The reaction was initiated by the addition of CDNB, and the increase in absorbance due to the formation of GS-DNB conjugate was recorded at 340 nm. Always a nonenzymatic reaction was run in parallel, which contained all components of the reaction mixture except the enzyme and the value obtained for nonenzymatic reaction was substracted from the value of enzyme-containing reaction mixture.
The inhibition of human placental GST-π by different concentrations of SER was measured as discussed above, and the data obtained were plotted as remaining activity versus [SER] and percentage remaining activity versus log[SER] to determine the half maximal inhibitory concentration (IC50) values. 35 To determine the effect of substrate concentrations on IC50 values of human GST-π inhibition by SER, percentage remaining activity versus log [SER] at fixed [CDNB] and variable [GSH] at fixed [GSH] variable [CDNB] and the IC50 versus [GSH] and [CDNB] plots were performed.
Kinetic experiments with SER
The stock solution of SER was prepared in dimethylsulfoxide (DMSO) and appropiate amounts were added to the incubation mixture. The final concentration of DMSO was always adjusted to 5% (v/v) and the same amount was also added to the controls. Incubation mixture contained 500 µl of 100 mM potassium phosphate buffer (pH 6.5) and 4 mM EDTA ± various concentrations of SER, fixed 1 mM [GSH] varied [CDNB] or fixed 1 mM [CDNB] varied [GSH]. The data obtained with antidepressants were calculated as discussed above and they were evaluated with different kinetic models. 35
Statistical analysis
The data were evaluated using Statistica 9.00 for Windows (StatSoft Inc., Tulsa, OK, USA).
Results and discussion
All the results presented below were obtained with the human placental GST-π previously purified from placenta at term and stored at −80°C. 30
The V m values for [CDNB] fixed–[GSH] varied and for [GSH] fixed–[CDNB] varied were 46.4 ± 1.7 and 61.3 ± 2.3 U/mg protein, respectively. The affinity of human placental GST-π for GSH (K m = 0.09 ± 0.03 mM) was about 4 times higher than the affinity for CDNB (K m = 0.37 ± 0.04 mM; Table 1). At variable [GSH]–fixed [CDNB] and at variable [CDNB]–fixed [GSH], the k cat values of 3.63 × 106 and 4.79 × 106 s− 1and the k cat/K m values of 2.59 × 1010 and 1.29 × 1010 M− 1 s− 1 were obtained (Table 1). At varied [CDNB]–fixed [GSH], the k cat value obtained was 1.3 times higher than the k cat value obtained at varied [GSH]–fixed [CDNB] but in contrast, the catalytic efficiency was 2 times higher than that of fixed [GSH]-varied [CDNB], implying that human placental GST-π, in the presence of a high concentration of GSH, converts it into the less active conformation (Table 1).
Kinetic parameters of GST-π inhibition effected by sertraline
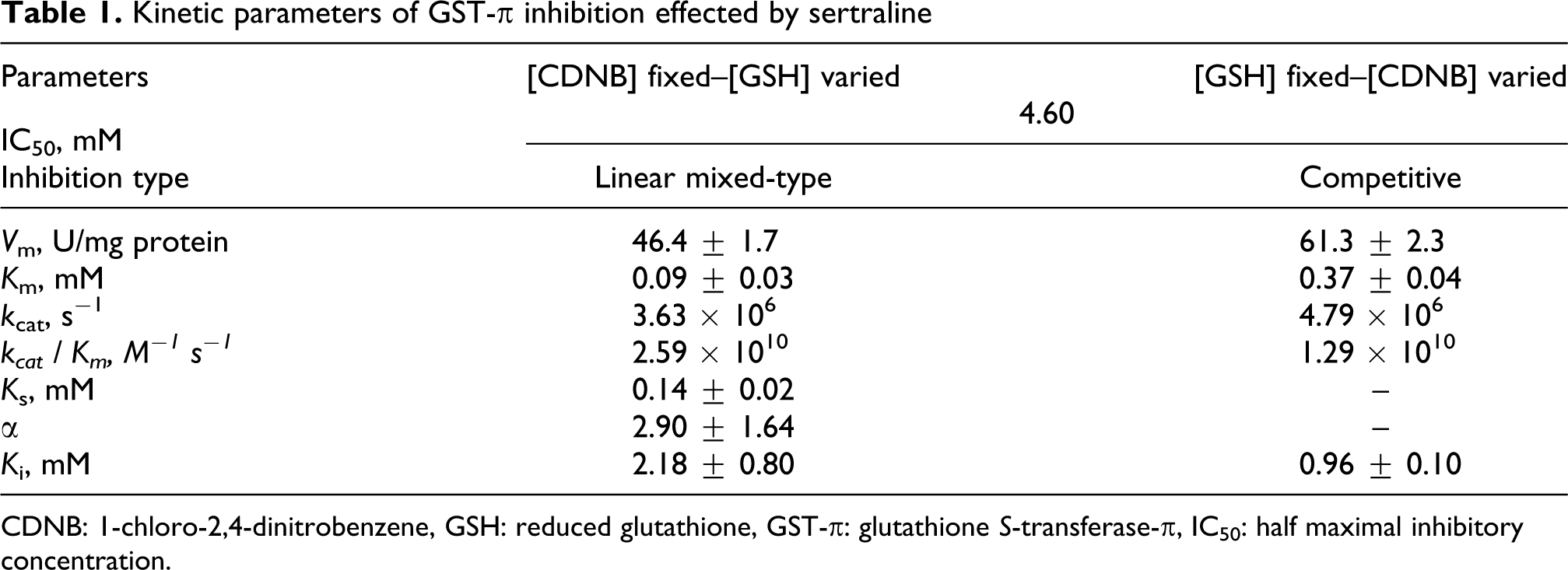
CDNB: 1-chloro-2,4-dinitrobenzene, GSH: reduced glutathione, GST-π: glutathione S-transferase-π, IC50: half maximal inhibitory concentration.
SER inhibited human placental GST-π in a concentration-dependent manner and an IC50 value of 4.60 mM was obtained from both remaining activity versus [SER] (Figure 1A) and percentage remaining activity versus log[SER] (Figure 1B) plots (Table 1). Although no detailed kinetic analysis was performed, the inhibition characteristics show that inhibition of GST-π by SER is reversible and it does not go to zero at the concentration range studied. The IC50 value obtained for SER is in good agreement with the results previously reported. 30,36 It was observed that changes in substrate concentration affected the IC50 values, so the effect of substrate concentration on IC50 values of human GST-π was elucidated. At fixed [CDNB]–varied [GSH], percentage remaining activity versus log[SER] gave parallel lines with a different IC50 value at each [GSH] (Figure 2A). When the IC50 values obtained from Figure 2A versus [GSH] were plotted, it was observed that the IC50 values increased with the increasing [GSH] concentration but above 0.4 mM [GSH] a decrease in IC50 values was observed (Figure 2B), implying that SER binds to a site on the GST-π isozymes that is different from the GSH-binding site (Table 1). On the other hand, when the varied substrate is CDNB, the IC50 values increased with the increasing [CDNB], parallel lines giving rise to different IC50 values at each [CDNB] (Figure 3A). The IC50 values obtained from Figure 3A versus [CDNB] plot gave a straight line, indicating that SER binds to the CDNB binding site in the xenobiotic-binding site on GST-π and competes with CDNB (competitive inhibition; Figure 3B). 25
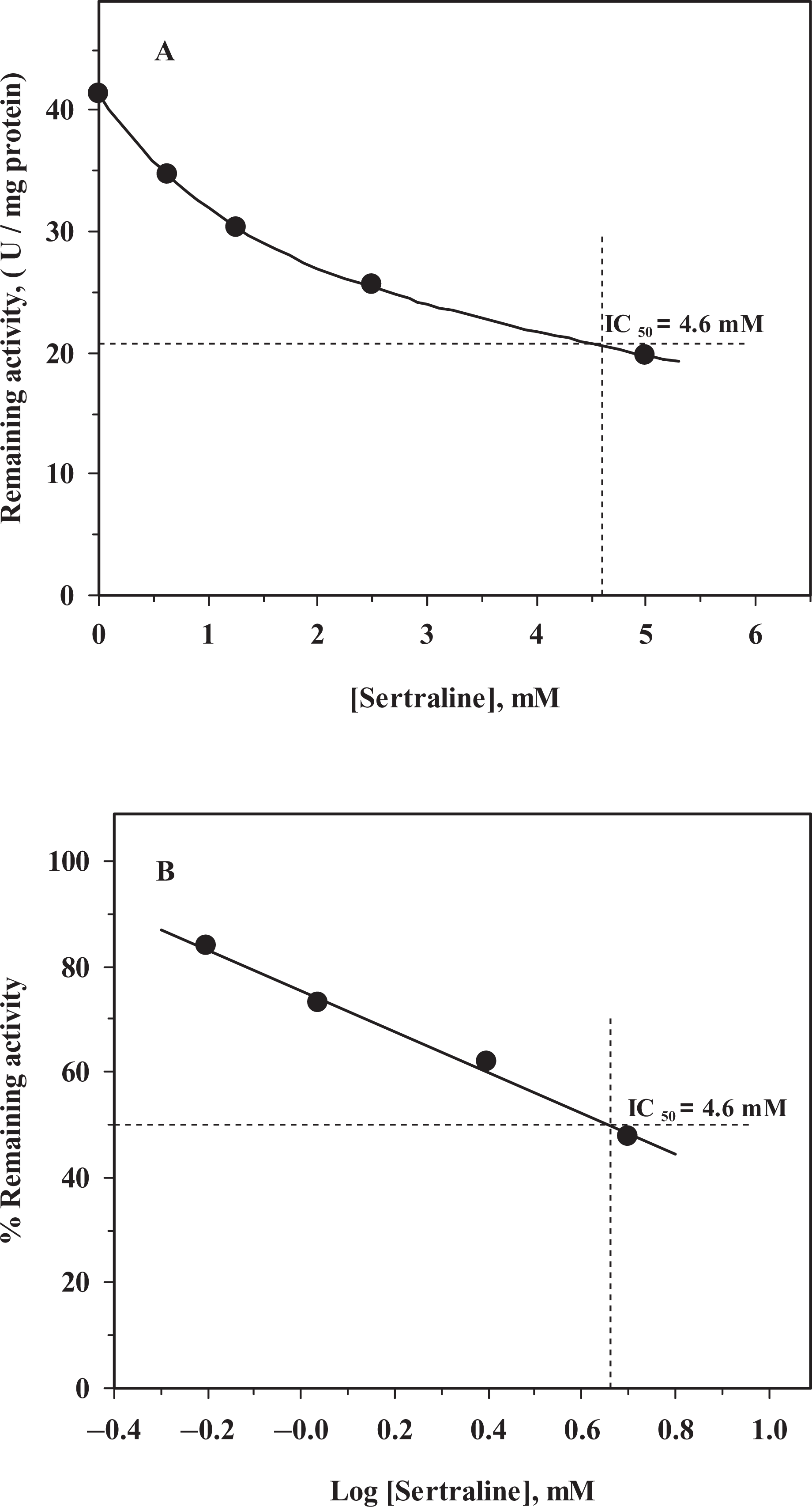
Inhibition of human glutathione S-transferase-π (GST-π) by sertraline. [GSH] = [CDNB] = 1 mM. (A) Remaining activity versus [Sertraline]; (B) percentage remaining activity versus log[Sertraline].
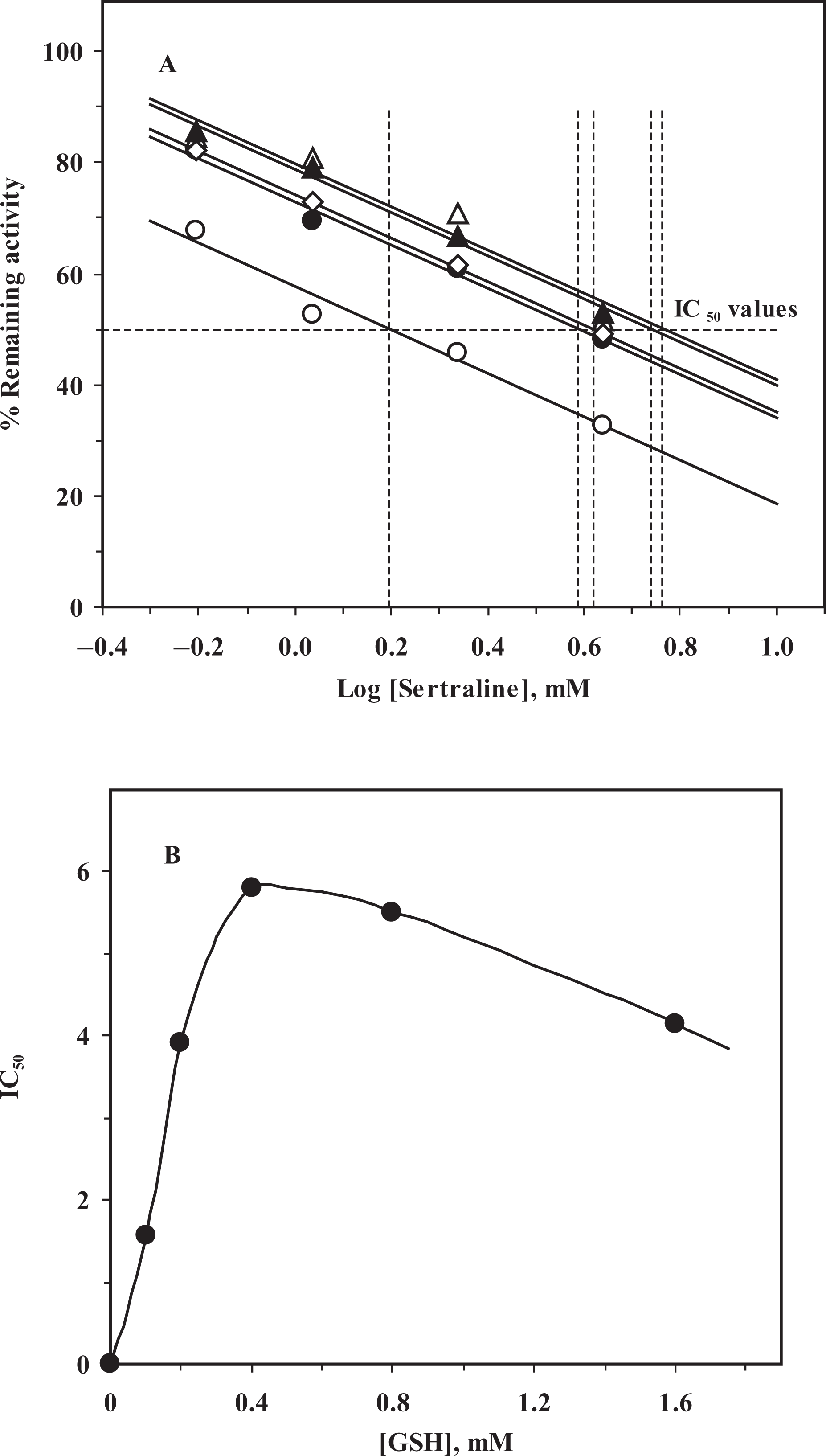
The effect of substrate concentration on half maximal inhibitory concentration (IC50) values of human GST-π inhibition by sertraline. (A) Percentage remaining activity versus log[Sertraline] plot: at fixed [CDNB] = 1 mM, and variable [GSH]: [GSH]: (○), 0.1 mM; (•), 0.2 mM; (▵), 0.4 mM; (▴), 0.8 mM; (⋄), 1.6 mM. (B) IC50 versus [GSH] plot.

The effect of substrate concentration on half maximal inhibitory concentration (IC50) values of human glutathione S-transferase-π (GST-π) inhibition by sertraline. (A) Percentage remaining activity versus log[Sertraline] plot: at fixed [GSH] = 1 mM, and variable [CDNB]: [CDNB]: (○), 0.1 mM; (•), 0.2 mM; (▵), 0.4 mM; (▴), 0.8 mM; (
At constant [CDNB]–varied [GSH], a linear mixed-type inhibition pattern for GST-π with SER was observed (Figure 4A). The linear mixed-type inhibition behavior implies that, although binding of SER to ligand site cannot be excluded, SER may also bind to a site which may be in the vicinity of G-site and cause the conformational change in GST-π. From the statistical analysis, the K s, α, and K i values calculated for GSH were 0.14 ± 0.02, 2.90 ± 1.64, and 2.18 ± 0.80 mM, respectively (Table 1). The slopes and intercepts obtained from Figure 4A versus [SER] were plotted and the slopes versus [SER] line intersected the x-axis at the −αK i, whereas intercepts versus [SER] line intersected the x-axis at −K i (Figure 4B). The same values for K s, α, and K i were obtained from Figure 4B and statistical analysis (Table 1).

Inhibition of glutathione S-transferase-π (GST-π) by sertraline. (A) Lineweaver and Burk plot: [CDNB] = 1 mM, [GSH] variable. [Sertraline]: (○), 0 mM; (•), 0.625 mM; (▵), 1.25 mM; (▴), 2.5 mM; (□), 5 mM; (B) Replot of the slopes (○) and intercepts (•) of (A) versus [Sertraline].
On the other hand, at constant [GSH]–varied [CDNB], GST-π exhibited a competitive inhibition pattern with SER (Figure 5A). This competitive type inhibition implies that SER binds to CDNB-binding site at the xenobiotic-binding site on the GST-π isozyme. 25 The K i value obtained for SER was 0.96 ± 0.10 mM (Table 1). The replots of slopes and the K m(app) versus 1/[CDNB] obtained from Figure 5A gave straight lines intersecting the x-axis at the −K i (Figure 5B) and the same value was obtained from the statistical analysis (Table 1).

Inhibition of glutathione S-transferase-π (GST-π) by sertraline. (A) Lineweaver and Burk plot: [GSH] = 1 mM, [CDNB] variable. [Sertraline]: (○), 0 mM; (•), 0.625 mM; (▵), 1.25 mM; (▴), 2.5 mM; (□), 5 mM; (B) Replot of the slopes (○) and K m(app) (•) of (A) versus [Sertraline].
It is well known that all GST isozymes have at least two binding sites: G-site for binding its natural substrate, GSH, and H-site for binding of xenobiotics. 25 The linear mixed-type inhibition pattern observed at fixed [CDNB]–varied [GSH] implies that SER, in addition to the ligand-binding site, may also bind to a site in the vicinity of G-site and creates partial competitive inhibition. 25,30 The competitive type of inhibition observed at varied [CDNB] is supported by the finding of Ralat and Colman. They have reported that there are at least three sites for binding of xenobiotics on GST-π and only one of them is for the binding of CDNB. 37 Although at high fixed [CDNB], the CDNB-binding site is saturated, the other xenobiotic-binding sites are still available for the binding of SER, and the binding of SER may cause the conformational change in GST-π leading to linear mixed-type inhibition. 30,35,37
It is well documented in the literature that in various stress-induced disorders, oxidative or nitrosative stress is the major cause of damage and the antidepressants are the primarily prescribed drugs for treatment. 38 –42 In addition to GSTs, several antioxidant enzymes such as catalase (CAT), superoxide dismutase (SOD), glutathione reductase (GR), and glutathione peroxidase (GPx) also function in the detoxification of endobiotics and xenobiotics. 41 The half-life of SER in women is 1.5 times longer than in men (an average of 32 h vs. 22 h). 43 Moreover, due to the immature hepatic clearance of the fetus the antidepressants ingested by the mother will cross through the placenta and accumulate in in utero and hence its effects on the fetus will be more severe. 14,20 –22 It was clearly shown above that SER is an effective inhibitor of human placental GST-π, which means extended exposure of the fetus to toxic electrophiles. Although the K i values obtained for the inhibition of GST-π are at micromolar to millimolar range and in the blood probably its concentration never reaches toxic levels in adults (the highest plasma SER concentration observed was 0.52 μM; cf. K i’s are 0.96±0.10 and 2.18±0.80 mM, Table 1), but due to its uneven distribution and ability to accumulate it may create an adverse situation in the fetus. 14 Moreover, it was recently shown that 10−6–10−7 M antidepressant concentrations are sufficient to induce the mRNAs of SOD, CAT, GPx, γ-glutamylcysteine synthetase, GR, and GST. 42 The controversial observation about GSTs is that transcriptional induction and inhibition by SER may lead one to speculate that the use of SER at very early stages of pregnancy might result in some mutations due to the insufficient clearance of the electrophiles. 14,21,22,30 Indeed, the role of GSTT1 and GSTM1 genotypes in determining the individual sensitivity to sister chromatid exchange induction by diepoxybutane in cultured human lymphocytes has been shown. 44,45 Several conflicting reports on congenital malformations attributed to antidepressants have been published in the literature, an aspect which clearly requires further study. 8
Footnotes
The authors declare that there are no conflicts of interest.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
