Abstract
In this study, the protective effects of vanillin were evaluated against carbon tetrachloride (CCl4)-induced kidney damages in Wistar albino rats. CCl4 (1 ml/kg, intraperitoneally [i.p.]) caused a significant induction of renal disorder, oxidative damage and DNA fragmentation as evidenced by increased plasma creatinine, urea and uric acid levels, increased lipid peroxidation (malondialdehyde [MDA]) and protein carbonyl. Furthermore, glutathione levels, catalase, superoxide dismutase, glutathione transferase and glutathione peroxidase activities were significantly decreased. A smear without ladder formation on agarose gel was also shown, indicating random DNA degradation. Pretreatment of rats with vanillin (150 mg/kg/day, i.p.), for 3 consecutive days before CCl4 injection, protected kidney against the increase of MDA and degradation of membrane proteins compared to CCl4-treated rats and exhibited marked prevention against CCl4-induced nephropathology, oxidative stress and DNA damage. Kidney histological sections showed glomerular hypertrophy and tubular dilatation in CCl4-treated rats, however, in vanillin pretreated rats, these histopathological changes were less important and present a similar structure to that of control rats. These data indicated the protective role of vanillin against CCl4-induced nephrotoxicity and suggested its significant contribution of these beneficial effects.
Introduction
Carbon tetrachloride (CCl4) is a manufactured chemical and most of it is used in the production of chlorofluorocarbons (CFCs) and other chlorinated hydrocarbons.
CCl4 intoxication in animals is an experimental model that mimics oxidative stress in many physiological situations. 1 The initial step in the tissue injury induced by CCl4 is its cytochrome P450-mediated transfer of a single electron to the C–Cl bond, giving a radical anion as a transient intermediate that eliminates chlorine to form a carbon-centered radical, the trichloromethyl radical (•CCl3) and chloride. 2,3 The trichloromethyl radical can dismutate to chloroform, bind to macromolecules or attack polyenoic fatty acids in cellular membranes. The double allylic hydrogen atoms in these acids are particularly susceptible to abstraction by free radicals, giving chloroform and secondary lipid radicals which react rapidly with molecular oxygen to form lipid peroxyradicals. The trichloromethyl radical can also react with oxygen to form the peroxytrichloromethyl free radical (•CCl3O2), which is more reactive than the 3,2 trichloromethyl radical and produces similar kinds of damage. The eventual decomposition of peroxidized fatty acids gives rise to a number of stable end products, such as carbonyls including malondialdehyde (MDA), ethane and pentane. 4 With respect to its molecular characteristics, CCl4 is considered a hepatotoxic and nephrotoxic chemical. Radicals generated by CCl4 induce damages and dysfunctions of DNA and proteins in addition to lipid peroxidation. The renal alterations caused by CCl4 are possibly mediated by oxidative stress. The key enzyme involving the CCl4-induced nephrotoxicity is P450 cytochrome, which is localized in the cortical tubule cells, 5 and then increased lipid peroxidation is evident in the renal brush border. 6 CCl4 treatment also affects renal mitochondrial function, including calcium flux across mitochondrial membranes. 6
In order to cope with the excess of free radicals produced upon oxidative stress, organisms have developed enzymatic and nonenzymatic antioxidant systems to scavenge or detoxify reactive oxygen species (ROS), block their production or sequester transition metals which are the source of free radicals. 7 The generation of small amounts of free radicals appears to have an important biological function, but oxidative stress is caused by the excessive production of ROS, which are implicated in the pathogenesis of several diseases. Excessive production of free radicals and oxidative stress can be induced by a variety of factors such as ionizing radiation or exposure to drugs and xenobiotics (e.g. CCl4). Various investigations have established that CCl4 causes renal injuries in rats8,9 in addition to hepatic toxicity through the generation of free radicals. 10 Alteration of antioxidant status with CCl4 may potentially cause nephropathies in rats.
The administration of antioxidants could conceivably protect tissues from the effects of free radicals, ROS and lipid peroxidation and thereby retard the progress of many chronic diseases. 11,12 A number of plants have been shown to possess protective properties by improving the antioxidant and anti-inflammatory status, for example flax, pumpkin, sesame and peanut.13–15 However, there is still a lack of scientific evidence to authenticate the protective properties of some natural products which are traditionally used to treat disorders. Vanillin, for example, is a chemical compound that confers the smell and flavor of vanilla, an important flavoring agent used in various confectionaries, sleep prevention agent, and aphrodisiac. 16 Currently, a lot of new functional uses of vanillin have been discovered. It exhibits chemopreventive effects on multiorgan carcinogenesis models in rats 17 and suppresses the invasion and migration of cancer cells 18–20 and could be used to treat sickle cell anemia. 21 Recently, vanillin has been shown to inhibit lipopolysaccharide-stimulated nuclear factor kappa B (NF-κB) activation and cyclooxygenase 2 gene expression in murine macrophages. 22 In addition, according to Sasaki et al., 23 vanillin is reported to be effective in reducing substance-induced mutations. As an antimutagen product, it is also able to prevent and treat cancer, a disease closely related to mutations. 24

Structure of 4-hydroxy-3-methoxybenzaldehyde (vanillin).
Despite the beneficial biological properties of vanillin, its protective effect against CCl4 nephrotoxicity has not so far been explored. In the present study, we investigated the antioxidant effects of vanillin against CCl4-induced nephrotoxicity in rats on relevant oxidative stress parameters, including thiobarbituric acid reactive substances (TBARS), protein carbonyl (PCO), glutathione (GSH) levels, antioxidant enzyme (catalase [CAT], superoxide dismutase [SOD], glutathione peroxidase [GPx] and glutathione transferase [GST]) activities, renal injury biomarkers (creatinine, uric acid and urea), genotoxicity (DNA damage) and renal histopathology.
Materials and methods
CCl4 intoxication, vanillin pretreatment and samples collection
The investigation was conducted in accordance with the international principles for laboratory animals use and care as found in the guidelines. 25 The investigation was approved by the ethics committee of the faculty of science of Sfax, Tunisia.
A total of 32 male Wistar rats (weighing between 180 and 200 g) were housed in cages at 22 ± 2°C on a 12-h day/night regimen with access to a diet and water ad libitum. They were divided into 4 groups of 8 rats each: group 1 (control group [C]); group 2 (CCl4 group [CCl4]); group 3 (vanillin group [Va]) and group 4 (vanillin + CCl4 group [Va + CCl4]).
The rats in groups 2 and 4 were i.p. injected with 1.0 ml/kg body weight of CCl4 dissolved in olive oil, v/v. Rat of the group 1 received an equal volume of olive oil. The administered dose was according to the reference dose for chronic oral exposure (RfD) as recommended for CCl4 (CASRN 56-23-5). 26 Groups 3 and 4 were i.p. injected daily with vanillin (150 mg/kg body weight), during 3 days before CCl4 injection. Twenty-four hours after the CCl4 intoxication, all rats of each group were killed. Kidney tissue was quickly removed, cleaned and washed in ice-cold saline solution. It was finely minced and homogenized in phosphate buffer (0.1 M; pH 7.4) and centrifuged at 8000g for 20 min at 4°C. The supernatant was used to assay biochemical parameters.
Determination of protein carbonyl content in kidney
PCOs were measured using the method of Reznick and Packer. 27 The absorbance of the sample was measured at 370 nm. The carbonyl content was calculated based on the molar extinction coefficient of 2,4-dinitrophénylhydrazine ([DNPH] ∊ = 2.2 × 104 cm−1 M−1) and expressed as nmol/mg protein.
Measurement of malondialdehyde in kidney
Concentrations of MDA in kidney, an index of lipid peroxidation, were determined spectrophotometrically according to Draper and Hadley. 28 The absorbance of TBA-MDA complex was determined at 532 nm. Lipid peroxidation was expressed as nmol of TBARS, using 1,1,3,3-tetraethoxypropane as standard.
Antioxidant enzymes and glutathione assays in kidney
Total superoxide dismutase activity
SOD activity was estimated according to Beauchamp and Fridovich. 29 The blue color developed in the reaction was measured at 560 nm. Units of SOD activity were expressed as the amount of enzyme required to inhibit the reduction of nitroblue tetrazolium by 50% and the activity was expressed as units per mg of protein.
Catalase activity
CAT activity was assayed by the method of Aebi. 30 Changes in absorbance were recorded at 240 nm. CAT activity was calculated in terms of nmol H2O2 consumed/min/mg of protein.
Glutathione peroxidase
GPx activity was measured according to Flohe and Gunzler. 31 The enzyme activity was expressed as nmoles of GSH oxidized/min/mg protein.
Glutathione transferase
GST activity was determined following the procedure of Habig et al. 32 The change in color was monitored by recording the absorbance (340 nm) at 30 s intervals for 3 min. The enzyme activity was calculated as nmoles 1-chloro-2,4-dinitrobenzene (C-DNB) conjugate formed/min/mg protein.
Glutathione levels
GSH in tissues was determined by the method of Jollow et al. 33 based on the development of a yellow color when 5,5-dithiobis-2 nitro benzoic acid was added to compounds containing sulfhydryl groups. The absorbance was measured at 412 nm after 10 min. Total GSH content was expressed as µg/mg of tissue.
Estimation of urea, uric acid and creatinine
The levels of urea, uric acid and creatinine in plasma were estimated spectrophotometrically using commercial diagnostic kits, respectively (Refs. 20151, 20143, and 20091), purchased from Biomagreb (Ariana, Tunisia).
DNA fragmentation analysis
The extent of DNA fragmentation in the kidney tissue was determined by the method described by Kanno et al. 34 Briefly, kidney tissue was homogenized in lysis buffer. The lysate was incubated for 3 h with proteinase K at 56°C. DNA was purified with an equal volume of Tris-saturated phenol/chloroform/isoamyl alcohol (25:24:1), incubated in ice and centrifuged at 10,000g at 4°C. The clear supernatant containing DNA was transferred to another tube and mixed and saturated sodium acetate was added, with the same volume of ice-cold isopropanol. The precipitated DNA was separated by centrifugation; it was washed twice with ethyl alcohol (70%) and finally dissolved in 100 µl of TE buffer (10 mM Tris, 1 mM EDTA, pH 8). The suspended DNA was incubated with RNase A for 60 min at 37°C in a water bath. DNA samples (10 µg of DNA/lane) were kept at 80 V for 1 h on 0.8% agarose gel in Tris-acetate-EDTA buffer, containing 0.5 μg/ml ethidium bromide. The gel was observed under an ultraviolet lamp and a photograph has been taken.
Histological examination
Kidney samples, intended for histological examination by light microscopy, were removed and immediately fixed in formalin solution, embedded in paraffin, serially sectioned at 5 μm and stained with hematoxylin–eosin.
Statistical analysis
The data were analyzed using the statistical package program stat view 5 software for windows (SAS Institute, Berkley, CA, USA). Statistical analysis between all groups was performed with one-way analysis of variance followed by Student’s t test. All data were expressed as means ± SD. The results were considered significant if p ≤ 0.05.
Results
Table 1 shows the effects of vanillin on the plasma levels of urea, uric acid and creatinine in rats. Nephrotoxicity was evidenced by a significant alteration in these plasma parameters in the CCl4 group when compared with those of controls. Pretreatment with vanillin significantly ameliorated the concentrations of urea, uric acid and creatinine in the plasma of Va-CCl4 rats when compared with CCl4-treated rats. The Va group showed no significant variation in these biomarkers as compared to the control group.
Plasma levels of creatinine, urea and uric acid of C-, Va-, CCl4- and Va + CCl4-treated rats a

C: control, CCl4: carbon tetrachloride, Va: vanillin.
aValues are expressed as mean ± SD (n = 8).
bSignificant differences between the CCl4 and C groups, p < 0.001.
cSignificant differences between the Va + CCl4 and CCl4 groups, p < 0.001.
dSignificant differences between the CCl4 and C groups, p < 0.05.
eSignificant differences between the Va + CCl4 and CCl4 groups, p < 0.01.
A significant increase in PCO and MDA content (44% and 59%, respectively), an end product of protein and lipid oxidation, respectively, in the kidney of CCl4 rats was observed when compared with the control group. Pretreatment with vanillin of CCl4 group significantly reduced the PCO and MDA content in the kidney (27% and 29%, respectively), when compared with that of rats administered CCl4 only, thus restoring it back to the range in the controls (Table 2).
Effect of vanillin pretreatment on the lipid peroxidation (MDA) and protein carbonyls levels (PCO) in the kidneys of rats a

C: control, CCl4: carbon tetrachloride, MDA: malondialdehyde, PCO: protein carbonyl, Va: vanillin.
aValues are expressed as mean ± SD (n = 8).
bSignificant differences between the CCl4 and C groups, p < 0.001.
cSignificant differences between the Va + CCl4 and CCl4 groups, p < 0.01.
dSignificant differences between the CCl4 and C groups, p < 0.01.
eSignificant differences between the Va + CCl4 and CCl4 groups, p < 0.05.
Antioxidant enzyme activities (CAT, SOD, GPx and GST) and GSH level in kidney of control and tested groups are shown in Table 3. In CCl4 group, significant decreases in enzymes activities (CAT, SOD, GPx and GST by 46%, 45%, 64% and 63%, respectively) and GSH level (53%) were observed in kidney, as compared to the control group. Pretreatment with vanillin improved, in Va + CCl4 rats, GSH levels by 109%, CAT, SOD, GPx and GST activities by 34%, 49%, 135% and 99%, respectively, in kidney. The Va group showed no significant variations in these parameters as compared to control group.
Glutathione levels and enzymes activities in the kidneys of C-, Va-, CCl4- and Va + CCl4-treated rats a
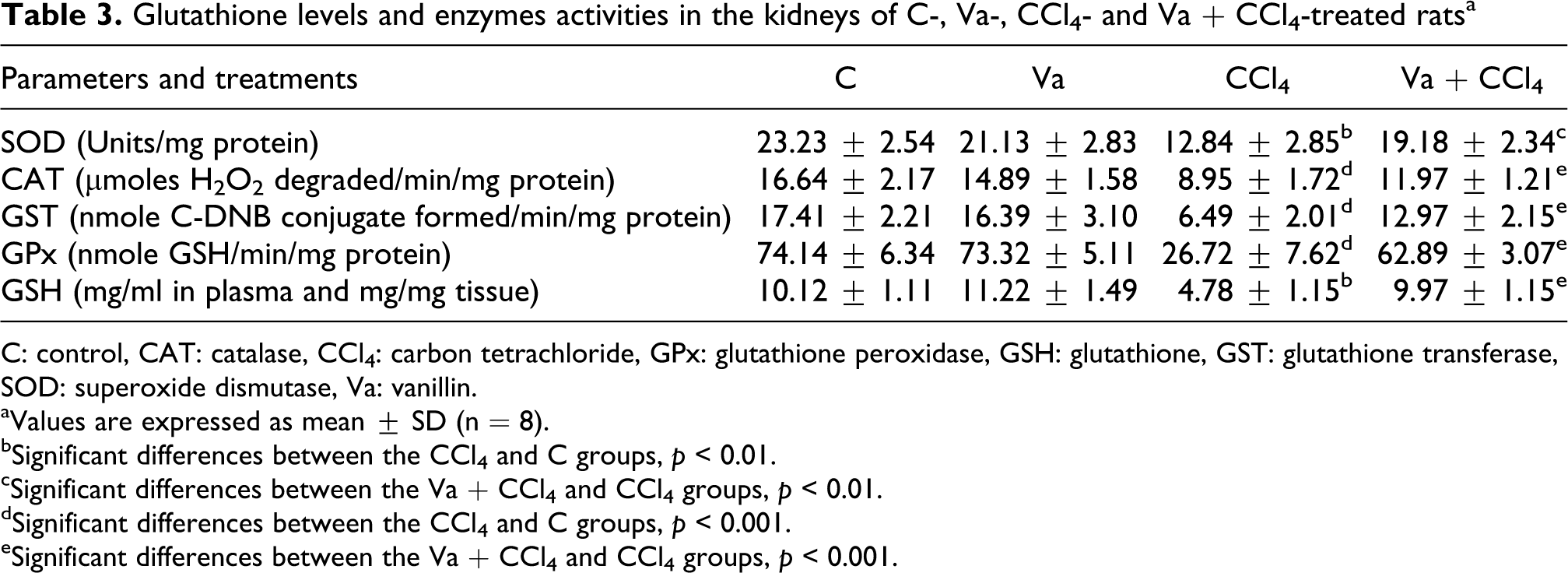
C: control, CAT: catalase, CCl4: carbon tetrachloride, GPx: glutathione peroxidase, GSH: glutathione, GST: glutathione transferase, SOD: superoxide dismutase, Va: vanillin.
aValues are expressed as mean ± SD (n = 8).
bSignificant differences between the CCl4 and C groups, p < 0.01.
cSignificant differences between the Va + CCl4 and CCl4 groups, p < 0.01.
dSignificant differences between the CCl4 and C groups, p < 0.001.
eSignificant differences between the Va + CCl4 and CCl4 groups, p < 0.001.
As shown in Figure 2, a smear (hallmark of necrosis) without ladder formation on agarose gel, indicating random DNA degradation, was observed in the kidney tissue of the CCl4-treated rats. Vanillin pretreatment exerted a protective effect against CCl4 by reducing the smear formation. No difference could be observed in DNA damage among the control and the vanillin groups.

Agarose gel electrophoresis of DNA fragmentation. M: marker (1 kb DNA ladder); lane 1: C group; lane 2: Va + CCl4 group; lane 3: CCl4 group; lane 4: Va group.
Histopathological studies of the kidney of control, Va and Va + CCl4 animals showed normal histology. In CCl4-treated rats, significant kidney injury was observed confirmed by damage in renal structure, showing marked glomeruli and tubular damages (Figure 3).

Kidney histological sections of C, Va, CCl4 and Va + CCl4 rats (hematoxylin and eosin, ×400).
Discussion
The present in vivo study demonstrated the protective potential of vanillin by reversing CCl4-induced nephrotoxicity. CCl4 has been commonly used in rat experimental models to investigate the oxidative stress induced in various organs. In recent years, there has been considerable interest in free radical-mediated damage in biological systems due to pesticide exposure35,36 and the search for herbal and natural drugs with antioxidant activity has gained importance as the dietary intake of antioxidants obtained from natural sources is considered to be relatively safe and involves no side effects. 37
CCl4 intoxication generates free radicals that trigger a cascade of events resulting in nephrotoxicity in rat. The pathogenesis of kidneys is a major public health problem. It is well known that the kidneys play a pivotal role in the regulation of various chemicals. Administration of CCl4 causes nephrotoxicity as indicated by the modification in the plasma levels of urea, creatinine and uric acid. These pathological changes signify a potential damage in liver and/or kidneys induced with CCl4 treatment.38,39 The increased plasma level of creatinine can be attributed to the damaged structural integrity of nephron. The present study revealed that pretreatment with vanillin of CCl4-treated rats ameliorated the toxic effects of CCl4 by restoring the markers mentioned above to normal levels.
Toxicity was also evidenced by a significant increase in kidney PCO and MDA content after the administration of CCl4 only. These parameters are also used according to Tokyay et al. 40 to determine early liver oxidative stress. They are used to investigate the oxidative damage of proteins and lipid peroxidation of the membrane and lipoproteins as a possible pathogenic mechanism for tissues injury. 41 Lipid peroxidation, an autocatalytic process and a common consequence of cell death, may cause peroxidative tissue damage in inflammation, cancer and aging. 42 It has been reported that lipid peroxidation is one of the major causes of CCl4-induced nephrotoxicity mediated by the production of free radical derivatives of CCl4. In the present study, the administration of CCl4 resulted in a significant elevation in renal PCO and MDA levels, indicating increased protein and lipid oxidation as indicated by the severe injuries in the histopathology of kidneys. Interestingly, preadministration of vanillin markedly reduced the extent of protein and lipid oxidation by decreasing the PCO and MDA levels, which confirms the nephroprotective effect of vanillin against the renal lipid peroxidation induced by CCl4.
Generally, antioxidant enzymes such as CAT and SOD are easily inactivated by lipid peroxides or ROS, which results in a decrease in their activities as reported by our results in CCl4-treated rats. These enzymes are also extremely effective antioxidant enzymes responsible for catalytic dismutation of highly reactive toxic superoxide radicals to H2O2 and for the catalytic decomposition of H2O2 to oxygen and water, respectively. 43 Thus, antioxidant enzymes play an important role in the detoxification of xenobiotics, catalyzing their conjugation with the reduced GSH. The ability of vanillin to modulate the activity of renal antioxidant enzymes such as SOD and CAT has been demonstrated in our study, indicating the antioxidative action of this molecule.
The GSH system includes reduced glutathione, GPx and glutathione-S-transferase. The status of these antioxidant enzymes is an appropriate indirect way to assess the prooxidant–antioxidant status in tissues. These enzymes are lowered due to enhanced lipid peroxidation and are also accompanied by a decrease in GSH level. GSH concentration is known to influence the redox status of cells. Therefore, GSH-dependent enzymes will be affected when its level is depleted in the cells. GSH acts as a nonenzymatic antioxidant both intracellularly and extracellularly in conjunction with various enzymatic processes, which reduces hydrogen peroxides and hydroperoxides by redox and detoxification reactions. 44 We obtained a significant depletion of GPx, glutathione-S-transferase and GSH in renal tissues of CCl4-treated rats. These results support the view that renal injury induced by CCl4 is the best characterized system of xenobiotic-induced nephrotoxicity, as it causes extensive oxidative stress in renal tissues. Administration of vanillin attenuated the CCl4 toxicity, thereby increasing the level of GSH and activities of GPx and glutathione-S-transferase compared to the CCl4 group.
Agarose gel electrophoresis showed undetectable DNA laddering (DNA fragmentation) in the kidney of the control rats. The DNA intact band appears to be condensed near the application point with no DNA smearing, suggesting no DNA fragmentation. On the other hand, in our study, CCl4-treatment resulted in massive DNA fragmentations with a subsequent formation of a DNA smear on agarose gel, a hallmark feature of necrosis 45 without ladder formation, suggesting CCl4-induced renal cell damage. Vanillin pretreatment was found to be effective in preventing this CCl4-induced smear formation.
The biochemical parameters were correlated with the renal histological studies. In fact, we revealed that CCl4 caused a significant damage to renal structure, showing marked glomeruli and tubular damages, probably due to the generation of reactive radicals and to subsequent lipid peroxidation induced by its generated metabolites. So, hydroperoxides accumulated in kidney could cause cytotoxicity associated with membrane phospholipids peroxidation, the basis for renal cellular damage and necrotic renal cells. Pretreatment of rats with vanillin improved the histological alterations induced by CCl4, which could be attributed to its antiradical/antioxidant activities.
The mechanism by which vanillin prevented generation of free radicals against CCl4-induced oxidation could be explained by (i) a decrease in the metabolic activation of CCl4 or (ii) acting as a chain-breaking antioxidant for scavenging free radicals or (iii) a combination of these effects. Recently, Tai et al. 46 demonstrate that vanillin shows a stronger antioxidant activity than ascorbic acid and Trolox, and it reacts with radicals via adduct formation or self-dimerization mechanism, which contribute to the high reaction against ABTS•+ and AAPH-derived radicals.
In conclusion, our results demonstrate that vanillin present a protective effect on kidney toxicity induced by CCl4. The mechanisms of protection include the inhibition of protein and lipid oxidation processes, the increase in antioxidant enzymes activities and the inhibition of DNA damage, which results in the recovery of biological parameters and the integrity of kidney histological aspects.
Footnotes
M Makni and Y Chtourou contributed equally to this work.
Acknowledgements
The authors thank the skilful technical assistance of Miss Kchaou Dalinda (Histopathology Laboratory of CHU Habib Bourguiba, Sfax, Tunisia). We also extend our thanks to Mr Bejaoui Hafed, teacher of English at the Sfax Faculty of Science, who helped proofread and edit this manuscript.
Funding
The present work was supported by the grants of DGRST (Appui à la Recherche Universitaire de base, ARUB 99/UR/08-73), Tunisia.
