Abstract
Sulphur mustard (SM) is a bifunctional alkylating agent that causes cutaneous blisters in human and animals. Remedies to SM-induced dermatotoxicity are still in experimental stage. Due to inevitable requirement of a wound-healing formulation against SM-induced skin lesions, efficacy of formulations including povidone iodine, Aloe vera gel, betaine or framycetin sulphate was evaluated in present study. SM was applied percutaneously (5 mg/kg) once on back region of Swiss albino mice; and after 24 hours, DRDE/WH-02 (Defence Research and Development Establishment/ Wound Healant- 02, containing polyvinylpyrrolidone [PVP], A. vera gel and betaine), Ovadine, Soframycin or A. vera gel were applied topically, daily for 3 or 7 days in different groups. Skin sections were subjected to histopathology, histomorphologic grading, tissue leukocytosis, terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) assay and immunohistochemistry of inflammatory-reparative biomarkers. DRDE/WH-02 treated mice received highest score on the basis of histomorphologic scale and lowest number of TUNEL-positive cells compared to other groups. DRDE/WH-02 showed better wound healing as evidenced by widespread re-epithelialization, homogenous fibroplasias well supported by the expression of transforming growth factor-α, endothelial nitric oxide synthase (eNOS) and fibroblast growth factor. Upregulation of interleukin 6 in DRDE/WH-02-treated mice skin resulted in increased tissue leukocytosis and an early removal of tissue debris that initiated reparative process at faster rate compared to other groups. In conclusion, DRDE/WH-02 provided better healing effect and can be recommended as an effective wound healant against SM-induced skin injury.
Keywords
Introduction
Sulphur mustard (SM), chemically bis(2-chloroethyl)sulphide, is a bifunctional alkylating agent that causes cutaneous blisters or systemic toxicity in human or animals. SM-induced blisters are characterized by edema, dermal infiltration of inflammatory cells, premature death of basal layer of epidermal cells and dermoepidermal separation.1–4 The easy availability of precursors, the simple method of synthesis and extremely stable nature make SM a chemical weapon of choice by the military and terrorist groups. Although decontamination is one of the important aspects of protection against chemical warfare agents, it is effective only when done in first 2 minutes of the exposure, which is not always practical in field conditions. However, there is always a possibility of wound formation due to absorption of warfare agents during time interval between exposure and decontamination. 5
A variety of formulations have been developed for the treatment of mechanical and burn injuries. These formulations were alternatively used in the management of vesicants-induced skin injury but with limited protection. Most of the commercially available burn wound-healing formulations contains antibiotics, antifungal or antiseptics that are preferred in the management of SM-induced skin lesions. The widespread uses of antimicrobial products have prompted some speculation on the development of microbial resistance and occurrence of cross-resistance among all these products.6–8 Moreover, use of cream, lotion or gel containing silver sulfadiazine, chlorhexidine digluconate and others leads to the production of altered form of microvessels and reduced density of dendrocytes and fibroblasts. 9 Other cosmetic formulations have organic molecules such as cetyl alcohol, glyceryl stearate, mineral oil, glycerine, stearic acid, triethanolamine, dimethicon FEG-40 (dimethyl polysiloxane), sorbitan lanolate, methyl paraben, quaternium 15 and propyl paraben. 10 The main drawback of these organic molecules is that they enhance the systemic absorption of vesicants and produce additive effect to their toxicity. Also, these formulations lack moisturising property, which is a prerequisite for enhancing wound healing.
There is no specific therapy for SM-induced skin injury, but procedures such as LASER debridement of established burns have shown to increase the rate of burn resolution in animal models and may, therefore, benefit clinically. 11 But limitation of LASER debriment is the requirement of advanced infrastructure, which is practically not possible in field conditions. Many compounds were evaluated as therapeutic approaches to SM-induced dermatotoxicity, 12 but recommendations regarding their therapeutic efficacy still remain in experimental stage.
Povidone iodine has been reported to enhance angiogenesis and increase the healing rate of mechanical wounds. 13 Brodsky et al. 14 hypothesize that topical treatment with an oxidising agent may protect the skin against SM toxicity. Indeed, postexposure decontamination with topical iodine or povidone iodine preparations significantly reduced SM-induced skin lesions in guinea pigs. 15 Besides the wound-healing properties, povidone iodine is a well-known antimicrobial agent that does not show microbial resistance. Wound-healing efficacy of A. vera is well known for curing mechanical wounds 16 and second-degree burn wound 17 in rats. Subramanian17, Chithra18 and Takzare19 elicited the prophylactic effect of A. vera leaf gel extract, supporting its wound-healing properties. Topical application of A. vera gel may be beneficial for protecting the SM-induced skin lesions. 20
Betaine, a derivative of choline, is found in dietary sources such as wheat germ, spinach, shrimp, wheat bread and raw mushrooms. Betaine functions as an osmolyte and methyl donor in many pathways, it prevents the induction of inducible nitric oxide synthase, attenuates the production of nuclear factor κβ and increases the production of glutathione, which in turn is involved in cytokine regulation and is related to anti-inflammatory response. 21 Betaine is also used in detergents at various concentrations for reducing irritation. 22 However, slight or no information regarding the use of povidone iodine, A. vera and betaine alone or in combination is known for their possible role in protecting SM-induced skin lesions.
The formulations that are recommended during alternative clinical practices by the clinicians for the symptomatic treatment of blisters provide only antisepsis, and wound healing occurs naturally. Inclusion of wound healant in the formulation is a prerequisite but the formulators have ignored this till now. Thus, in view to develop an effective dermal treatment against SM-induced skin lesions, an attempt was made in the present study to evaluate the efficacy of wound-healing formulations including povidone iodine, A. vera gel, betaine or Framycetin sulphate in a resourceful and consistent mouse model described by us. 23
Material and methods
Chemicals
SM was synthesized in the Synthetic Chemistry Division of the Establishment and was found to be above 99% pure by gas chromatographic analysis. Polyvinylpyrrolidone iodine (PVP-I) was procured from Sigma (St. Louis, MO, USA) and Betaine (anhydrous, 98%) was procured from Alfa Aesar (Chennai, India). Commercially available (5% w/v) solution of PVP-I (Ovadine, Ind-Swift Ltd., India) and Framycetin sulphate (Soframycin, Aventis, India) ointment were procured from local medical shop.
Animals
All experiments were performed on randomly bred female Swiss albino mice (approximately 2 months old) weighing 25–30 g. Animals were obtained from the Animal Facility of Defence Research and Development Establishment. Care and management of the animals were as per the approved guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA, India). The Institutional Animal Ethical Committee (IAEC) approved the protocol for the experiment. The animals were fed standard pellet diet (Ashriwad Feeds, India) and water ad libitum. A day before the application of SM, hairs from back region of the mice were closely clipped using a pair of scissors. Animals were randomly allocated to various groups of six mice in each group and SM diluted in acetone was applied once dermally at a dose of 5 mg/kg body weight. All the safety precautions were taken while handling SM during dermal application. Based on the observations made in pilot studies, three mice were housed in a single cage of 430 × 290 × 150 mm size to avoid licking or self-licking of applied formulations by mice. This may be probably due to enough space in the cage that avoided close contacts of mice with each other. Out of six mice in a group, three mice were euthanized on day 3 and remaining was euthanized on day 7 post SM application. Same set of experiment was repeated and data complied for six mice at each time point are discussed in the results section. Details of the groups and treatments are mentioned in Table 1.
Details of experimental groups and treatments
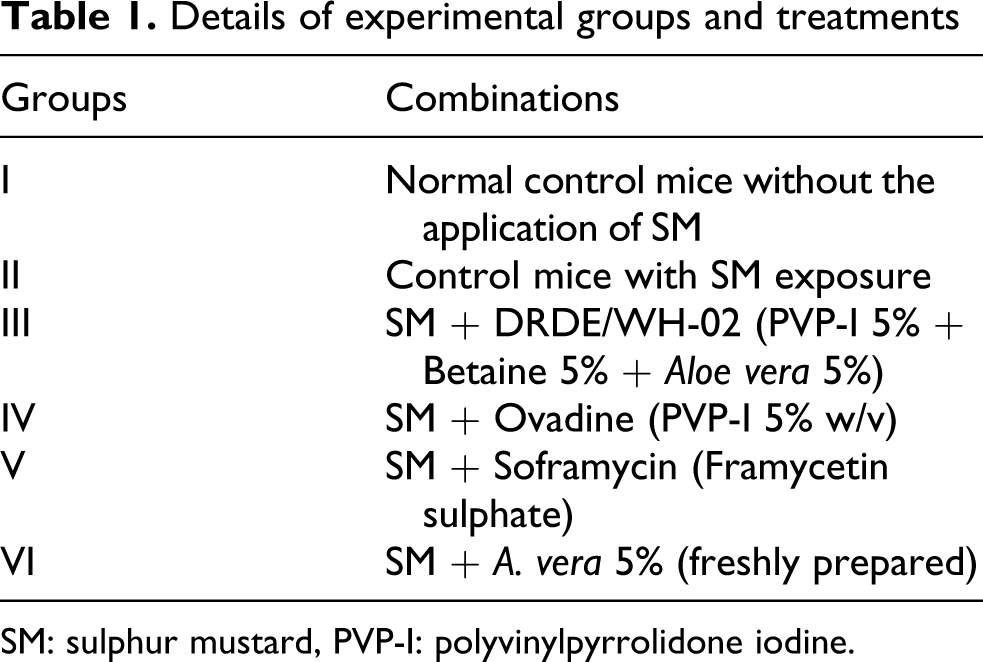
SM: sulphur mustard, PVP-I: polyvinylpyrrolidone iodine.
Preparation of DRDE/WH-02 formulation
Fresh leaves of A. vera were used throughout the experiment as a source of A. vera leaf gel. Central Research Institute (A Branch of Central Council for Research in Ayurveda and Siddha, Ministry of Health, Government of India), Gwalior, provided the plant material (specimen identification number: F.No.5-4/09-10/CRI-GWL./Tech./Survey/), which was harvested in October 2009. Five grams of PVP-I, 5 g of A. vera leaf gel and 5 g of betaine were triturated with distilled water in mortar pestle and weight was adjusted to 100 g by adding distilled water to make the final formulation. Storage, stability and homogeneity of the formulation were carried out as per International Conference on Harmonisation (ICH) Q1A (R) guidelines, which showed that the formulation was homogenously prepared and remain stable for more than 3 years (data not shown).
Treatment
Twenty-four hour after the application of SM, mice were treated with dermal application of the formulations. The formulation was spread on the exposed area using a sterilized glass rod once daily throughout the experimental period.
Histopathology
SM-exposed skin from the back region of mice was dissected out and punched on 2 × 2 cm card paper with dorsal side facing downwards and fixed in Bouin’s fluid. Furthermore, the skin was processed in ascending concentrations of ethanol, cleared with toluene (Leica tissue processor, TP1020; Leica, Germany) and embedded in paraffin (Leica embedding station, EG1160; Leica, Germany). Serial sections (4–5 µm thick) of the paraffin-embedded tissue blocks were cut with a Microm HM 360 (Microm, Germany) microtome and processed for haematoxylin and eosin (H&E), Masson trichrome (Accustain Trichrome Stains [Masson], Sigma-Aldrich Inc, USA) and toluidine blue (mast cells and basophils) stain using standardized programme in Leica autostainer XL (Leica, Germany).
Tissue leukocytosis
Tissue leukocytosis was quantified to assess the cellular reaction. A total of six lesions of the same age were obtained from six different mice and processed for histopathology. In each section, the cells (neutrophils, monocytiod cells and mast cell/basophils) were counted in five representative fields as described earlier. 24 The cited results, thus, represent the mean value ± standard error of 30 microscopic fields of each time interval. Intravascular and intramural leukocytes were excluded.
Histomorphologic scale
Lesions were scored in a blind fashion based on a published histomorphologic scale for cutaneous SM injury of weanling pig 25 modified for the evaluation of tissues as mentioned in Table 2. The maximum total score for best possible outcome (eg, normal skin) was 16.
Histomorphologic Scale

TUNEL assay
In situ apoptosis detection by staining with terminal deoxynucleotidyl transferase dUTP nick end labelling ([TUNEL] Dead End™ Colourimetric TUNEL System; Catalogue No. G7131 and G7132; Promega, Madison, WI, USA) was performed using manufacturers protocol. Apoptotic cells were identified as TUNEL-positive cells and counted randomly in at least five selected fields (×40 magnification) of control and SM-treated skin tissue sections under Leica DMLB microscope using Qwin Version-3 image analysis software (Leica Inc, Germany). Apoptotic index was calculated as per the formula given below.

Immunohistochemistry
The avidin–biotin immunohistochemical staining using Vectastain Elite PK-6200 universal kit (Vector Laboratories Inc, Burlingame, CA, USA) was performed to localize various markers viz. transforming growth factor-α (TGF-α), fibroblast growth factor (FGF), endothelial nitric oxide synthase (eNOS) and interleukin 6 (IL-6) in the skin sections. Primary antibodies (Mouse monoclonal [213-4.4] to TGF-α at 1:20 dilution; Rabbit polyclonal to eNOS at 1:100 dilution; Rabbit polyclonal to FGF and IL-6 at 1:500 dilution) were procured from Abcam, Cambridge Science park, Cambridge, UK. Sections were developed using diaminobenzidine ([DAB] Peroxidase Substrate kit; Vector Laboratories Inc) and sections were counterstained with light green or haematoxylin.
Statistical analyses
Tissue leukocytosis, histomorphologic scores and apoptotic index were analysed using one-way analysis of variance with Student–Newman–Keuls multiple comparison. A p ≤ 0.05 was taken as statistically significant. The analyses were carried out using Sigma stat for Windows version 2.03 (SPSS Inc, USA).
A decision analysis was carried out to describe the severity of lesions in histopathological and immunohistochemical variables. Based on the affected area in microscopic field, arbitrary scale of 0–4 (0 = Nil [absent]; 1 = Minimal [1–10% area]; 2 = Mild [11-25% area]; 3 = Moderate [26-50% area]; 4 = Severe [50% < area]) was used for scoring in H&E, Masson trichrome and immunohistochemical staining. The results are presented in the form of summary incidence table for H&E and Masson trichrome stained skin sections and bar diagram for immunohistological localization grading.
Results
No signs of infection were noticed at SM-exposed site in any of the control and treated mice, and all the animals survived throughout the course of study.
Histopathology and tissue leukocytosis
Histopathological findings of routine H&E and Masson trichrome stains of skin sections from treated and control mice 3 and 7 days post SM exposure are presented in Table 3 and Figures 1 and 2.
Histopathological grading of mice skin sections after sulphur mustard (5 mg/kg) exposure in various treatment groups a

a Values represent the number of animals showing lesions/total number of animals (severity).
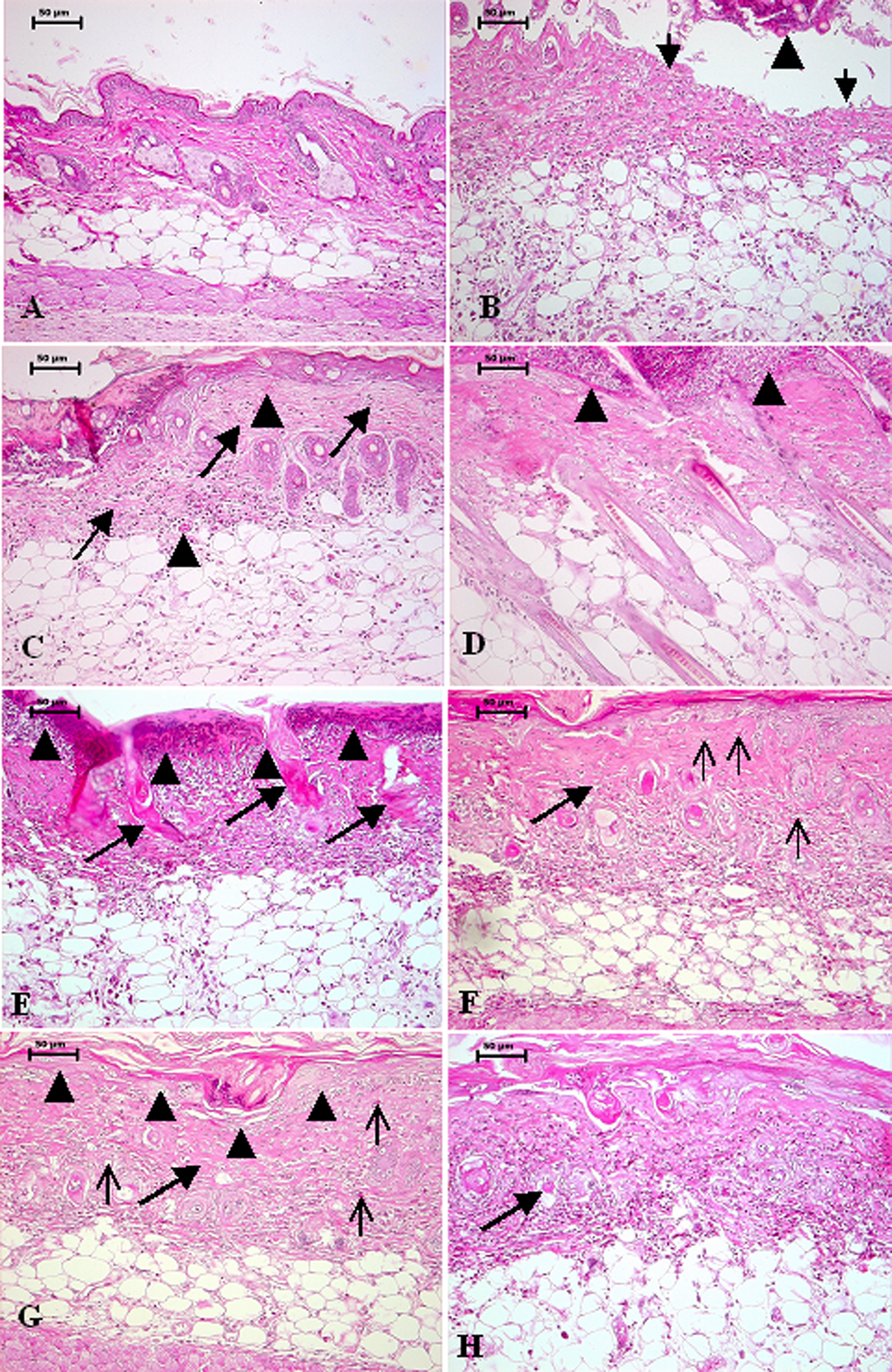
Photomicrograph of mouse skin with and without treatment, 3 or 7 days post SM (5 mg/kg, diluted in acetone) exposure, H&E (A) Control mice skin without SM exposure showing normal epidermis consisting of stratified epithelium, dermis and hair follicle. (B) Section of mice skin 3 day post SM exposure (without any treatment) showing sloughed off necrosed epidermis (arrowhead) leading to ulcer formation (arrow). (C) Section of mice skin, 3 day post SM exposure treated with DRDE/WH-02. Note homogenous distribution of immature fibroblast and collagen throughout the dermis (arrow), normal hair follicles and angiogenesis (arrowhead). (D) Section of mice skin, 3 day post SM exposure treated with soframycin. Note mild proliferation of fibroblast and collagen in upper reticular dermis and degenerated hair follicles and epidermis (arrowhead). (E) Section of mice skin showing coagulative necrosis of epidermis extending into the dermis (arrow head), follicular degeneration (arrow), necrosed inflammatory cells entrapped in fibrinous exudates along with edema noticed by separation of dermal tissue, 7 days post SM exposure. (F&G) Section of mice skin, 7 day post SM exposure treated with DRDE/WH-02. Note homogenous distribution of proliferated fibroblast and collagen throughout the dermis (arrow) and complete re-epithelialisation (arrow head in G) along with angiogenesis (thin arrow). (H) Mice skin section 7 days post SM exposure treated with ovadine. Note proliferation of fibroblast and collagen in upper reticular dermis and degenerated hair follicles (arrow).

Photomicrograph of mouse skin with and without treatment, 3 or 7 days post SM (5 mg/kg) exposure (A, B, C, D, E, F) Masson's trichrome stain and (G, H) Toluidine blue stain (A) Control mice skin without SM exposure showing normal collagen fibroblast in extracellular matrix of dermis. (B) Mice skin section 3 day post SM exposure without any treatment showing minimal collagen (in blue colour). (C) Mice skin section 3 days post SM exposure treated with ovadine showing same collagenisation as showed by SM control. (D) Mice skin section 3 days post SM exposure treated with DRDE/WH-02. Note homogenous proliferation of fibroblast collagen throughout the dermis, which is comparatively higher than other treatment groups. (E) Section of mice skin, 7 day post SM exposure without any treatment showing mild collagen in blue colour. (F) Section of mice skin, 7 day post SM exposure treated with DRDE/WH-02 showing complete reepithelialisation and normal extracellular matrix deposition comparable to normal control as shown in A. (G) Section of mice skin 3 days after SM exposure with DRDE/WH-02 treatment showing three metachromatic basophils (arrow). (H) Section of mice skin 7 days after SM exposure with DRDE/WH-02 treatment showing two metachromatic basophils (arrow).
Microscopic observation of unexposed mice skin showed normal architecture (Figures 1A and 2A). SM-exposed untreated mice skin showed severe necrosis of superficial epidermal cells, vacuolation and necrosis of basal cells and dermoepidermal separation. Some sections showed sloughing off necrotic epidermal cells with fibrinous exudate entrapping necrotic inflammatory cells leading to the formation of ulcer roofed by eschar tissue (Figure 1B). Increased thickness of epidermis was evidenced in the area circumscribing necrosis. This increased thickness circumscribing the ulcer was characterized by increase in the number of proliferating basal cells, which were arranged in three to five layers and showed pyknosis along with cytoplasmic swelling. Severity and incidence of occurrence of lesions was substantially reduced in mice treated with DRDE/WH-02 followed by Ovadine treatment on 3 and 7 days post SM exposure compared with SM control. Proliferating cells seem to migrate over the roof of ulcer under the eschar tissue to restore the damaged epidermis (re-epithelialization), 3 and 7 days post SM exposure. Broad re-epithelialization was noticed 7 days post SM exposure in mice treated with DRDE/WH-02 (Figure 1G) and Aloe gel, compared with other treatment groups and control, which showed narrow proliferation pattern of epidermal cells.
Minimal-to-mild proliferation of endothelial cells representing the process of angiogenesis was evident in all groups (Figure 1) irrespective of the treatment 3 days post SM exposure. However, there was a marked increase in newly developed blood vessels after DRDE/WH-02 treatment (Figure 1F and G), 7 days post SM exposure compared with other groups and SM control.
Necrosis of various grades was observed in external root sheath, matrix, internal root sheath and other supporting parts of hair follicle along with the sebaceous glands, 3 and 7 days after SM exposure in different treatment groups and SM control mice skin. Severity of adenexal atrophy was distinctly reduced with DRDE/WH-02 treatment compared with SM control mice skin 3 or 7 days after SM exposure. Proliferation of follicular cells was evidenced from healthy or apparently healthy hair follicle of mice skin with or without treatment 3 or 7 days post SM application. These cells from outer sheath wall of hair follicle migrated upwards for the restoration of lost epidermis. Restoration of epidermis (re-epithelialization) from outer sheath of hair follicle was a striking finding in DRDE/WH-02-treated mice skin compared with other treatment groups 3 and 7 days post SM exposure (Figures 1G and 2F).
H&E- and Masson trichrome-stained sections showed minimal-to-mild fibroblast proliferation and collagen synthesis 3 days post SM exposure in all mice irrespective of treatments (Figure 2). However, marked fibroblast proliferation was noticed in DRDE/WH-02-treated group compared with SM control and other groups (Figure 2D). Ovadine and Soframycin showed better fibroblast proliferation compared with SM control 7 days post SM exposure. Collagen was restricted to lower reticular dermis in SM control mice (Figure 2). However, mice with DRDE/WH-02 treatment showed diffused collagen proliferation (Figures 1F, G and 2F), which was evenly distributed throughout the dermis. In contrast to this, collagen was restricted to upper reticular dermis in Ovadine- (Figure 1H) and Soframycin (Figure 1D)-treated groups.
Minimal-to-mild edema of dermal interstitium was noticed in SM-exposed mice skin sections with or without treatment. The number of infiltrating leukocytes (neutrophils and monocytoid cells) was significantly higher in DRDE/WH-02-treated mice skin compared with other treatment groups and SM control group 3 days after SM exposure (Table 4). Skin sections from mice, 7 days post SM exposure with DRDE/WH-02 treatment, showed significant increase in the number of monocytoid cells compared with mice in Soframycin-treated, A. vera gel-treated and SM control group. There was no change in the number of basophils, 3 or 7 days post SM exposure.
Leukocyte response after sulphur mustard (5 mg/kg) application on the mouse skin a

SM: sulphur mustard.
a Same superscripts on values in column (on same day) indicate nonsignificant difference between different treatment groups at p < 0.05.
Histomorphologic scoring
Grading on histomorphologic scale showed significantly high scores in DRDE/WH-02-treated mice, 7 days post SM exposure followed by A. vera gel-, Ovadine- and Soframycin-treated groups compared with SM control. Scores of all the SM-exposed groups with or without treatment were significantly lower when compared with scores of normal skin (Figure 3).

Histomorphologic scoring of SM (5 mg/kg) induced skin lesions treated with various formulations (A). 3 days after SM exposure (B). 7 days after SM exposure. Same alphabets on bars indicate non-significant difference between different treatment groups at P < 0.05. Value are Mean ± S.E. n = 6.
TUNEL assay
Occasional presence of TUNEL-positive apoptotic cells was noticed in skin of unexposed mice; whereas, there was a substantial increase in the number of apoptotic cells 3 days after SM exposure (Figure 4). There was a significant reduction in apoptotic cells 3 days post SM exposure in DRDE/WH-02-treated mice skin compared with other groups. No change in TUNEL-positive cells was observed in SM control and other treatment groups 7 days post SM (5 mg/kg) exposure. Histopathological features of cells were used to differentiate apoptosis from necrosis. In apoptosis, the cells showed nuclear chromatin condensation along with fragmentation and multiple cytoplasmic protrusions; however, in necrosis, there was cloudy swelling of the cells prior to death including vacuolization.

(A, B) TUNEL index of SM (5 mg/kg) induced skin lesions treated with various formulations taken on day 3 and 7 respectively. Same alphabets on bars indicate non-significant difference between different treatment groups at P < 0.05. Value are Mean ± S.E. n = 6. (C) TUNEL positive cells (arrowhead) in epidermis of mice skin section 3 days after SM exposure without any treatment. (D) TUNEL positive cells in epidermis of mice skin section (arrowhead) 3 days after SM exposure with DRDE/WH-02 treatment. Note the decreased number of apoptotic cells.
Immunohistological localization
Semiquantitative immunohistologic grading and immunohistological localization of FGF, eNOS, IL-6 and TGF-α 3 and 7 days post SM (5 mg/kg) exposure are shown in Figures 5 –8. Unexposed mice skin showed expression of IL-6 and TGF-α restricted to epidermis and outer wall of hair follicles, while eNOS was localized in vascular endothelium. In SM-exposed mice skin, immunoreactivity of FGF, IL-6 and TGF-α was evident in the interstitium of skin by the proliferating fibroblast and infiltrating inflammatory cells. Epidermal cells showed immunoreactivity with IL-6 and TGF-α, whereas skin interstitium showed diffused expression of eNOS by macrophages and proliferating and resting vascular endothelium. There was a marginal increase in immunoreactivity of FGF and IL-6 in mice skin 3 days post SM exposure, which increased markedly after 7 day of DRDE/WH-02 treatment compared with other treatment groups and SM control. There was no treatment-related change in immunoreactivity of eNOS 3 days post SM exposure compared with SM control. However, marked increase in eNOS immunoreactivity was noticed in DRDE/WH-02-treated mice, 7 days post SM exposure compared with control and other groups. Significant increase in immunoreactivity of TGF-α was noticed in skin sections from DRDE/WH-02-treated mice, 3 and 7 days post SM exposure compared with other treatment groups and SM control.

(A, B) Semiquantitative immunohistological grading of FGF expression in mice skin sections, 3 and 7days post SM (5 mg/kg) application treated with various formulations. (C) Negative control (without primary FGF antibody). (D) 7 days post SM exposure without any treatment showing immunoreactivity of FGF in upper dermis and in migrated leukocytes. (E) 3 days post SM exposure with DRDE/WH-02 treatment showing increased immunoreactivity of FGF in proliferating fibroblast (arrow). (F) 7 days post SM exposure with DRDE/WH-02. Note increased immunoreactivity of FGF in proliferating fibroblast compare to D&E.
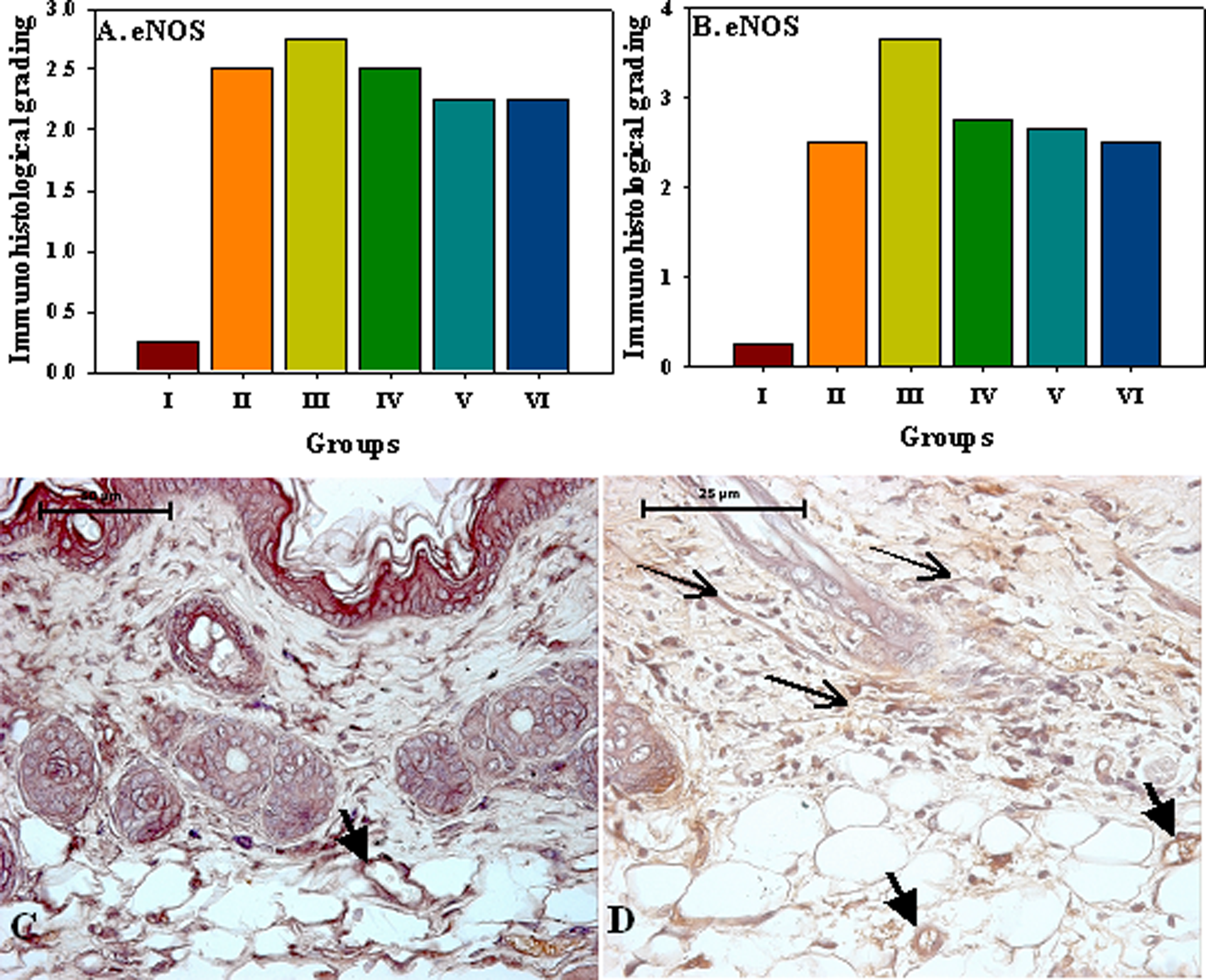
(A, B). Semiquantitative immunohistological grading of eNOS expression in mice skin sections, 3 and 7days post SM (5 mg/kg) application treated with various formulations. (C) eNOS immunoreactivity with vascular endothelium (arrow) in normal control mice without SM exposure (D) increase in eNOS immunoreactivity with vascular endothelium (arrow) and in dermal interstitium (thin arrow) 7 days after SM exposure.

(A, B). Semiquantitative immunohistological grading of IL-6 expression in mice skin sections, 3 and 7days post SM (5 mg/kg) application treated with various formulations. Photomicrograph of mouse skin with and without treatment post SM (5 mg/kg) exposure showing immunoreactivity with IL-6 (C) Normal control mice without SM exposure showing minimal streaks of immunosignals in epidermal cells (arrow). (D) 3 days after SM exposure without any treatment showing IL-6 positive signals by epidermal cells, hair follicles (arrow) and few transmigrated inflammatory cells. (E) 3 days after SM application showing IL-6 positive signals by few transmigrated inflammatory cells (arrow) and by some epidermal keratinocytes comparatively more than SM control as shown in photomicrograph D (F) 3 days after SM exposure and treatment with DRDE/WH-02 showing IL-6 positive signals by epidermal cells (arrow heads), hair follicles (arrow) and few transmigrated inflammatory cells comparatively more than SM control as shown in photomicrograph D & E.
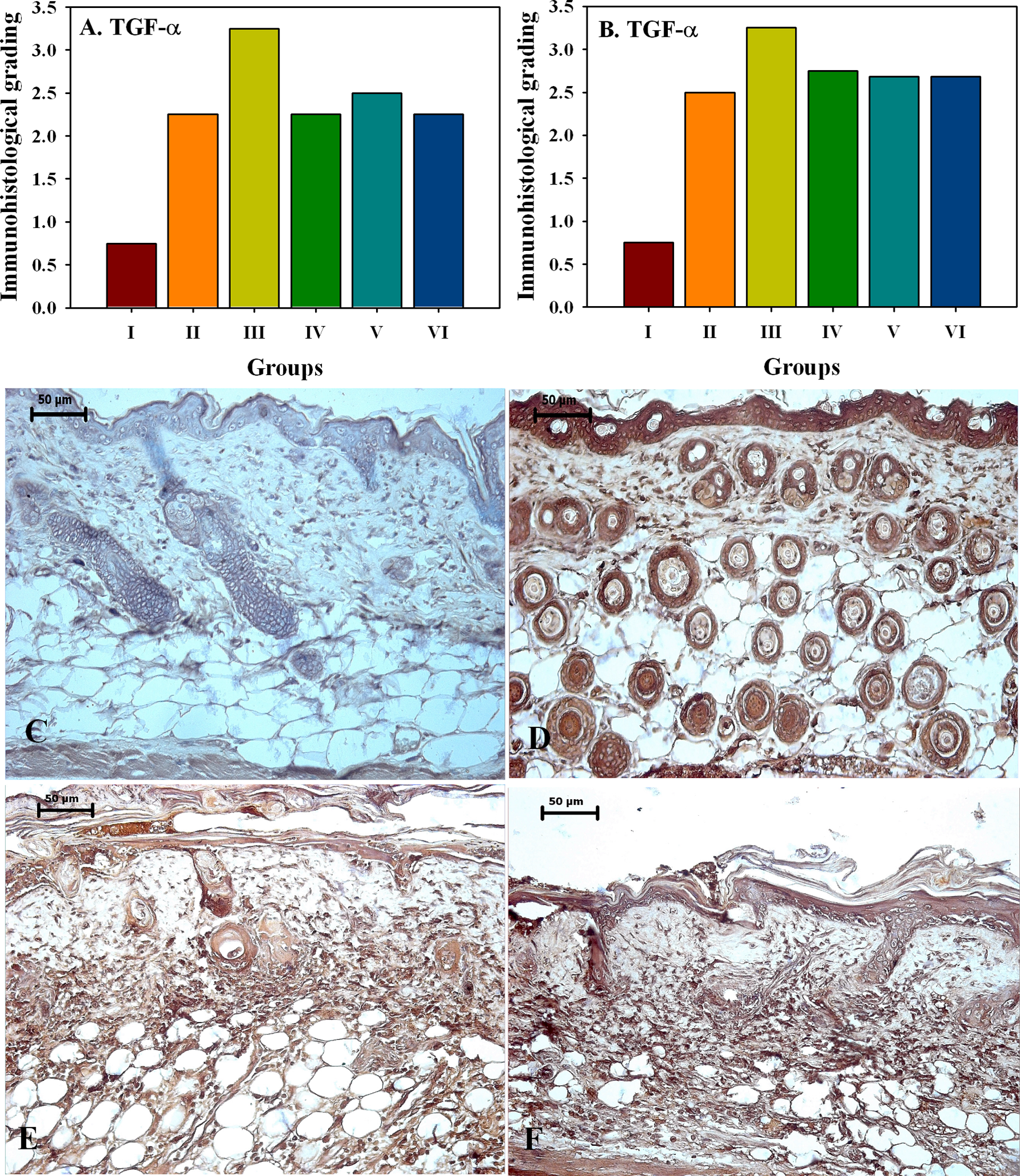
(A, B). Semiquantitative immunohistological grading of TGF-? expression in mice skin sections, 3 and 7days post SM (5 mg/kg) application treated with various formulations. Localization of TGF-α in mouse skin with and without treatment, 3 and 7 days post SM (5 mg/kg) (C). Negative control mice skin without SM exposure showing no immunosignals. (D) Control mice skin without SM exposure showing TGF-α immunosignals by epidermal keratinocytes and outer layer of follicular cells. (E) SM (5 mg/kg) exposed mice skin with DRDE/WH-02 treatment, after 3 days showing moderate immunosignals of TGF-α in dermis by migrating fibroblasts and inflammatory cells. (F) SM (5 mg/kg) exposed mice skin with DRDE/WH-02 treatment, after 7 days showing extensive immunosignals of TGF-α in dermis compared to sections of day 3 as shown in E.
Discussion
Development of an antidote against SM is still awaited, although possible threat from various sources to human and animals and risk during destruction of stockpiled mustard agent has imposed the need of suitable wound healant as a prime requirement. In view of this, present study was carried out for evaluating preclinical wound healing efficacy of DRDE/WH-02 in female mice. As per the OECD (Organisation for Economic Co-operation and Development) test guideline 425, there is little difference in sensitivity between sexes, but females are generally slightly more sensitive than male and hence normally females should be used in toxicity studies. Moreover, males are aggressive and fight with each other and produce mechanical wounds that may interfere in judging the efficacy of the wound-healing formulation. Thus, female mice were employed in the present study.
Wound healing is a biological process triggered by tissue injury and directed towards the restoration of tissue continuity and its function. The early phase of healing process is inflammation followed by fibroplasia and re-epithelialization and finally tissue remodeling. 26 These phases overlap and their separation is arbitrary. During the past, a vast amount of new information concerning cellular and molecular mechanism have been documented that has a direct bearing on the understanding of SM-induced skin injury, inflammation and healing processes in various animal models. However, these findings provided partial information due to the limitation of the availability of physiologically normal animal models for SM-induced skin injury. In SM injury to human skin, recognizable skin pathology does not usually occur for several hours to a day after exposure. 27 Oral pretreatment with drugs against SM has been studied in experimental models for accessing their protective efficacy. 5,20 However, reports on topical pretreatment with drugs for protection against SM-induced skin injuries are lacking. We have conducted this study in newly developed physiologically normal animal model as described earlier, 23 confirming reliability and reproducibility of the results and their subsequent implementation for clinical subjects. Furthermore, results of present study confirm the general pattern of SM-induced skin lesions as described by us 23 in the same animal model and in other mammalian species. 4,12,25,28 –35
It was a common belief that the stronger bactericidal effect of an antiseptic agent has more deleterious effect on living tissue, and earlier studies have shown impaired wound healing with the use of iodine. 36 ,37 Contradictorily, Mayer 38 showed that PVP-I ointment when used in conjunction with the newer gel-type occlusive dressings enhances the healing process. Povidone iodine has been reported to enhance angiogenesis and is a potent microbicidal. 13 A. vera is another important ingredient, which is widely used for its healing, soothing and moisturising qualities. A. vera augments re-epithelialization and fibroplasia, thereby increasing the healing rate of mechanical wounds. 39 In the present study, combination of PVP-I, A. vera gel and betaine in DRDE/WH-02 showed increased intensity of re-epithelialization, fibroplasia and angiogenesis over other treatment groups in SM-induced skin lesions. Present results are in agreement with Wormser et al. 15 who proposed povidone-iodine ointment as an efficient protective agent against skin toxicity caused by mustards and other alkylators in guinea pig.
IL-6 has been identified as a key in inflammatory cell recruitment and activation at the site of injury 40 and in proliferation and remodelling phases of wound healing by promoting collagen deposition and angiogenesis. 41 Further, neutrophils acts by killing and phagocytosing bacteria and damaged matrix proteins within the wound bed. 42 Monocytes transmigrate into tissue spaces and transform into macrophages that soon become the predominant cell type during the latter part of the inflammatory phase. Macrophages phagocytize, digest and kill pathogenic organisms, scavenge tissue debris and destroy any remaining neutrophil. These all-important processes performed by the monocyte/macrophage allow for induction of angiogenesis and formation of granulation tissue. 42 Increased immunoreactivity of IL-6 along with increase in the number of leukocytes in DRDE/WH-02-treated mice compared with other groups and SM control in present study suggest better wound-healing property of this formulation. Moore 43 suggested that the delivery of iodine in the chronic wound induces influx of macrophages and T helper cells, which are considered to play a positive role in modulating wound healing. The increased influx of tissue leukocytosis in the present study might have resulted in early removal of tissue debris at faster rate, ultimately paving way for the initiation of reparative process.
The proliferative phase in the process of wound healing is characterized by increase in the number of immature fibroblast. This cell of mesenchymal origin is responsible for producing the new matrix needed to restore structure and function of the injured tissue. Fibroblasts attach to the cables of the provisional fibrin matrix and begin to produce collagen. 44 In this study, collagen was restricted to lower reticular dermis in SM-exposed mice without any treatment. However, DRDE/WH-02-treated mice showed diffused collagen proliferation, which was distributed homogenously throughout the dermis. In contrast to this, collagen was restricted to upper reticular dermis in Ovadine- and Soframycin-treated groups. A. vera gel in the present formulation in DRDE/WH-02 might have participated in collagen synthesis and its subsequent maturation in the healing process of SM-induced skin lesion. These findings are supported by the results of Takzare 19 in mechanical wounds, Somboonwong 16 in burn wounds of rats and Gautam et al. 20 in protection studies of SM-induced skin lesions of mice. Furthermore, findings of Subramanian 17 and Chithra 18 supports wound healing activity of A. vera in terms of fibroblast proliferation and promotion of epithelialization and collagen synthesis.
Results of the decision analysis helped in comparing all treatments on histomorphologic scale listed from the most efficacious to least efficacious formulation. DRDE/WH-02-treated mice received significantly highest score followed by A. vera gel, Ovadine, and Soframycin with similar scores and SM control showed least score.
SM-induced apoptosis of basal keratinocytes was reported earlier45,46 and was evident during SM-induced skin injury in our studies. Histological analysis of SM-treated skin sections in the present study demonstrated that epidermal thickening was associated with cytoplasmic swelling of epidermal cells and cell consisting of condensed nuclei, which indicated pyknosis and apoptotic cell death. These findings were confirmed by TUNEL assay. It can be speculated that the SM-induced apoptosis, observed in the present study, is due to the DNA damaging effect of SM as suggested by various other reports.11,46 –48 In the present study, DRDE/WH-02-treated mice showed significant decrease in the number of apoptotic cells compared with SM control and other treatment groups. However, antiapoptotic mechanisms of the formulation or its component need further investigation.
Morbidelli et al. 49 and Ziche et al. 50 reported that nitric oxide mediates the effects of vascular endothelial growth factor, an important mitogen for vascular endothelial cells, which is essential for angiogenesis. There was increased expression of eNOS in transmigrated inflammatory cells, macrophages and fibroblasts during healing of SM-induced skin injury in mice treated with DRDE/WH-02. Interestingly, there was a pronounced increase in the newly developed blood vessels sustaining the higher expression of eNOS after DRDE/WH-02 treatment, indicative of its beneficial role in wound healing.
TGF-α leads to keratinocyte migration, re-epithelialization and angiogenesis. FGF modulates fibroblast proliferation and its migration into damaged tissue for the restoration of lost extracellular matrix and it further stimulates angiogenesis and affects cell differentiation. 42 ,51 DRDE/WH-02-treated mice showed higher immunoreactivty with TGF-α and FGF supported by increased proliferation of fibroblast, improved re-epithelialization and enhanced angiogenesis compared with control and other groups in this study suggesting its beneficial role in wound healing. However, further gene expression studies are required to open the molecular mechanism of wound healing by DRDE/WH-02 in SM-induced skin lesions.
In summary, semiquantitative estimation of re-epithelialization, fibroplasias and neovascularization by histomorphologic scoring along with biological markers of inflammatory and reparative stages have revealed that DRDE/WH-02 provided better healing effect over other formulations in dermatotoxicity caused due to SM exposure. Thus, DRDE/WH-02 can be recommended as an effective wound healant against SM-induced skin injury.
Footnotes
Acknowledgements
The authors thank Dr. S.J.S. Flora, Head, Pharmacology and Toxicology Division, for his support and suggestions and also Dr. K. Ganesan for synthesis and analysis of purity of sulphur mustard.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
