Abstract
Little is known about effects of alcohol consumption on dendritic cell (DC) function and resultant immune response. However, quantitative and qualitative disturbances of DCs are speculated to be involved in alcohol-related as well as in other liver pathology. The present study aimed to evaluate changes in circulating DC subsets in alcoholic liver disease (N = 43), autoimmune hepatitis (N = 26) and primary biliary cirrhosis (N = 20). DCs isolated from the peripheral blood of recruited participants were stained with monoclonal antibodies against blood dendritic cell antigens (BDCAs) and estimated using the flow cytometry. Myeloid DCs were defined as BDCA-1+/CD19− cells, and lymphoid DCs as BDCA-2+/CD123+ cells. Total numbers of circulating DCs in subjects with some liver diseases were markedly lower than in the healthy participants (p = 0.03). There was a significantly lower percentage of circulating BDCA-2+/CD123+ (p = 0.02), and a tendency for the percentage of circulating BDCA-1+/CD19− cells to decrease in patients with liver diseases compared to the controls (p = 0.09). These results may suggest that decreased numbers of DCs may be responsible for reduced adaptive immune responses and increased susceptibility to infections and cancer development observed in patients exposed to alcohol. Moreover, numerical abnormalities of DCs may contribute to the breakdown of self-tolerance, a feature of autoimmune diseases.
Keywords
Introduction
Alcoholic and autoimmune liver diseases are known to be associated with the immune system dysfunction. Reduced adaptive immune responses and increased susceptibility to infections and cancer development observed in individuals exposed to alcohol are probably related to the status of ethanol intake and existence of liver injury. 1,2 Acute alcohol exposure has been linked to reduced antigen presentation by monocytic cells and lower secretion of inflammatory cytokines. 3–6 In turn chronic alcoholism has been considered to result in T cell activation, higher secretion of inflammatory cytokines by monocytes and upregulation of costimulatory molecules on macrophages. 7–9 As for autoimmune liver diseases, the nature of underlying self-tolerance breakdown still remains an enigma. Autoantibodies, T cell infiltration in the portal tracts and hepatic parenchyma, and destruction of the hepatocytes and biliary epithelia reflect dysregulated immune mechanisms. 10–12
Dendritic cells (DCs) are considered to be central regulators of immune responses. These cells are highly specialized for uptake, processing and presentation of foreign and self-antigens, both to naive and to antigen-specific T lymphocytes. 13,14 They are also presumed to participate in the immune tolerance. 15,16 The substantial heterogeneity of DCs has been demonstrated, and it is probably akin to differences in their function. 17 How exactly DC phenotype correlates with their function, and how complex interactions between DCs and T cells shape the immune response in vivo have not been fully characterized yet. However, of two main circulating DC subsets, myeloid peripheral blood DCs are usually referred as pivotal activators of naive T cells in a primary antigenic response, whereas lymphoid peripheral blood DCs are strongly implicated in the peripheral tolerance and events in the course of atopy, viral infection or cancer development. 13,14 Furthermore, the latter are regarded as immunomodulating cells, and both subsets are potent regulators of the cytokine milieu for T cell activation and differentiation. 13,18
Given the key role of DCs in the immune system, their abnormalities may underlay a variety of diseases. The aim of the current study was to assess the changes in populations of circulating DCs in various liver disorders, including alcoholic liver disease (ALD), autoimmune hepatitis (AIH) and primary biliary cirrhosis (PBC). The respective experiments were among the first attempts to clarify an impact of chronic alcohol consumption on human DCs in vivo.
Material and methods
Study population
The study comprised 89 newly diagnosed patients with ALD (N = 43), AIH (N = 26) and PBC (N = 20) hospitalized in the Gastroenterology Department of the Medical University of Lublin. In parallel, 19 age-matched healthy volunteers with no history of chronic alcohol use, liver disease or any autoimmune processes were recruited (the controls).
Diagnosis of ALD was based on the history of chronic alcohol consumption, and ascites, hepatic encephalopathy or variceal bleeding, and the presence of other symptoms and signs of chronic liver disease, as well as on gastroscopic and/or ultrasonographic findings. None of the patients had acute alcoholic hepatitis or decompensated cirrhosis at the time of enrolling. Alcohol use habits of the study participants were determined using a health questionnaire. 19 The average alcohol consumption in ALD group was required to be ≥90 g of EtOH/day. The inclusion criteria to the AIH group combined clinical, laboratory (i.e., high levels of serum aminotransferases and immunoglobulin G, and the presence of serum antinuclear antibodies) and histologic components and were consistent with the international guidelines and scoring system for AIH diagnosis codified by International Autoimmune Hepatitis Group. 20,21 PBC subjects were required to meet the established criteria for the disease diagnosis, including high serum levels of alkaline phosphatase and immunoglobulin M, the presence of serum antimitochondrial or anti-M2 antibodies, and specific liver biopsy histology. 22 Patients recruited to the AIH, PBC and control groups had to satisfy no excess alcohol intake criterion (i.e., the average alcohol consumption <15 g of EtOH/day). None of study participants had viral hepatitis or HIV infection, or had been taking any drug known to be hepatotoxic or to affect evaluated immunological parameters (e.g., steroids or immunosuppressants). Other exclusion criteria were comorbid liver conditions, any ongoing infection, malnutrition or history of blood transfusion. The data on age, sex and clinical characteristics of the study population with regard to the etiology of the liver disorder are detailed in Table 1.
Age and sex distribution and clinical characteristics of the study population (N = 108) a
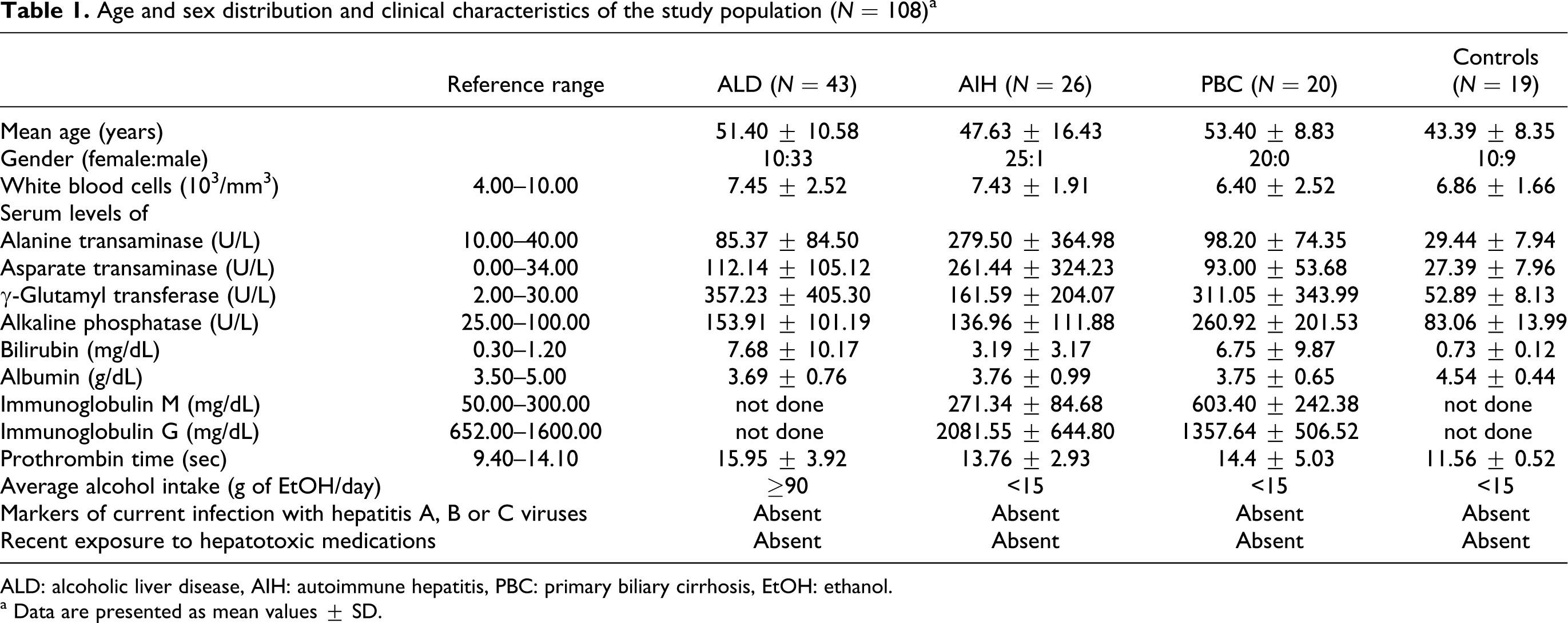
ALD: alcoholic liver disease, AIH: autoimmune hepatitis, PBC: primary biliary cirrhosis, EtOH: ethanol.
a Data are presented as mean values ± SD.
The study protocol, being in accordance with the ethical standards of the revised Helsinki Declaration, was approved by the Human Subjects Ethics Committee at the Medical University of Lublin. Following detailed explanation of nature and purpose of the study, written informed consent was obtained from each participant.
Blood sampling, cell preparation and phenotyping
Blood samples were taken by venepuncture into sterile, heparinized tubes after an overnight fast. Mononuclear cells were immediately isolated by density gradient centrifugation on Lymphoprep (Nycomed, Norway) at 400g at room temperature for 20 min. Interphase cells were removed and washed twice in phosphate-buffered saline without Ca2+ and Mg2+ containing 0.5% bovine serum albumin and 2 mM EDTA. The cells were then counted with a hemocytometer and resuspended in the mentioned buffer. Subsequently, the double-color immunofluorescent staining was performed according to the manufacturers’ protocols using monoclonal antibodies: mouse anti-human blood dendritic cell antigen (BDCA)-1-FITC (Miltenyi-Biotec, Germany), CD19-CyChrome (Pharmingen, USA), BDCA-2-FITC (Miltenyi-Biotec, Germany), and CD123-PE (Becton Dickinson, USA). Mouse anti-human IgG2a isotype control was used for anti-BDCA-1 staining. Mouse anti-human IgG1 isotype control was used for anti-BDCA-2 staining. Briefly, the cells (aliquots of ≤1×107 cells) were incubated with optimum amounts of monoclonal antibodies in the dark at 4°C for 10 min, then washed in the buffer and centrifuged at 300g and 4°C for 10 min. To minimize nonspecific FcR (Fragment crystallizable receptor)-monoclonal antibody binding, the immunostaining was performed in the presence of FcR blocking reagent (Miltenyi-Biotec, Germany).
Flow cytometric analysis
The immunolabeled cells were collected (300,000 events) using a FACSCalibur flow cytometer equipped with 488-nm argon laser (Becton Dickinson) and analyzed with Cell-Quest Software. Cell debris and dead cells were excluded from the analysis based on scatter signals. The number of DCs was expressed as the percentage of mononuclear cells. Peripheral blood DCs were identified by unique expression of cell surface markers. Namely myeloid DCs were defined as BDCA-1-positive and simultaneously CD19-negative cells (BDCA-1+/CD19−), whereas lymphoid DCs were defined as double BDCA-2- and CD123-positive cells (BDCA-2+/CD123+), as described by Dzionek et al. 23 Cytometric differentiation of circulating myeloid and lymphoid DCs of selected study participants is presented in Figure 1.
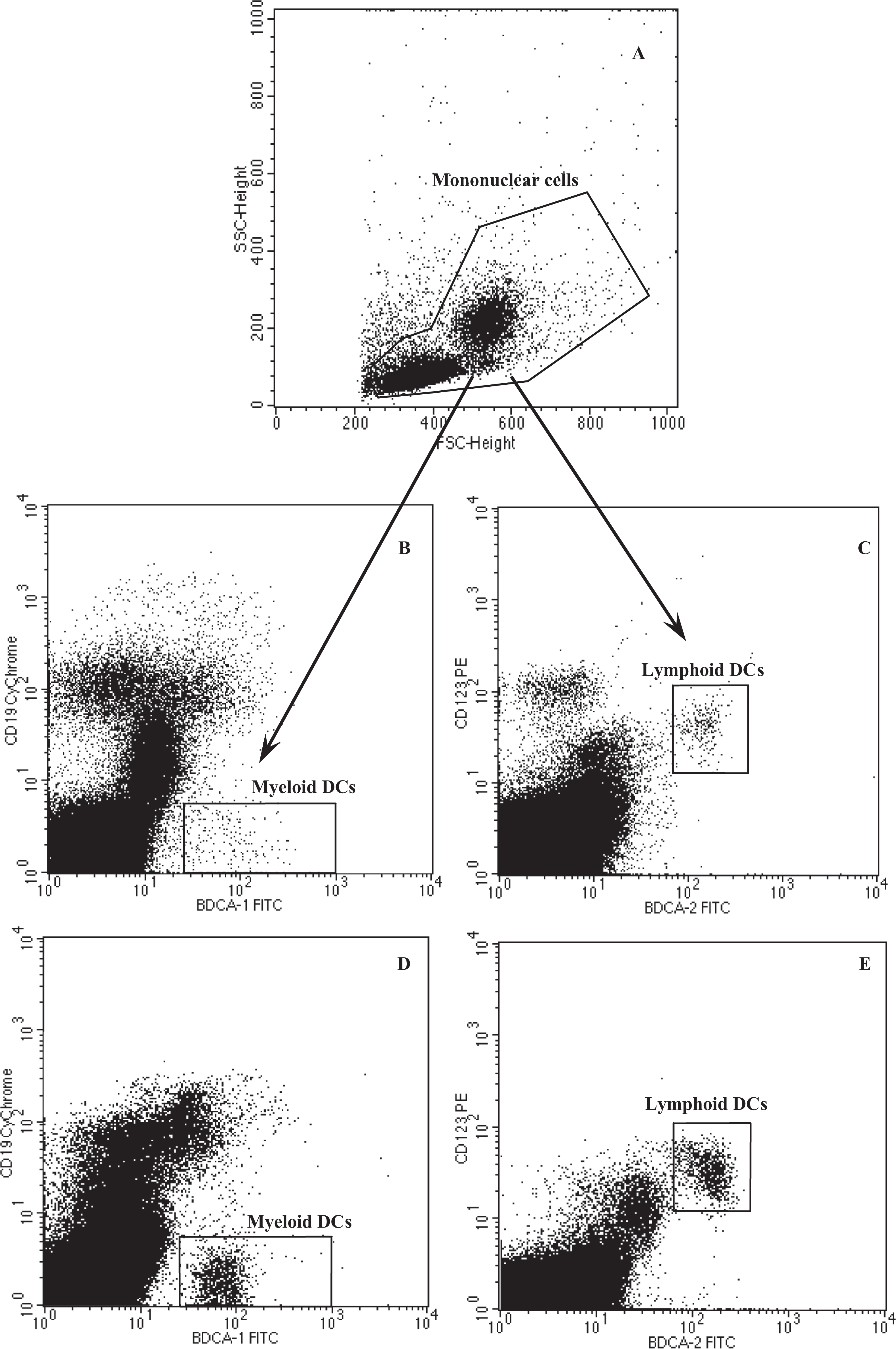
Flow cytometric analysis of circulating DCs of one patient with ALD (B, C) and one healthy donor (D, E). The mononuclear cell region, an example depicted in a scatter plot (A), was analyzed for BDCA-1 FITC and CD19 Cy-Chrome staining (dot plots B, D) to identify myeloid DCs defined as BDCA-1+/CD19− cells, and for BDCA-2 FITC and CD123 PE staining (dot plots C, E) to identify lymphoid DCs defined as BDCA-2+/CD123+ cells. Flow cytometry consistently demonstrated lower percentages of myeloid and lymphoid DCs in peripheral blood of patients with ALD compared with healthy volunteers. ALD: alcoholic liver disease; BDCA: blood dendritic cell antigen; DCs: dendritic cells; FITC: fluorescein isothiocyanate (fluorochrome); PE: phycoerythrin (fluorochrome); SSC: side scatter.
Statistical analysis
Data were analyzed using STATISTICA Data Analysis Software, version 8.0 (StatSoft, Inc. Poland). For variable distribution normality Shapiro-Wilk’s test was applied. Given the nonnormal distribution of involved variables, the differences between groups were assessed with Kruskal–Wallis test. Nonlinear correlation of coefficient between continuous variables was calculated using Spearman’s rank test. For all analyses, a probability value of p < 0.05 was considered statistically significant.
Results
Circulating DCs in ALD, AIH and PBC
The percentages of DC subsets in peripheral blood of the study groups are presented in Table 2. Significant differences in the numbers of circulating DCs were observed between examined groups (p = 0.02). Total numbers of DCs (myeloid DCs + lymphoid DCs) present in peripheral blood of patients with some liver disease were markedly lower than in healthy subjects (p = 0.03 for ALD and AIH groups vs. control group; and p = 0.06 for PBC group vs. control group; Figure 2). Patients with AIH displayed the lowest total number of circulating DCs compared to patients with other liver disorders. However, the differences were not statistically significant (p > 0.05). There was a trend toward slightly lower total number of DCs in autoimmune liver diseases (AIH or PBC) compared with ALD (p > 0.05).

Comparison of circulating DC subpopulations between the study groups. Data are presented as median and range (minimum–maximum). DCs: dendritic cells; ALD: alcoholic liver disease; AIH: autoimmune hepatitis; PBC: primary biliary cirrhosis, *: the number of DCs was expressed as the percentage of mononuclear cells, **: p < 0.05 versus controls.
DCs subpopulations in peripheral blood of the study groups a

DCs, dendritic cells; ALD, alcoholic liver disease; AIH, autoimmune hepatitis; PBC, primary biliary cirrhosis, BDCA, blood dendritic cell antigen.
a Data are presented as mean values ± SD and range (given in parentheses).
b The number of DCs was expressed as the percentage of mononuclear cells.
c p < 0.05 versus controls.
A detailed analysis of DC subsets revealed a tendency for the percentage of BDCA-1+/CD19− cells (i.e. myeloid DCs) to decrease in peripheral blood of patients with liver disease compared to healthy individuals, but the differences were not significant (p > 0.05; Figures 1 and 2). The lowest percentage of BDCA-1+/CD19− cells was observed in PBC group, but there were no marked differences in the parameter between the examined groups of patients with liver disorder of different etiology. In contrast, the differences in the percentage of circulating BDCA-2+/CD123+ cells (i.e. lymphoid DCs) between the evaluated patients and control subjects reached the level of statistical significance (p = 0.02). Namely, there was markedly lower percentage of lymphoid DCs in peripheral blood of patients with ALD (p = 0.02; Figure 1) and AIH (p = 0.03) in comparison with the healthy subjects, and there was an apparent, though statistically insignificant (p > 0.05), trend toward lower percentage of lymphoid DCs in patients with PBC compared to control group (Figure 2). The lowest percentage of circulating BDCA-2+/CD123+ cells was observed in AIH group and the highest in PBC group. The comparison of percentage of BDCA-2+/CD123+ cells in peripheral blood between patients with liver disease of different etiology revealed no significant differences.
As for the ratio of myeloid DCs to lymphoid DCs, no statistically significant differences were observed between the groups (p > 0.05; Figure 2). The myeloid DC/lymphoid DC ratios in ALD, AIH and control groups were comparable and estimated to be >1, which generally implied that predominant subset of circulating DCs were BDCA-1+/CD19− cells. In contrast, the changes in the percentages of the respective DC subsets in peripheral blood of patients with PBC resulted in the myeloid DC/lymphoid DC ratio <1, indicating the relative prevalence of BDCA-2+/CD123+ cells.
Correlations between circulating DCs and liver function tests in ALD, AIH and PBC
No relationships between the percentages of circulating BDCA-1+/CD19− and BDCA-2+/CD123+ cells and most liver function parameters were found. The only noted correlations were between the percentage of circulating myeloid DCs and asparate transaminase and γ-glutamyl transferase serum levels for patients with ALD (R = –0.33, p = 0.03 and R = –0.31, p = 0.04, respectively), and the percentage of circulating lymphoid DCs and albumin serum level in patients with AIH (R = –0.39; p = 0.04).
Discussion
The adverse immunomodulatory effects of the excess alcohol consumption on immune responses related to DC activation are an issue of ongoing research. Animal studies demonstrated that acute and chronic alcohol exposure affected numbers and allostimulatory capacity of these most potent antigen-presenting cells. 24–26 A few reports regarding the influence of alcohol on human DCs have been published recently. Buttari et al. 27 demonstrated that acute alcohol exposure disrupted differentiation and maturation of healthy human monocyte-derived DCs in response to lipopolysaccharide (LPS) in vitro and the obtained cells were incapable of priming and polarizing naive allogeneic T cells into Th1 cells. According to Szabo et al. 4,5 and Mandrekar et al. 28 moderate single alcohol intake was sufficient to downregulate CD80 and CD86 costimulatory molecules on healthy human monocyte-derived DCs and to decrease interleukin-12 (IL-12) levels and increase IL-10 levels in DC cultures. No abnormalities were observed with respect to cell morphology, apoptosis, viability or surface expression of human leukocyte antigen (HLA) Class I, HLA-DR, CD14, and CD1a molecules. Most importantly, abnormal costimulatory molecules expression on DCs correlated with defective antigen presentation. In vitro experiments demonstrated that DCs derived from monocytes obtained after acute alcohol exposure presented with significantly reduced allostimulatory capacity compared to DCs from the same individuals generated from alcohol-naive monocytes. 4,5,28 Furthermore, Mandrekar et al. 28 found that naive CD4+ T cells cocultured with alcohol-treated DCs produced significantly lower amounts of interferon-γ compared with CD4+ T cells cocultured with normal DCs.
So far only isolated studies evaluated potential DC defects in chronic alcohol consumption and related liver diseases. Buttari et al. 27 generated fewer CD1a+ cells from monocytes obtained from alcoholics. These immature DCs displayed weaker CD86 and higher HLA-DR expression, with resultant impairment of endocytosis and allostimulatory capacity compared to the cells generated from healthy subjects, but they still produced large amounts of tumor necrosis factors (TNF)-α and IL-6. Of note, LPS stimulation failed to induce their full phenotypical and functional maturation. 27 In research by Laso et al. 1 alcoholics without related liver disease presented with normal numbers of circulating myeloid, plasmocytoid and CD16+ DCs but with a more immature phenotype (reflected by decreased expression of the HLA-DR molecule) and with increased secretion of inflammatory cytokines, such as IL-1β, IL-6, IL-12, and TNF-α. It would be in contrast with the previously reported in vitro and in vivo inhibitory effect of acute alcohol exposure on the IL-12 secretion by DCs. 4,5,28 Importantly, IL-12 and resultant Th1 cytokines favor activation and expansion of CD8+ cytotoxic T cells that might contribute to ALD development. 29 In turn established alcohol liver cirrhosis was associated with decreased numbers of circulating DCs and reduced secretion of the inflammatory cytokines, even after ≥1 year of sobriety. 1 Consistently with the Laso et al., 1 the present study participants diagnosed with ALD displayed decreased numbers of circulating DCs. Unfortunately, the percentages of DC subpopulations were highly variable among individual patients, but the same was also observed among the controls. Given the literature data on the liver immunopathology, possible mechanisms that may underlie presented changes in numbers of peripheral blood DCs in patients with ALD may include their increased migration and compartmentalization to the liver, 1,30–32 trapping and destruction in the spleen, as it happens with other white blood cells and platelets in patients with advanced chronic liver disease, 1,33 and decreased bone marrow output. 1 The exact nature and mechanisms involved in abnormalities of DC subpopulations in patients with ALD still await elucidation.
The present study seems also to link DC aberrations and autoimmune liver diseases. Although DC abnormalities have been presented in isolated reports regarding the pathogenesis of AIH and PBC, the full extent of DC involvement in liver destruction due to autoimmune processes has not been fully explored so far. In a small study, Hiasa et al. 34 observed lower numbers of myeloid and plasmocytoid DCs in AIH or PBC patients, both treatment naive and receiving the specific therapy (corticosteroid or ursodeoxycholic acid), than in healthy subjects. However, as in the current study, the differences did not reach the statistical significance level, and there were no correlations between DC numbers and liver dysfunction. Of note, plasmocytoid DCs from treatment-naive AIH and PBC patients expressed significantly lower levels of HLA-DR and CD123 than the cells from the controls. 34 Another study reported significantly lower stimulatory capacity of DCs from patients with PBC compared with that of DCs from normal controls. 35 Interestingly, the expression of costimulatory and major histocompatibility complex molecules (CD86, HLA-DR) and cytokine production (IL-10 and IL-12) were not significantly different between PBC and healthy individuals. The observed functional defect of DCs correlated with increased production of nitric oxide by these cells. 35 Reduced circulating DC percentages in patients with liver diseases revealed in the present study are also consistent with the Wertheimer et al. 30 findings. Moreover, Wertheimer et al. 30 reported an inverse correlation between serum alanine aminotransferase and DC numbers in subjects with liver disease, and some relationships between the respective DC subsets and the liver function parameters were also found in the present study. Unfortunately, Wertheimer et al. 30 combined data from patients diagnosed as having various liver diseases, including viral hepatitis, PBC, primary sclerosing cholangitis, nonalcoholic fatty liver disease, alcohol cirrhosis, hemochromatosis, alpha-1 trypsin disease or cryptogenic cirrhosis. The nature of dysregulated immune mechanisms observed in patients with autoimmune liver diseases is elusive, but the present research coupled with recently published studies suggests that abnormalities in quantity and function of DC subsets might contribute to the autodestructive immune processes within the liver.
Conclusions
The changes in the percentages of the circulating DC subsets found in the current study shed light on the role of these cells in the modulation of immune functions by alcohol. Importantly, decreased numbers of DCs may be reflected by reduced adaptive immune responses and increased susceptibility to infections and cancer development, observed in patients exposed to alcohol. Moreover, the study implicates DCs in the pathogenesis of AIH and PBC, as their numerical abnormalities may contribute to the breakdown of tolerance to self-antigen—a feature of autoimmune diseases. Further studies to establish the probable pathogenic links and elucidate a possible chain of cause and effect between DCs and respective liver disorders are warranted.
Footnotes
This article is presented in part as a poster at the 44th Annual Meeting of the European Association for the Study of the Liver (EASL), April 22-26, 2009, Copenhagen, Denmark.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declared no conflicts of interest.
