Abstract
The present study investigated the protective effect of kolaviron, a biflavonoid from the seed of Garcinia kola, on ethylene glycol monoethyl ether (EGEE)–induced reproductive toxicity in male rats. The protective effect of kolaviron was validated using vitamin E, a standard antioxidant. EGEE was administered at a dose of 200 mg/kg. Other groups of rats were simultaneously treated with kolaviron (100 and 200 mg/kg) and vitamin E (50 mg/kg) for 14 days. EGEE treatment resulted in significant decrease in glutathione (GSH) level, superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) activities but markedly increased the glutathione-S-transferase (GST) and lactate dehydrogenase (LDH) activities in the testes. In the spermatozoa, administration of EGEE caused significant decrease in the activities of CAT, GPx, GST and LDH as well as in the level of GSH but significantly increased SOD activity with concomitant increase in hydrogen peroxide and malondialdehyde levels in both testes and spermatozoa. EGEE-exposed rats showed marked testicular degeneration with concomitant decrease in spermatozoa quantity and quality. Overall, EGEE causes reproductive dysfunction in rats by altering antioxidant systems in the testes and spermatozoa. Kolaviron or vitamin E exhibited protective effects against EGEE-induced male reproductive toxicity by enhancement of antioxidant status and improvement in spermatozoa quantity and quality.
Introduction
Ethylene glycol monoethyl ether (EGEE) is used in a variety of industrial and house products. It is used in a number of paints, varnishes, resins, engine fuels, hydraulic fluids and also in many household products including floor polishes and glass, leather and upholstery cleaners. 1 Up to date, EGEE is used as a raw material in Nigeria, where most operations such as mixing and cleaning in the factories are done manually with 8-h minimum exposure period daily. In addition, occupational painters and artisans that use products containing EGEE routinely carry out their work without gloves and respirators, thereby exposing such workers to dermal contact and absorption of EGEE.
The scale of infertility is overwhelming as a substantial amount of the global economy relies on environmental contaminants that are widely used in modern everyday life. The adverse effects of EGEE on male reproductive system include reduced testis weight and testicular degeneration in rabbits, 2 dramatic suppression of fertility and increased spermatozoa abnormalities in male mice 3 and caused significant decrease in the total and progressive motility of spermatozoa in both the cauda epididymis and spermatozoa ducts of rats. 4 In humans, reduced spermatozoa counts and testicular atrophy have been observed following occupational exposure to EGEE. 5,6 Recent studies from our laboratory demonstrated that exposure to EGEE induced oxidative damage in the testes and spermatozoa in rats. 7 Free radical production and lipid peroxidation from exposure to environmental contaminants 8 have been identified as potentially important mediators in testicular physiology and toxicology 9 and male infertility 10 in rodents 11 and humans. 12
Garcinia kola is a medium-sized tree found in moist forest and widely distributed throughout West and Central Africa. The nut is highly valued in these countries for its edible nuts. 13 The seed commonly known as bitter kola is a masticatory and is a major kola substitute offered to guests at home and shared at social ceremonies. The seeds are used in folk medicine and in many herbal preparations for the treatment of ailments such as laryngitis, liver disorders and bronchitis. 14 G. kola extract has been documented to possess antioxidant property. 15 Recently, it has also been reported to possess analgesic/anti-inflammatory effects resulting in the improvement of locomotors function and significant pain reduction in patients with knee osteoarthritis. 16
The biflavonoid isolated from the seeds of G. kola is known as kolaviron 14 (Figure 1). The chemopreventive effects of kolaviron against carbon tetrachloride, 2-acetylamino fluorine (2-AAF), aflatoxin B1, potassium bromate and dimethyl nitrosamine have been reported. 17–21 The ability of kolaviron to inhibit aflatoxin B1-induced genotoxicity in a human liver-derived cell line (HepG2) 22 and oxidant-induced DNA damage in human lymphocytes 23 has been reported.

Chemical structure of kolaviron.
Recently, much attention has been focused on the protective effects of antioxidants and naturally occurring substances against oxidant-induced testicular damage that results from excessive generation of free radicals from environmental compounds. Our recent data demonstrated the ameliorative effect of plant-derived antioxidants on di-n-butylphthalate-induced testicular damage in rats. 24 In continuation with our interest on the role of naturally occurring antioxidants on drug-induced testicular dysfunction, we investigated the possible protective effects of kolaviron against EGEE-mediated reproductive toxicity in rats and the effects validated with vitamin E, a major radical chain-breaking antioxidant.
Materials and methods
Chemicals
EGEE, epinephrine, glutathione (GSH), 5,5′-dithio-bis-2-nitrobenzoic acid, hydrogen peroxide, nicotinamide adenine dinucleotide (NADH), thiobarbituric acid, sodium pyruvate and 1-chloro-2,4-dinitrobenzene (CDNB) were purchased from Sigma Chemical Co. (St Louis, MO, USA). All other reagents were of analytical grade and were obtained from the British Drug Houses (Poole, Dorset, UK).
Isolation of kolaviron
Kolaviron was isolated according to the published procedure. 25 Briefly, the fresh G. kola seeds were sliced, air dried and powdered. The powdered seeds were extracted with light petroleum ether (boiling point [bp] 40–60°C) in a soxhlet for 24 h. The defatted dried marc was repacked and extracted with acetone. The extract was concentrated and diluted twice with water and extracted again with ethylacetate (6 × 300 ml). The concentrated ethylacetate yielded a golden yellow solid termed kolaviron which was identified by direct comparison of the 1H nuclear magnetic resonance (NMR), 13 C NMR and electron ionization (EI)–mass spectral results with previously published data. 25
Animal model
A total of 50 healthy adult male Wistar rats weighing approximately 220 ± 25 g obtained from the Department of Biochemistry, University of Ibadan, Ibadan, Nigeria, were used for this study. They were housed in plastic suspended cages placed in a well-ventilated rat house, provided rat pellets and water ad libitum, and subjected to natural photoperiod of 12-h light:dark cycle and an ambient temperature of 31 ± 2°C. All the animals received humane care according to the criteria outlined in the ‘Guide for the Care and Use of Laboratory Animals’ prepared by the National Academy of Science and published by the National Institute of Health. The ethic regulations have been followed in accordance with National and institutional guidelines for the protection of animal welfare during experiments. 26
Experimental protocol
The rats were randomly assigned to 5 groups of 10 rats each. Group I rats received corn oil alone at 2 ml/kg body weight (bw) and served as control. While group II rats were treated with 200 mg/kg bw EGEE alone by gavage for 14 days, groups III and IV were pretreated with kolaviron at 100 (KV1) and 200 (KV2) mg/kg/day in corn oil, respectively, by gavage for the first 7 days followed by a daily administration of both kolaviron and EGEE simultaneously for the last 14 days of the experiment. Group V rats were pretreated with vitamin E at 50 mg/kg bw 9 in corn oil by gavage for the first 7 days followed by a daily administration of both vitamin E and EGEE simultaneously for the last 14 days of the experiment. The doses of kolaviron and EGEE and the duration of experiment were selected based on our previous studies. 7,21 The basis for the selected dose of EGEE is because 200 mg/kg is the lowest dose that caused significant changes in stress and haematological parameters within 14 days of exposure to rats, whereas doses of kolaviron and vitamin E are the highest effective doses against oxidative indices from our preliminary studies. Twenty-four hours after the last treatment, all the animals were killed by cervical dislocation and the testes, epididymis, seminal vesicles and prostate glands were quickly removed and weighed. Samples from testes were fixed in Bouin’s solution, paraffin embedded, sectioned and stained routinely with haematoxylin and eosin for microscopic analysis. The body weights of rats were taken before exposure to various treatments and prior to killing.
Biochemical assay
Totally 10 right testes from each group were homogenized in 50 mM Tris-HCl buffer (pH 7.4) containing 1.15% potassium chloride and the homogenate was centrifuged at 10,000g for 15 min at 4°C. The supernatant was collected for the estimation of catalase (CAT) activity using hydrogen peroxide as substrate, according to the method of Clairborne. 27 Superoxide dismutase (SOD) was assayed by the method described by Misra and Fridovich. 28 Hydrogen peroxide generation was assessed by the method of Wolff. 29 Glutathione peroxidase (GPx) activity was determined by the method of Rotruck et al., 30 which is based on the reaction between GSH remaining after the action of GPx and 5′,5′-dithio-bis(2-nitrobenzoic acid) to form a complex that absorbs maximally at 412 nm. Glutathione-S-transferase (GST) was assayed by the method of Habig et al. 31 Lactate dehydrogenase (LDH) was assayed by the method of Vassault. 32 Protein concentration was determined by the method of Lowry et al. 33 Reduced glutathione (GSH) was determined at 412 nm using the method described by Jollow et al. 34 Lipid peroxidation was quantified as malondialdehyde (MDA), according to the method described by Farombi et al., 17 and expressed as micromoles of MDA/g tissue.
Collection of epididymal spermatozoa
The spermatozoa were collected as quickly as possible after a rat was killed. The 10 right cauda epididymis was placed in cold phosphate buffer saline solution and cut by surgical blades into pieces. The solution was pipetted several times to obtain the spermatozoa suspension and then filtered through a nylon mesh. The spermatozoa suspensions (8 × 106 spermatozoa cell/ml) were subsequently homogenized at 4°C with a glass Teflon homogenizer for 10 s and centrifuged at 2000×g for 10 min to obtain the supernatant which was used for biochemical assays.
Spermatozoa motility assay
Spermatozoa motility was assessed by the method described by Zemjanis. 35 One drop of the spermatozoa samples from the cauda epididymis from each group were used for the motility assay. The suspension was placed on a slide, covered by a 24 × 24 mm coverslip, and evaluated under a phase contrast microscope at ×200 magnification. The motility was evaluated microscopically within 2–4 min of their isolation from the cauda epididymis and data were expressed as percentages.
Spermatozoa count
Epididymal spermatozoa count was obtained by mincing the 10 left cauda epididymis in distilled water and filtering through a nylon mesh. The spermatozoa were counted by haemocytometer using the improved Neubauer (Deep 1/10 m, LABART, Darmstadt, Germany) chamber as described by Pant and Srivastava. 36
Determination of daily spermatozoa production and testicular spermatozoa number
Daily spermatozoa production was determined using five frozen left testes from control and treated rats according to Joyce et al. 37 Briefly, after the testes had been removed and weighed, they were homogenized for 3 min in 25 ml of physiological saline containing 0.05% (volume/volume [v/v]) Triton X-100. Sample aliquots of 5.5 μl were then placed on the haemocytometer and counted twice at ×100 magnification under microscope to determine the average number of spermatids per sample. These values were used to obtain the total number of spermatids per testis and this number was then divided by the testes weight to obtain spermatids per gram of testes. Developing spermatids spend 4.61 days in the testes in rats. Thus, the values for the number of spermatids per testis were divided by 4.61 to obtain daily spermatozoa production.
Morphological abnormalities and percentage viability assay
A portion of the spermatozoa suspension placed on a slide glass was smeared out with another slide and stained with Wells and Awa’s stain (0.2 g of eosin and 0.6 g of fast green dissolved in distilled water and ethanol in the ratio 2:1) for morphological examination and 1% eosin and 5% nigrosine in 3% sodium citrate dehydrate solution for live/dead ratio, according to the method described by Wells and Awa. 38 A total of 400 spermatozoa from each rat were examined for morphological examination. The major abnormalities consisted of headless tails, bent tails, curved mid-piece, bent mid-piece and cytoplasmic droplets.
Statistical analysis
Statistical analyses were carried out using one-way analysis of variance (ANOVA) to compare the experimental groups followed by the Student’s t test using SPSS (student version 7.5, SPSS Inc., UK) and values less than 0.05 were considered statistically significant.
Results
Body weight and organ weight
The body weight gain and absolute weights of the organs of control animals and those treated with EGEE alone, EGEE plus kolaviron and EGEE plus vitamin E are shown in Table 1. Significant decrease in body weight gain was observed in the EGEE-treated animals compared with the control (p < 0.05). However, the weights of absolute testes, epididymis, prostate glands and seminal vesicles in all treatment groups did not significantly differ from control. Body weight was unchanged in animals treated with EGEE plus kolaviron or vitamin E compared with the control group.
The body weights (g) and relative organ weights (g/100 g body weight) of rats treated with EGEE, kolaviron and vitamin E a

EGEE: ethylene glycol monoethyl ether, KV1: kolaviron 100 mg/kg; KV2: kolaviron 200 mg/kg, Vit E: vitamin E.
a The data are expressed as mean ± SD for 10 rats per group.
b p < 0.05 against control.
c p < 0.05 against EGEE.
Spermatozoa analysis
Table 2 reveals that the epididymal spermatozoa number, testicular spermatozoa number, daily spermatozoa production and percentage of motile spermatozoa were significantly decreased, whereas the percentage of spermatozoa abnormalities was significantly increased following exposure of animals to EGEE alone compared with the control group. However, treatment with kolaviron or vitamin E significantly prevented the decline in the quantity and quality of spermatozoa in EGEE-treated rats.
Spermatozoa parameters and total percentage abnormalities in spermatozoa of rats treated with EGEE, kolaviron and vitamin E a
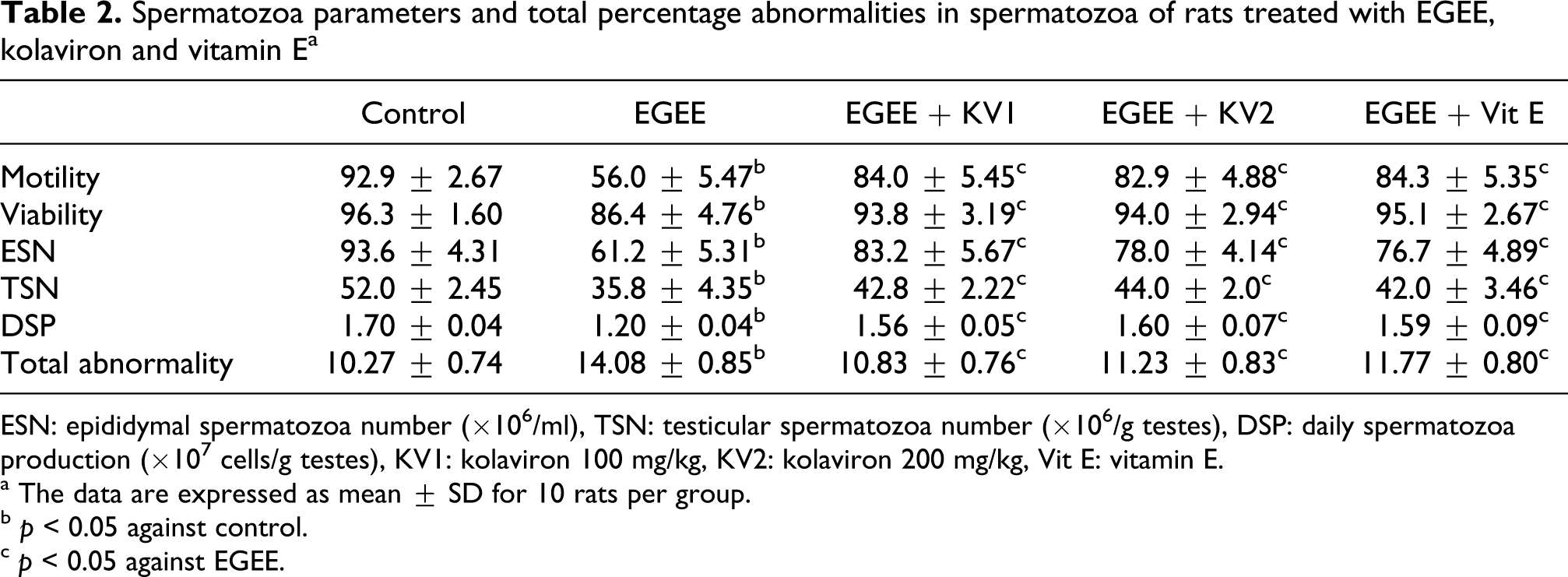
ESN: epididymal spermatozoa number (×106/ml), TSN: testicular spermatozoa number (×106/g testes), DSP: daily spermatozoa production (×107 cells/g testes), KV1: kolaviron 100 mg/kg, KV2: kolaviron 200 mg/kg, Vit E: vitamin E.
a The data are expressed as mean ± SD for 10 rats per group.
b p < 0.05 against control.
c p < 0.05 against EGEE.
Antioxidant system
Following exposure to EGEE, activity of SOD decreased dramatically in the testis but was significantly increased in spermatozoa compared with control animals. While GST and LDH activities were significantly increased in the testes, they were decreased significantly in the spermatozoa. Kolaviron or vitamin E treatment prevented the perturbation in the activities of these enzymes and maintained their normalcy in EGEE-treated animals. In addition, EGEE treatment resulted in significant decrease in GSH level as well as in the activities of CAT and GPx but significantly increased MDA and H2O2 levels both in the testes and spermatozoa compared with the control group. Treatment with kolaviron or vitamin E significantly elevated CAT and GPx activities and GSH level and completely reversed the increase in MDA and H2O2 levels to normalcy (Figures 2–9).

Effects of kolaviron and vitamin E (Vit E) on super-oxide dismutase (SOD) activity in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on catalase (CAT) activity in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on H2O2 level in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on glutathione (GSH) level in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on glutathione S-transferase (GST) activity in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on glutathione peroxidase (GPx) activity in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on lactate dehydrogenase (LDH) activity in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).

Effects of kolaviron and vitamin E (Vit E) on lipid peroxidation (LPO) level in testes and sperm of ethylene glycol monoethyl ether (EGEE)–exposed rats. Each bar represents mean ± SD of 10 animals. aValues differ significantly from control (p < 0.05). bValues differ significantly from EGEE group (p < 0.05).
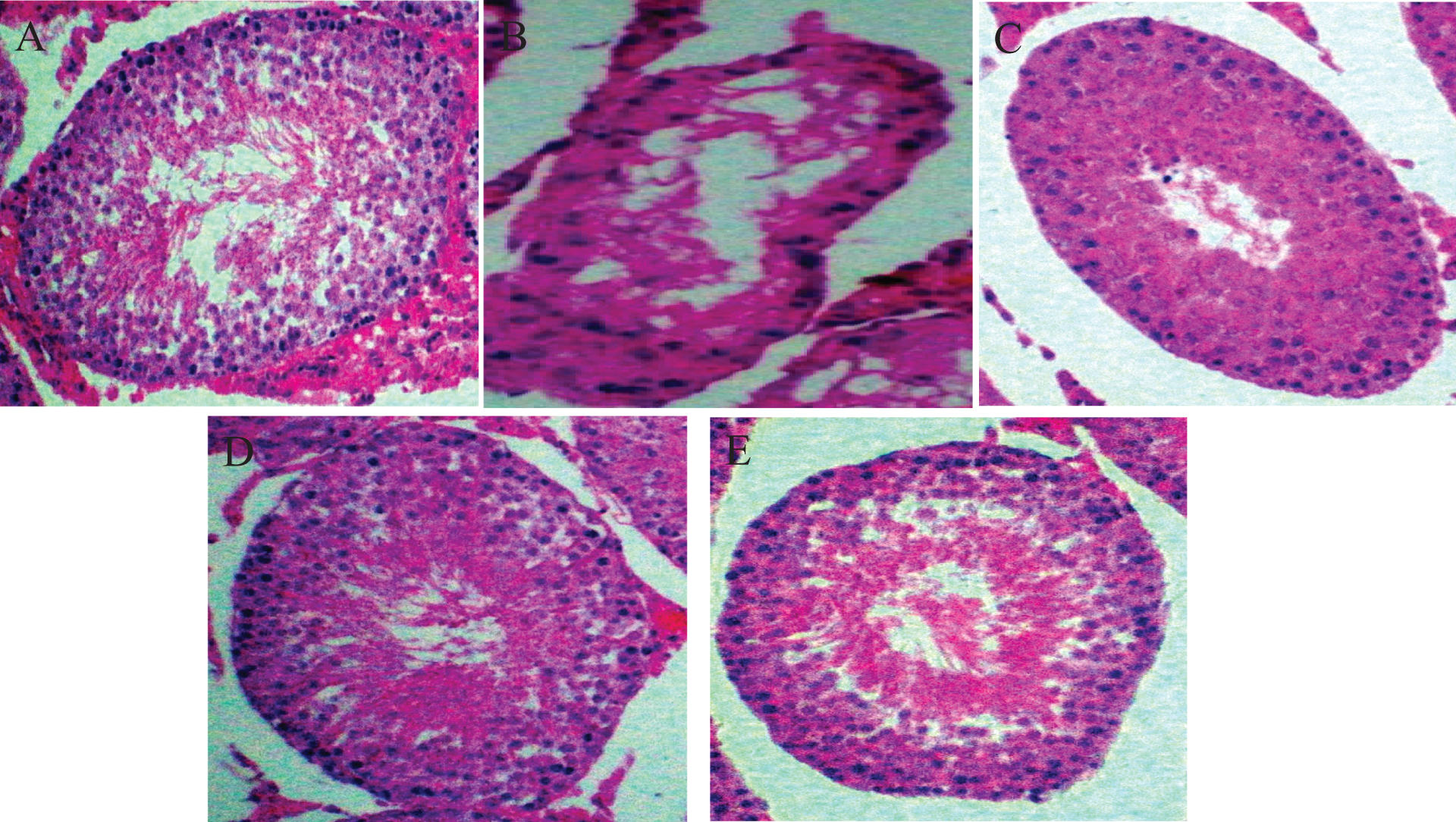
Representative photomicrographs of control and treated testes. (A) Control testes with normal architecture and viable spermatozoa. (B) Ethylene glycol monoethyl ether (EGEE)–treated testes showing generalized erosion, necrosis of the germinal epithelium and reduction in Sertoli cell population. (C) EGEE plus 100 mg/kg kolaviron-treated testes showing normal seminiferous tubules with appropriate spermatozoa cells. (D) EGEE plus 200 mg/kg kolaviron-treated testes showing normal seminiferous tubules with adequate testicular spermatozoa cells. (E) EGEE plus 50 mg/kg vitamin E-treated testes showing normal architecture with appropriate spermatozoa cells. Haematoxylin and eosin (H & E) stain at ×400 magnification.
Histological evaluation
Control animals showed normal testicular architecture with Sertoli cells. The testes of animals treated with EGEE were severely degenerated characterized by generalized erosion, necrosis of the germinal epithelium and reduction of Sertoli cell population. However, the morphologic characteristics of testes were preserved and comparable to those in control groups and in animals treated with EGEE plus kolaviron or vitamin E (Figure 10).
Discussion
Male factor infertility accounts for up to half of all cases of infertility and affects 1 person out of 20 in the general population. 39 Reactive oxygen species (ROS) impact testicular tissue causing degeneration and thus affect gonadal functions. Oxidative stress is a result of the imbalance between ROS and antioxidants in the body, which can lead to spermatozoa damage, deformity and eventually male infertility. 40 The testes contain an elaborate array of antioxidant enzymes and free radical scavengers to ensure that the twin spermatogenic and steroidogenic functions of the testes are not impacted by oxidative stress. 41 SOD is a family of metalloenzymes that is known to accelerate the dismutation of endogenous cytotoxic superoxide radicals to H2O2, which are deleterious to polyunsaturated fatty acids and structural proteins of plasma membrane. 42 The elimination of H2O2 is either effected by CAT or GPx, with the latter predominating in the case of the testes. 43,44
The present study demonstrates that EGEE administration significantly decreased SOD activity in the testes, whereas it increased significantly in the spermatozoa. The decreased activity of testicular SOD observed in EGEE-treated rats may lead to an increased steady-state level of the deleterious superoxide radicals. This may also indicate an enzyme inhibition by the H2O2 as a result of corresponding decrease in testicular CAT activity. The dramatic increase in spermatozoa SOD suggests a means of protecting spermatozoa from oxidative stress at a point when it can be particularly disruptive to their integrity. The present finding corroborates with our previous results. 7 Interestingly, amelioration by kolaviron or vitamin E was evident by the reversal in the alteration of SOD activity and the decreased CAT activity in EGEE-treated animals. We have previously demonstrated in vitro the ability of kolaviron to inhibit hydroxyl and superoxide anion radicals. 45 Kolaviron has also been shown to be highly bioavailable because of its ability to survive first-pass metabolism which inactivates most flavonoids. 46 The present results could therefore be constructed to mean that there was a quick scavenging of superoxide radicals by kolaviron, possibly due to its high bioavailability, to keep their normal levels, thereby allowing the testes and spermatozoa to combat oxidative stress efficiently at least up to moderate concentrations.
GPx is the most important peroxidase for the detoxification of hydroperoxides. It catalyses the GSH-dependent reduction in hydroperoxides and hydrogen peroxide. GST is directly responsible for the elimination of electrophilic oxidants at the expense of GSH 31 to form more water-soluble products that are readily excreted from the system. Administration of EGEE significantly increased testicular GST activity but decreased spermatozoa GST activity. The significant decrease in the testicular and spermatozoa GPx activity was accompanied with a considerable depletion in GSH levels in EGEE-treated animals. Increased activities of GST are known to serve as protective responses to eliminate xenobiotics. 47,48 The increase in the activity of testicular GST in EGEE-treated animals could be due to its induction to counter the effect against increased oxidative stress possibly generated during EGEE metabolism, whereas the decrease in the activities of GPx and spermatozoa GST suggests that these antioxidant enzymes were inhibited and their protective effects against free radicals and lipid peroxidation were reduced. The decreased level of GSH in both testes and spermatozoa may be due to their utilization by the enhanced production of ROS. Administration of kolaviron or vitamin E was effective in increasing the GSH levels as well as restoring the activities of GPx and GST in the testes tissue and spermatozoa.
LDH activities, a testicular germ cell marker enzyme, increased in the testes but decreased in the spermatozoa in EGEE-treated animals in the present study. Lactate is one of the compounds produced by the Sertoli cells and utilized primarily by the germ cells for ATP production in the mitochondrial oxidative phosphorylation. 49–51 It is considered that testicular organs need great energy to continuously maintain spermatogenesis. Lactate effectively inhibited apoptosis of testicular germ cells in a dose-dependent manner. 52 The increase in testicular LDH may therefore reflect its protective mechanism against germ cell death, whereas the reduction in the LDH activity observed in the spermatozoa may contribute to low spermatozoa motility due to insufficient ATP generation. The present results are consistent with previous reports. 7,36 The alteration of lactate content in the testes has been attributed to perturbation in the hormonal control and supply. 53 The restoration in the activity of LDH to near normalcy upon administration of kolaviron or vitamin E possibly suggest that kolaviron or vitamin E in a way could have prevented the degeneration of Sertoli cells which supplies lactate during spermatogenesis.
Lipid peroxidation is a degenerative pathway of membrane components mediated through free radicals produced in the cell. 54 The present study demonstrates that EGEE treatment resulted in increased MDA level, an end product of lipid peroxidation, in both testes and spermatozoa. The increased lipid peroxidation level observed in EGEE-exposed animals might be a consequence of increased free radical formation as well as the disruption of antioxidant status. The protective effect of kolaviron or vitamin E against lipid peroxidation can be attributed to its ability to directly scavenge the free radicals and thus prevent their attack on the membrane. Kolaviron has been reported to elicit its protective action by acting as membrane stabilizer and thus can break the chain reaction of the lipid peroxidation initiated by hydroxyl radicals. 17,55
In the present study, administration of EGEE significantly decreased spermatozoa motility, the daily spermatozoa production, testicular and epididymal spermatozoa numbers with significant elevation in spermatozoa abnormalities. We had previously reported that the decrease in the testicular and epididymal spermatozoa numbers coupled with the decreased daily spermatozoa production may indicate that EGEE affects the early stages of spermiogenesis. 7 In addition, the observed alterations in spermatozoa motility and elevated spermatozoa abnormalities in EGEE-treated animals might be the outcome of altered and hostile internal milieu of epididymis caused by ROS. Direct ROS damage to mitochondria resulting in decreasing energy availability and ROS-induced peroxidation of the spermatozoa membrane decreasing its flexibility and therefore tail motion and impairs motility. Significant increases in spermatozoa count and motility and decreases in the number of dead and abnormal spermatozoa were observed upon coadministration of kolaviron or vitamin E. The improvement observed in spermatozoa production and quality may be attributed to prevention in excessive generation of free radicals by means of the antioxidant property of kolaviron or vitamin E.
Although EGEE administration produced a severe testicular degeneration, the relative weights of the testes, epididymis, seminal vesicles and prostate glands of EGEE-treated animals were comparable with the control. The observed alterations in the testicular architecture could disrupt its functions. Spermatogenic inhibition has been reported to be the detrimental effects of free radical products in testicular tissue. 56 Our result is inconsistent with Lamb et al. 3 who reported a significant decrease in the testis and epididymis of the mice exposed to EGEE in drinking water for 14 weeks. In their report, it was indicated that the male mice which had 1% EGEE in water received about 1500 mg/kg/day and mice receiving 2% EGEE in water received about 2600 mg/kg/day. The differences in these data may be due to differences in treatment duration, dose and mode of administration and/or to species differences. However, our result is consistent with Wang et al. 4 who reported that administration of EGEE by oral gavage at 100 and 600 mg/kg/day for 1 week did not induce any significant change in the weight and body weight ratios of the testes, prostate and epididymis in mice. The damage observed in the testicular architecture in this study may be attributed to the direct or indirect effect of increased ROS following EGEE administration, which consequently induces lipid peroxidation that is a chemical mechanism capable of disrupting the structure and function of the organ. The biochemical evidence of the protective potential of kolaviron or vitamin E against EGEE toxicity is well supported by the histological evaluation. Kolaviron or vitamin E maintained the normalcy of the testicular architecture upon administration to EGEE-treated animals.
The protective effects of kolaviron or vitamin E against EGEE-induced toxicity observed in this present investigation appear to correlate with the structural variations in the two natural antioxidants. In mechanistic terms, early studies on the structure–activity relationships of phenolic compounds related to kolaviron revealed that the presence of a hydroxyl group in the position three (3-OH) of the C-ring can make it a potent inhibitor of lipid peroxidation. 57–59 Furthermore, antiperoxidative effect of kolaviron may be linked to the presence of a C-4 carbonyl and C-5 and C-7 hydroxyl groups of the A-ring. 24 These structural features confer on kolaviron the antioxidant activity of radical scavenging utilizing its many hydroxyl groups on the dual B-rings and metal ion chelation utilizing an abundant availability of 3- or 5-hydroxyl and 4-ketosubstituents or hydroxyl groups in the ortho position in the B-ring. 60 Vitamin E because of the reactivity of the phenolic hydrogen on its C-6 hydroxyl group and the ability of the chromanol ring system to stabilize an unpaired electron 61 can provide hydrogen for the reduction in peroxyl radicals before deleterious interaction with the cell membranes and other cell components. We therefore hypothesize that kolaviron and vitamin E, by way of maintaining testicular and spermatozoa antioxidant capacity, are able to combat EGEE-induced oxidative damage.
Overall, the findings of the present investigation demonstrate that oxidative stress plays a major role in the development of EGEE-induced reproductive dysfunction and that kolaviron or vitamin E, as a result of its intrinsic antioxidant potential, protects against the reproductive toxicity of this xenobiotic. The protective effect of kolaviron and vitamin E against enhanced ROS generation by EGEE is achieved through stimulation of antioxidant status. It is therefore reasonable to suggest that clinical application of kolaviron may be a potentially helpful new approach for chemotherapy of EGEE-induced reproductive toxicity.
Footnotes
Acknowledgements
The authors gratefully acknowledge Professor Canice C Asonye of the University of Benin, Nigeria, for the generous gift of ethylene glycol monoethyl ether. The technical assistance of Messrs Omoko Ejiro of the Department of Veterinary Surgery and Reproduction, University of Ibadan, is appreciated.
This work was supported by the Multidisciplinary Research Grants under the Staff Training and Research Capacity Building Programme of the John D and Catherine T MacArthur Foundation Grant (USA) endowment from the University of Ibadan, Nigeria, awarded to Professor EO Farombi.
The authors declare that there are no conflicts of interest.
