Abstract
In the present study, we have evaluated the effects of eicosapentaenoic acid (EPA) on Na+-K+-ATPase in sheep pulmonary artery. Acute (30 min) and prolonged (24 h) exposure of arterial rings to EPA (30 μM) significantly decreased potassium chloride (KCl)-induced relaxation, an index of functional Na+-K+-ATPase activity. In acute exposure, the pD2 and E max (the maximal response) values for KCl-induced relaxation were 3.21 ± 0.33 and 61.58 ± 11.30% (n = 5) versus control 3.58 ± 0.07 and 82.44 ± 2.36% (n = 24), respectively. The pD2 and E max values for KCl-induced relaxation in arterial rings exposed to EPA for 24 h in organ culture were 2.52 ± 0.11 and 55.00 ± 5.72% versus control 3.04 ± 0.19 and 80.74 ± 11.96%, respectively; n = 4. Exposure of the arterial rings to EPA (30 μM) for 24 h in organ culture, significantly decreased (17.58 ± 2.15%) the protein expression of α1 isoform of Na+-K+-ATPase. Acute exposure to EPA for 30 min significantly decreased (21.06 ± 5.89%) the Na+-K+-ATPase activity as measured by inorganic phosphate (Pi) release. EPA, up to 100 μM concentration, marginally (<10% of 80 mM KCl contraction) increased the basal tone of the pulmonary artery. Additionally, EPA (10–30 μM) had no effect on Mg2+-ATPase activity as well as on cyclic guanosine monophosphate (cGMP) production. All these results show that EPA has inhibitory effect on Na+-K+-ATPase in sheep pulmonary artery but prolonged exposure had no additional effect on sodium pump, and EPA-induced inhibition of Na+-K+-ATPase may be due to attenuation in protein expression of α1 isoform of Na+-K+-ATPase independent of cGMP production.
Introduction
Fish oils and n-3 polyunsaturated fatty acids (PUFAs) protect against several types of cardiovascular diseases such as, myocardial infarction, arrhythmia, atherosclerosis and hypertension. It is widely accepted that eicosapentaenoic acid (EPA) is the one of the active biological components of these effects.1–3 EPA has got both vasodilator (bovine coronary artery 4 and rat aorta 5 ) and vasoconstrictor (rabbit pulmonary vascular bed 6 ) effects. While the vasodilator action of the fatty acid has been shown to be mediated by endothelium-dependent (sheep pulmonary artery 7 ) and -independent mechanisms, the pathways involved in the vasoconstrictor action are not clearly understood. Nevertheless, dietary supplementation with EPA has been shown to impair endothelium-dependent dilations to acetylcholine (ACh) in the coronary microvessels of guinea pig. 8 Further, fish oil supplementation produces an increase in arterial pressure via peripheral vasoconstriction and that the vascular response to α-adrenergic stimulation is exaggerated. 9 The Na+-K+-ATPase has been hypothesized to play an important role in the regulation of vascular tone.10,11 Na+-K+-ATPase modulation is important in vascular smooth muscles, because it re-establishes the resting membrane potential by regulating intracellular Na+, which in turn regulates the intracellular Ca2+. Thus, the enzyme is important in the regulation of vascular tone and contractility. 12 For instance, stimulation of the plasmalemmal Na+-K+-ATPase in vascular smooth muscle causes membrane hyperpolarization that consequently reduces Ca2+ influx through voltage-gated Ca2+ channels leading to vasodilation. Alternately, vascular smooth muscle relaxation through stimulation of the Na+-K+-ATPase is mediated by the activation of Na+-Ca2+ exchange mechanism, which in turn reduces the intracellular Ca2+. On the contrary, inhibition of the enzyme produces vascular contractility. 13 EPA has inhibitory effect on Na+-K+-ATPase in different tissues like human endothelial cells, synaptosomal membranes isolated from rat cerebral cortex and isolated basolateral membranes from rat duodenal enterocytes.14–16 However, there is limited information on the effect of EPA on vascular Na+-K+-ATPase. Therefore, the first objective of the present study was to evaluate the effect of EPA on functional Na+-K+-ATPase and its activity in sheep pulmonary artery. A high concentration of ouabain (500 nM), which would inhibit the α1 isoform of Na+-K+-ATPase, produced smooth muscle depolarization, indicating that this isoform is active under basal conditions in the rat mesenteric artery. 17 Recently, data published from our laboratory showed that inhibition of the α1 isoform of Na+-K+-ATPase by ouabain significantly increased the basal tone and 5-hydroxytryptamine hydrochloride (5-HT)-induced contractility in the sheep pulmonary artery. 18 Based on these observations, it was hypothesized that the low-affinity isoform α1 regulates basal tone, as well as agonist-induced contractility in the sheep pulmonary artery. Keeping these points in view, the second objective was to study the effect of EPA on α1 isoform of Na+-K+-ATPase in respect to protein expression. The third question was that whether EPA would modify the vascular tone in this vessel.
Methods
Blood vessel preparation
Lungs from freshly slaughtered adult sheep were collected from the local slaughterhouse in cold (4–6°C) oxygenated modified Krebs-Henseleit solution (MKHS) of the following composition: sodium chloride (NaCl) 118 mM, potassium chloride (KCl) 4.7 mM, calcium chloride (CaCl2) 2.5 mM, magnesium sulfate (MgSO4) 1.2 mM, sodium bicarbonate (NaHCO3) 11.9 mM, potassium dihydrogen phosphate (KH2PO4) 1.2 mM and
Tension experiments
Arterial rings of 2–3 mm were prepared from second-generation intrapulmonary arteries. Rings were mounted between two L-shaped stainless steel hooks (30 gauge) under a resting tension of 1.5 g in a thermostatically controlled (37.0 ± 0.5°C) organ bath of 10 ml capacity containing MKHS and continuously aerated with carbogen (95% oxygen [O2] + 5% carbon dioxide [CO2]). Experiments were done in endothelium-denuded pulmonary artery rings. Endothelium was removed by mechanical rubbing with a cotton swab. Removal of endothelium was confirmed by demonstrating the absence of relaxation to ACh (10 μM). Following the removal of endothelium, most of the tissues contracted with ACh. The arterial rings were equilibrated for 90 min with a repeated replacement of bath solution every 15–20 min. A high sensitivity force displacement transducer (model MLT0202/D; PowerLab, CastleHill, Australia) measured the change in tension, and the data were recorded in a personal computer (PC) using chart version 4.1.2 software program (PowerLab).
Functional sodium pump analysis
KCl-induced relaxation in vascular rings, contracted with K+-free solution plus agonist has been used for the assessment of functional sodium pump. 19 In brief, after equilibration, pulmonary arterial rings were exposed to K+-free solution (MKHS in which KCl and KH2PO4 were replaced with equimolar concentration of NaCl and NaH2PO4, respectively) for 30 min. At the declining phase of the K+-free contracture, 5-HT (0.1 μM) was added to maintain a steady level of contraction. At the plateau phase of contraction, KCl (10−5–10−2 M) was added cumulatively at an increment of 0.5 log unit to elicit relaxation which was expressed as per cent reversal of contraction elicited with K+-free solution plus 5-HT. To study the influence of EPA in modifying sodium pump function, vessels were incubated with 30 μM of this drug for 30 min in K+-free solution before steady state contraction with 5-HT was elicited.
Organ culture
Second-generation pulmonary arteries were isolated in laminar flow to avoid the contamination. After cleaning the artery in modified Krebs–Hensleit’s solution, the arteries were cut into rings (approximately 2–3 mm). Arterial rings were then placed in 2 ml Dulbecco Modified Eagle Medium (DMEM) supplemented with 10% fetal calf serum (FCS) and 1% penicillin–streptomycin in 24-well culture plate. The arterial rings were maintained at 37°C in an atmosphere of 95% O2 and 5% CO2 for 24 h (water jacketed CO2 incubator).
Western blot analysis
Pulmonary arterial rings treated with EPA (30 μM) for 24 h in organ culture were homogenized (10% weight/volume [w/v]) on ice in a homogenization buffer (250 mM sucrose in 10 mM 3-(N-morpholino) propanesulfonic acid [MOPS], pH 7.4) using polytron homogenizer to evaluate the effect on α1 isoform of Na+-K+-ATPase. The homogenate was then centrifuged at 10,000g for 10 min at 4°C. The supernatant was subjected to ultracentrifugation at 112,000g at 4°C for 1 h. The pellet consisting of microsomes was resuspended in appropriate (depending on pellet size) volume of homogenization buffer and protein concentration was determined by Lowry’s method. 20 For Western blot, microsomal fractions were electrophoresed at a constant voltage on a linear gradient polyacrylamide gel (5–15%) with a stacking gel of 3.3% polyacrylamide. 21 Proteins were transferred from gel onto nitrocellulose membrane (NCM) and the electroblot performed at 200 V for 1.5 h in a water-cooled transfer apparatus. The membrane was then blocked in blocking buffer (5% skim milk in 0.05% phosphate buffered saline [PBS]-Tween) for 12 h at 4°C. The NCM was incubated with primary polyclonal antibody (0.4μg/ml PBST) against α1-Na pump for 5 h at room temperature. After three washings of NCM in PBS-Tween (0.05%), detection was achieved using horseradish peroxidase (HRPO)-conjugated chicken anti-goat IgG at a concentration of 0.4 μg/ml (Santacruz Biotechnology, USA) and diaminobenzidine (DAB) substrate solution. Quantification of α1 isoform of Na+-K+-ATPase was done using the densitometric analysis of the band, and the data are presented in per cent of the integrated density value (IDV) in control and treatment groups.
Estimation of Na+-K+-ATPase and Mg2+-ATPase activities in plasma membrane
Isolation of sarcolemmal membranes from pulmonary arteries was performed as per the procedure described. 22 Na+-K+-ATPase activity was determined by measuring the liberation of inorganic phosphate (Pi) from ATP as per the method described by Chanda and coworkers (2008). 18 For total ATPase assay, ouabain was omitted from the reaction mixture, which also included Mg2+-ATPase. After 1-h incubation in both the cases, the reaction was stopped with 0.1 ml of 5% sodium dodecyl sulfate and color was developed with 3 ml of acidic ammonium molybdate and 0.1 ml of 1-amino-2-naphthol-4-propane sulfonic acid (ANSA) reagent. The Pi in the reaction mixture was estimated as per the method. 23 Enzyme activity is expressed as nmol of Pi liberated per min per mg of protein. To determine the effect of EPA on the Na+-K+-ATPase and Mg2+-ATPase activities, tissues were exposed to 30 μM concentrations of EPA for 30 min.
cGMP measurement
Pulmonary arterial rings (n = 6) were equilibrated in MKHS for 30 min at 37°C under continuous bubbling with 95% O2+5% CO2, were exposed to 30 μM dipyridamole for next 30 min and quickly snap frozen in liquid nitrogen. Arterial rings treated similarly in all respects were used to estimate the basal cyclic guanosine monophosphate (cGMP) concentrations. Arterial rings snap frozen with liquid nitrogen were homogenized in 1 ml chilled trichloroacetic acid (6%) and centrifuged at 10,000g for 10 min. Supernatant, thus obtained was extracted for five times with water-saturated diethyl ether and residual ether was removed from aqueous layer by heating the sample at 70°C for 5–10 min. The neutralized supernatant was then used for cGMP assay using EIA kit (Cayman Chemicals, Ann Arbor, MI, USA) following the manufacturer’s instructions. Tissue pellets obtained following centrifugation were dissolved in 1 N sodium hydroxide (NaOH) for protein estimation by Lowry’s method. Concentrations of cGMP are expressed as pmol/mg protein.
Drugs
Acetylcholine chloride, 5-HT, dipyridamole and sodium nitroprusside (SNP) were purchased from Sigma Chemicals (St Louis, MO, USA). EPA was purchased from Cayman Chemicals (Ann Arbor, MI, USA). EPA was dissolved in absolute alcohol. All other drugs were dissolved in distilled water. The vehicles had no effect on the responses of the tissue at the concentrations used. Primary polyclonal antibodies against α1 (goat polyclonal IgG against epitope mapping within an internal region of Na+-K+-ATPase α1 of human origin) isoform of Na+-K+-ATPase were purchased from Santacruz Biotechnology, USA.
Statistical analysis
Results are expressed as mean ± SEM and multiple comparisons were done using two-way analysis of variance (ANOVA) followed by Bonferroni post hoc test. Student’s t test was used when comparisons were made between control and drug treatment. A p value < 0.05 was considered statistically significant. pD2 and E max (the maximal response) were determined using nonlinear regression analysis of Graphpad Prism. pD2 is defined as −log EC50 of the agonist. ‘EC50’ refers to the median effective concentration and ‘n’ refers to the number of animals.
Results
Effect of acute exposure of EPA on functional Na+-K+-ATPase in sheep pulmonary artery
The raw tracing in Figure 1A shows the KCl (10−5–10−2 M) elicited concentration-dependent relaxation of pulmonary artery rings precontracted (1.80 ± 0.20 g, n = 12; 24 rings) with K+-free solution plus 5-HT (0.1 μM). The pD2 and E max values for KCl-induced relaxation of control were 3.58 ± 0.07 and 82.44 ± 2.36%, respectively (n = 12; 24 rings). As illustrated in the experimental tracing in Figure 1B, EPA significantly (p < 0.05) decreased the KCl-induced relaxation in sheep pulmonary artery. In the presence of EPA (30 μM), the absolute tension generated by the K+-free solution plus 5-HT (0.1 μM) was 2.65 ± 0.46 g; n = 5. As summarized in Figure 1C, EPA caused concentration-dependent inhibition of KCl-induced relaxation and the pD2 and E max values for KCl-induced relaxation were 3.21 ± 0.33 and 61.58 ± 11.30%, respectively (n = 5).

(A) Representative raw tracings show concentration-dependent relaxation of KCl (10−5–10−2 M) in the isolated sheep pulmonary artery taken from vehicle control. (B) Acute effect (30 min incubation) of eicosapentaenoic acid (EPA; 30 μM) on the arterial rings mounted in the organ bath to assess the functional sodium pump activity by KCl-induced relaxation in vessels precontracted with K+-free solution plus 0.1 μM 5-hydroxytryptamine hydrochloride (5-HT). (C) The line diagram depicts the mean concentration–response curves elicited with cumulatively added KCl on the pulmonary arterial rings obtained from control and EPA-treated rings. Vertical bars represent SEM. Data were analyzed by two-way analysis (ANOVA) followed by Bonferroni post hoc tests (*p < 0.05).
Effect of prolonged exposure of EPA on functional Na+-K+-ATPase in sheep pulmonary artery
To study the prolonged exposure of EPA on sodium pump, the arterial rings were treated with EPA (30 μM) for 24 h in organ culture and thereafter, they were mounted in the organ bath to study the functional sodium pump. KCl (10−5–10−2 M) elicited concentration-dependent relaxation of pulmonary artery rings precontracted (0.77 ± 0.09 g, n = 4) with K+-free solution plus 5-HT (0.1 μM) of control. The pD2 and E max values for KCl-induced relaxation of control were 3.04 ± 0.19 and 80.74 ± 11.96%, respectively (n = 4). Pretreatment of tissues for 24 h with EPA (30 μM) had significant effect on K+-free solution plus 5-HT (0.1 μM)-induced contraction (1.20 ± 0.11 g). As raw tracing in Figure 2A illustrates, EPA (30 μM) caused significant (p < 0.05) inhibition of KCl-induced relaxation in this artery. The pD2 and E max values are 2.52 ± 0.11 and 55.00 ± 5.72%, respectively (Figure 2B).
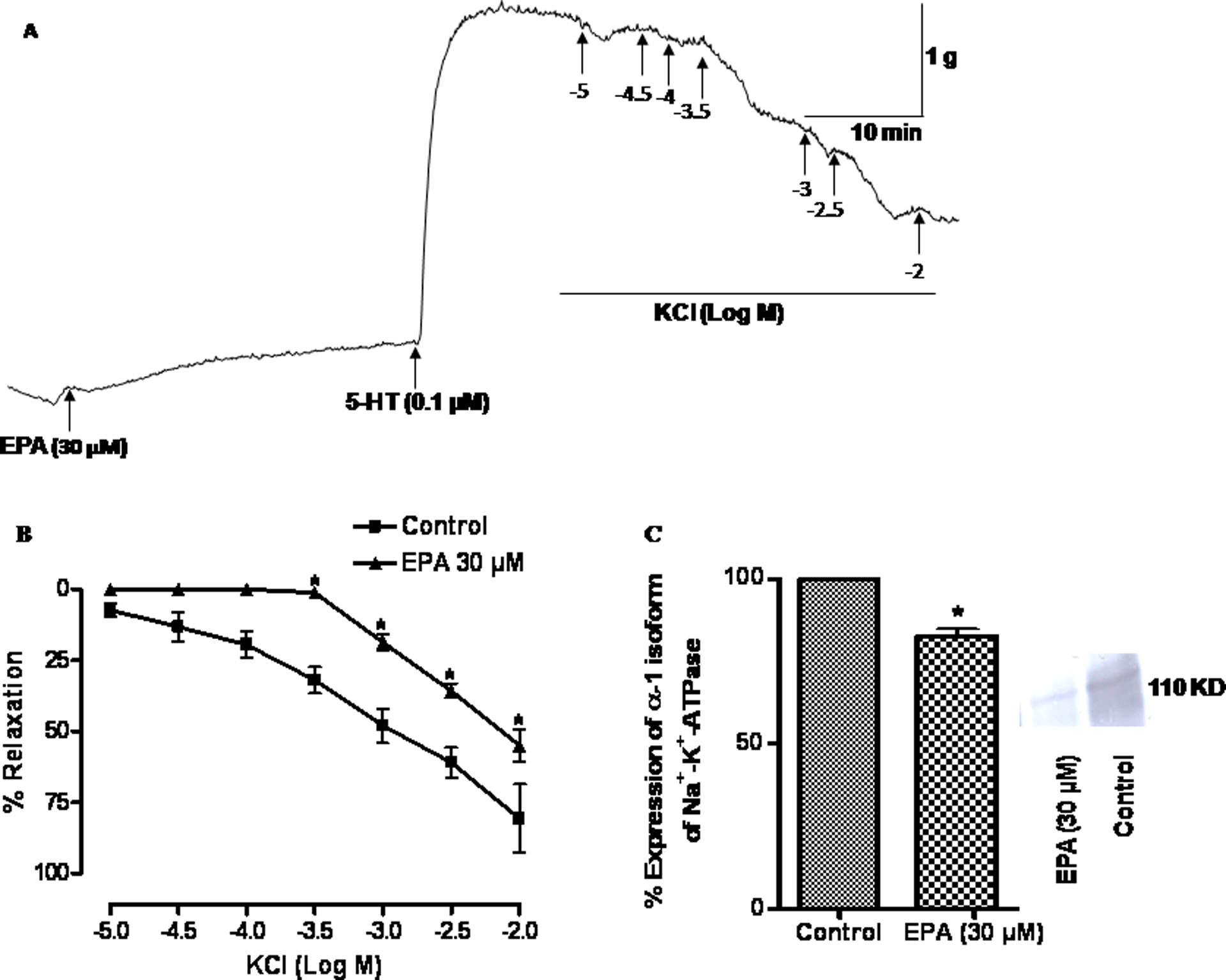
(A) Raw tracing shows that arterial rings treated with eicosapentaenoic acid (EPA; 30 μM) for 24 h were mounted in the organ bath to assess the functional sodium pump activity by KCl-induced relaxation in vessels precontracted with K+-free solution plus 0.1 μM 5-hydroxytryptamine hydrochloride (5-HT). (B) The line diagram depicts the mean concentration–response curves elicited with cumulatively added KCl on the pulmonary arterial rings obtained from control and EPA-treated rings. Vertical bars represent SEM. Data were analyzed by two-way analysis of variance (ANOVA) followed by Bonferroni post hoc tests (*p < 0.05). (C) Bar diagram shows the effect of EPA on the protein expression of Na+-K+-ATPase α-1 isoform in sheep pulmonary artery. The arterial rings were organ cultured for 24 h in the absence (control) and presence of EPA (30 μM) then the rings were homogenized and used for the quantitative Western blot analysis. Proteins were separated by SDS-polyacrylamide gel electrophoresis (PAGE) and transferred to nitrocellulose. The α-1 isoform was identified using goat polyclonal IgG against epitope mapping within an internal region of Na+-K+-ATPase α-1 of human origin. The comparison has been shown between different bands of α-1 isoform of Na+-K+-ATPase which had been treated with EPA. The proteins were quantitated using densitometry and the level of expression is expressed as per cent of integrated density value (IDV). Statistical comparison was done using Student’s t test (*p < 0.05).
Effect of prolonged exposure of EPA on protein expression
Figure 2C shows the influence of 24-h treatment with EPA on the expression of α1 isoform of Na+-K+-ATPase. The EPA (30 μM)-treated tissues significantly (p < 0.05) decreased (17.58 ± 2.15%) the expression of α1 isoform when compared to control (n = 3).
Effect of EPA on Na+-K+-ATPase activity
As a part of functional studies of sodium pump and regulation of pump function by EPA, plasmalemmal Na+-K+-ATPase activity was estimated in sheep pulmonary artery. Purified plasma membrane was prepared (5–10%) using tissue homogenate. Na+-K+-ATPase activity was measured in EPA-treated arterial rings. EPA (30 μM) significantly (p < 0.05) decreased (21.06 ± 5.89%) the enzyme activity when compared with control (Figure 3), while 10 μM of EPA had no effect on enzyme activity.

Effect of different concentrations (10 and 30 μM) of eicosapentaenoic acid (EPA) on Na+-K+-ATPase activity in the sheep pulmonary artery. The individual arterial rings were treated for 30 min with or without (10 and 30 μM) EPA in organ bath chamber at 37°C. Then arterial rings were homogenized with pestle and mortar and then centrifuged at 112,000g for 2 h to separate microsomes. The microsomes were used for the estimation of Na+-K+-ATPase activity. The activity is expressed in percentage. Statistical comparison was done using one-way analysis of variance (ANOVA) as Tukey’s multiple comparison test (*p < 0.05).
Effect of EPA on Mg2+-ATPase activity
To rule out the role of Mg2+-ATPase in EPA-induced inhibition of Na+-K+-ATPase, Mg2+-ATPase activity was estimated in tissue samples treated with EPA (10 and 30 μM) which was compared with the control. Pulmonary arterial rings were treated with 10 and 30 μM concentrations of EPA. Mg2+-ATPase activity was not significantly different from the control at all different concentrations (Figure 4).
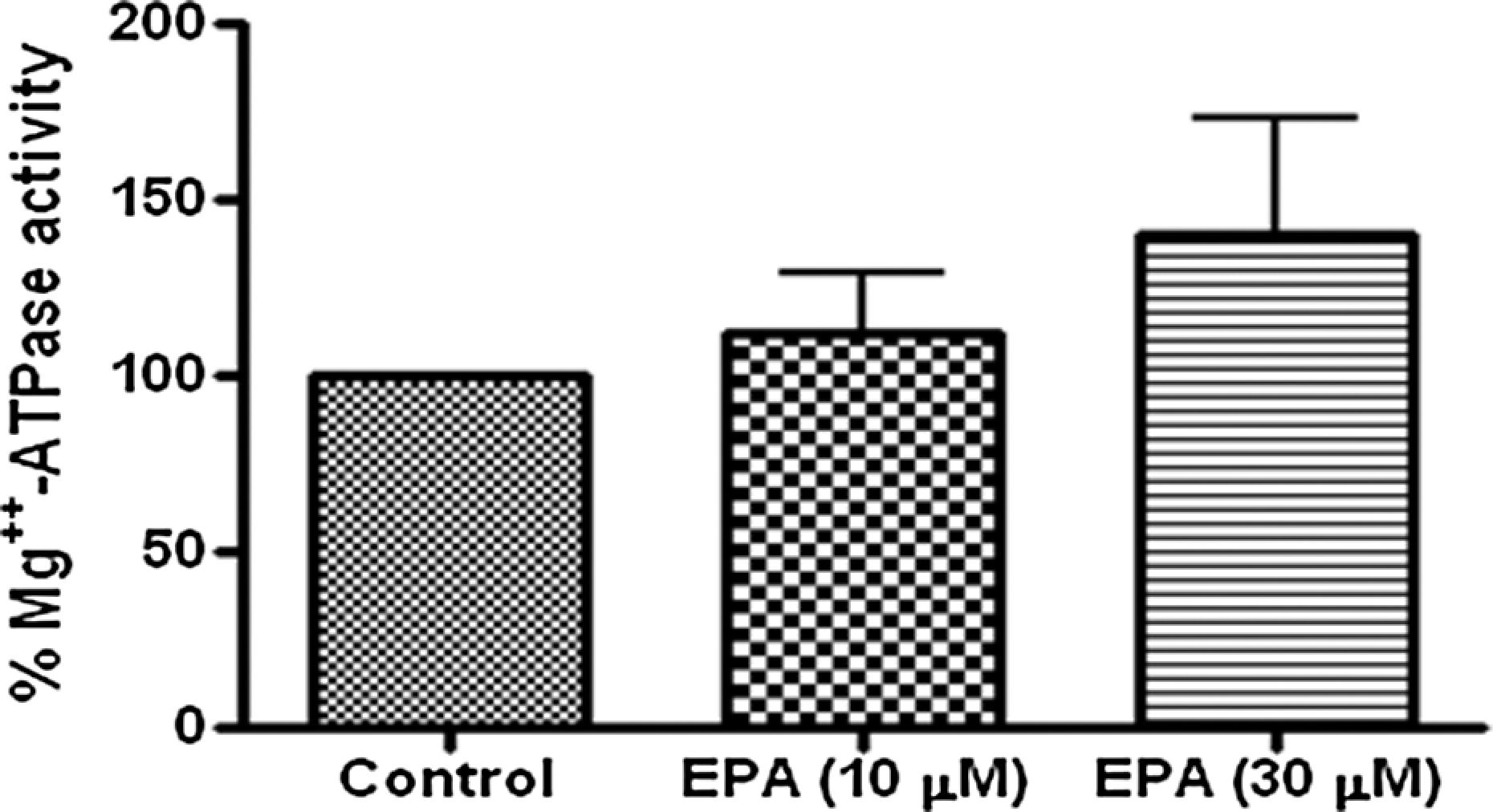
Effect of different concentrations (10 and 30 μM) of eicosapentaenoic acid (EPA) on Mg2+-ATPase activity in the sheep pulmonary artery. The individual arterial rings were treated for 30 min with or without (10 and 30 μM) EPA in organ bath chamber at 37°C.
Effect of EPA on basal tone of sheep pulmonary artery in the presence of ouabain
EPA (10−7–10−4 M) added cumulatively at an increment of 0.5 log unit caused a concentration-dependent contraction of pulmonary artery rings. The contraction at 30 μM concentration of the fatty acid was 4.26 ± 0.80% of K+ (80 mM)-induced contraction. The maximum contraction was achieved at 100 μM concentration of EPA that was 10.33 ± 1.14% of K+ (80 mM)-induced contraction (n = 5; rings = 10). There was no significant difference in maximum contraction of pulmonary artery by EPA in the presence of ouabain (0.1 μM). The maximum contraction was 19.80 ± 3.10% of K+ (80 mM)-induced contraction in the presence of ouabain (Figure 5).

Effect of eicosapentaenoic acid (EPA; 0.1–100 μM) on basal tone of the sheep pulmonary artery rings in the presence and absence of ouabain (0.1 μM). The change in tone by the fatty acids is expressed as per cent contraction of 80 mM K+-depolarizing solution. Results are presented as mean ± SEM.
Effect of EPA on basal cGMP production in sheep pulmonary artery
Figure 6 depicts the effect of EPA on basal cGMP production in pulmonary artery. Dipyridamole (3 × 10−5 M) was used to prevent the breakdown of cGMP by phosphodiesterase 5 (PDE5). In the presence of phosphodiesterase inhibitor, the basal cGMP level was 38.50 ± 8.65 pmol/mg protein. At 30 μM concentration, EPA (54.30 ± 9.437 pmol/mg) had no significant effect on intracellular cGMP levels. SNP (10 μM) significantly increased tissue cGMP (76.30 ± 7.89 pmol/mg protein; n = 6).

Eicosapentaenoic acid (EPA; 30 μM) increased cyclic guanosine monophosphate (cGMP) level in the sheep pulmonary arterial segments, but it was not significantly different from the control. The data were analyzed by one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test. Vertical bars represent SEM.
Discussion
This study demonstrates that (1) acute exposure to EPA (30 μM) inhibited KCl-induced relaxation in K+-free MKHS, (2) prolonged exposure (24 h) to EPA (30 μM) decreased the protein expression of the α1 isoform of Na+-K+-ATPase of organ cultured vascular rings as well as KCl-induced relaxation, (3) acute exposure (30 min) to EPA (30 μM) caused an inhibition of the vascular Na+-K+-ATPase activity, (4) EPA (10 and 30 μM) concentrations had no effect on Mg2+-ATPase activity, (5) concentration of EPA (1–100 μM) caused a dose-dependent increase in basal tone of the sheep pulmonary artery, but ouabain (0.1 μM) had no effect on EPA-induced contraction in this artery and (6) EPA (30 μM) had no effect on cGMP production.
EPA reduced KCl-induced relaxation in sheep pulmonary artery which is an index of functional sodium pump activity. Our findings regarding inhibition of Na+-K+-ATPase are supported by the inhibitory effect of EPA on other tissues such as human endothelial cells and synaptosomal membrane of cerebral cortex of rat).14–16 This functional study is also supported by the inhibition of Na+-K+-ATPase activity. Evaluation of enzyme kinetics (dose–response curves for ouabain) in membrane preparations allowed correlating the ouabain affinity of α1 isoform with fatty acid composition. The affinity of the α1 isoform for ouabain was significantly increased with accretions in the total amount of fatty acids of the n−6 series. 24 Polyunsaturated fatty acids are major components of membrane phospholipids and can modify membrane function. For example, EPA is known to increase membrane fluidity by increasing membrane cholesterol efflux. 3 Since the dietary free fatty acid concentrations can reach 20–30 μM, the inhibitory effect induced by EPA on Na+-K+-ATPase in vitro may have a physiological relevance.25,26 In addition to evaluating the acute effects of EPA on Na+-K+-ATPase activity, we also examined the influence of prolonged exposure to EPA in pulmonary artery kept in organ culture medium for 24 h, but there was no significant difference between acute and chronic exposures to EPA in isolated pulmonary artery. EPA decreased the protein expression of the α1 isoform of Na+-K+-ATPase in organ culture. Western blot data together with functional data relating to K+-induced relaxation showed inhibition of protein expression due to EPA. It is, therefore, possible that prolonged exposure to EPA may alter gene regulation of Na+-K+-ATPase. The change in the density of the receptor may be one of the mechanisms for the change in the expression. This change in the density of the receptor may lead to change in the gene expression and regulation. EPA appears to inhibit Na+-K+-ATPase in the pulmonary artery through attenuation of expression of α-1 isoform of Na+-K+-ATPase. Organ culture is a useful method for analyzing the long-term effects of various pharmacological agents on vasculature and basic biology. 27
It is known that inhibition of Na+-K+-ATPase increases basal tone in several vascular smooth muscles. Therefore, it is reasonable to predict that the EPA used in the present study may increase the tone of sheep pulmonary vessel through inhibition of Na+-K+-ATPase. However, such a possibility appears to be remote from the observation that near maximal inhibition of Na+-K+-ATPase by ouabain (0.1 μM) barely had any effect on the basal tone of pulmonary artery. 18 Therefore, a marginal increase in pulmonary artery vascular tone by 30 μM EPA may not relate to inhibition of Na+-K+-ATPase. We recently reported that ouabain enhanced the contractility to the vasoconstrictor 5-HT in the sheep pulmonary artery. 18 EPA had no effect on cGMP production that means the role of soluble guanylyl cyclase (sGC) activity is ruled out in the inhibitory mechanism.
In conclusion, the results of the present study show that EPA inhibits functional Na+-K+-ATPase, its activity and protein expression of the α1 isoform of Na+-K+-ATPase in pulmonary vessel. EPA-induced inhibition of Na+-K+-ATPase may be due to attenuation in protein expression of the α1 isoform of Na+-K+-ATPase, but it is independent of cGMP production. Mg2+-ATPase may have no role in the KCl-induced relaxation. But prolonged exposure had no additional effect on sodium pump.
However, the influence of this EPA on the myogenic tone appears not to be related to the inhibition of the sodium pump. The activity of this fatty acid observed in the present study is of clinical significance in the management of vascular diseases.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
