Abstract
The inhibitory effect of vernolide-A (C21H28O7) on lung metastasis induced by B16F-10 melanoma cells was studied using C57BL/6 mice. Vernolide-A was administered in three different modalities such as simultaneously with tumor, prophylactic to tumor and after tumor development. Maximum inhibition in the metastasis was observed when vernolide-A was administered simultaneously with tumor. There was 89.39% inhibition of lung tumor nodule formation and 88.51% increase in the life span of metastatic tumor-bearing animals. Highly elevated levels of lung hydroxyproline, lung uronic acid, lung hexosamine, serum sialic acid, serum γ-glutamyl transpeptidase (GGT) and serum vascular endothelial growth factor (VEGF) in the metastatic control animals were found to be significantly lowered in the vernolide-A-treated animals. Histopathological analysis of lung tissues also correlated with these results. Vernolide-A administration downregulated the expression of matrix metalloproteinase-2 (MMP-2), MMP-9, extracellular-signal-regulated kinase-1 (ERK-1), ERK-2 and VEGF in the lung tissue of B16F-10 melanoma challenged animals. In the in vitro system, vernolide-A showed a significant inhibition of invasion of B16F-10 melanoma cells across the collagen matrix. Vernolide-A treatment also inhibited the migration of B16F-10 melanoma cells across a polycarbonate filter in vitro. Vernolide-A could inhibit MMP-2 and MMP-9 protein expression in gelatin zymographic analysis of B16F-10 cells. 3 H-thymidine proliferation assay showed that vernolide-A could inhibit the proliferation of B16F-10 melanoma cells in vitro. These results indicate that vernolide-A could inhibit the metastatic progression of B16F-10 melanoma cells in mice.
Introduction
Cancer is a major health problem in the world. Globally, 9 million deaths result from cancer each year, a figure the World Health Organization (WHO) anticipates will rise to 20 million by 2020, again due to increased life expectancy and lifestyle changes. A total of 1,529,560 new cancer cases and 569,490 deaths from cancer are projected to occur in the US in 2010. 1 Based on the National Cancer Registry (India) reports, there are nearly 800,000 new cancer cases in India every year. 2 Metastasis, the spread of cancer from the primary site of tumor growth to other organs is the leading cause of cancer-related morbidity and mortality. Death from cancer is most often due to metastases that are resistant to conventional therapies. 3 It is the single most important obstacle to curative cancer therapy. Although there are several drugs available to encounter cancer growth in humans, there are no drugs presently available to specifically inhibit the metastasis of cancer cells. Tumor cell metastasis is a complex, multistep process that involves cell separation from the primary tumor, entry into the vascular and lymphatic systems, transport to and arrest within the microcirculation of distant organs, and extravasation. 4 Any drug, which can inhibit one of the steps in the cascade, will be useful in the inhibition of tumor metastasis.
MMPs are a family of endopeptidases which digest proteins of the extracellular matrix (ECM). They are important for regulating normal tissue development in morphogenesis, angiogenesis, and wound healing 5 ; however, unregulated activities of MMPs can play a role in many disease states including cancer expansion, invasion and metastasis, arthritis and atherosclerosis. 6 MMP-2 (gelatinase A, 72 kDa) and MMP-9 (gelatinase B, 92 kDa) are the most vital enzymes for the degradation of the main constituent of the basement membrane, Type IV collagen, and are therefore deeply involved in cancer invasion and metastasis. Therefore, the inhibition of migration or invasion mediated by MMPs could be a preventive way of cancer metastasis. 7
Plant-derived compounds have been an important source of several clinically useful anti-cancer agents. 8 Several antimetastatic drugs or compounds for cancer have been found in natural products. Sesquiterpene lactones (SLs) are the active constituents of a variety of medicinal plants used in traditional medicine for the treatment of inflammatory diseases. 9 Various SLs have been demonstrated to execute their anti-cancer capability via inhibition of inflammatory responses, prevention of metastasis and induction of apoptosis. 9 All SLs contain a common functional structure, α-methylene-γ-lactone group, and this important chemical characteristic means that the thiol reactivity of SLs is an underlying mechanism responsible for their bioactivities. 9 Vernolide-A (C21H28O7) is a SL present in the plant Vernonia cinerea L. (Asteraceae). Biological evaluation showed that vernolide-A has potent cytotoxicity against human KB, DLD-1, NCI-661, and HeLa tumor cell lines. 10 We have already reported the antioxidant and anti-inflammatory activity of methanolic extract of Vernonia cinerea. Study shows that administration of Vernonia cinerea can inhibit the elevated proinflammatory cytokine level in carrageenan-induced paw edema model. 11 Vernolides are major sesquiterpenoids reported in this plant and are considered as the active principle.10,12 In the present study, the effect of vernolide-A on the inhibition of pulmonary metastasis induced by B16F-10 melanoma cells in C57BL/6 mice was evaluated with special emphasis on the mechanism of action using in vitro models.
Materials and methods
Animals
Balb/c and C57BL/6 mice (20–25 g body weight [b.wt.], 6–8-weeks-old males) were purchased from National Institute of Nutrition, Hyderabad, India. The animals were fed with mouse chow (Sai Feeds, India) and water ad libitum. All the animal experiments were carried out with the prior approval of the Institutional Animal Ethics Committee (IAEC) and were conducted strictly adhering to the guidelines of the Committee for the purpose of Control and Supervision of Experiments on Animals (CPCSEA No: 2009/12) constituted by the Animal Welfare Division of Government of India.
Cell line
B16F-10 melanoma, a highly metastatic cell line, was obtained from the National Centre for Cell Sciences, Pune, India. The cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and antibiotics.
Chemicals
DMEM was obtained from Himedia (Mumbai, India). FBS was procured from Life Technologies (Grand Island, NY, USA). 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT), hydroxyproline and glucuronic acid lactone were purchased from Sigma Chemicals (St. Louis, MO, USA). N-acetylneuraminic acid (sialic acid) was purchased from Sisco Research Laboratory, Mumbai. Oligonucleotide primer sequences were purchased from Maxim Biotech Inc (San Francisco, CA, USA; Table 1 ). Radioactive thymidine [3H] was purchased from BRIT (Mumbai, India). Highly specific quantitative sandwich enzyme-linked immunosorbent assay (ELISA) kit for mouse vascular endothelial growth factor (VEGF) was purchased from R&D system, USA. All other reagents were of analytical grade.
Primer sequences of different genes

bp: base pairs, ERK: extracellular signal-regulated kinase, GAPDH: reduced glyceraldehyde phosphate dehydrogenase, MMP: matrix metalloprotease, VEGF: vascular endothelial growth factor
Isolation of vernolide-A
Vernolide-A (Figure 1 ), a SL, was isolated from Vernonia cinerea as explained previously. 13 Vernonia cinerea was collected from Thrissur district and a voucher specimen with number ACRC (Astr-08) was deposited at the herbarium of Amala Cancer Research Centre (Kerala, India) after authentication. The whole plant was air dried and powdered. The air-dried powder (3.0 kg) was extracted with 95% ethanol, three times at room temperature. After removal of the ethanol under reduced pressure, the crude extract was dissolved in water to form a suspension, and partitioned with ethyl acetate to get ethyl acetate soluble extract. The ethyl acetate soluble extract was subjected to a silica gel column chromatography eluted with gradient solvents of petroleum–ethyl acetate (10:0–0:10), and then methanol to afford eight fractions. The Fraction 7 containing mainly sesquiterpenoids was fractionated by a column packed with Mitsubishi Chemical Infonics (MCI) gel (MeOH-H2O; 30–80%) to obtain three major Fractions 7a-7c. Fraction 7b was chromatographed over a silica gel column using chloroform (CHCl3)-MeOH (10:0–10:1) as the eluting solvent to obtain vernolide-A. The physical and chemical data, including infrared, 1H nuclear magnetic resonance, carbon-13 nuclear magnetic resonance spectroscopy and high-resolution mass spectrum of vernolide-A, were identical with the results published in previous report. 10
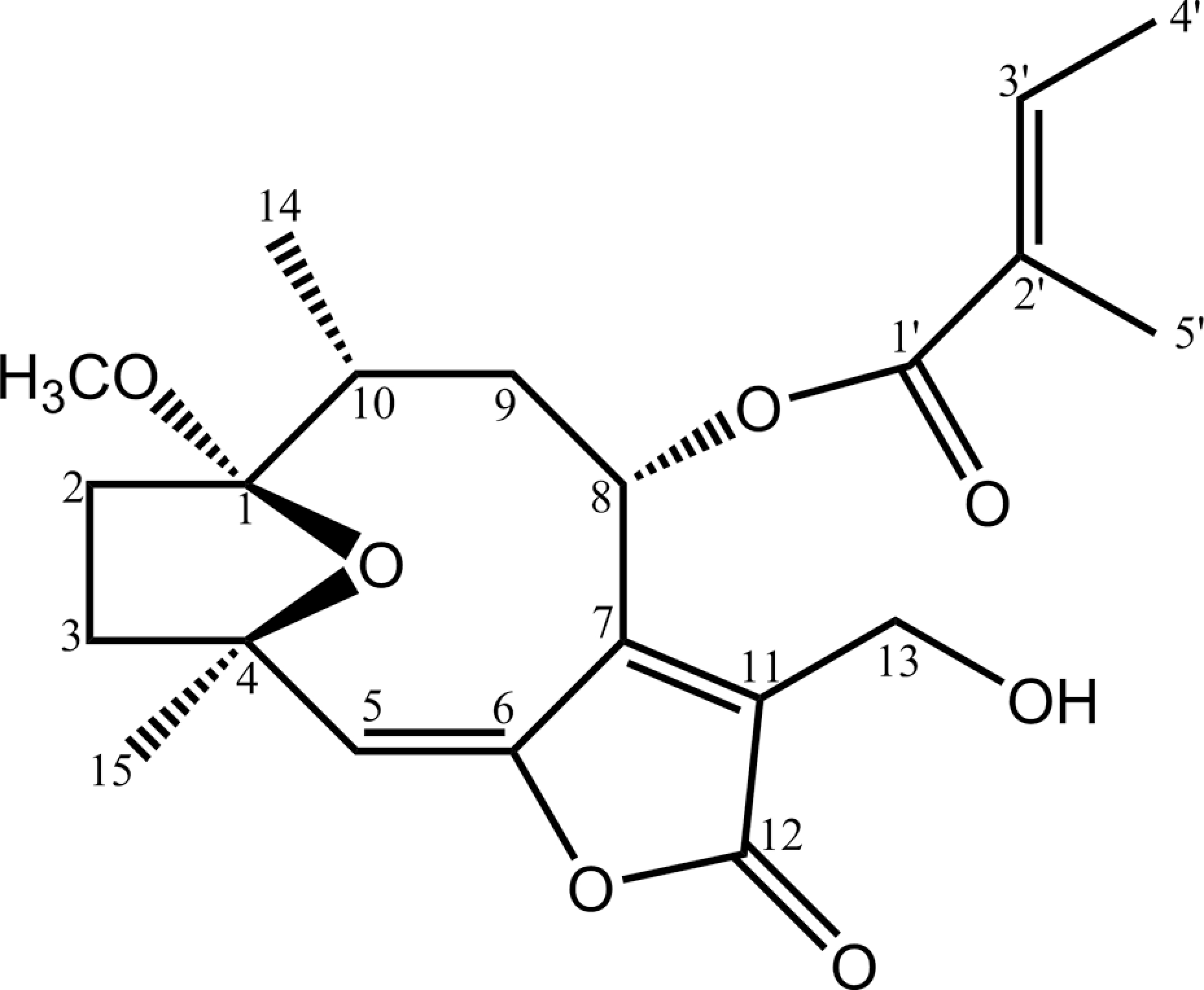
Structure of vernolide-A.
Toxicological evaluation
Balb/c mice were divided into five groups (N = 10). The control group received 1% gum acacia in Phosphate buffered saline, while the experimental groups received 10 doses of vernolide-A (0.5, 1, 2 and 4 mg/kg b.wt.) intraperitoneally for 10 days. Animals were observed for the mortality, behavior changes and change in body weight. All the animals were killed by cervical dislocation after 14 days of drug administration. Selected organs such as liver, spleen, thymus, kidney, and lungs were dissected out and weights were recorded. Blood was collected by heart puncture immediately after killing; serum separated and was used for the analysis of hepatic and renal functions. Liver function markers, such as alkaline phosphatase (ALP) 14 and glutamate pyruvate transaminase (GPT), 15 and kidney function markers, such as creatinine 16 and blood urea nitrogen (BUN), 17 were determined.
Drug administration
The dosage of vernolide-A for in vivo studies was determined by toxicity analysis and was given at a concentration of 500 μg/kg b.wt. intraperitoneally. The drug was administered in three different modalities as follows: Prophylactic administration: Animals were treated with 10 consecutive doses of the drug prior to tumor inoculation. Simultaneous administration: The drug was given to the animals simultaneously with metastatic tumor cells and continued for10 consecutive days. Developed administration: A total of 10 days after tumor inoculation the drug was administered for 10 consecutive days.
Determination of the antimetastatic activity in the in vivo system
Pulmonary colonization assay
C57BL/6 mice were divided into four groups (14 mice/group). All the animals were induced metastasis by injecting B16F-10 melanoma cells (10 6 cells/ animal) through lateral tail vein.18,19 To three groups of animals, vernolide-A was administered intraperitoneally at a dose of 500 μg/kg b.wt. for 10 consecutive days in three different modalities—prophylactic administration (Group I); simultaneous administration (Group II) and developed administration (Group III). Group IV animals were kept as untreated metastatic tumor-bearing control. Eight animals from each group were killed on the 21st day after tumor challenge, the lungs were excised and the blood was collected. Lungs were used for morphological examinations of metastatic tumor nodules and for the estimation of collagen hydroxyproline, 20 hexosamine 21 and uronic acid 22 contents. Serum was separated from the blood and used for determining the sialic acid 23 and γ-glutamyl transpeptidase (GGT) 24 levels. Serum VEGF was estimated using highly specific ELISA kit purchased from R&D system, USA, as per manufacture’s protocol. A portion of the lung was used for histopathological analysis. The rest of the six animals in each group were observed for their survival. The mortality of the animals was observed and the percentage increase in life span (%ILS) was calculated using the formula %ILS = T − C/C × 100, where T represents the number of survival days of treated animals and C represents the number of survival days of control animals.
Determination of the gene-expression levels of MMP-2, MMP-9, ERK-1, ERK-2 and VEGF
From the excised lungs, total RNA was isolated by the method of Chomczynski and Sacchi. 25 Reverse transcription reaction mixtures (10× polymerase chain reaction [PCR] buffer; 25 mM magnesium chloride, MgCl2; 10 mM dinucleotide triphosphates [dNTPs]; Oligo (dT) RNase inhibitor, Avian Myeloblastosis Virus (AMV) Reverse Transcriptase) were incubated with 5 μl of RNA sample using a thermocycler at 42°C for 60 min, 92°C for 5 min and 4°C for 5 min. After the reaction, the vials were stored at –70°C. Complementary DNA (cDNA) was amplified with specific primers of ERK-1, ERK-2, MMP-2, MMP-9 and VEGF against the house-keeping gene, glyceraldehyde phosphate dehydrogenase reduced (GAPDH), using PCR. The following PCR reaction profile was followed in the minicycler: 1 min at 94°C, 1 min at 55°C and 1 min at 72°C for 39 cycles followed by a 10-min extension at 72°C. Amplified samples were subjected to electrophoresis in an agarose gel (1.5%) containing 0.5 mg/ml ethidium bromide and photographed under ultra violet (UV) light. Band intensity was measured using software provided with Vilber Lourmat (Photo-Capt Version 12.3 for Windows) gel documentation system and percentage expression was calculated.
Histopathological analysis
Lung tissues were fixed in 10% formalin, dehydrated in different concentrations of alcohol and embedded in paraffin wax. Sections (4 μm) were stained with hematoxylin and eosin (H&E).
Determination of antimetastatic activity using in vitro models
The following parameters were used to assess the antimetastatic mechanism of action in the in vitro system.
Cell Viability
B16F-10 melanoma cells were seeded (5000 cells/well) in a 96-well flat bottomed titer plate and incubated for 24 h at 37°C in 5% carbon dioxide (CO2) atmosphere. Different concentrations of vernolide-A (0.01–10 μg/ml) were added and incubated further for 48 h. Before 4 h of completion of incubation, 20 μl MTT (5 mg/ml) was added.26,27 Percentage of viable cells was determined using an ELISA plate reader set to record absorbance at 570 nm.
Proliferation assay
B16F-10 melanoma cells (5000 cells/well) were plated in a 96-well culture plate and incubated at 37°C in 5% CO2 atmosphere. After 24 h, various concentrations of vernolide-A (0.01, 0.05 and 0.1 μg/ml) were added and further incubated for 48 h. 3 H-thymidine was added to each well (1 μCi/well) and incubation was continued for additional 18 h. After completing incubation, the plate was centrifuged and the culture supernatant was removed, the cells were washed three times with PBS and then treated with ice-cold perchloric acid for 15 min. The resulting precipitate was dissolved in 0.5 N sodium hydroxide (NaOH) and was added to the scintillation fluid and kept overnight in the dark. The radioactivity was counted using a Rack Beta liquid scintillation counter.
Collagen matrix invasion assay
The invasion assay was carried out in modified Boyden chambers as described by Albini et al. 28 The lower compartment of the chamber was filled with serum-free DMEM and a polycarbonate filter coated with 25 μg Type I collagen was placed above this. B16F-10 melanoma cells (10 5 cells/150 μl DMEM) were then seeded on to the upper chamber in the presence and absence of vernolide-A (0.01, 0.05 and 0.1 μg/ml) and incubated at 37°C in 5% CO2 atmosphere for 10 h. After incubation, the filters were removed, fixed with methanol and stained with crystal violet. Cells migrating to the lower surface of the polycarbonate filters were counted under a microscope. The results were expressed as percentage inhibition of invasion.
Migration assay
The effect of vernolide-A on the migration of the tumor cells was carried out using the Boyden chamber. The polycarbonate filters were placed in Boyden chambers. The lower compartment of the chamber was filled with serum-free DMEM, and B16F-10 cells (10 5 cells/chamber) in DMEM were then seeded on to the upper chamber in the presence and absence of vernolide-A (0.01, 0.05 and 0.1 μg/ml) and incubated for 10 hours at 37°C in 5% CO2. The number of migrated cells in the lower compartment was measured by hemocytometer counting.
Gelatin zymography
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was performed with 5% gelatin incorporated in the separating gel. 29 B16F-10 melanoma cells of subconfluent cultures were incubated with serum-free medium for 24 h at 37°C in 5% CO2 atmosphere. The conditioned medium was then collected and subjected to zymographic analysis. Fifty microliters of sample (equivalent to 100 μg protein) was activated with 5 μl trypsin solution (75 μg/ml) in the presence and absence of vernolide-A (0.01, 0.05 and 0.1 μg/ml) in 0.1 M Tris-HCl, 10 mM calcium chloride (CaCl2) buffer (pH 8.0) and incubated for 1 h at room temperature. Samples were mixed with an equal volume of 2× sample buffer and loaded on to 11% polyacrylamide gels containing 5% gelatin. Electrophoresis was carried out at 4°C with constant current of 2 mA/tube until the tracking dye reached the periphery. The gels were then washed with 2% Triton X-100 in 0.1 M Tris-HCl, 10 mM CaCl2 at 37°C for 18 h followed by staining with Gelcode Blue stain reagent for 2 h. Gels were then destained to visualize the clear area against the dark background.
Statistical analysis
Values are expressed as mean ± SD. The statistical analysis was done using one-way analysis of variance (ANOVA) followed by Dunnett’s test.
Results
Toxicological evaluation
Short-term administration of vernolide-A at concentrations of 0.5 and 1 mg/kg b.wt. for 10 days did not cause any mortality, change in behavior, body weight, relative organ weight, hepatic and renal functions compared to normal untreated animals. Vernolide-A at 2 mg/kg showed mild toxicity, whereas vernolide-A at 4 mg/Kg exhibited an observable toxicity showing behavior changes, mortality, weight loss and decrease in the organ weight and also alterations in both hepatic and renal functions (Table 2 ).
Toxicity profile of vernolide-A a
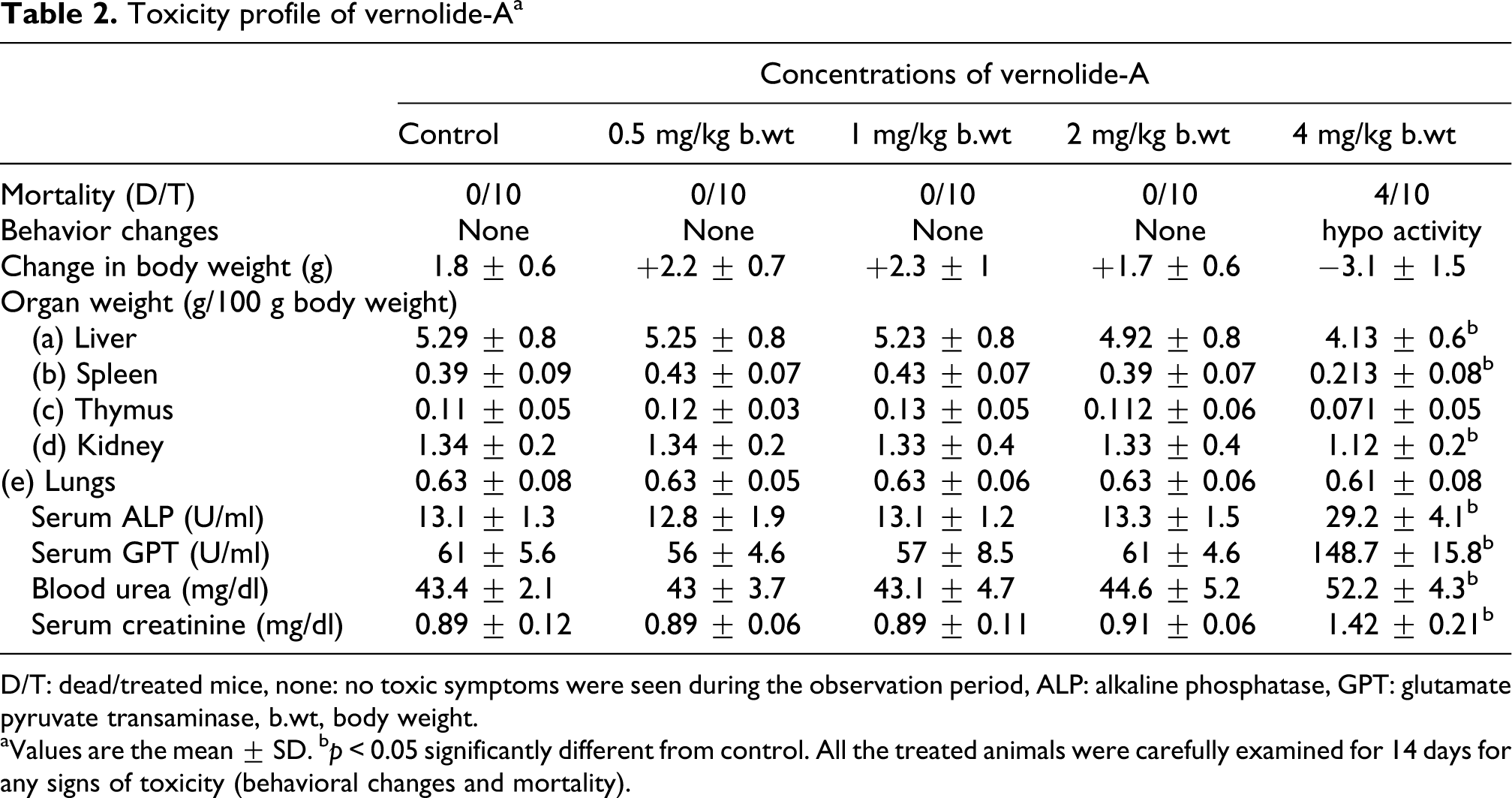
D/T: dead/treated mice, none: no toxic symptoms were seen during the observation period, ALP: alkaline phosphatase, GPT: glutamate pyruvate transaminase, b.wt, body weight.
aValues are the mean ± SD.
b p < 0.05 significantly different from control. All the treated animals were carefully examined for 14 days for any signs of toxicity (behavioral changes and mortality).
Antimetastatic activity in the in vivo system
Effect of vernolide-A on the inhibition of lung metastasis and survival
Metastatic tumor-bearing animals treated with vernolide-A showed significant reduction in tumor nodule formation (Table 3 ). Metastatic control animals had massive tumor growth and were assigned an arbitrary number of 250. 30 The three different modalities of compound administration were found to be significantly (p < 0.001) effective. Of this, simultaneous mode of administration of vernolide-A produced maximum inhibition of lung nodules (89.39%) followed by prophylactic mode of administration (82.95%) and administration after tumor development (76.54%).
Effect of vernolide-A on lung colonization of B16F-10 melanoma cells and survival of animals a
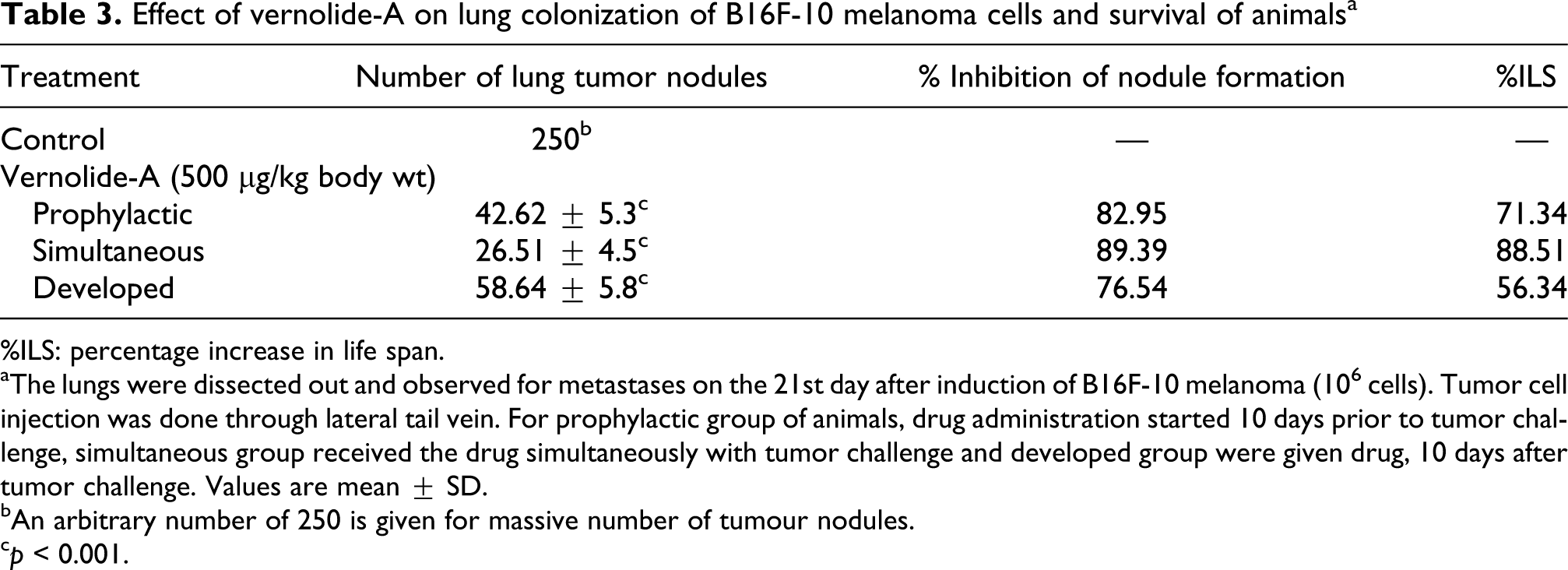
%ILS: percentage increase in life span.
aThe lungs were dissected out and observed for metastases on the 21st day after induction of B16F-10 melanoma (106 cells). Tumor cell injection was done through lateral tail vein. For prophylactic group of animals, drug administration started 10 days prior to tumor challenge, simultaneous group received the drug simultaneously with tumor challenge and developed group were given drug, 10 days after tumor challenge. Values are mean ± SD.
bAn arbitrary number of 250 is given for massive number of tumour nodules.
c p < 0.001.
Administration of vernolide-A significantly increased the life span of metastatic tumor-bearing animals (Table 3). The life span was highly enhanced when vernolide-A was administered simultaneously to 88.51%. Prophylactic administration of vernolide-A enhanced the life span by 71.34%, whereas administration after tumor development increased the life span by 56.34%.
Effect of vernolide-A on lung and serum biochemical parameters of metastatic tumor-bearing animals
The effect of vernolide-A on lung collagen hydroxyproline, hexosamine and uronic acid content is shown in Table 4 . Control animals showed an increased level of lung collagen hydroxyproline (22.56 ± 2.41 μg/mg protein) which was significantly (p < 0.001) reduced in animals treated with vernolide-A in prophylactic (10.51 ± 1.2 μg/mg protein), simultaneous (6.62 ± 0.82 μg/mg protein) and developed (15.83 ± 1.6) modalities.
Effect of vernolide-A on the lung biochemical parameters of metastatic melanoma cell-bearing animal a
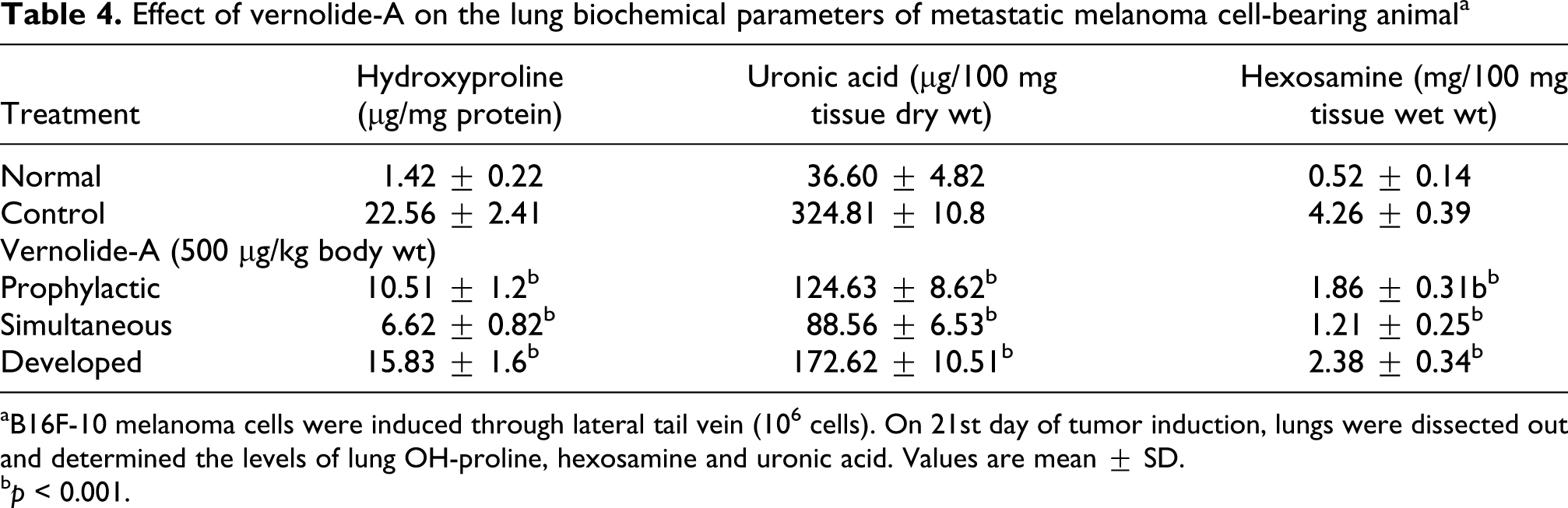
aB16F-10 melanoma cells were induced through lateral tail vein (106 cells). On 21st day of tumor induction, lungs were dissected out and determined the levels of lung OH-proline, hexosamine and uronic acid. Values are mean ± SD.
b p < 0.001.
Tumor-bearing control animals had a high level of lung uronic acid content (324.81 ± 10.8 μg/100 mg tissue wet wt.; p < 0.001) compared to normal animals (36.60 ± 4.82 μg/100 mg tissue wet wt.). Treatment of vernolide-A reduced the uronic acid content in all the three modalities; prophylactic (124.63 ± 8.62 μg/100 mg tissue wet wt.), simultaneous (88.56 ± 6.53 μg/100 mg tissue wet wt.) and developed (172.62 ± 10.51 μg/100 mg tissue wet wt.) modalities.
Hexosamine content was also high in the lungs of tumor control (4.26 ± 0.39 mg/100 mg tissue dry wt.) compared to normal animals (0.52 ± 0.14 mg/100 mg tissue dry wt.). Vernolde-A treatment significantly (p < 0.001) reduced the lung hexosamine content in both prophylactic (1.86 ± 0.31 mg/100 mg tissue dry wt.) and simultaneous (1.21 ± 0.25 mg/100 mg tissue dry wt.) modalities, whereas it was only 2.38 ± 0.34 mg/100 mg tissue dry wt. in developed modalities.
The effect of vernolide-A on serum biochemical parameters and serum VEGF is presented in Table 5 . The serum sialic acid level of control metastatic tumor-bearing animals was highly increased (112.52 ± 8.31 μg/ml serum) as compared to normal (24.83 ± 2.64 μg/ml serum). Here also the simultaneous administration of vernolide-A significantly (p < 0.001) reduced the elevated serum sialic acid level to 34.62 ± 2.85 μg/ml serum, followed by prophylactic modality (58.41 ± 3.34 μg/ml serum); and in developed modality it was reduced only to 86.51 ± 5.46 μg/ml serum.
Effect of vernolide-A on serum biochemical parameters and serum VEGF a

VEGF: vascular endothelial growth factor, GGT: γ-glutamyl transpeptidase.
aB16F-10 melanoma cells were induced through lateral tail vein (106 cells). On 21st day of tumor induction, blood was collected by heart puncture, serum separated to determine the levels of sialic acid, GGT and VEGF. Vernolide-A treatment was done in different modalities (10 doses at 24 h interval, intraperitoneally).Values are mean ± SD.
b p < 0.001.
Serum GGT level was also significantly (p < 0.001) enhanced in metastatic control animals (115.61 ± 6.14 U/ml) compared to normal animals (26.43 ± 2.38 U/ml). After the administration of vernolide-A, the elevated γ- GGT level was reduced significantly (p < 0.001) to 54.86 ± 3.85 U/ml in prophylactic and 38.62 ± 2.54 U/ml in simultaneous modalities. In animals with developed tumor, the serum GGT level was reduced to 82.45 ± 4.96 U/ml after vernolide-A treatment.
Effect of vernolide-A on the VEGF level in metastases-bearing animals
Serum VEGF level is shown in Table 5. The normal serum VEGF level was only 16.8 ± 2.4 pg/ml. This was significantly increased (p < 0.001) to 154.2 ± 16.2 pg/ml in metastatic tumor-bearing mice. This elevated level of serum VEGF was reduced significantly (p < 0.001) in vernolide-A administered group with prophylactic (86.5 ± 8.4 pg/ml) and simultaneous (75.3 ± 6.5 pg/ml) modalities. The elevated serum VEGF level was also reduced in the animals with developed tumor (113.4 ± 9.4 pg/ml) by vernolide-A administration.
MMP-2, MMP-9, ERK-1, ERK-2 and VEGF expression
MMP-2, MMP-9, ERK-1, ERK-2 and VEGF genes were clearly expressed in metastases control animals (Figure. 2 ). Expression of all these genes were downregulated or suppressed in the treated group where vernolide-A was administered both prophylactically and simultaneously with the tumor induction. But there were a diminished expression of these genes in animals treated with vernolide-A after tumor development (Figure 2 (A)).

Expression of MMP-2, MMP-9, ERK-1, ERK-2 and VEGF. (A) Lane 1: metastatic bearing control; Lane 2: vernolide-A treated prophylactically; Lane 3: vernolide-A treated simultaneously; Lane 4: vernolide-A treated 10 days after tumor induction. (B) Band intensity was measured using software provided with Vilber Lourmat gel documentation system and percentage expression of messenger RNA (mRNA) was calculated. All the values are mean ± SD (mean of triplicate). *p < 0.001, compared to control.
In the untreated control, the percentage expression of MMP-2, MMP-9, ERK-1, ERK-2 and VEGF were found to be 85.38%, 81.42%, 95.3%, 49.69% and 91.33%, respectively, compared to an internal control GAPDH (100%). Vernolide-A treatment downregulated the gene expression of MMP-2, MMP-9, ERK-1, ERK-2 and VEGF in all the three treatment modalities (Figure 2 (B)).
Histopathological analysis of lungs
The H&E-stained sections of lung tissues are shown in Figure 3 (×100). Lungs in the control animals showed infiltration of the neoplastic cells around the main bronchioles extended to the pleura. This together with fibrosis reduces alveolar space, which in turn leads to reduced vital capacity. Prophylactic and simultaneous administration of vernolide-A showed significant reduction in tumor mass. Alveoli and pleura were tumor free, alveolar passage lined with healthy ciliated columnar epithelial cells and almost similar to normal lung. Considerable reduction in tumor mass was also observed in developed modalities of administration.

Histopathology of the lung of metastatic tumor-bearing animals (×100). Lungs of the metastasis-induced animals were fixed in neutral buffered formalin, and 4 µm sections were taken and stained with hematoxylin and eosin: (A) normal lung, (B) metastasis-bearing control, (C) vernolide-A treated prophylactically, (D) vernolide-A treated simultaneously, (E) vernolide-A treated 10 days after tumor induction.
Determination of antimetastatic activity in the in vitro system
Cell viability assay
Cytotoxicity of vernolide-A toward B16F-10 melanoma cells in culture is shown in Table 6 . At concentration of 0.01–0.1 μg/ml vernolide-A was found to be non-toxic to B16F-10 melanoma cells and these concentrations were used for further in vitro experiments.
Cell viability assay

B16-F10 melanoma cells (5000 cells/well) were plated in a 96-well titer plate with different concentrations of vernolide-A and incubated for 48 h. Percentage of viable cells were determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (MTT assay).
Tumor cell proliferation
Proliferation rate was determined by the 3 H-thymidine incorporation by the DNA of B16F-10 melanoma cells. Thymidine incorporation is proportional to the potential of the cells to synthesize DNA. Proliferation was expressed as radioactive count per minute. Untreated B16F-10 cells had very high rate of proliferation (4652.6 ± 262 cpm). Administration of vernolide-A at a concentration of 0.1 μg/ml significantly (p < 0.001) reduced the proliferation (1635.4 ± 184 cpm, 64.84%) of B16F-10 melanoma cells. Considerable inhibition in the proliferation of tumor cells was also observed when vernolide-A was administered at a concentration of 0.05 μg/ml (2475.3 ± 175 cpm, 46.79%) and 0.01 μg/ml (3528.4 ± 218 cpm, 24.16%; Table 7 ).
Effect of vernolide-A on tumor cell proliferation a

aB16F-10 melanoma cells (5 × 103 cells/well) were grown in 96-well flat bottom plate. After 24 h, various concentrations of vernolide-A were added and incubation was continued for 48 h. After incubation, 3H-thymidine was added to each well (1 μCi/well) and incubation was continued for 18 h. Cells were lysed and radioactivity was counted using Rack Beta liquid scintillation counter. Values are mean ± SD.
b p < 0.001
c p < 0.01.
Collagen matrix invasion assay
Metastatic B16F-10 melanoma cells show highly invasive property through the collagen matrix. Very high numbers of cells were found in the lower surface of the polycarbonate membrane, but administration of vernolide-A produced significant inhibition in the invasion of the collagen matrix by the tumor cells in a dose-dependent manner. At a concentration of 0.1 μg/ml, vernolide-A significantly inhibited the invasion of B16F-10 melanoma cells by 88.62%, whereas at 0.05 μg/ml and 0.01 μg/ml the percentage of invasion inhibition was found to be 72.93% and 31.42%, respectively (Figure 4 ).
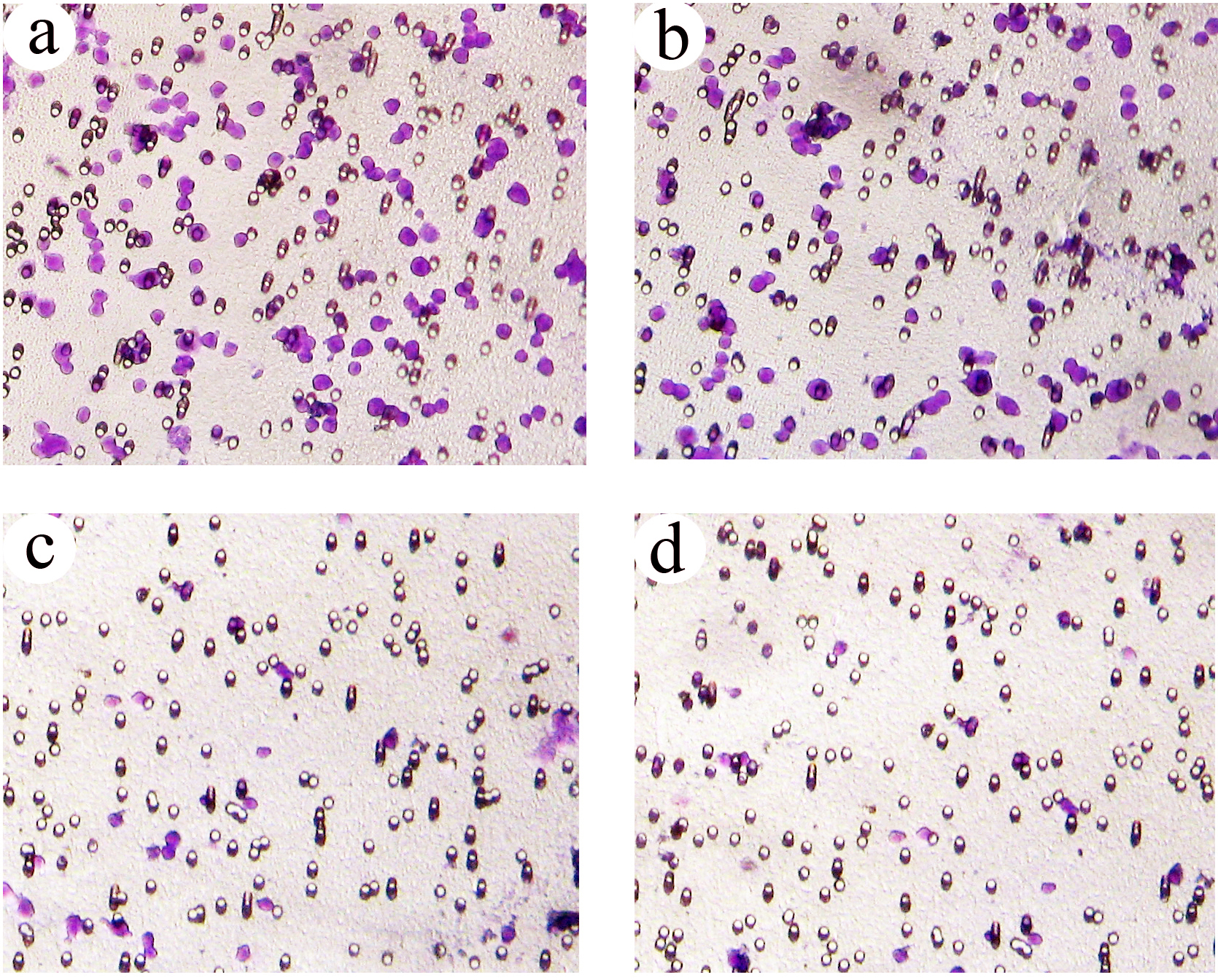
The effect of vernolide-A on the collagen matrix invasion. (A) Untreated control; (B) treatment with vernolide-A (0.01 μg/ml); (C) treatment with vernolide-A (0.05 μg/ml); (D) treatment with vernolide-A (0.1 μg/ml).
Migration
Inhibition of tumor cell migration by vernolide-A is given in Figure 5 . Vernolide-A significantly inhibited the migration of B16F-10 melanoma cells across the polycarbonate filters in a dose-dependent manner. In untreated control 1885 ± 68 cells were observed under the polycarbonate membrane and it was significantly reduced to 1479 ± 56, 976 ± 48 and 517 ± 43 cells when the cells were treated with vernolide-A at 0.01, 0.05 and 0.1 μg/ml, respectively. The percentage inhibitions in the invasion of B16F-10 cells after the treatment with vernolide-A at these concentrations were found to be 21.5%, 48.2% and 72.6%, respectively.

The effect of vernolide-A on tumor cell migration. B16F-10 cells were seeded on to the upper surface of a polycarbonate filter and incubated for 10 h. Cells that had undergone migration were quantified after fixation and staining and photographed at ×200 magnification and percentage inhibition was calculated. All the values are mean ± SD (mean of triplicate). ***p < 0.001, compared to control.
Gelatin zymographic analysis
Vernolide-A inhibited the activation of MMPs produced by B16F-10 melanoma cells as shown in Figure 6 . Conditioned medium, after trypsin activation, showed digested clear areas at 92 and 72 kDa, which was identical to MMP-9 and MMP-2 activity (Figure 6 (B)). Gels loaded with conditioned medium, without trypsin activation, did not show any clear areas, indicating the inactive form of the enzyme collagenase (Figure 6 (A)). Trypsin-activated conditioned medium loaded gels, after incubation with 10 mM EDTA, did not show clear areas which indicates that the enzyme responsible for degradation is metalloproteinase (Figure 6 (C)). When the conditioned medium was treated with vernolide-A during trypsin activation no clear bands were observed (Figure 6 (D) and (E)), indicating that vernolide-A inhibited the activation of procollagenase to active collagenase at concentrations of 0.1 and 0.05 μg/ml. Conditioned medium from cells treated with vernolide-A at a concentration of 0.01 μg/ml showed two faded bands (Figure 6 (F)).
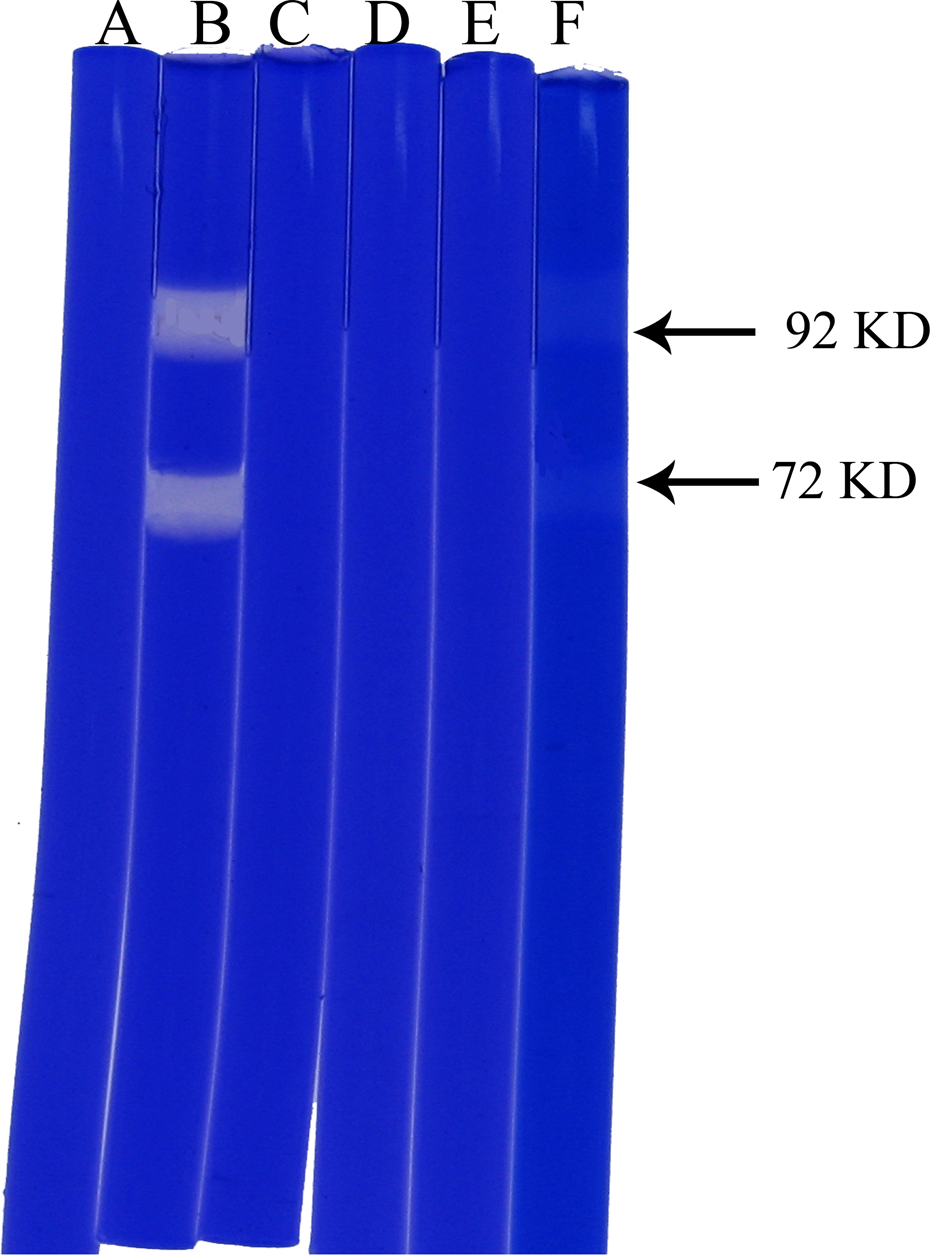
Effect of vernolide-A on MMP-2 and MMP-9 production by B16F-10 melanoma cells. (A) Conditioned medium from untreated B16F-10 melanoma cells without trypsin activation. (B) Conditioned medium from untreated B16F-10 melanoma cells after trypsin activation. (C) Conditioned medium from untreated B16F-10 melanoma cells after trypsin activation + EDTA. (D) Conditioned medium from pretreated B16F-10 melanoma cells (0.1 µg/ml vernolide-A) after trypsin activation. (E) Conditioned medium from pretreated B16F-10 melanoma cells (0.05 µg/ml vernolide-A) after trypsin activation. (F) Conditioned medium from pretreated B16F-10 melanoma cells (0.01 µg/ml vernolide-A) after trypsin activation.
Discussion
Invasion and metastasis are the most insidious and life-threatening aspects of cancer. Metastasis of cancer cells is a threat to the available cancer therapy and is a major reason for the treatment failure. Medicines available presently are cytoreductive drugs which are non-specific. Use of natural products against metastasis is less toxic and more effective. Several antimetastatic compounds have been found as natural products of plant origin. 31 Few number of novel antimetastatic agents have been published from our lab.32–40
In the present study, we analyzed the antimetastatic effect of vernolide-A. Introducing highly metastatic B16F-10 melanoma cells into C57BL/6 mice through the tail vein can produce metastatic tumor colonies on lungs and that promotes the fibrosis and collagen deposition in the lung. Intraperitonial administration of vernolide-A prophylactically and simultaneously with tumor inoculation showed maximum reduction in lung colonization and also increased the life span of metastatic tumor-bearing animals. Continuous treatment beyond 10 days might have protected at least some of the tumor-challenged animals. Vernolide-A also showed significant reduction in tumor nodule formation even when it was administered after tumor induction.
The state of lung fibrosis was evaluated by estimating the lung collagen hydroxyproline content, because during lung fibrosis collagen is deposited massively in the alveolus of lungs; 15–30% of collagen is hydroxyproline, 41 which results in the reduction of pulmonary function. Treatment with vernolide-A was very effective in reducing lung fibrosis, which was confirmed by the lowered levels of lung collagen hydroxyproline content.
Hexosamine is a significant sugar derivative present in the tumor cells. It has an important role in the synthesis of sialic acid, which is a component of glycolipids present on the surface of tumor cells. 41 Sialic acid, a family of acetylated derivative of neuraminic acid, occurs as terminal component of carbohydrate chains of glycolipids and glycoproteins. Increased levels of β1-6 branched N-linked oligosaccharides have been observed in many metastatic tumor cells, 42 and progression from a tumorigenic to metastatic phenotype in both rodent and human cancers has been associated with corresponding upregulation of sialic acid content. 43 Uronic acid and hexosamines are the basic structures of sialic acids. 41 The index of tumor cell proliferation was also evaluated by estimating hexosamine and uronic acid contents. The lungs of the metastatic tumor-bearing animals showed higher levels of uronic acid and hexosamine. Administration of vernolide-A significantly lowered the elevated contents of uronic acid and hexosamine. Total sialic acid levels in serum have been recognized as a valuable non-specific monitor of tumor burden in melanomas. 44 The elevated serum sialic acid levels in the control metastatic tumor-bearing animals was significantly reduced in the animals treated with vernolide-A.
GGT, a marker of cellular proliferation, was increased in the serum of untreated animals compared with its level in normal animals. Glutathione (GSH) is synthesized intracellularly and is present in the tumor cell. GGT catalyzes the GSH breakdown and provides energy to the tumor cells by γ-glutamyl cycle. 41 The increased serum GGT level was reduced by the treatment with vernolide-A. All of these results correlated with the histopathological analysis and ILS of treated animals.
The metastatic process is comprised of multiple events involving cell motility, cell invasion, surface-adhesion properties and degradation of ECM. Thus, degradation of the ECM and components of the basement membrane by a concerted action of proteinases, such as matrix metalloproteinases (MMPs), play a critical role in tumor invasion and metastasis45,46; MMP-2 (gelatinase A) and MMP-9 (gelatinase B) are Type IV collagenases that degrade basement membrane collagen. 47 These two MMPs are also unique in their ability to degrade denatured interstitial collagen or gelatin. Both gelatinases are expressed in many different types of cancer cells; however, they are predominately produced in stromal cells located adjacent to the tumors. 48 In human malignancies, increased MMP-2 and MMP-9 expression and activity correlate with poor prognosis and decreased survival.49,50 Nowadays, MMPs have been considered a promising target for anti-cancer drugs, and a number of synthetic MMP inhibitors have been developed and tested. 51 Here in our study, incubation of B16F-10 melanoma cells with different concentrations of vernolide-A downregulated the MMP-2 and MMP-9 protein expression in a dose-dependent manner as shown in gelatin zymography. Vernolide-A also downregulated the expression of MMP-2 and MMP-9 genes in both prophylactic and simultaneous drug-treated animals, supporting the above observations. It was previously shown that VEGF gene expression level is positively correlated with those of MMP-2 and MMP-9. 52 VEGF expression was downregulated when vernolide-A was treated after tumor development compared to the melanoma-bearing untreated control. In prophylactically and simultaneously treated groups, there was a clear suppression of VEGF, which positively correlate the MMP-2 and MMP-9 expression.
Tumor cell migration and invasion to the ECM is an important step in the process of tumor metastasis and involves the attachment of tumor cells to the ECM.53,54 In this study, we showed that vernolide-A inhibited the metastasis of the highly metastatic melanoma cell line, B16F-10 cells, by inhibiting the expression of MMP-2, MMP-9 and VEGF thereby decreasing the tumor cell migration and invasion in a dose-dependent manner. The ERK signaling pathway, also known as mitogen-activated protein kinase (MAPK) pathway, is a major determinant in the control of cell growth, cell differentiation and cell survival. This pathway, which operates downstream of Ras, is often upregulated in tumors and represents an attractive target for anticancer therapy. 55 Inhibition of the ERK pathway is expected to result in antimetastatic and antiangiogenic effects. 55 Our results indicate that vernolide-A administration inhibited the expression of ERK-1 and ERK-2 in all the three modalities. From the above observations, it is clear that vernolide-A regulated the expression of MMP-2, MMP-9, VEGF, ERK-1 and ERK-2 genes and thereby inhibited the tumor cell proliferation, invasion and metastasis.
The results of the above experiments strongly suggest the antimetastatic activity of vernolide-A. Further detailed studies have to be conducted to elucidate the exact mode of action of this compound in the inhibition of metastasis.
Footnotes
Acknowledgements
The authors would like to thank Dr Ramadasan Kuttan, Director, Amala Cancer Research Centre, for his consistent support during the study.
This research received no specific grant from any funding agency in the public commercial or not-for-profit sectors.
