Abstract
Objective: The effects of electromagnetic radiation (EMR) produced by a third-generation (3G) mobile phone (MP) on rat brain tissues were investigated in terms of magnetic resonance spectroscopy (MRS), biochemistry, and histopathological evaluations. Methods: The rats were randomly assigned to two groups: Group 1 is composed of 3G-EMR-exposed rats (n = 9) and Group 2 is the control group (n = 9). The first group was subjected to EMR for 20 days. The control group was not exposed to EMR. Choline (Cho), creatinin (Cr), and N-acetylaspartate (NAA) levels were evaluated by MRS. Catalase (CAT) and glutathione peroxidase (GSH-Px) enzyme activities were measured by spectrophotometric method. Histopathological analyses were carried out to evaluate apoptosis in the brain tissues of both groups. Results: In MRS, NAA/Cr, Cho/Cr, and NAA/Cho ratios were not significantly different between Groups 1 and 2. Neither the oxidative stress parameters, CAT and GSH-Px, nor the number of apoptotic cells were significantly different between Groups 1 and 2. Conclusions: Usage of short-term 3G MP does not seem to have a harmful effect on rat brain tissue.
Keywords
Introduction
The mobile phone (MP), one of the fastest growing technological developments, has become popular and necessary in modern life. The third generation (3G) is the latest application of MP technologies. The 3G mobile telecommunication system is developed for a variety of services such as voice and high-speed data transmission, Internet, and multimedia applications which require high speed and bandwidth.1–3 The 3G-launched applications can emit continuous electromagnetic radiation (EMR) to the environment. Other systems are generally used in close contact to ear or head but the 3G-specific MPs are usually used distinct from speakers or listeners’ ear. This can make 3G-specific MP safer. On the other hand, these phones work in higher frequency than second-generation (2G) cell phones. The higher frequency means that they emit much more EMR.2,3
Side effects of MP usage increased the concerns on health and quality of life. Potential consequences after EMR exposure have been reported in many studies and brain is one of the most commonly discussed tissues.4,5 Recently, neuronal damage in the brain, DNA mutations, deterioration of blood–brain barrier permeability, and tumor development after EMR exposure have been shown by several studies.6–8 But controversial results have been reported in some other studies.9–13
Magnetic resonance spectroscopy (MRS) is a non-invasive technique that can measure several metabolites such as N-acetylaspartate (NAA), choline (Cho), and creatine (Cr), which reflects the neuronal and glial condition, membrane metabolism, and energy status of brain. Its advantage is to be able to detect changes in the chemical status of tissues before macroscopic morphological changes occurs, compared to magnetic resonance imaging (MRI) and other imaging techniques. 14 We could find only one study using MRS for detecting the harmful effect of EMR exposure due to MP on human brain. In this study, there were no changes in brain metabolites such as Cho, Cr, and NAA, according to MRS findings. 15
The EMR can create oxidative stress by triggering free radicals. 16 The response of organism to oxidative stress can be evaluated by measuring antioxidant system and apoptosis. 16 The antioxidant enzymes catalase (CAT) and glutathione peroxidase (GSH-Px) activity levels are used as antioxidant parameters. Caspase 3 activity is used in the assessment of apoptosis in brain tissue.
In the literature, there is no study detecting the effects of EMR due to 3G-specific MP on brain tissue. Therefore, the current study was designed to observe acute changes in the brain caused by EMR from 3G-specific MP with MRS findings, antioxidant parameters, and histopathological evaluation.
Materials and methods
Animals and groups
A total of 18 male Wistar rats weighing 200–300 g were placed in a quiet controlled room with a suitable temperature (21 ± 2°C), humidity (60 ± 5%), and light–dark cycle (12–12 h). All of the rats were housed in polypropylene cages on wood-chip bedding and fed with pelleted diet and drinking water ad libitum. They were maintained and used in accordance with the ‘Animal Welfare Act and the Guide for the Care and Use of Laboratory’. The procedure was approved by the Animal Ethical Committee of Inonu University School of Medicine, Malatya, Turkey.
The rats were randomly divided into two groups: Group 1 is 3G-EMR-exposed rats (n = 9) and Group 2 is the control group (n = 9).
Exposure experiments of third-generation electromagnetic radiation
Before the experiment, two fluent medical students (male and female) separately read the same chapter from a textbook 17 and their speeches were recorded into digital recorders (Premier, PDR-6015) for 20 min in a silent room. These recordings (male and female voices) were alternately used as the speech of speaker group that linked to the microphone on 3G MP, while MP in the video-streaming mode transmitting rat’s profile to another 3G-EMR-exposed group, as listener, demonstrating an interactive conversation for 20 min. Afterward, the roles of the groups (listener and speaker) were reversed and the same procedure was conducted for another 20 min. This procedure was repeated every day from 2nd to 21st day (between 17:00 and 19:00 daily) of the experiment. The employed 3G MP was a Nokia N-70 MP (900, 1800, and 1900 MHz with General Packet Radio Service (GPRS) and Enhanced General Packet Radio Service (EGPRS) EDGE (Enhanced Data rates for GSM [Global System for Mobile Communications] Evolution) data rate capabilities; Nokia Corporation, Helsinki, Finland), and the commercial mobile communication operating system was operating approximately at 2 MB/sec data rate in 1.9–2.2 GHz frequency band. During the experimentation, the 3G-EMR exposure groups (Group 1) were kept in specifically designed polypropylene cages (length: 40 cm, width: 30 cm, height: 15 cm, and without wood-chip bedding) and the MPs for both speaker and listener were placed in contact with the transparent underside of cages. The rats were placed in close-contact above the cell phone and the cage ventilated to decrease the stress of the rats. The speaker’s and listener’s cages were placed 10 m away from each other. The MPs were manually turned on for activating (40 min) incessant conservation and turned off (standby mode) for the rest of the day (23 hours and 20 min), while their batteries were charged continuously. The digital recorders were kept away from the MPs to eliminate the possible effects of the recorder on the experiment. In the nonexposure group (Group 2), the MP was kept in standby mode and the batteries of the MPs were charged continuously. The experiment was performed on the 3G-EMR-exposed and nonexposed groups at the same time in separate and similarly conditioned rooms.
In order to reduce the possible environmental effects on the rat’s emotional state, the ambient temperature was controlled to provide comfort to all rats during the experimental sessions. Also, to reduce the effects of other electromagnetic sources, all other MPs and electronic instruments including computers were turned off and removed from the exposure zone of the experimental room. The MP was set into silent mode (not vibrate or illuminate) during the experiment. For achieving the same experimental protocol conditions in all groups, all of the equipment and tools used in the experiment did not include metal or iron which could affect electromagnetic conduction, and the fluorescent lights were turned off during the experiment.

(a) Magnetic resonance spectroscopy (MRS) of third-generation (3G)-specific mobile phone (MP)—exposed rat. The imaging volume included almost the entire brain in all cases. The NAA, Cho, and Cr signals were detected in all rats (TR = 1500 ms, TE = 136 ms). (b) Magnetic resonance spectroscopy (MRS) of control subject. The imaging volume included almost the entire brain in all cases. The NAA, Cho, and Cr signals were detected in all rats (TR = 1500 ms, TE = 136 ms).

(a) Normal cortical neurons (hematoxylin and eosin [H&E] ×200). (b) Normal histology, negative caspase 3 activity in neurons (×200).
Due to the shortage of microwave measuring instrument, unfortunately, it was not possible to measure the intensity of microwave emitted by the MP. In the literature, the available reports concerning the classical MP exposure duration schemes are very different such as 15 min–1.5 h for 4 days–4 weeks.18–20 In the current study, the EMR exposure time was chosen as 40 min a day (20 min speaker and 20 min listener).
Magnetic resonance spectroscopy
The MRS was performed with ketamine anesthesia. The MRI was conducted on a 1.5-T system (32 mT/m) and a quadrature birdcage coil (Philips, Gyroscan Intera Master, Best, Netherland). It consisted of conventional imaging sequences and MRS. The three-dimensional (3D) T2-weighted turbo spin echo images were obtained in the axial, coronal, and sagittal planes; slice thickness was 2 mm. The MRS was conducted with all rats of Group 1 (3G exposure) and Group 2 (control group) using a point-resolved spectroscopy sequence. The parameters were TR/TE: 2000/136 ms, 1024 samples, and 10 × 10 × 10 mm3 voxels that covered the entire brain. Shimming was performed prior to MRS for optimizing the field homogenity. Water suppression was optimized using automated routines provided by the manufacturer. The water signal was suppressed with a chemical-shift-selective saturation pulse. A spectral sweep width of 1000 Hz was used with a data size of 1024 points. The spectra were processed automatically using baseline correction and curve-fitting procedures in order to determine the resonance areas of NAA, Cr, and Cho. Resonance areas were assigned as follows: NAA, 2.0 p.p.m.; Cr, 3.02 p.p.m.; and Cho, 3.2 p.p.m. Signal intensities of NAA, Cho, and Cr were defined in the form of arbitrary units. Analysis of the spectra was performed using the built-in software package of the MR system (Figures 1a and b). Using the technique described by Khiat et al., 15 the Cr peak served as reference for the spectrum. Mean and standard deviation (SD) values were calculated for the metabolite ratios (NAA/Cr, Cho/Cr, and NAA/Cho) for all the subjects in each brain voxel.
Histopathologic examination of brain
At the end of study, the animals were anesthetized with ketamine and perfused with heparinized iso-osmotic phosphate buffer saline (PBS; 0.1 M, pH 7.4). After perfusion, rat brains were removed and the tissue samples were taken for histopathologic, immunohistochemical, and biochemical analyses.
Histopathologic and immunohistochemical analyses were performed in all cases on formalin-fixed and paraffin-embedded brain tissues. Sections of 5 μm thickness were cut, deparaffinized, hydrated, and stained with hematoxylin and eosin (H&E). Then, slices were dyed with caspase 3 and the apoptotic cells (hypoxic neurons) were assessed. Sections on poly-L-lysine-coated slides were used after drying in an oven for 1 h at 60°C. The sections were dewaxed in xylene, rehydrated in ethanol, and incubated for 8 min in 3% hydrogen peroxide to block endogenous peroxidase. After washing in PBS, the sections were incubated for 5 min in ultraviolet block. Each slide was then incubated for 30 min at room temperature with the primary polyclonal rabbit anti-mouse antibody to caspase 3 (Santa Cruz Biotechnology, Santa Cruz, USA) at appropriate dilutions (1:100). After washing in PBS, the slides were incubated with large-volume streptavidin peroxidase for 13 min. Finally, the preparations were developed in AEC chromogen, counterstained with hematoxylin, and mounted with Aqueous-Mount.
Biopsies were evaluated at ×400 magnification by a minimum of 10 fields for each brain slide and a score from 0 to 3 was given for hypoxic neuron profiles involving an intersection: grade 0, normal histology (Figures 2a and 2b); grade 1, mild injury consisting of red neurons (Figure 3); grade 2, moderate, as for score 1, but greater than one-third and less than two-thirds of hypoxic neurons (Figure 4); grade 3, severe hypoxic neurons. 21
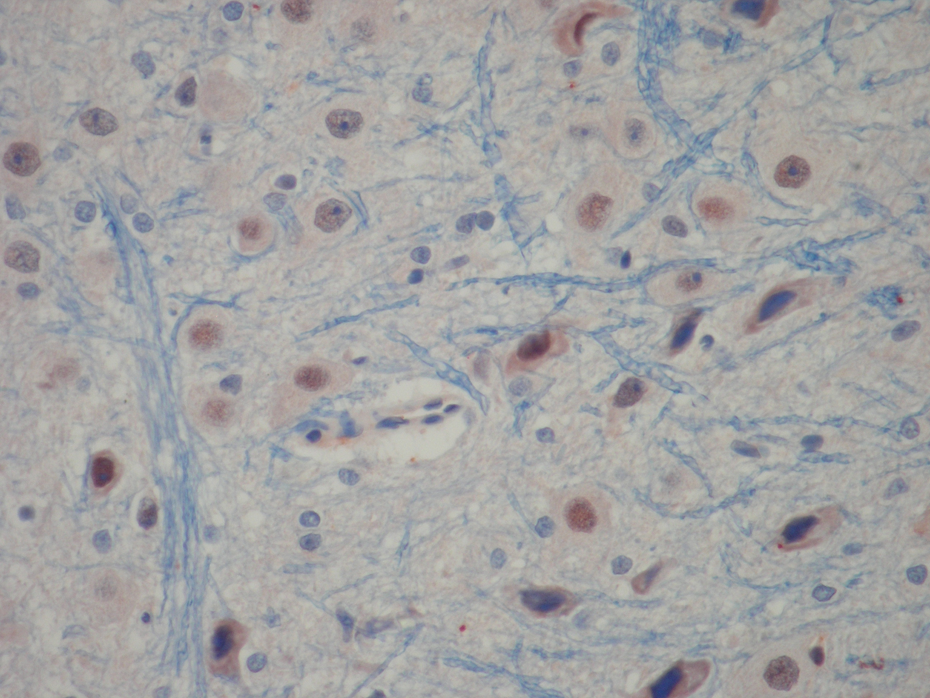
Grade 1, mild injury consisting of red neurons, positive caspase 3 activity in neurons (×200).

Grade 2, moderate injury, positive caspase 3 activity in neurons (×200).
Biochemical analysis
The tissue samples taken after perfusion with heparinized iso-osmotic PBS (0.1 M, pH 7.4) were frozen at –70°C until assayed.
The samples were homogenized in cold phosphate buffer (20 mmol, pH 7.4, containing a protease inhibitor, phenylmethylsulfonyl flouride, 1 mM) with a homogenizer (IKA ultra turrax T 25 basic) at 16,000 rpm for 3 min at +4°C. The homogenates were centrifuged at 10,000g for 20 min at +4°C and the obtained supernatants were used to measure the levels of GSH-Px and CAT.
Catalase activity assay
The CAT activity was determined according to the method of Aebi 22 by monitoring the initial rate of disappearance of hydrogen peroxide (initial concentration 10 mM) at 240 nm in a spectrophotometer. Results were reported as constant rate per gram protein (K/g protein).
Glutathione peroxidase activity assay
The GSH-Px activity was measured according to the method of Paglia and Valentine 23 by monitoring the oxidation of reduced nicotinamide adenine dinucleotide phosphate (NADPH) at 340 nm. Results were reported as units per gram protein (U/g protein).
Protein concentrations in samples were measured by the method of Lowry et al. 24
Statistical analyses
Data were expressed as mean ± SD. Normality was confirmed using Shapiro-Wilk test. Independent sample t test was used for the comparison of two groups in MRS, biochemical, and histopathological evaluations. Categorical data were analysed by the Pearson’s chi-square test. A p value of <0.05 was considered as statistically significant. Data were analysed using SPSS software program, version 15.0, for Windows (SPSS Inc., Chicago, Illinois).
Results
At the beginning of the experiment, each simple-randomized group contained 9 rats. Unfortunately, one of the rats from Group 2 (control) died and the rat was excluded from the experiment. Therefore, the current study was completed with 17 rats.
In MRS examination, there were no significant differences in Cho/Cre (p = 0.58), NAA/Cre (p = 0.39), and NAA/Cho (p = 0.56) ratios between Groups 1 and 2. Table 1 gives the descriptive statistics for the Groups 1 and 2.
Magnetic resonance spectroscopy metabolite ratios for third-generation (3G)-specific mobile phone exposed and control rats

NAA: N-acetylaspartate, Cho: coline, Cre: creatinine.
a Independent samples t test.
Biochemical parameters and histopathological findings of Groups 1 and 2 are shown in Tables 2 and 3. According to our biochemical and histopathological results, there were no significant differences in CAT and GSH-Px levels and apoptotic cell numbers (p = 0.60) between Groups 1 and 2.
Biochemical variables of brain tissue for catalase (CAT) and GSH-Px

CAT: catalase, GSH-Px: glutation peroxidase.
a Independent samples t test.
Apoptosis grade in cortical neurons, purkinje cells, and brain stem neurons

Discussion
To our knowledge, this study is the first animal research conducting radiological (MRS), biochemical (CAT and GSH-Px), and histopathological (apoptotic cell numbers) evaluations due to EMR exposure with 3G MP. Many studies have detected various biological effects of EMR exposure from MPs in animals and humans. According to our results, in all three methods, there is no significant finding in rat brain due to EMR exposure with 3G MP use.
The MRS is an imaging technique with increasing use and application in different diseases of the central nervous system. This technique supplies information on neuronal/axonal viability, cellular energetics, and cellular membrane status.25–27 The NAA, Cho, and Cr are major resonances detected on MRS in normal brains. The NAA is an important predictor of neuronal dysfunction because decreased neuronal density or viability leads to reductions in NAA. The Cr has an important role in the cellular energy metabolism. It is more concentrated in glia than in neurons. The Cr levels tend to remain relatively unchanged except for trauma, stroke, tumor, and Cr-deficiency syndromes. Therefore, Cr is often used as a putative internal standard against which the other metabolites can be compared. 27
Salford et al. 28 found evidence for neuronal damage in the cortex, hippocampus, and basal ganglia in the brains of rats exposed to EMR due to MPs. However, another study observed no effect on neuronal structure. 9 In our study, in rat brains exposed to EMR with 3G MPs, NAA/Cr ratios were normal compared to control subjects in MRS, which could indicate an absence of neuroaxonal loss.
Major components of the Cho resonance are phosphocholine and glycerophosphocholine that form a pool involved in membrane synthesis and degradation. Intracranial neoplasms have extensively been studied in vitro and in vivo. Recently, many investigators reported that spectral changes in brain tumors are found at an increasing level of Cho due to elevated cell turnover and decreased NAA level related to tumoral tissue replacing or infiltrating neuronal tissue.29–31
Potential carcinogenic effects of EMR are reported in several studies in the literature.9,32 In 1997, Salford et al. 12 reported no carcinogenic effect of EMR. Surprisingly, Adey et al. 13 showed a decreased risk in their study. In our study using MRS, Cho/Cr, NAA/Cr, and NAA/Cho ratios were compared with Groups 1 and 2 and the results do not support tumor development. These results are similar with that of Salford’s study. 12 Hardell et al. 8 and Repacholi et al. 32 have been studying the EMR exposure and its intensity for a long period.
The MRS can show secondary effects of radiation in brain parenchyma. It has an important clinical role in the differentiation of necrosis and tumor. Chan et al. 33 has shown high Cho and lactate levels with 28 Gy single-dose irradiation for 12 months in the damaged region of the brain tissue of rats. There are many studies indicating decreased metabolite levels due to radiation necrosis. 34 However, our study showed that EMR exposure did not cause an alteration in metabolite levels in the brain tissue of rats.
Regarding the oxidant injury induced by EMR, the cascade of events follows the generation of the reactive oxygen species (ROS) through the hydrolysis of the cellular water content. 35 Although the detection of the ROS such as O2 –, OH, and H2O2 might be successfully accomplished using in vitro systems, the detection of these species in real time using in vivo systems is rather difficult. 36 For this reason, we have adopted an indirect approach in which we have used detection of antioxidant enzyme activities and caspase 3 activity as markers of oxidative stress and apoptosis.
In general, MP-induced EMR might decrease the antioxidant enzyme activities or increase the production of ROS both of which disturb the antioxidant protection system. However, the antioxidant system of the organism creates various responses against the oxidative stress produced by ROS. 37 It is known that free radicals at a low level of intensity might enhance the antioxidant defense system via stimulation of gene expression of antioxidant enzymes, while an oxidant stress at a high level of intensity might exceed the capacity of the antioxidant defense system because of oxidant damage especially in the active sites of the 3D structure of antioxidant enzymes. 38 Our results are parallel to this hypothesis. Even though this is not statistically significant, the occurrence of free radicals at a low level of intensity due to low EMR level may have a stimulating effect that caused a slight increase in Group 1 compared to the control group. This result may depend on exposure to EMR at a short period and low intensity. Dasdag et al. 16 reported that there was an increase in CAT activity levels in brain tissue after EMR exposure. Additionally, Irmak et al., 39 in a similar study, showed that EMR exposure did not show any difference in CAT and GSH-Px levels in the brain tissue of rats. Besides, there is no study reported with the use of 3G MP, our results are parallel to the findings of studies done with other generations of MP. The differences in the obtained results may be related to the implementation time and dose differences.
Apoptosis is a programmed cell death. Increased oxidative stress leads to apoptotic process. According to our antioxidant parameter levels, we can say that EMR intensity and exposure time used in our study did not cause a dramatic oxidative stress in the cell level. And parallel to this, there was no significant difference in apoptotic cell numbers between groups.
The limitations of our study is the short time period, slight EMF exposure, and small number of subjects. There was no close proximity of 3G MP to the head during its use. This may lead to low exposure to EMR caused by 3G MP. This may make 3G MP more secure than other generation telephones.
As a conclusion, we could not find any detrimental effect of radiation from 3G phones in our experimental set up; however, in the future, studies with more subjects using 3G MP for a long time period and with high intensity could show hazardous effects on brain tissue. At this point, studies with more molecules and different doses are needed.
Footnotes
All authors declare that they have no conflicts of interest.
The authors received no financial support for the research and/or authorship of this article.
