Abstract
Studies on the neurobehavioral toxicity of monocrotophos, an organophosphate, have been carried out on rats following their exposure from postnatal day (PD) 22 to PD 49 to investigate whether neurobehavioral changes are transient or persistent. Exposure of rats to monocrotophos (0.50 or 1.0 mg/kg body weight, p.o.) decreased body weight (10% and 30%) and impaired grip strength (28% and 32%) and learning ability (65% and 68%) at both the doses, respectively in comparison to controls. A trend of recovery was observed in body weight and learning, while decrease in grip strength persisted in rats 15 days after withdrawal. Activity of acetylcholinesterase was decreased in frontal cortex (36% and 67%), hippocampus (21% and 49%) and cerebellum (29% and 51%) in monocrotophos-treated rats at both the doses. The decrease in the activity of acetylcholinesterase persisted in frontal cortex and hippocampus; however, a trend of recovery was observed in cerebellum 15 days after withdrawal. Binding of 3 H-quinuclidinyl benzilate ( 3 H-QNB) to frontocortical (19% and 35%), hippocampal (32% and 39%) and cerebellar (19% and 28%) membranes was decreased in monocrotophos-treated rats compared to controls. The decrease in the binding of 3 H-QNB persisted in frontocortical, hippocampal and cerebellar membranes 15 days after withdrawal. The results suggest that repeated exposure to monocrotophos in rats may cause behavioral and neurochemical modifications which may persist even after withdrawal. The findings are of concern in view of the high consumption of monocrotophos in many countries.
Introduction
Organophosphates are widely used insecticides in agriculture, home, garden and veterinary practices to control a large number of pests.1,2 Although these pesticides are classified in to different subclasses, they share a common structure.3,4 Due to the extensive use of organophosphate pesticides all over the world, these are widely distributed in the environment and exert adverse effects on the biological system. Cases of organophosphate poisoning have been reported both in India and abroad.5–9 Chronic exposure to organophosphate pesticides has been reported to cause extrapyramidal symptoms and psychiatric disturbances.10–15 As organophosphates have been used as nerve gases in the war zones either alone or in combination, their use has been a matter of great concern.16,17
Inhibition of acetylcholinesterase and neurotoxic esterase are the primary consequences in the neurotoxicity of organophosphate pesticides affecting both peripheral and central nervous system.3,18 Besides, non-cholinergic mechanisms have also been found to be involved in the neurobehavioral toxicity of organophosphate pesticides.3,19–24 Potential role of oxidative stress in the neurotoxicity of organophosphate is well accepted. 25 Alterations in dopaminergic and serotonergic activity have also been reported in rats exposed to chlorpyrifos prenatally or neonatally.20,26 Several experimental studies have been carried out on different organophosphates to understand the potential of their developmental neurotoxicity.27–30 Low-level exposure to organophosphate pesticides has been found to be detrimental and lead to developmental abnormalities.27,31–34 Interestingly, the impact of these pesticides in producing neurotoxicity differs. Also, their effect on events in brain development may relatively differ. Fetuses and children have been found to be more sensitive than adults depending on the pesticide to which they are exposed. Physiological differences in developing organisms also contribute in enhancing the vulnerability to toxic insults of pesticides as compared to adults.33–35 Brain controls various behavioral, pharmacological and physiological functions of the body and is a soft target of these pesticides. Exposure to pesticides could occur for a short period or over the entire period of life and the outcome is generally dependent upon the time and duration of exposure. Cognitive and motor dysfunctions are quite common following exposure to pesticides including organophosphates.6,7,9 At times, the changes persist for a longer period of time and therefore a number of clinical and experimental studies have been carried out on different classes of organophosphate pesticides to understand their health effects.3,9,20,21
Monocrotophos (dimethyl (E)-1-methyl-2-(methylcarbamoyl) vinyl phosphate), an organophosphate insecticide, is in continuous use in several developing nations, although its use has been banned in the Western world and restricted in many countries. Production and consumption of monocrotophos in India is tremendously high compared to other organophosphates and classes of pesticides. 36 It is extensively used in crop protection as a non-specific systemic insecticide and acaricide. 37 Monocrotophos produces systemic and toxic action in insects and is toxic to mammals.37–39 In a study on mice, acute exposure to monocrotophos caused hyperthermia and reduced locomotor activity and affected the ability of trained mice to ride on the rotating rod. 38 Because of the extensive use of monocrotophos in agriculture and animal husbandry, humans are also exposed to it. Levels of monocrotophos were found to be four times higher in blood. 40 In a study on farmers in Egypt using organophosphates including monocrotophos, 50% of the workers exhibited neurological symptoms including loss of reflexes.41–43 Degradation and persistence of pesticides including monocrotophos in the soil used in cotton crops were studied. 44 It was found that monocrotophos persists for a much longer time in soil because its degradation decreases with depth and at low temperatures. 44
Although the use of monocrotophos is substantially high, comprehensive data on the neurobehavioral toxicity of monocrotophos is not available. Also the mechanism of its toxicity and its potential on the developing brain is not understood. The present study has therefore been undertaken to investigate the mechanism of neurobehavioral toxicity of monocrotophos on brain cholinergic system of rats following their exposure from postnatal day (PD) 22 to PD 49. To further understand whether such changes are transient or persistent, the effect on these parameters was studied after 15 days of withdrawal or cessation of treatment.
Materials and methods
Animals and treatment
Female rats (PD 21) of Wistar strain were obtained from the animal breeding colony of Indian Institute of Toxicology Research (IITR), Lucknow, India. The rats were housed in polypropylene cages under standard animal house conditions in a 12-h light–dark cycle and fed pellet diet and water ad libitum. Rats were divided in to three groups. On PD 22, rats in two groups were administered monocrotophos, purchased from the local market (commercial formulation—Phoskill 36%, United Phosphorus Limited, Ankleshwar, Gujarat, India), suspended in groundnut oil, either at 0.50 or 1.0 mg/kg body weight dose orally until PD 49. The third group of rats was fed with groundnut oil in an identical manner until PD 49, which served as control. The effect on behavioral parameters—grip strength and learning—was studied on PD 50 in young rats. Separate set of rats in each group was sacrificed on PD 50 for the assay of neurotransmitter receptors and acetylcholinesterase activity. The brain was removed and dissected into regions—frontal cortex, hippocampus and cerebellum following the standard protocol. 45 The brain regions were kept frozen at −20°C till assay. The study was approved by the Institutional Animal Ethics Committee (IAEC) of Indian Institute of Toxicology Research, Lucknow, India, and all experiments were carried out in accordance with the guidelines laid down by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests (Government of India), New Delhi, India.
To study whether neurobehavioral changes are transient or persistent, a set of rats in all the treatment groups were left as such till PD 64. The rats were monitored daily and the effect on selected behavioral and neurochemical parameters was studied on PD 65.
Behavioral studies
Grip strength
Forelimb grip strength was measured in the control and monocrotophos-treated rats using a computerized grip strength metre (TSE Germany) following the standard procedure. 1
Learning ability
The learning ability of rats was measured by assessing conditioned avoidance behavior using a shuttle box apparatus (Techno, India) following the standard protocol 46 with minor modification. Briefly, rats from the control and monocrotophos-treated groups were placed individually in one chamber of the shuttle box and habituated for 5 minutes. Rats received a conditioned stimulus in the form of a buzzer for 10 seconds followed by a buzzer and foot shock of 0.5 mA for 10 seconds. The avoidance was considered positive if the rats jumped to a shock-free chamber to avoid the foot shock. All the rats in the treatment and control groups were subjected to 20 trials per day for 3 days. The intertrial interval was 1 minute. Of the 20 trials per day, the total number of avoidances and the number of trials for the first avoidance are recorded for each rat. The data of the final day are presented in the results. The percentage of conditioned avoidance response has been recorded as a measure of learning ability in treated groups and compared with controls.
Biochemical studies
Muscarinic–cholinergic receptor binding assay
Assay of muscarinic–cholinergic receptors in frontal cortex, hippocampus and cerebellum was carried out in synaptic membrane by the radioligand receptor-binding technique following the standard procedure described in detail by Khanna et al. 47 Briefly, 3 H-QNB (42 Ci/mmole, Perkin Elmer, USA, 1 × 10−9 M) was used as a radioligand and atropine sulphate (1 × 10−6 M) as a competitor to determine the extent of non-specific binding for the assay of muscarinic–cholinergic receptors. Specific binding has been expressed as picomoles ligand bound/g protein. Saturation experiments were carried out using varying concentrations of radioligands (normally 1/10 to 10 times of the affinity of radioligand) to determine whether the change in the binding is due to alteration in the affinity or number of receptor-binding sites in each case.
Assay of acetylcholinesterase activity
The activity of acetylcholinesterase has been estimated in frontal cortex, hippocampus and cerebellum using acetyl thiocholine iodide as substrate following the standard procedure. 48
Protein estimation
Protein concentration in tissue homogenates and membranes was measured using bovine serum albumin as the reference standard. 49
Statistical analysis
Mean significant difference in the treatment groups has been determined using one-way analysis of variance followed by Newman–Keuls test. Values up to p < 0.05 have been considered statistically significant.
Results
Effect on body and brain weight of rats following post-lactational exposure to monocrotophos
Post-lactational exposure (PD 22–PD 49) of rats to monocrotophos at both the doses (0.50 or 1.0 mg/kg body weight) caused a decrease in body weight to 10% and 30%, respectively, as compared to controls (Figure 1A). Body weight of rats exposed to monocrotophos at a higher dose (1.0 mg/kg body weight for 28 days) was found to decrease to 21% as compared to those exposed to monocrotophos at a lower dose (0.50 mg/kg body weight for 28 days). However, a trend of recovery in the body weight of monocrotophos-treated rats was observed 15 days after withdrawal as compared to respective controls. No significant change in brain weight was observed in rats treated with monocrotophos for 28 days or 15 days after withdrawal at any of the doses (Figure 1B).

(A) Effect on body weight of rats following post-lactational exposure to monocrotophos. Values are mean ± SE of 15 animals in each group. (a) Compared to control group and (b) compared to monocrotophos (MCP)-I-treated group. *Significantly differs from control (p < 0.05). (B) Effect on brain weight of rats following post-lactational exposure to monocrotophos. Values are mean ± SE of 15 animals in each group.
Effect on grip strength in rats following post-lactational exposure to monocrotophos
The effect on forelimb grip strength of rats treated with monocrotophos is shown in Figure 2. A significant decrease in forelimb grip strength was observed in rats treated with monocrotophos at both the doses (0.50 or 1.0 mg/kg body weight) to 28% and 32%, respectively, in comparison to rats in the control group. No significant change in fore limb grip strength was, however, observed between rats exposed to monocrotophos at both the doses. The fore limb grip strength remained decreased to 21% and 30% even 15 days after withdrawal in monocrotophos-treated rats at both the doses as compared to respective controls (Figure 2).

Effect on grip strength of rats following post-lactational exposure to monocrotophos. Values are mean ± SE of six animals in each group. (a) Compared to control group. *Significantly differs from control (p < 0.05).
Effect on active avoidance response in rats following post-lactational exposure to monocrotophos
A significant impairment in learning, assessed by monitoring the active avoidance response, was observed in rats exposed to monocrotophos (0.50 or 1.0 mg/kg body weight) from PD 22 to PD 49 to 65% and 68%, respectively, in comparison to controls (Figure 3). No significant change in the active avoidance response was observed between rats exposed to monocrotophos at both the doses. Although a trend of recovery in learning was observed in monocrotophos-treated rats, 15 days after cessation of treatment, the learning remained impaired to 23% and 30%, respectively, as compared to controls (Figure 3).

Effect on active avoidance response of rats following post-lactational exposure to monocrotophos. Values are mean ± SE of five animals in each group. (a) Compared to control group. *Significantly differs from control (p < 0.05).
Effect on acetylcholinesterase activity in brain regions of rats following post-lactational exposure to monocrotophos
Post-lactational exposure of rats to monocrotophos (0.50 or 1.0 mg/kg body weight) caused a significant decrease in the activity of acetylcholinesterase in frontal cortex (36% and 67%), hippocampus (21% and 49%) and cerebellum (29% and 51%), respectively, as compared to controls (Figure 4). Treatment with monocrotophos at a higher dose (1.0 mg/kg body weight for 28 days) in rats caused a further decrease in acetylcholinesterase activity in frontal cortex (48%), hippocampus (35%) and cerebellum (30%) as compared to those exposed at a lower dose. The decrease in the activity of acetylcholinesterase persisted in monocrotophos-treated rats even 15 days after withdrawal in frontal cortex (30% and 56%) and hippocampus (12% and 32%; Figure 4). Although a trend of recovery was observed in cerebellum, the activity of acetylcholinesterase remained decreased (14% and 28%) 15 days after withdrawal in monocrotophos-treated rats as compared to respective controls.

Effect on acetylcholinesterase activity in brain regions of rats following post-lactational exposure to monocrotophos. Values are mean ± SE of four animal in each group. (a) Compared to control group and (b) compared to monocrotophos (MCP)-I-treated group. *Significantly differs from controls (p < 0.05).
Effect on muscarinic–cholinergic receptors in frontal cortex, hippocampus and cerebellum of rats following post-lactational exposure to monocrotophos
Exposure of rats to monocrotophos (0.50 or 1.0 mg/kg body weight) from PD 22 to PD 49 caused a decrease in the binding of 3 H-QNB to frontocortical (19% and 35%), hippocampal (32% and 39%) and cerebellar (19% and 28%) membranes, known to label muscarinic–cholinergic receptors as compared to controls (Figure 5). Rats treated with monocrotophos at a higher dose (1.0 mg/kg body weight for 28 days) caused a further decrease in the binding of muscarinic–cholinergic receptors in frontocortical (19%) and marginal changes in hippocampal (9%) and cerebellar membranes (11%) as compared to those exposed at a lower dose. The decrease in the binding was more pronounced in rats exposed to monocrotophos at a higher dose. Scatchard analysis revealed that the decrease in binding of 3 H-QNB to frontocortical, hippocampal and cerebellar membranes was due to decreased number of receptor-binding sites (Bmax) and no change in the affinity (Table 1 ). The decrease in the binding of 3 H-QNB to frontocortical membranes (10% and 35%) persisted, while it remained decreased in hippocampal (12% and 35%) and cerebellar (8% and 23%) membranes 15 days after withdrawal in monocrotophos-treated rats (Figure 5).
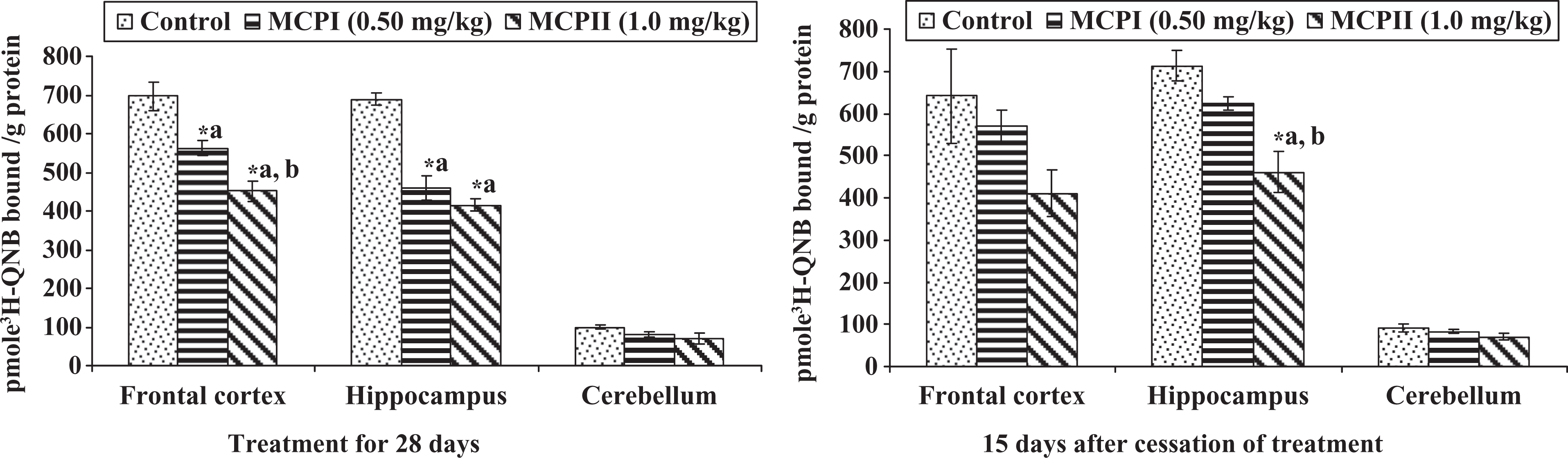
Effect on muscarinic–cholinergic receptors in different brain regions of rats following post-lactational exposure to monocrotophos. Values are mean ± SE of five animals in each group. (a) Compared to control group and (b) compared to monocrotophos (MCP)-I-treated group. *Significantly differs from control (p < 0.05).
Scatchard analysis of 3H-QNB to rat frontocortical, hippocampal and cerebellar membranes following exposure to monocrotophos

3H-QNB: 3H-quinuclidinyl benzilate, MCP: monocrotophos, Kd: dissociation constant expressed in nM, Bmax: maximum number of binding sites expressed in pmoles bound/g protein.
Values are mean ± SE of five animals in each group. (a) compared to control group.
* Significantly differs from control (p < 0.05).
Discussion
Inhibition in the acetylcholinesterase activity associated with altered levels of acetylcholine in the brain is the primary mechanism in organophosphate neurotoxicity.3,28,50,51 Exposure of rats to methyl parathion 51 and dichlorvos 28 significantly decreased the activity of acetylcholinesterase in brain regions. The decrease in the acetylcholinesterase activity was more marked in rats exposed for a longer duration 51 or even after withdrawal. 28 Raheja and Gill 52 also observed decreased acetylcholinesterase activity in cerebrum, cerebellum and brain stem of rats exposed to dichlorvos for 8 weeks, with no symptoms of excessive cholinergic activity. Developmental increase in brain cholinesterase activity has been reported in rats from PD 6 to PD 22. 30 A significant decrease in the brain cholinesterase activity was observed in rats following exposure to methyl parathion at low, medium and high doses from PD 1 to PD 21. The decrease was found to persist even after 9 and 19 days of withdrawal. In a recent study, marginal changes in the activity of brain acetylcholinesterase and monoamine oxidase have been observed in rats treated with dichlorvos or monocrotophos or in combination. 53 However, a significant decrease in the levels of brain biogenic amines and reduced glutathione (GSH):oxidized glutathione (GSSG) was observed in rats treated with dichlorvos or monocrotophos alone or in combination in these studies. 53 The rate of inhibition in the acetylcholinesterase activity following exposure to organophosphates is not consistent in the brain regions. Regional variations in the density of cholinergic innervations in the brain could be one of the possible reasons for the differences in the level, synthesis and turnover rate of acetylcholine. 54 Also, the rate of inhibition of brain acetylcholinesterase activity varies with the potential of chemical/formulation used and differences in their toxicokinetic pattern. Dose, duration and route of exposure are also important determinants regulating the neurotoxic response of different chemical/formulation. In the present study, a significant decrease in the activity of acetylcholinesterase in frontal cortex, hippocampus and cerebellum was observed following post-lactational exposure of rats to monocrotophos. The effect was more marked in rats exposed to monocrotophos at a high dose (1.0 mg/kg) and persisted in frontal cortex and hippocampus 15 days after withdrawal. Although a trend of recovery in the activity of acetylcholinesterase in cerebellum was observed in monocrotophos-treated rats 15 days after withdrawal, it still remained decreased when compared to controls.
Repeated and chronic exposure to organophosphate pesticides has been reported to cause significant decrease in the binding of brain cholinergic receptors28,51,52 which are differentially distributed in brain. 55 Differential effects on the sensitivity of cholinergic receptors in rats exposed to dichlorvos have been reported. 28 A decrease in the binding of 3 H-QNB in cortex was observed 1 day after repeated treatment with dichlorvos for 7 and 14 days. It was also found that downregulation of cholinergic receptors in cortex returned to normal 6 and 11 days after withdrawal in rats exposed to dichlorvos for 7 days but not in those exposed to dichlorvos for 14 days. Based on this finding, it was suggested that shorter treatment with dichlorvos may induce a reversible downregulation of muscarinic–cholinergic receptors; however, the changes may be prolonged or irreversible in case of prolonged exposure. In another study, chronic exposure to dichlorvos in rats for 8 weeks decreased the muscarinic–cholinergic receptors in cerebellum and brain stem and caused no change in cerebrum. 52 Repeated exposure to methyl parathion in rats during the neonatal period (PD 1–PD 21) also caused a significant decrease in the binding density of brain cholinergic–muscarinic receptors on PD 22. 30 The binding of muscarinic–cholinergic receptors was found increased in methyl parathion-treated rats 9 and 19 days after withdrawal, while it could not be correlated whether the changes are transient or permanent. 30 Volpe et al. 56 in an interesting study correlated the decreased muscarinic receptors following exposure to dichlorvos in vitro with clinical neurological abnormalities in humans exposed to organophosphate pesticides. In the present study, post-lactational exposure to monocrotophos for 28 days caused a significant decrease in the binding of 3 H-QNB in frontal cortex, hippocampus and cerebellum. The decrease in the binding of cholinergic–muscarinic receptors in frontal cortex and hippocampus was more marked in monocrotophos-treated rats and persisted 15 days after withdrawal indicating adverse effects on the brain cholinergic circuitry. Such an effect on the sensitivity of muscarinic–cholinergic receptors as observed in the present study could be due to the decreased activity of acetylcholinesterase, which persisted even 15 days after withdrawal, associated with age. Downregulation of muscarinic–cholinergic receptors is a key mechanism of adaptation to increased brain acetylcholine levels. 57 As the decrease in the muscarinic–cholinergic receptors has been found to be agonist induced, 30 inhibition in the acetylcholinesterase activity as observed in the present study may affect cholinergic receptors by increasing the acetylcholine levels at the site. Further, exposure to oxotremorine, muscarinic agonist, downregulates muscarinic–cholinergic receptors in brain regions by internalizing the receptors situated on the external cell surface.58,59,28 Another possibility that organophosphates may have direct effect on muscarinic–cholinergic receptors also exists as acetylcholinesterase inhibitors have been shown to target the muscarinic receptors directly. 60 Interestingly, downregulation of muscarinic–cholinergic receptors due to prolonged stimulation by acetylcholine or muscarinic agonists may affect microtubular transport of receptors to their intracellular degradation sites. 58
Decrease in body weight following exposure to organophosphate pesticides has been reported. 52 Studies on systemic and developmental neurotoxicity of diazinon, chlorpyrifos and parathion were carried out to compare the dose–effect relationship.61,62 The systemic toxicity of parathion was highest among the three organophosphate pesticides studied. 61 In the present study, the decrease in body weight was more marked in rats treated with monocrotophos at a higher dose and a recovery in body weight was observed 15 days after withdrawal. The adverse effect on body weight as observed could be the toxic effect of monocrotophos. It also suggests that exposure to monocrotophos may affect the growth of developing organism if the exposure continues. The dose of monocrotophos does not appear to be high as no apparent signs of salivation, lachrymation or ataxia were observed in the treated rats in the present study.
Integrity of central cholinergic system is important for learning, memory and cognition in humans and animals. 63 The decrease in cholinergic receptors particularly in hippocampus may significantly reduce the transmission of cholinergic impulse affecting the learning ability. Process of learning and memory has been found to be affected by anticholinesterases. 64 Exposure to dichlorvos significantly impaired the cognitive behavior in rats and was attributed to decreased cholinesterase activity. 65 Masoud et al. 66 observed that acute exposure to monocrotophos at a high dose (20 mg/kg body weight) impaired the learning and memory in rats 1 week after treatment and the effect persisted even 28 days after post exposure. The functional changes were due to mitochondrial dysfunction associated with enhanced oxidative stress. Earlier, enhanced oxidative stress in brain has been linked with impaired learning in mice. 67 Impaired performance in learning ability in rats treated with monocrotophos as observed in the present study may be due to decreased cholinergic receptors. It is, however, difficult to comment on the observation that decrease in the learning ability in monocrotophos-treated rats exhibited a trend of recovery 15 days after withdrawal. Exposure to neurotoxicants may significantly affect the grip strength, leading to muscle weakness. Low-level exposure to chlorpyrifos, an organophosphate, has been found to significantly impair grip strength in rats. 1 Although the effect on the grip strength was found to be partially recovered 6 days after withdrawal in the chlorpyrifos-treated rats, it was suggested that chlorpyrifos has the potential to produce neuropathic changes with long-term exposure. An impairment in grip strength in rats following monocrotophos exposure as observed in the present study is indicative of muscle weakness, which may prolong for a long period of time. The results of the present study exhibit vulnerability of developing rats to monocrotophos exposure, leading to cholinergic dysfunctions. Further, these results are of concern in view of high consumption of monocrotophos in many countries.
Footnotes
Acknowledgements
The authors thank Director, Indian Institute of Toxicology Research (IITR), Lucknow, India, for his keen interest in the present study. The study is a part of Supra-institutional project (SIP - 008) of Indian Institute of Toxicology Research (A constituent laboratory of Council of Scientific and Industrial Research, New Delhi, India.). Madhu Lata is grateful to the Council of Scientific and Industrial Research (CSIR), New Delhi, India, and Rajesh Singh Yadav to University Grants Commission (UGC), New Delhi, India, for the award of research fellowships. The technical assistance provided by Dr. Pramod Kumar and Mr. B.S. Pandey is acknowledged (IITR communication number 2681).
The authors have no competing financial interest.
