Abstract
The study is to explore the toxicity of organic extracts and water-soluble fraction of fine particles on human umbilical vein endothelial cells (HUVECs). The exposure doses were 100, 200 and 400 μg/ml, respectively, for two kinds of fractions. Moreover, atorvastatin was used for intervention study. HUVECs were stimulated by 400 μg/ml organic and water soluble extracts, respectively, immediately followed by treatment with atorvastatin in concentrations of 0.1 μmol/L, 1 μmol/L and 10 μmol/L, respectively. Cell viability, malondialdehyde (MDA), nitric oxide (NO), superoxide dismutase (SOD), reactive oxygen species (ROS) and the expression of interleukin-6 beta (IL-6), tumor necrosis factor-α (TNF-α), endothelin-1 and P-selectin were determined in cells. The results showed that MDA and ROS increased in HUVECs after exposed to organic extracts and water-soluble fraction, whereas cell viability, NO and SOD decreased. The mRNA expression of IL-6, TNF-α, endothelin-1 (ET-1) and P-selectin increased after exposed to different fractions. Meanwhile, at the same exposure dose, water-soluble fraction caused more significant increase of MDA, IL-6, TNF-α and P-selectin and decrease of cell viability and NO when compared to organic extracts. Compared to no atorvastatin group, the levels of MDA, ROS and the expression of IL-6, TNF-α, ET-1 and P-selectin decreased in HUVECs in adding atorvastatin group, but cell viability, NO and SOD increased, which indicated that atorvastatin attenuated fine particle-induced inflammatory response, oxidative stress and endothelial damage. The results hinted that the inflammatory response, oxidative stress and endothelial dysfunction might be the mechanisms of cardiovascular injury induced by different fractions of ambient fine particles.
Introduction
Particulate air pollution remains a threat to public health. It has been estimated by the WHO that particulate matter (PM) exposure accounts for 800,000 deaths per year worldwide. 1 Long-term exposure to PM is an important environmental risk factor for cardiopulmonary disease and lung cancer morbidity and mortality, especially for particles with aerodynamic diameters under 2.5 μm fine particles.2,3 Previous studies have also reported that fine particles are more strongly associated with mortality than inhalable particles.4,5 Epidemiological study has shown that an increase of 10 μg/m3 fine particles is associated with a 4.6% increase in asthma admissions in children and a 4.3% increase in admissions for chronic obstructive pulmonary disease and 1.5% for ischemic heart disease in the elderly. 6 In many cities in China, the concentrations of fine particles are far excess the American air quality standards 35 μg/m 3 (24 h averaging time) and WHO air quality guideline 25 μg/m3 (24 h averaging time). 7 Therefore, it is necessary to study the effects of fine particles on cardiovascular system and explore the potential mechanisms to protect people’s health.
Fine particle is a complex mixture which includes various chemical, physical and biological compositions. Our previous study had demonstrated that the chemical compositions of fine particle included Fe, Ni, Zn, Pb, Cu, etc. 8 Epidemiologic studies suggested that the physical and chemical compounds of PM were responsible for its toxicity in causing adverse health effects. 9 Among the fractions, metal elements10,11 and organic extracts 12 were the most important fractions which could cause adverse health effects. However, the underlying pathophysiological mechanisms linking fine particles air pollution and cardiovascular morbidity and mortality remain to be elucidated. Various mechanisms have been proposed to explain the cardiovascular health effects of particulate matter. These hypotheses include pulmonary and systemic oxidative stress and inflammation, 13 direct vasoconstriction, 14 enhanced coagulation pathways 15 and altered cardiac autonomic function. 16 In our previous study, results also showed that the inflammation, endothelial dysfunction and coagulation disorders might be the mechanisms of cardiovascular injury induced by fine particles. 17 Moreover, as the wall of blood vessels and the blood stream, the pathophysiology of endothelium is complex and involves multiple mechanisms. Results have shown that endothelial dysfunction has been proposed to be an early of pathophysiologic importance in pulmonary inflammation caused by ambient pollution. 18 Endothelial cells could be playing an important role in the response to airborne particles. Endothelial dysfunction is associated with, and may contribute to, an increased risk of cardiovascular events. 19 Meanwhile, endothelial dysfunction can promote inflammation and platelet adhesion which maybe aggravated by the cardiovascular injury. Previous study demonstrated that systemic endothelial dysfunction was induced in animals when exposed to particulate matter by instillation. 20 In this study, the human umbilical vein endothelial cells (HUVECs) are used to observe the effects of organic extracts and water-soluble fraction of fine particles on endothelial function and explore the potential mechanisms.
Statin can improve endothelial function, elevate NO biological activity, inhibit inflammation, modulate immune function and reduce oxidation in clinical studies. In this study, atorvastatin was used for intervention studies on the possible toxicological mechanisms of fine particle fractions on HUVECs.
Materials and methods
Fine particles collection
Fine particle sample was collected using a Thermo Anderson G-2.5 air sampler (Model GV 2630 Series, USA) from ambient air from October 2006 to April 2007 in a non-industry district in Shanghai, China. The filters were heated for 24 h at 200°C before sampling. After sampling, filters were cut into small pieces and immerged in 0.9% saline and probe sonicating 3 × 10 min with a sonicator (Model JL-120DT, China), then the suspension was treated by vacuum-freeze dry, the concentrated fractions were weighted and stored at −20°C.
Water-soluble and organic extracts of fine particles
Fine particles were suspended with sterilized 0.9% physiological saline. Water-suspended fine particles were diluted to 1000 μg/ml, 2000 μg/ml and 4000 μg/ml, centrifuged for 30 min at 20,000 × g, and the supernatants were collected as the water-soluble fractions. 21 Extraction of organic compounds was carried out by 30 ml acetonitril during 1/2 h under ultrasounds. Dimethyl sulfoxide (DMSO, Sigma, USA) was added and the mixture was concentrated under a nitrogen flux in order to remove acetonitril. 12 Organic extracts were collected and diluted to 1000 μg/ml, 2000 μg/ml and 4000 μg/ml with 0.9% physiological saline.
Analysis of the chemical constitutes in water-soluble and organic extracts
Levels of soluble metals for water-soluble fraction of fine particles were measured by ICP-MS instrument (VGX7, Thermo Electron Corporation, USA). The components of organic extracts of fine particles were characterized by high-performance liquid chromatography (HPLC; Agilent 1100 seriers). The column was a DIKMA (5 μm, 4.6 × 250 mm). Standards for phenanthrene, anthracene, fluoranthene, pyrene, benzo[a]anthracene, benzo[b]fluoranthene, benzo[a]pyrene and benzo[g, h, i]perylene (Sigma, USA) were used for qualitative analysis.
Preparation of the atorvastatin
Atorvastatin (Pfizer Inc., New York, USA) was dissolved in DMSO to obtain a stock solution of 10 mM. Atorvastatin was added to the cells at the final concentrations of 0.1 μmol/L, 1 μmol/L and 10 μmol/L, respectively. The final concentration of DMSO in the cell culture media was less than 0.1% and did not affect cell viability.
Cells culture and fine particles fractions treatment
Human umbilical vein endothelial cells were purchased from NanJing KeyGen Biotech. Inc. Cells were grown in RPMI 1640 medium (PAA laboratories GmbH, Austria) supplemented with 10% fetal calf serum (PAA laboratories GmbH, Austria). Cells were cultured at 37°C in a 5% CO2 atmosphere. Cell culture was done according to the manufacturer’s manual. Cells were grown to confluence in culture plastic (Costar, USA) and used for experiment in the second to fifth cell passages. Cells were grown to 80% confluency in 6-well or 24-well plates, then cells were treated with the organic, water-soluble extracts of fine particles (final concentrations were 100, 200 and 400 μg/ml) for 24 h, respectively. Moreover, cells were stimulated with the organic and water-soluble extracts of fine particles (400 μg/ml) and immediately treated with atorvastatin in concentrations of 0.1 μmol/L, 1 μmol/L and 10 μmol/L, respectively. Then the cells were incubated for 24 h for analysis.
MTT assay for cell viability
MTT assay is a quantitative colorimetric method to determine cell proliferation. It utilizes the yellow tetrazolium salt 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium-bromide which is metabolized by mitochondrial succinic dehydrogenase activity of proliferating cells to yield a purple formazan reaction product. After HUVECs were treated with atorvastatin (0.1, 1 and 10 μmol/L), organic extracts (100, 200 and 400 μg/ml) and water-soluble fraction (100, 200 and 400 μg/ml) for 24 h, respectively, MTT (Sigma, USA) was added to a final concentration of 0.5 mg/ml and incubated for 4 h at 37°C. The culture medium was then removed and the remaining blue precipitate was solubilized in dimethyl sulfoxide, followed by reading absorbance at 492 nm (318MC, Shanghai 3 Division Appliance Co., Ltd., China). This reading was divided by the adjusted absorbance reading of untreated cells in control wells to obtain the percentage of cellular survival. 22
Measurement of NO, MDA and SOD
The assays for SOD, MDA and NO were carried out using assay kits (Nanjing Jiancheng Bioengineering Institute) and performed exactly according to the manufacturer’s instructions.
Measurement of ROS
The intracellular reactive oxygen species (ROS) level is an important biomarker for oxidative stress. An increased ROS level generally indicates increased oxidative stress. Probe 2,7-dichlorodihydrofluorescein diacetat (DCFH-DA, Sigma) penetrates the cells and is hydrolyzed by intracellular esterases to the nonfluorescent DCFH, which can be rapidly oxidized to the highly fluorescent 2,7-dichlorofluorescein (DCF) in the presence of ROS. Relative ROS production was determined by the formation of a fluorescent dichlorofluorescein (DCF) compound. 23 After incubation, cell counts of living and dead cells were examined by trypan blue staining and cell viability was calculated. The numbers of cells were 2 × 105 cells/well. After cells were stimulated with 100, 200 and 400 μg/ml organic extracts or water-soluble fraction for 1 h, the cells were incubated with 10 μM DCF-DA at 37°C for 30 min and washed twice with PBS. Relative fluorescence was measured using a fluorescence plate reader at 485 nm excitation and 535 nm emission wavelength (Hitachi, Japan). 22
Reverse transcriptase polymerase chain reaction (RT-PCR)
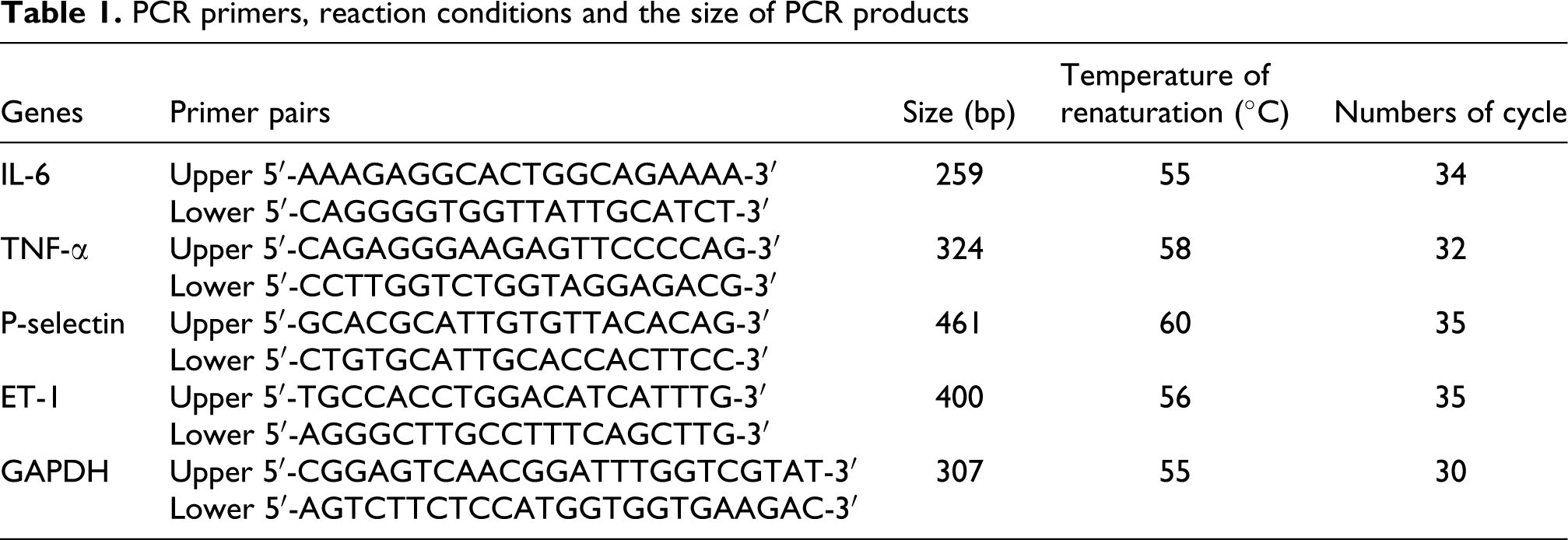
After HUVECs were treated with different fractions of fine particles for 24 h, total RNA from cultivated endothelial cells was extracted according to the RNeasy protocol (Fermentas). One microgram of total RNA was reversely transcribed into cDNA according to the instruction of reagent kit. The PCR primers were designed based on the sequencing of a cDNA encoding the human ET-1, IL-6, TNF-α and P-selectin protein using the primer design software, and GAPDH PCR primer sequences were obtained from the published literature. Table 1 depicts the PCR primers, reaction conditions and the size of PCR products. In reverse transcription reaction system, for each PCR, 1 μL of reverse transcribed RNAs (cDNA) was added directly into a PCR mixture, and 1× PCR master mix, 0.5 μM forward primer, 0.5 μM reverse primer, and water nuclease-free were contained in a total 25 μL reaction volume according to the manufacturer’s (Fermentas) instructions. The conditions of PCR were as follows: the initial denaturation step was at 94°C for 8 min, and denaturation for 45 s at 94°C, annealing for 45 s at 55–60°C, 30–35 cycles of 94°C for 45 s, extension at 72°C for 1 min and a final extension at 72°C for 10 min was applied to all the reactions and the PCR products were electrophoresed on a 2% agarose gel and pictures taken by UV illumination. The densitometry of each PCR band was measured and analyzed by BandScan 5.0 software. The densitometric values obtained from amplification of genes were corrected for background and divided by the density of parallel amplified GAPDH from the same samples to obtain normalized unit values. 24
PCR primers, reaction conditions and the size of PCR products
Statistical analysis
The results were presented as means ± standard deviation (SD). Comparisons of control and particle-treated groups were made by one-way analysis of variance (ANOVA), which is also used to evaluate the differences among non-treated group and treated with 0.1 μmol/L, 1 μmol/L and 10 μmol/L of atorvastatin group. The differences between water-soluble fraction and organic extracts were evaluated using paired t-test. The level of significance at 5% and the statistical package employed was the SPSS 11.5 for Windows.
Results
The chemical constitutes of water soluble and organic extracts
The levels of metals were determined in the water-soluble fraction of fine particles (Table 2). At least 10 elements were detected including some of transition metals. The predominance of transition metals such as Fe, V, Cu and Ni were detected. Among the transition metals, higher amounts of Fe, Cu and Cr were found. Eight polycyclic aromatic hydrocarbons (PAHs) were identified in the organic extracts of fine particles (Figure 1): phenanthrene, anthracene, fluoranthene, pyrene, benzo[a]anthracene, benzo[b]fluoranthene, benzo[a]pyrene and benzo[g, h, i]perylene, phenanthrene, anthracene, fluoranthene, pyrene, benzo[a]anthracene and benzo[b]fluoranthene. High levels of benzo[a]pyrene and benzo[g, h, i]perylene were observed in organic extracts of fine particles.
The content of elements in water-soluble fraction of fine particles (ng/m3, n = 3)


The components were characterized in organic extracts of fine particles by high-performance liquid chromatography (HPLC). 1–8 represent phenanthrene, anthracene, fluoranthene, pyrene, benzo[a]anthracene, benzo[b]fluoranthene, benzo[a]pyrene and benzo[g, h, i]perylene.
Cytotoxicity
The cell viability was determined in HUVECs after stimulated with atorvastatin alone (Figure 2a). The cell viability in HUVECs stimulated with 0.1 μmol/L, 1 μmol/L and 10 μmol/L doses of atorvastatin had no significant change when compared to no atorvastatin, which indicated the treatment of atorvastatin had no cytotoxic effect on cell viability. The cell viability of HUVECs after exposure to organic extracts and water-soluble fraction of fine particles for 24 h is shown in Figure 2b. Different fractions of fine particles caused decrease of cell viability when compared with the untreated control cells. The results showed that the water-soluble fraction of fine particles significantly decreased the cell viability in a dose-dependent manner. When exposed to the highest dose of the organic extracts and water soluble fraction, cell viability of HUVECs decreased to 70.1% and 61.2%, respectively. Meanwhile, the cell viability was significantly improved by immediate treatment with atorvastatin. When simultaneously treated with 10 μmol/L atorvastatin, cell viability of HUVECs in organic extracts and water-soluble fraction group elevated to 82.3% and 73.3%, respectively.

Effects of atorvastatin (a), organic extracts (b) and water-soluble (b) fraction of fine particles on cell viability of human umbilical vein endothelial cells (HUVECs). HUVECs were treated with different concentrations (0, 0.1, 1.0, 10 μmol) of atorvastatin, different concentrations (100, 200 and 400 μg/ml) of organic extracts and water-soluble fraction of fine particles for 24 h, respectively. And another 400 μg/ml organic extracts or water-soluble fraction exposure group was immediately treated with atorvastatin in concentrations of 0.1 μmol/L (A), 1 μmol/L (B) and 10 μmol/L (C), respectively. Each bar represents mean ± standard deviation (SD) of three independent experiments. Significant difference (*p < 0.05 and **p < 0.01) between different concentration of atorvastatin group and no atorvastatin group. Significant difference (*p < 0.05 and **p < 0.01) between fine particles fractions exposed group and control. Significant difference ( # p < 0.05) between organic extracts and water-soluble fraction at the same dose exposed. Significant difference (☆ p < 0.05, ☆☆ p < 0.05) between 400 μg/ml organic extracts (or water soluble fraction) and adding atorvastatin group.
NO, MDA, SOD and ROS in HUVECs
The levels of NO, MDA, SOD and ROS in HUVECs after stimulated with water-soluble and organic extracts of fine particles are shown in Figure 3. Fine particle fractions caused a significant increase in MDA and ROS in a dose-dependent manner, but NO and SOD decrease. Meanwhile, at the same dose group, water-soluble fraction induced more significant increase of MDA when compared to organic extracts. On the other hand, organic extracts caused more significant increase of ROS than-water soluble fraction. Meanwhile, water soluble extracts strongly diminished NO and SOD expression in HUVECs than that of organic-soluble extracts. Compared to no atorvastatin group, adding atorvastatin could attenuate fine particle-induced increase of MDA and ROS and suppress the decrease of SOD and NO in HUVECs in a dose-dependent manner.
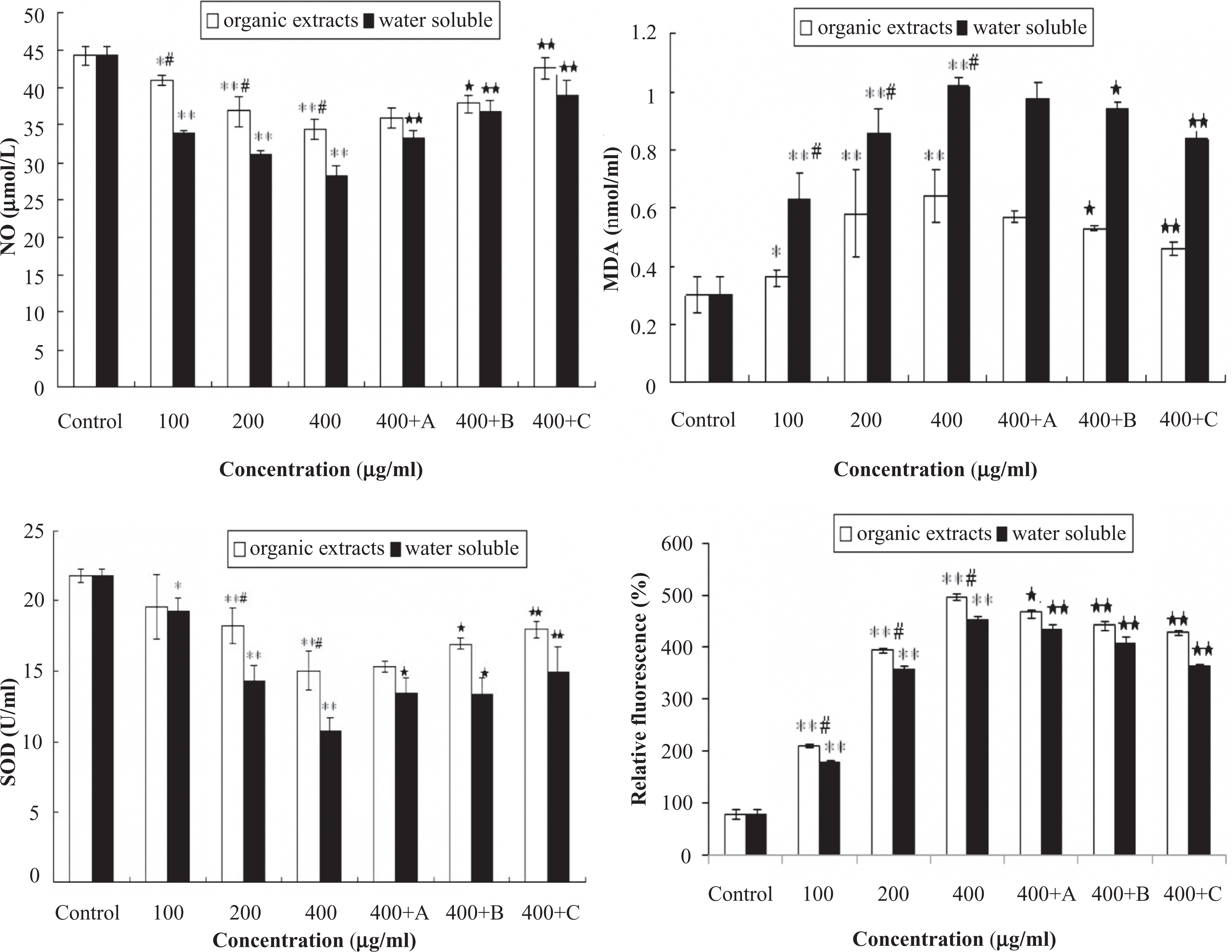
The levels of nitric oxide (NO), malondialdehyde (MDA), superoxide dismutase (SOD) and reactive oxygen species (ROS) in human umbilical vein endothelial cells (HUVECs) after exposed to organic extracts and water-soluble fraction of fine particles. HUVECs were exposed to organic extracts and water-soluble fraction (100, 200, and 400 μg/ml) for 24 h, and another 400 μg/ml organic extracts or water-soluble fraction exposure group was immediately stimulated by astorvastatin in concentrations of 0.1 μmol/L (A), 1 μmol/L (B) and 10 μmol/L (C), respectively. Each bar represents mean ± standard deviation (SD) of three independent experiments. Significant difference (*p < 0.05, **p < 0.01) between exposed group and control. Significant difference (# p < 0.05) between organic extracts and water-soluble fraction at the same dose exposed. Significant difference (☆ p < 0.05, ☆☆ p < 0.05) between 400 μg/ml organic extracts (or water soluble fraction) and adding atorvastatin group.
Cytokine mRNA expression
RT-PCR assay was used to determine mRNA expression of cytokines involved in inflammatory response, endothelial function and coagulant function in HUVECs (Figure 4). The results showed that water soluble and organic extracts of fine particles caused increase in mRNA expression of IL-6, TNF-α, ET-1 and P-selectin in a dose-dependent manner. In the same dose group, water-soluble fraction caused more expression of IL-6, TNF-α and P-selectin in HUVECs than that of the organic extracts, but organic extracts caused more expression of ET-1 in HUVECs. After atorvastatin was added to the HUVECs, the mRNA expression of all cytokines significantly decreased in HUVECs.

The mRNA expression of IL-6 (A), tumor necrosis factor-α (TNF-α) (B), P-selectin (C) and ET-1 (D) cytokines in human umbilical vein endothelial cells (HUVECs) after exposed to organic extracts and water-soluble fraction for 24 h. In the pictures of agarose gel, the exposed dose from left to right is 0.0 (PBS), water-soluble fraction dose group (100, 200, 400 μg/ml and 400 μg/ml + 10 μmol/L atorvastatin), organic extracts dose group (100, 200, 400 μg/ml and 400 μg/ml+10 μmol/L atorvastatin). *Significant difference (p < 0.05) between exposed group and control. # Significant difference (p < 0.05) between organic extracts and water-soluble fraction at the same dose. ☆ Significant difference (p < 0.05) between 400 μg/ml exposure group and adding atorvastatin group.
Discussion
The epidemiological studies have shown that cardiovascular disease is associated with an increased risk of morbidity and mortality from particulate matter air pollution. Recent studies have demonstrated that epithelial lung cells, macrophages, monocytes and granulocytes stimulated by particulate matter produce oxidative stress, apoptosis, mitochondrial damage and NF-κB activation.25–27 However, these effects have not been related to the systemic effects that epidemiologists described in many different studies. Organic extracts and metals as the major fractions in ambient fine particles play an important role in cardiovascular injury. Water-soluble fraction contain many metals (such as Ni, V and Zn), which can influence biological availability and may be a crucial factor when considering PM toxicity after inhalation. 28 Our study showed that the predominance of transition metals such as Fe, Cu, V, Cr, Cd and Ni were detected in water-soluble fraction. Among the transition metals, higher amounts of Fe, Cu, V and Ni were found. Previous studies showed that metals associated with PM may play an important role in particle-induced gene changes 29 and cardiac oxidative stress. 30 Meanwhile, organic extracts of fine particles and diesel exhaust particles induced oxidative stress and reduced the plasminogen activator inhibitor-1 (PAI-1) production of endothelial cells. 31 To better understand how the components of organic extracts may affect vascular endothelial cells, this study was to determine the PAHs in the organic extracts of fine particles. Eight PAHs were found in the organic extracts of fine particles. High levels of benzo[a]pyrene and benzo[g, h, i]perylene were observed. These PAHs are crucial factors in the DNA damage induced by PM. 32 This study determined the effects of water-soluble fraction and organic extracts of fine particles on vascular endothelial cell. Meanwhile, atorvastatin was used to further observe the toxicity of fine particles. The doses of atorvastatin used in this study (0.1–10 μmol/L) relate closely to blood levels of atorvastatin in patients treated with 10–80 mg of the drug. Other result showed that atorvastatin at the concentrations of 0.1–10 μmol/L could moderately enhance the expression of endothelial nitric oxide synthase (eNOS) and diminished the production of interleukin IL-8 in a dose-dependent manner. 33 Therefore, the concentrations of atorvastatin we used in intervention study were 0.1 μmol/L, 1.0 μmol/L and 10 μmol/L. In this study, cell viability was assayed in cells after stimulated with atorvastatin alone. The results showed that 0.1 μmol/L, 1.0 μmol/L and 10 μmol/L doses of atorvastatin had no cytotoxic effect on cell viability.
NO, derived from the action of NOS, is one of the most important mediators in the regulation of endothelial functions. 34 It is reported that down-regulation of NO production is implicated in the pathogenesis and clinical course of all known cardiovascular diseases and is related to future risk of adverse cardiovascular events. 35 In our previous study, the results showed that NO decreased in the serum of rats when instilled with fine particles. 17 In this study, stimulation of HUVECs by organic extracts and water-soluble fraction of fine particles induced a dramatic decrease of NO. Meanwhile, it is likely that endothelial dysfunction is more severe in HUVECs after exposure to water-soluble fraction when compared to organic extracts. Simultaneous treatment with atorvastatin could suppress the decrease of NO induced by fine particles fraction in a dose-dependent manner. Other study supported that the cardioprotective effects of statins are partly independent of their serum lipid lowering effects and may be due to the activation of eNOS in the vascular endothelium, 36 namely, statins protected against cardiovascular injury possibly through NO-dependent mechanisms. 37 Furthermore, in this study, increase of the mRNA expression of ET-1 in HUVECs also indicated that organic extracts and water-soluble fraction caused endothelial dysfunction, and atorvastatin could attenuate fine particle fractions-induced ET-1 production. Similarly, other results showed that the rise in ET-1 mRNA levels was prevented in HUVECs after treated with 1.0 μmol/L or 10 μmol/L of rosuvastatin. 38 The results declared that organic extracts and water-soluble fractions caused the endothelial dysfunction of HUVECs.
Lipid peroxidation is one of the primary events in free radical-mediated cell injury. MDA as a thiobarbituric acid reactive substance is the end-product of lipid preoxidation and is widely used as a biomarker of oxidative stress. On the other hand, other enzymatic antioxidants, such as SOD and GSH-Px play an important role in preventing cellular damage caused by ROS. 39 In this study, significant increase in MDA and decrease in SOD were observed in HUVECs after exposure to organic extracts and water-soluble fraction in a dose-dependent manner. Meanwhile, water-soluble fraction caused the greater increase of MDA and decrease of SOD when compared to organic extracts. More interesting, when HUVECs were simultaneously treated with atorvastatin, these fine particles fractions-induced cellular events were blocked to a great extent, which verified that atorvastatin was able to diminish the oxidative stress in HUVECs. The results further revealed that organic extracts and water-soluble fraction could cause the endothelial oxidative injury. ROS could lead to oxidative stress, and then affected endothelial and vascular function. This study showed that organic extracts and water-soluble fraction of ambient fine particles induced oxidative stress mediated by increase of ROS. Similar to this result, other study has shown that fine particles induce the increase of ROS. 40 Different to MDA and SOD, the changes of ROS induced by organic extracts were more significant than by water-soluble fraction. The difference needs to be explored in further research. Moreover, simultaneous treatment with atorvastatin alleviated the ROS and MDA and enhanced SOD in a dose-dependent manner. These results jointly suggested that enhancement of endogenous antioxidant preservation and attenuation of lipid peroxidation may represent a major mechanism of cellular protection by atorvastatin. The effects of atorvastatin on the levels of SOD, MDA and ROS supported the results of the oxidative damage of fine particle fractions on HUVECs.
Fine particles could induce inflammatory effects on human epithelial lung cells. 41 In this study, increase of the expression of IL-6 and TNF-α in HUVECs indicated that organic extracts and water-soluble fraction caused the inflammatory response. Moreover, the production of IL-6 and TNF-α in HUVECs were stronger for water-soluble fraction than for organic extracts. The conclusion is in contrast to results from other study of fine particles toxicity. 42 P-selectin is a cell adhesion molecule found in platelets and endothelial cells and appears to play a key role in response to tissue injury and inflammation. The mRNA expression of P-selectin increased in HUVEC after treatment with water-soluble fraction and organic extracts, especially in water soluble fraction. Meanwhile, atorvastatin diminished the inflammatory relative factors IL-6, TNF-α and P-selectin. Other reports also showed that statins could reduce the TNF-α-induced surface expression of ICAM-1, VCAM-1, IL-8 and IL-6 in HUVECs.43,44 In agreement with these reports, simultaneous treatment with atorvastatin could distinctly alleviate fine particle fractions-induced inflammatory response. It supported that fine particle compositions induced endothelial dysfunction and inflammatory response.
In summary, the increase of ROS, MDA, IL-6, TNF-α, ET-1 and P-selectin and the decrease of NO and SOD in human endothelial cells demonstrated that fine particles could be related with the development of inflammatory response, endothelial dysfunction and oxidative damage. More important, these factors were closely associated with the progression of cardiovascular diseases. High levels of Fe, Ni, V and benzo[a]pyrene, benzo[g, h, i]perylene were detected in water-soluble fraction and organic extracts, respectively. It indicated that these components might be the major factors caused inflammatory response, endothelial dysfunction and oxidative damage. It is possible to speculate that these different effects may be due to the different fractions of organic extracts and water-soluble extracts. Similarly, other result showed that high levels of V, Ni and Cu were found in water-soluble extracts of particulate matter, and these transition metals could be involved in PM-induced oxidative stress, promoting the formation of oxidative lesions.45,46 Organic extracts also induced significant oxidative stress in HUVEC. Jeng supported that redox activity induced by particulate matter compositions highly correlated with organic compounds, and insignificantly correlated with elemental carbon and metals, 47 which could be a major factor to explore the relationship of particulate matter compositions and redox activity. 47 Both water-soluble fraction and organic extracts resulted in inflammatory response, oxidative stress and endothelial dysfunction in endothelial cells at 24 h, which supported that water-soluble transition metals and the organic-soluble fraction of fine particles are both important to the endothelial damage induced by ambient fine particles. Meanwhile, this study pointed out that the effect of water-soluble fraction on endothelium was greater than organic extracts. The water-soluble fraction was more likely to influence endothelial cells than organic extracts. Such research would enhance the understanding of fine particles and health, particularly the question on whether fine particles-induced health effects depended on the specific physical and/or chemical characteristics of the particles. The findings hinted that inflammatory response, endothelial dysfunction and oxidative stress in endothelial cells might be the mechanisms of vascular endothelial dysfunction, which mediated cardiovascular injury induced by different compositions of fine particles.
Footnotes
Jinzhuo Zhao and Yuquan Xie contributed equally to this work and should be considered as co-first authors.
The authors declare that they have no conflicts of interest.
This work was supported by grants from the Natural Science Foundation of Shanghai, China (No. 09ZR1402400), National Natural Science Foundation of China (No. 81001229), Gong-Yi Program of China Ministry of Environmental Protection (No. 200809109), Program for New Century Excellent Talents in University (No. NCET-09-0314), and the National High Technology Research and Development Program of China (No. 2007AA06Z409).
