Abstract
Iodine tincture poisoning is uncommon regardless of its widespread use as an antiseptic in daily practice. Previously reported effects of iodine-containing antiseptic poisoning included topical irritation, corrosive effects, allergic response, and hepatic or renal injury, which mainly resulted from complications of topical use during surgical procedures. We herein reported an unusual case of severe hemolysis and acute renal failure following intentional ingestion of iodine tincture containing 60 mg/ml iodine and 40 mg/ml potassium iodide in 70% v/v ethanol. The patient completely recovered 8 weeks later after receiving supportive treatment, plasma exchange, and temporary hemodialysis.
Introduction
Although iodine-containing antiseptics are widely used in daily practice, the toxic mechanisms of iodine is poorly understood. 1 Iodine was found to be of extremely low toxicity in animals with a median lethal dose (oral) around 14,000 mg/kg. 2 On the contrary, the probable mean lethal dose of iodine in human was estimated to be only 2−4 g. 2 Currently, only a few human iodine poisoned cases had been published, in whom topical irritation, corrosive effects, allergic response, and hepatic or renal injury were the primary manifestations, which mainly resulted from complications of topical use during surgical procedures.3,4 Acute hemolysis had been infrequently reported in English literature after large dose oral exposures.5,6
We herein describe an unusual case of acute hemolysis following deliberate ingestion of an iodine tincture that contains 60 mg/ml iodine and 40 mg/ml potassium iodide in 70% v/v ethanol.
Case Report
A 30-year-old previously healthy male attempted suicide by ingesting 200 ml “iodine tincture” stored as an antiseptic in his house because of socioeconomic stress. He soon developed sore throat, epigastralgia as well as bilateral flank pain. However, he did not seek medical help until 12 hours later. He was referred to our service 18 hours post-ingestion due to acute renal failure.
On arrival, his vital signs were blood pressure 148/98 mmHg, pulse rate 91/min, respiratory rate 20/min, temperature 37.6°C. Physical examinations revealed slight icteric sclera while no cyanosis presented. His chest X-ray and electrocardiogram were within normal limits. Laboratory data were remarkable for white blood cell count 35,200 × 109/L with 91% neutrophils, hemoglobin (Hb) 14 g/dL; serum C-reactive protein 2.09 mg/dL, potassium 5.6 mEq/L, blood urea nitrogen 46 mg/dL, creatinine 3.76 mg/dL, aspartate aminotransferase 545 U/L, alanine aminotransferase 51 U/L, lactate dehydrogenase 5,563 U/L, total bilirubin 2.45 mg/dL with conjugated form of 0.56 mg/dL, and plasma free Hb of 222 mg/dL (reference range 1−5 mg/dL) (Table 1). Arterial blood gases under 3 L/min O2 supplement revealed pH 7.35, PaCO2 33.8 mm Hg, PaO2 71.5 mm Hg and HCO3 - 18.1 mEq/L. Toxicological screening for basic drugs and paraquat were negative.
Baseline laboratory data of a 30-year-old male patient with intentional iodine tincture poisoning.
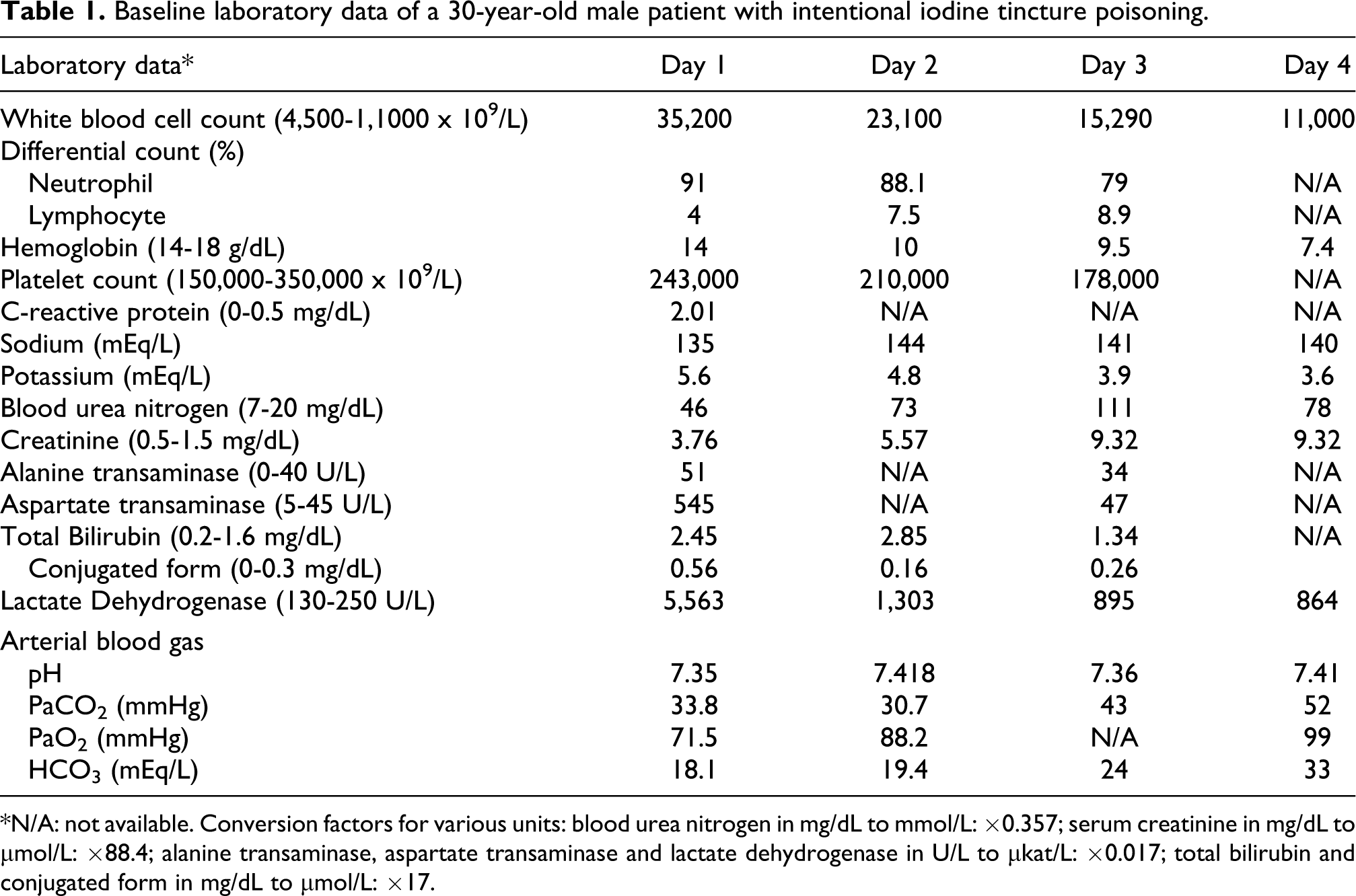
*N/A: not available. Conversion factors for various units: blood urea nitrogen in mg/dL to mmol/L: ×0.357; serum creatinine in mg/dL to μmol/L: ×88.4; alanine transaminase, aspartate transaminase and lactate dehydrogenase in U/L to μkat/L: ×0.017; total bilirubin and conjugated form in mg/dL to μmol/L: ×17.
Three sessions of plasma exchange (PE) with the dose of 2600 ml over 2 hours per day was arranged for suspected iodine-related severe intravascular hemolysis. Moreover, intermittent hemodialysis (HD) was required because of persistent anuria after day 3. Eighty hours post-ingestion, tarry stool with Hb decreasing to 7.4 g/dL was noted. Upper gastrointestinal (GI) endoscopy was not performed because he could not tolerate the procedure. He received conservative treatments only for probable corrosive injury of GI tract.
His hospital course was further complicated with aspiration pneumonia and respiratory failure that occurred 2 weeks post-ingestion, which necessitated 3 weeks of mechanical ventilation. His renal function gradually recovered 5 weeks later, and he was discharged 8 weeks post-ingestion.
Total serum iodine level excluding protein-bound iodine at our emergency department was found to be 1,155,901 μg/L (reference range 30.48−80.01 μg/L), measured by inductively coupled plasma mass spectrometry, a method described elsewhere. 7 The level then decreased steeply and was 63,336 μg/L and 4,849 μg/L on day 6 and day 11, respectively (Figure 1).

Serial serum iodine levels by number of days post-ingestion. There were pre- and post-PE iodine levels obtained during the first two sessions of PE. PE: plasma exchange, HD: hemodialysis.
Discussion
The effects iodine exerts on organisms are quite complex. When iodine is dissolved in water, at least 10 different iodine species are produced. In the presence of iodide and ethanol, additional species, including ethanol-I2, ethanol-I+, ethanol-I3 - are produced. 8 Some of them such as I+, I2, I3 -, and HOI have oxidizing capacity while iodide is not. 8 Iodine, in its molecular form, can readily penetrate the cell wall of microorganisms. Oxidation of the thiol (SH) group of amino acids by iodine can lead to inability of the proper formation of disulfide bonds, an important factor in the synthesis of functional enzymes. In addition, electron microscopy and biochemical observations suggested that the interaction between iodine and the double bonds of phospholipids could cause damage to cell membrane, which then led to the loss of intracellular materials in microbes. 8
Human erythrocyte is one of the cells first exposed to xenobiotics whether they are inhaled, ingested or applied directly to the skin. When the hemoglobin sustained uncorrected oxidative injuries, disulfide links with other cellular proteins result in intracellular precipitates. With a greater degree of oxidation, accompanied with loss of membrane integrity, the occurrence of intravascular hemolysis is possible. 9
There may be other mechanisms involved in iodine tincture-related hemolysis.5,6 Theoretically, iodide, though less toxic to human, 2 interacts much more effectively than either chloride or bromide with myeloperoxidase released by activated leucocytes. 10 When large amounts of iodide are present and hydrogen peroxide and chloride exist, myeloperoxidase can oxidize iodide to HOI, a more powerful oxidant, which leads to additional cellular damages. 11 Unfortunately, we could not perform speciation of iodine in our case; therefore, the potential role of different iodine species could not be further evaluated.
Controversy remains in the treatment of iodine poisoning. Hemodialysis was thought to efficiently remove iodine in acute renal failure since the main excretory route of iodine is kidney. 4 However, fatalities did occur when the initial serum iodine level was sufficiently high.3,12 We performed PE and found the total serum iodine level decreased substantially even between sessions of PE (Figure 1). Interaction of iodine and plasma protein (e.g. thiol group) may account for further decrease of toxic serum iodine level. 13 Severe hemolysis also rapidly subsided (Table 1). Furthermore, hepatic injury, refractory acidosis or altered consciousness upon high dose exposure1,12,14 did not occur in the patient. Therefore, PE may have some beneficial effects in the management of iodine-induced hemolysis.
Conclusion
The effects iodine species exert on tissues are quite complex. In addition to direct kidney injury, exposure to large dose iodine tincture may lead to severe hemolysis through complex and not well-understood mechanisms. We speculated that oxidative stress play one of the important roles. Plasma exchange may be effective in the management of iodine poisoning; however, its significance warrants further study.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
