Abstract
Sulfur dioxide is a typical air pollutant. Sulfite, which is formed at the bronchial mucosa from inhaled sulfur dioxide, might play a role in the exacerbation of asthma. In this study, we investigated the effects of sodium sulfite and its interaction with a house dust mite (
Introduction
Bronchial asthma is a chronic disease characterized by airway obstruction, eosinophilic airway inflammation and enhanced bronchial responsiveness. The disorder is mainly associated with T helper 2 (Th2)-mediated inflammation and allergic sensitization. 1 Allergic sensitization is characterized by environmental allergen exposure with subsequent production of allergen specific IgE antibody. 2 The increased incidence of bronchial asthma in many countries has been reported to be associated with the aeroallergen—house dust mites. A significant correlation between house dust mites and an increased rate of asthma has been observed. 3
Sulfur dioxide (SO2) is well recognized as a hazardous air pollutant. The main source of SO2 in the atmosphere is from the combustion of fossil fuels during industrial processes. SO2 and its derivatives (sulfides) are widely used in cosmetics, food preservatives, bleaching wood pulp or paper, waste or water treatment, and metal or oil refining. 4 Many sulfides release SO2 in the presence of water or acid.
In the human body, once SO2 is absorbed through the respiratory tract, it shows a wide tissue distribution. The amount of SO2 in the blood and other tissues increases proportionally with its concentration in inhaled air. 5 In the past few decades, multiple studies have shown that SO2 inhalation can cause multiorgan damage and impairment of the respiratory immune response. 6 In addition, DNA damage has also been observed which can result in cancer and other diseases. 7
Inhaled SO2 can be easily hydrated to sulfurous acid in the respiratory tract, which subsequently dissociates into sulfite, bisulfite and hydrogen ions. The toxicity of SO2 is, in fact, mediated by sulfite and bisulfite. 8 Many studies have shown the relationship between SO2 exposure and increasing prevalence of bronchial asthma. For instance, higher incidence of asthma was demonstrated among workers exposed to SO2 comparing to unexposed workers. 9 Moreover, SO2 exposure has also been associated with hospital admission in children with asthma. 10 In animal studies, SO2 inhalation has aggravated lung function impairment as well as enhancement of allergic sensitization. 11,12
Although there have been many studies regarding the relationship between bronchial asthma and mite allergen (or SO2), few studies had focused on the synergistic effects of SO2 and mite allergen on allergic sensitization and airway inflammation. We herein use an animal model to study the relationship. Mite (
Methods
Animals
A total of 46 male BALB/c mice (6–8 weeks of age) were purchased from the National Laboratory Animal Center, Nangang, Taipei, Taiwan. They were fed with a commercial diet and water.
Study protocol
The mice were divided into four groups: (1) control (

Study design. Experimental design for the treatment of BALB/c mice with Der p crude extract protein and/or sodium sulfite. IN: intranasal, mite: Dermatophagoides pteronyssinus crude extract protein, sulfite: sodium sulfite.
Mite protein preparation
Der p was purchased from Greer Lab (Lenior, NC, USA). The mite protein was extracted from the crude with phosphate buffered saline (PBS) using a glass-glass homogenizer (Kontes Glass Company, Vineland, NJ, USA). The mite protein concentration was then determined using the Bradford procedure (Bio-Rad protein Assay; Bio-Rad, Hercules, CA, USA).
Sensitization
Mite crude extract allergen (1.6 mg/ml) was prepared and well-emulsified in complete Freund’s adjuvant (CFA; Sigma, St Louis, MO, USA) at a ratio of 1:1 at 4°C. The mIN, sIN and mIN + sIN groups were immunized subcutaneously with 50 microliters of emulsified mite protein on day 1 and day 8.
Intranasal administration of mite protein
Ten microliters of mite crude extract allergen (5 mg/ml), dissolved in PBS, was administered to each mouse intranasally once daily from day 15 to day 22.
Intranasal administration of sodium sulfite
Sodium sulfite (Sigma) was dissolved in saline solution to the final concentration of 5 mM. Ten microliters of sodium sulfite was then administered to each mouse intranasally from day 1 to day 22.
Measurement of Der p-specific IgE and IgG2a antibodies
All 46 BALB/c mice were sacrificed, and the blood was obtained from inferior vena cava on day 23. Plasma Der p-specific IgE and IgG2a levels were measured by enzyme-linked immunosorbent assay (ELISA). The microplates were coated with mite crude extract allergen (50 μg/ml) overnight at 4°C. The plates were blocked with 10% fetal bovine serum (FBS) in PBS at room temperature for 2 hours. After washing with PBS containing 0.05% Tween 20 (PBST), diluted plasma samples (1/10 dilution) were added to the wells of the microplates and incubated at 4°C overnight. After washing with PBST, the plates were incubated with rat anti-mouse IgE biotinylated monoclonal antibody (specific for mouse IgE-heavy chain or IgG2a-heavy chain; Abcam) at room temperature for 2 hours. After the second wash with PBST, the wells were incubated with horseradish-peroxidase (HRP) streptavidin conjugate (Zymed for IgE and AbD for IgG2a) at room temperature for 1 hour. The wells were washed and then incubated with TMB substrate (BD Bioscience) in the dark, at room temperature for 30 minutes. The enzyme reaction was stopped by adding 2N H2SO4, and the absorption at 450 nm was measured using an ELISA reader. Since no Der p-specific IgE standard was available, all samples were tested at the same time and results were expressed as an optical density (O.D.) value. The final mean O.D. level of the control group was defined as 100% in final presentation.
Histopathological analysis
The lung section stained with hematoxylin and eosin (H&E) was assigned a unit value for alveolar, peribronchiolar and total inflammation by computing the means of three independent scores for three randomly selected view-fields. We followed a previous scoring system for airway inflammation. 13 Peribronchiolar inflammatory scores for each view-field were determined as follows: 0, normal; 1, a few cells; 2, a ring of inflammatory cells one cell layer deep; 3, a ring of inflammatory cells 2–4 cells deep; 4, a ring of inflammatory cells of > 4 cells deep. Alveolar inflammatory scores for each view-field were determined as follows: 0, normal; 1, alveolar walls normal, a few macrophages in alveoli; 2, mild thickening of alveolar walls and increased alveolar macrophages and eosinophils; 3, marked thickening of alveolar walls and alveolar multinucleated giant cells and eosinophils in 30%–50% of the field; 4, same as 3 but in >50% of the field; 5, complete consolidation. Total inflammatory scores were defined as the sum of the peribronchiolar and alveolar inflammatory scores.
Preparation of lung tissue supernatant
Four mice from each group underwent lung tissue supernatant cytokine analysis. Protein extraction reagent (PRO-PREPTM Protein Extraction Solution, iNtRON Biotechnology, Gyeonggi-do, Korea) was added (1 ml) to each lung tissue, then lung tissues were homogenized using a glass-glass homogenizer at 4°C. Tissue homogenates were centrifuged at 13000 rpm for 5 min at 4°C and the supernatants were transferred to clean microcentrifuge tubes on dry ice and then thawed on ice. Total protein concentrations in the lung tissue supernatants were determined using the Bradford procedure. The lung tissue supernatants were then diluted with PBS to a final protein concentration of 500 μg/ml and stored at –80°C.
Cytokine analysis of lung tissue supernatants by ELISA
The cytokine protein levels of interleukin-5 (IL-5) and interferon-γ (IFN-γ) were examined in the lung tissue supernatants using BD OptEIATM Set Mouse IL-5 and IFN-γ kits (BD Bioscience, San Jose, CA, USA). The kits were used according to the manufacturer's instructions. The microplates used in these assays were read at 450 nm with an ELISA reader (Thermo Labsystems, Waltham, MA, USA). The ratio of IL-5/IFN-γ in each group was also calculated and the mean ratio of the control group was defined as 100% for final presentation.
Statistical analysis
Data were expressed as mean ± standard error (SE). Analysis was performed with the Mann-Whitney U test for comparison between two groups. Differences with a probability value <0.05 were considered significant. Analysis was performed using the Statistical Package for the Social Sciences (version 10.1; SPSS, Chicago, IL, USA).
Results
Plasma Der p-specific IgE and IgG2a level
Figure 2A shows the Der p-specific IgE levels (the mean O.D. value of the control group defined as 100%) in the mice plasma. In comparison with the control group, Der p-specific IgE levels in the mIN, sIN and mIN + sIN groups were significantly increased (
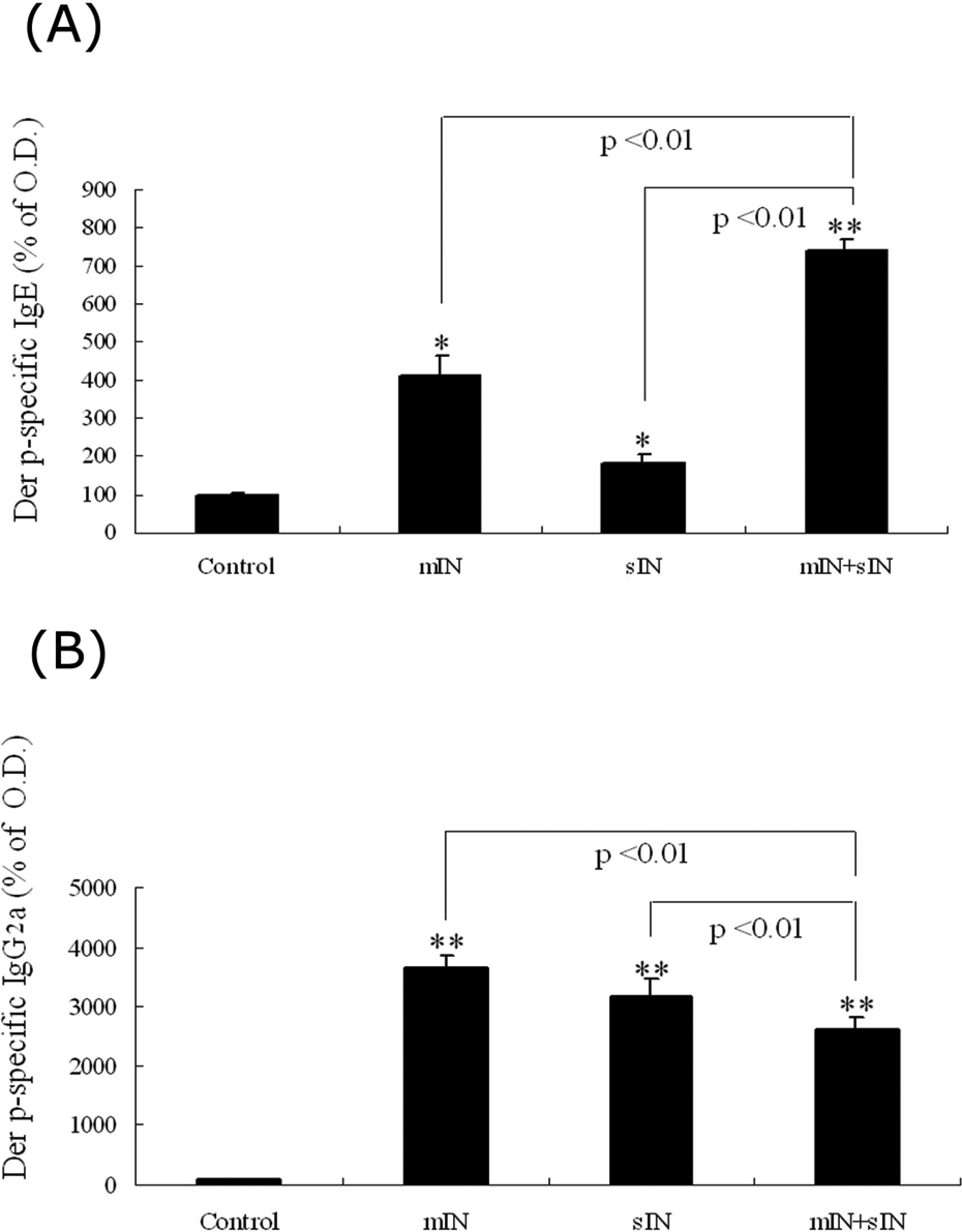
Plasma Der p-specific IgE and IgG2a levels. Plasma Der p-specific IgE levels (A) and IgG2a levels (B) in the four groups. (control:
Figure 2B demonstrated the Der p-specific IgG2a levels (the mean O.D. value of the control group defined as 100%) in the mice plasma. In comparison with the control group, the levels were significantly higher in mIN, sIN and mIN + sIN groups (all
Histopathological studies
Figure 3A showed significant airway inflammation in mIN, sIN and mIN + sIN groups by histopathological studies. The control group had clear alveolar space without alveolar wall thickening, while mIN group had mild alveolar wall thickening with some inflammatory cells in the alveolar space. In sIN group, marked alveolar wall thickening with many inflammatory cells in the alveolar space were observed. mIN + sIN group had many inflammatory cells infiltrating the alveolar space with the lung parenchyma totally consolidated. Figure 3B summarizes the mean scores in the four groups. The mean peribronchiolar inflammatory scores were significantly higher in mIN + sIN groups compared with the control group (
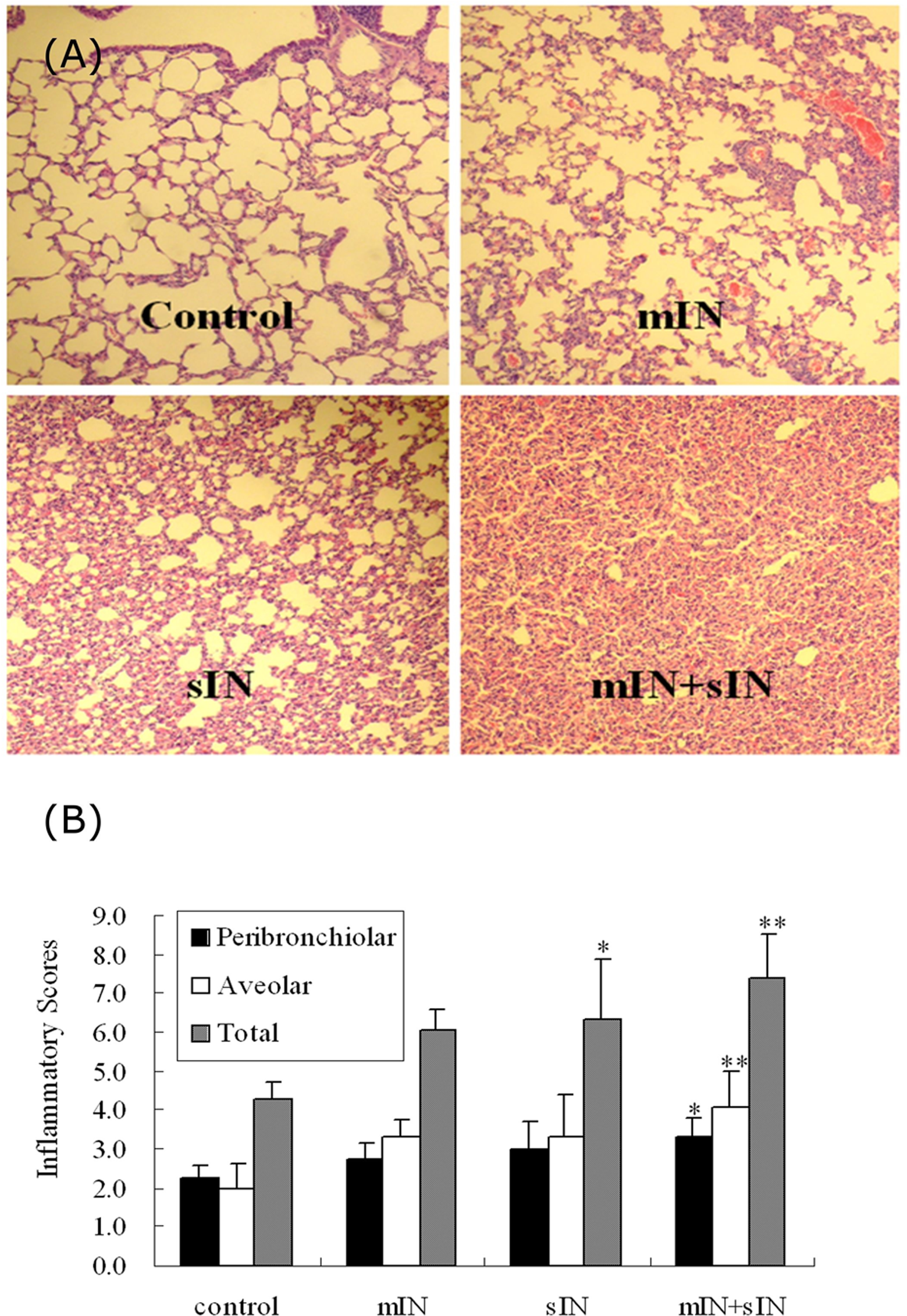
Histopathology and inflammatory scores. Histopathology of lung sections stained with H&E (100×) (A) and the scoring of these sections by peribronchiolar, alveolar and total inflammatory scores for each of the treatment groups (B). (control:
Lung tissue supernatant IL-5 levels
Figure 4A demonstrated lung tissue supernatant IL-5 levels in four groups. Only mIN + sIN group had significantly higher level than the control group (

Cytokines analyses for lung supernatant. Lung tissue supernatant (A) IL-5, (B) IFN-γ levels and (C) IL-5/IFN-γ ratio in 4 groups
Lung tissue supernatant IFN-γ levels
Figure 4B demonstrated lung tissue supernatant IFN-γ levels in four groups. The mIN, sIN and mIN + sIN groups had significantly lower levels than the control group (all
IL-5/IFN-γ ratio
Figure 4C demonstrated the ratio of IL-5/IFN-γ in four groups. The mIN, sIN and mIN + sIN groups had higher ratio than the control group (
Discussion
In the last decades, mite has been well-known as the most common aeroallergen around the world. 14 Meanwhile, air pollutant exposure is a significant risk factor for respiratory diseases in most urban and industrialized areas. The effects of air pollutant exposure on allergen-specific IgE production (allergic sensitization) have not been well studied. One study suggested that air pollutant, ozone or related ambient pollutants, up-regulate total IgE levels among asthmatic adults. However, no association was observed between total IgE levels and SO2 in this study. 15
Animal studies have shown that diesel exhaust particles enhance allergen-specific IgE production. 16 To our best knowledge, only one animal study regarding the effect of SO2 (or sulfite) on allergic sensitization has been reported. 12 It demonstrated that SO2 inhalation enhanced ovalbumin (OA)-specific antibodies both in serum and bronchoalveolar fluid in guinea pigs sensitized with inhaled OA. However, OA and house dust mites are vastly different allergens. The effects of SO2 (or sulfite) on house dust mite-induced allergic sensitization have never been studied.
In our study, plasma Der p-specific IgE was significantly increased in mIN + sIN group. This indicated that sulfur dioxide derivative—sulfite—may aggravate allergic sensitization in mite allergen sensitized BALB/c mice. Moreover, Der p-specific IgG2a declined significantly in mIN + sIN group than the mIN group. Since IgG2a was well recognized as a T helper 1 (Th1)-associated immunoglobulin, this result suggested that sulfite may attenuate the Th1 response. In addition, lung tissue supernatant cytokine levels, including IL-5 (Th2 cytokine) and IFN-γ (Th1 cytokine), were also analyzed in our study. The IL-5 level was significantly increased in the mIN + sIN group comparing with the control, mIN or sIN groups. The IFN-γ was significantly declined in sIN group. However, no more attenuation of IFN-γ level was observed in mIN + sIN group. To confirm the effect of sulfite toward Th1 or Th2 response, the IL-5/IFN-γ ratio, which has been previously used as a marker for Th2 polarized response, was also calculated. The ratio was significantly higher in mIN + sIN group than the control, mIN and sIN group. These results indicated that sulfite may enhance local Th2 polarized inflammation.
Although the direct relationship between air pollutants and airway diseases has not been fully established, airway epithelial cells were thought to play an important role. On contact with air pollutants, there may be increased ciliary activity and permeability in airway epithelial cells. 17 Besides, sodium sulfite has been reported to enhance mite-stimulated detachment of A549 airway epithelium cells. 18 As a result, increased ciliary activity and permeability as well as detachment of airway epithelium may lead to increased penetration and access of aeroallergens.
In addition, activation of transcriptional factors and the arachidonate pathway leading to the synthesis and release of large amount of inflammatory mediators in airway epithelium may result in recruitment of inflammatory cells and airway hyper-responsiveness. SO2 derivatives has been reported to increase the expression of epidermal growth factor, epidermal growth factor receptor, intercellular adhesion molecule-1 and cycloxygenase-2 on the transcription and translation levels in human bronchial epithelial cells. 19 It results in mucus over-production and inflammatory responses. Another possible effect of air pollutant for airway disease is their ability to induce a Th2 cytokines in airways. 17 Animal study has demonstrated sulfur dioxide inhalation facilitate local Th2-derived cytokines as well as the infiltration of eosinophils which subsequently enhance the development of allergic diseases. 20 The above mentioned possible mechanisms may partially explain the effect of SO2 in enhancement of allergic sensitization.
The airway inflammation was also evaluated in our study. There have been many studies regarding the ability of inhaled SO2 on airway inflammation. Increased human neutrophils and rat macrophages were observed in bronchoalveolar lavage fluids after SO2 inhalation. 21,22 Besides, there were significant increased IL-6 and tumor necrosis factor-α levels in lung tissues of SO2 inhaled mice. 23 In our study, we also confirmed the effects of sodium sulfite toward airway inflammation. The total inflammatory scores were higher in sIN and mIN + sIN groups. Comparing with sIN group, there were also significantly increased scores in the mIN + sIN group observed. Therefore, we also emphasize the synergistic effect of mite allergen and sulfite toward airway inflammation.
In conclusion, sodium sulfite, a derivative from air pollutant—sulfur dioxide, may aggravate allergic sensitization as well as airway inflammation in mite allergen sensitized BALB/c mice.
Footnotes
Acknowledgment
The authors wish to thank the Biostatistics Task Force of the Taichung Veterans General Hospital for statistical assistance.
This study was supported by a grant from Taichung Veterans General Hospital, Taichung, Taiwan (TCVGH-PU978109).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
