Abstract
Cigarette smoking is common in societies worldwide and has been identified as injurious to human health. Human red blood cells are important targets for electrophilic and oxidant foreign compounds. In the present study, the possible role of antioxidant status on smoking-induced erythrocyte hemolysis of smokers was studied. Erythrocyte superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) activities, reduced glutathione (GSH) level, erythrocyte membrane lipid peroxidation, total cholesterol and phospholipids were determined. Further, nitrite/nitrate levels (NO2/NO3) in both plasma and erythrocyte lysate were measured. Results showed increased plasma and erythrocyte membrane lipid peroxidation and nitrite/nitrate levels in smokers. The activities of SOD, CAT and GPx were also increased with reduced glutathione (GSH) level in smokers. No significant change was observed in smokers red cell hemolysis and cholesterol/phospholipid (C/P) ratio compared to controls. Erythrocyte membrane lipid peroxidation was positively correlated with SOD (r = 0.482, p < 0.01) and GPx (r = 0.368, p < 0.018) in smokers. Increased levels of nitrite/nitrate and antioxidant status of erythrocytes might be playing a crucial role in protecting red cell from free radical damage induced by cigarette smoke.
Introduction
Cigarette smoking adversely affects human health with a broad spectrum of harmful effects and diseases related to various organs and tissues. 1 The most affected are the cardiovascular and pulmonary systems leading to a number of diseases such as chronic obstructive pulmonary diseases (COPD), coronary heart diseases (CHD) and cancer. 2 Oxidative stress is believed to aggravate the symptoms of many diseases, including hemolytic anemias. 3 The adverse health consequences of smoking have been largely attributed to the abundance of reactive oxygen species (ROS) and reactive nitrogen species (RNS) that readily react with various biomolecules. 4 As humans are oronasal inhalers, the inhaled smoke affects not only pulmonary parts but also parts of digestive tract, which enters circulation affecting cardiovascular system and, all parts of the body through blood supply. 5 Plasma and red cells are frequently exposed for long time to cigarette smoke constituents for prolonged periods in smokers as smoking is a continuous puff after puff exercise. 6 Since blood plays an important role in the integration of metabolism, supplying oxygen and nutrients, blood analysis provides essential information regarding various physiological and biochemical processes. There is a paucity of information concerning the mechanisms of various biochemical events related to cigarette smoking.
It is well known that cigarette smoke contains many constituents including superoxide and a large number of other ROS.7,8 Free radicals can directly damage red blood cell (RBC) membranes by peroxidation of membrane polyunsaturated fatty acids. 9 Peroxidation of lipids can disturb the assembly of the membrane, causing changes in fluidity and permeability, alterations of ion transport and inhibition of metabolic processes. 10 Several studies suggested that cigarette smoke impair the antioxidant defense mechanisms of the cell membrane, and cause membrane deformity, especially by changing the lipid fluidity in the middle zone of the bilayer. 11 Removing superoxide ion and hydroxyl radical is probably one of the most effective defense mechanisms against a variety of diseases. 12 Superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) are among most important antioxidative enzymes in free radical scavenger system in normal conditions. Our previous reports also revealed that chronic cigarette smoking increases hemolysis. 13 There are several studies that showed an association between heavy smoking and oxidative damage.14,15 In spite of the abundance of evidence that links cigarette smoking-induced oxidative stress to several diseases, effect of moderate cigarette smoking on biochemical events of plasma, red cell and red cell membrane is not clear. The present study was conducted to obtain information on efficiency of erythrocyte antioxidants protective mechanism from moderate cigarette smoking-induced hemolysis.
Materials and methods
Subjects
Human male volunteers aged 32 ± 7 years smoking for 6 ± 1 years were chosen as experimental subjects. In the present study who smokes at least 4–6 cigarettes per day were considered as moderate smokers. Controls (age, sex and diet matched) who did not smoke were selected for the study. All the volunteers were well informed about the experimentation and their written consent was obtained. Institutional ethical committee approved this study. Blood samples from overnight fasted subjects were used for the study. All the volunteers in the present study were free from any chronic disease or illness and free from use of any tranquillizers, drugs and anesthetics. Blood samples drawn from human volunteers by venipuncture between 7 and 10 a.m. into heparinized test tubes were used immediately for plasma and red cell analysis.
Assay of plasma biochemical profile
Plasma aspartate aminotransferase (AST) and alanine aminotransferase (ALT) enzyme activities, 16 the levels of glucose, 17 triglycerides (TG), 18 total cholesterol (TC) 19 and high-density lipoprotein-cholesterol (HDL-C) 20 were determined by enzymatic colorimetric methods that were performed using appropriate kits (Span diagnostics, Surat, India) according to the manufacturer’s instructions. The concentration of low-density lipoprotein-cholesterol (LDL-C) was calculated according to the formula: LDL-C = (TC − HDL-C) − (TG/5). 21
Measurement of nitrite/nitrates levels
Erythrocyte lysate was prepared as described, 22 plasma and red cell lysate samples were treated with 30% zinc sulfate to deproteinize samples followed by centrifugation at 4000 g for 5 min. Nitrite was determined from 1.0 mL aliquots of plasma and erythrocyte lysate using Griess reagent (1% sulfanilamide, 2.5% phosphoric acid and 0.1% 1-naphthylethylene diamine). One milliliter aliquots of the supernatant were swirled for 90 min separately with activated cadmium granules for the conversion of nitrite to nitrate and then Griess reagent was added. Nitrite concentrations were estimated using a standard curve developed with sodium nitrite. 23
Osmotic fragility of red blood cells
The osmotic fragility test consists of exposing erythrocytes to lysis by osmotic stress to measure their resistance. Human erythrocytes were incubated in different NaCl concentration (0.1%–0.9%) for 30 min at 37°C, centrifuged and the absorbance was determined at 540 nm. The intensity of hemolysis was reported in percentage and plotted against NaCl concentration, in order to construct osmotic fragility curves. 24
Erythrocyte antioxidant enzyme activities
Erythrocytes were washed thrice with 0.9% NaCl and suspended in 1 volume of 0.9% NaCl. The packed cell volume was adjusted to 5% with PBS-pH7.5 (10 mM phosphate buffer saline). Hemoglobin content in erythrocytes was determined. 25 Reduced GSH content was estimated and expressed as µmol/gHb. 26 The SOD activity was measured based on the ability of the enzyme to inhibit the autoxidation of adrenaline and activity was expressed as Units/mg Hb/min. 27 The CAT activity in hemolysate was estimated and the activity of the enzyme was calculated using the extinction coefficient of H2O2 as 0.071 cm−1 mol−1 and expressed as IU × 104/g Hb at 37°C. 28 The GPx activity was measured and the activity was expressed as μmoles of glutathione oxidized/min/mg Hb. 29
Erythrocyte membrane studies
Erythrocyte membrane was prepared, 30 and the classical thiobarbiturate assay was used for the estimation of plasma and erythrocyte membrane MDA levels. 31 Erythrocyte membrane lipids were extracted. 32 To the lysed membrane preparations, 5 mL of methanol was added followed by chloroform. After 30 min, the same was filtered to collect filtrate and the residue was again subjected to same step and filtered again. The filtrates were pooled up and used for lipid analysis. Lipids were extracted from a portion of the erythrocyte membrane suspension with iso-propanol and chloroform and aliquots were taken for estimation of cholesterol 20 and phospholipids, 33 respectively. Erythrocyte membrane protein concentration was estimated. 34
Statistical analyses
Values are expressed as mean ± SD of each group. Student t-test was performed for finding significant difference between the groups. A p < 0.05 was considered statistically significant. Correlations between variables were assessed with Pearson’s correlation coefficients (r).
Results
Table 1 shows the general characteristics and plasma biochemical profile of controls and smokers. Smokers showed significant increase in total/HDL-cholesterol ratio and LDL/HDL-cholesterol ratios compared to controls. The AST and ALT enzyme activities also increased in smokers.
General characteristics and plasma biochemical profile of controls and cigarette smokers
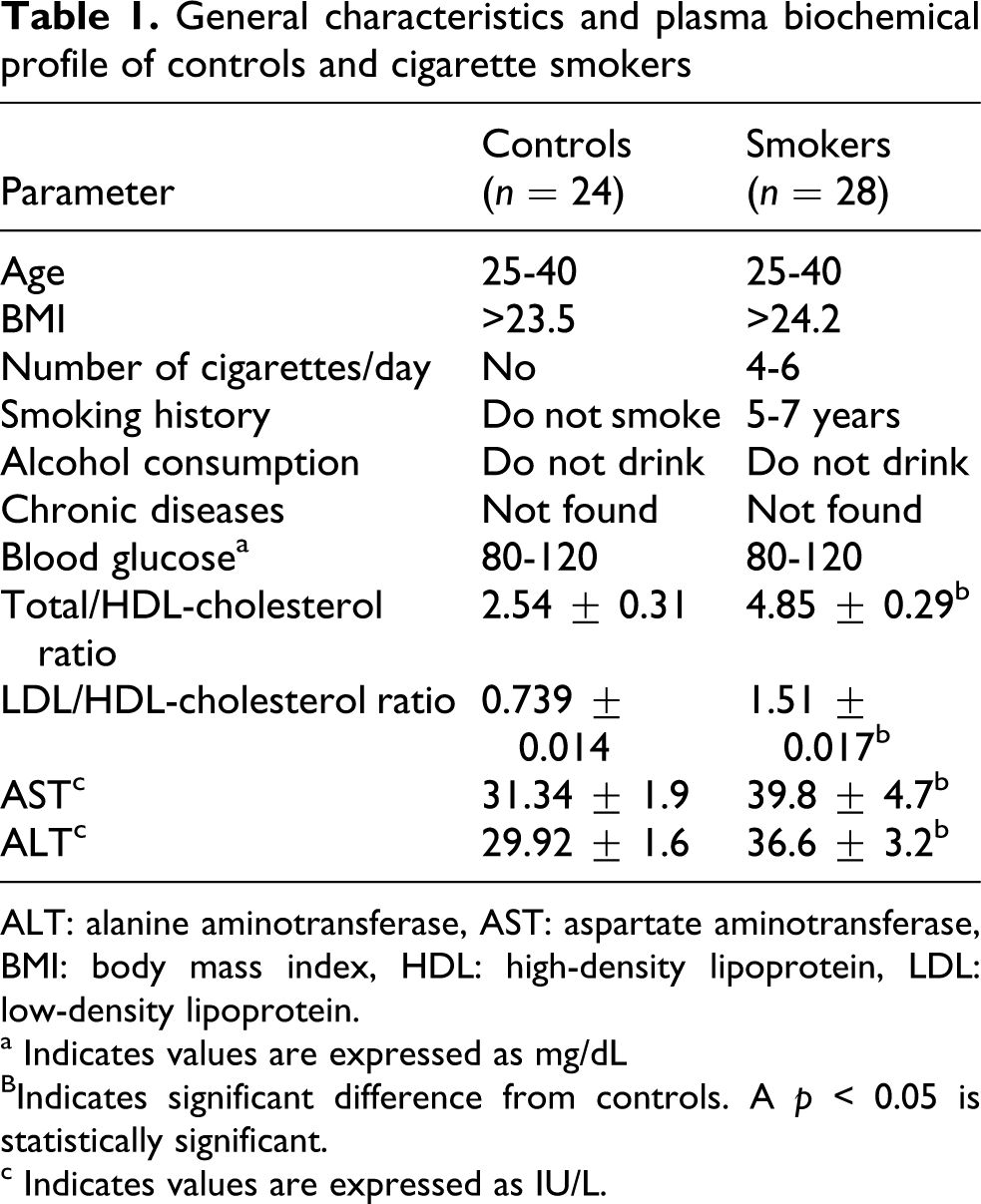
ALT: alanine aminotransferase, AST: aspartate aminotransferase, BMI: body mass index, HDL: high-density lipoprotein, LDL: low-density lipoprotein.
a Indicates values are expressed as mg/dL
BIndicates significant difference from controls. A p < 0.05 is statistically significant.
c Indicates values are expressed as IU/L.
Figure 1 shows increase in plasma and erythrocyte membrane lipid peroxidation in smokers compared to controls. Data presented in Table 2 reveals erythrocyte antioxidant enzyme activities and the content of GSH. In the present study, significant (p < 0.05) hike of erythrocyte CAT, SOD and GPx and decreased level of GSH were observed in smokers. Nitrite and nitrate levels are considered to be reliable indicators of nitric oxide generation. Increased concentrations of nitrite/nitrate in plasma and erythrocyte lysate of smokers were observed when compared to controls (Figure 2 ).

Effect of cigarette smoking on plasma and erythrocyte membrane lipid peroxidation. Values are mean ± SD of each group, *Indicates significant difference from controls. A p < 0.05 is statistically significant.
Effect of cigarette smoking on erythrocyte catalase, SOD, GPx and GSH content in controls and smokers a

GSH: reduced glutathione, GPX: glutathione peroxidase, SOD: superoxide dismutase.
a Values are mean ± SD of each group.
bIndicates significant difference from controls. A p < 0.05 is statistically significant.
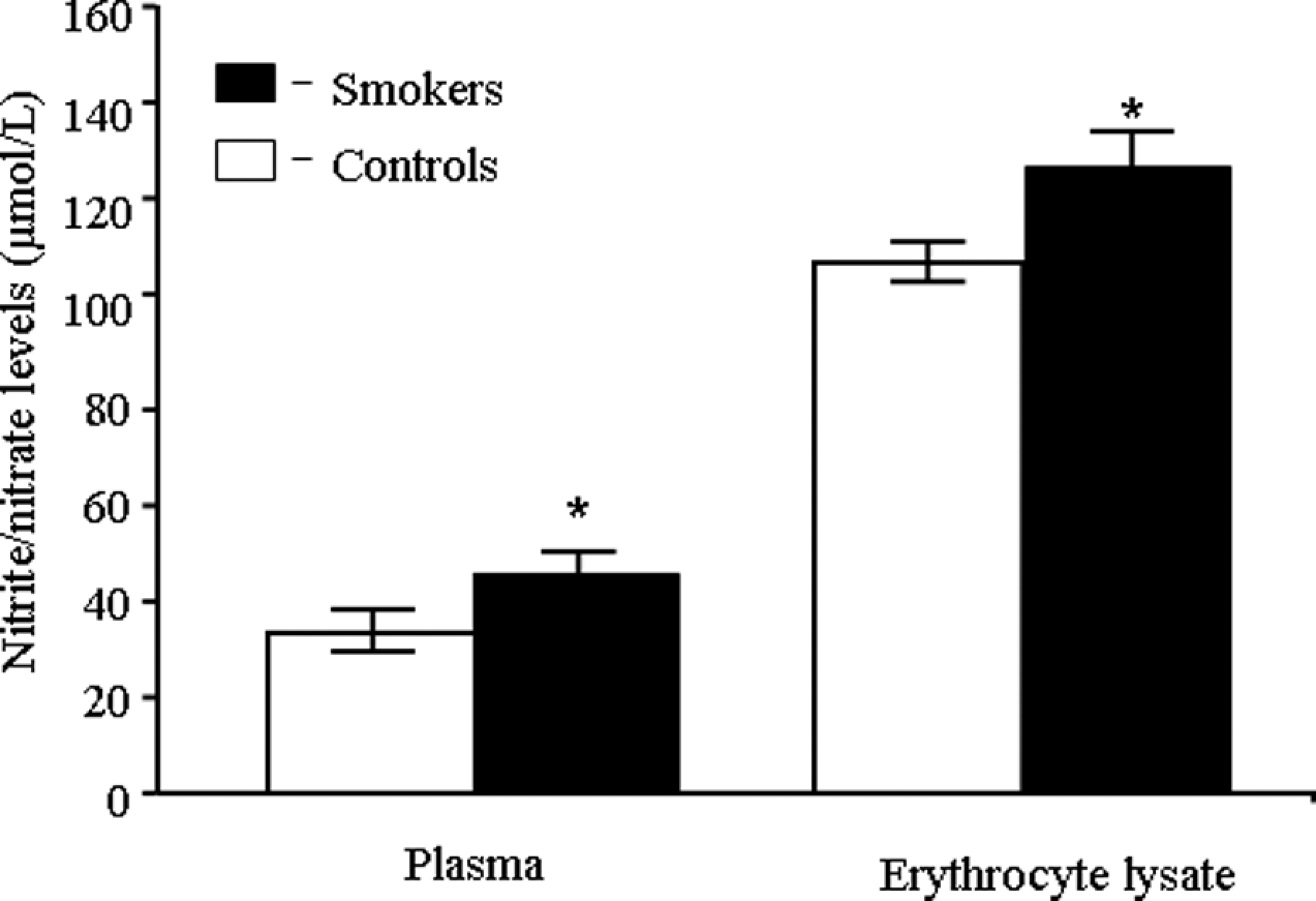
Effect of cigarette smoking on plasma and RBC lysate total nitric oxide (nitrite plus nitrate) production. The total nitrite levels are expressed as µmol/L, values are mean ± SD of each group. *Indicates significant difference from controls. A
The relationship between erythrocyte membrane lipid peroxidation and antioxidant enzymes were determined in smokers using Pearson correlation analysis. Erythrocyte membrane lipid peroxidation was positively correlated with catalase (r = 0.315, p < 0.016), SOD (r = 0.482, p < 0.01), GPx (r = 0.368, p < 0.018) and negatively correlated with GSH (r = −0.292, p < 0.016) in moderate smokers.
The mean values of the percentage hemolysis of both smokers and non-smokers are presented in Figure 3 . Results of the present study reveal that erythrocytes of smokers show no significant change in hemolysis when exposed to different concentrations of NaCl (0.1 to 0.9%) compared to controls. The erythrocyte membrane of smokers showed higher total cholesterol and phospholipid with lower protein content. However, there was no significant change in the consequent cholesterol and phospholipid (C/P) ratio of smokers than to controls (Table 3 ).

Effect of cigarette smoking on red blood cell osmotic fragility. Red blood cells were incubated in different concentrations of NaCl ranging from 0.1% to 0.9% for 30 min and hemoglobin released from red cells was determined in the supernatant. Values are mean ± SD of each group. A
Effect of cigarette smoking on erythrocyte membrane total protein, cholesterol, phospholipids and C/P ratio in controls and smokers a

C/P: cholesterol/phospholipids.
a Values are mean ± SD of each group.
bIndicates significant difference from controls and p < 0.05 is statistically significant. Total erythrocyte membrane protein expressed as mg/dL, total cholesterol and phospholipid values are expressed as μg/mg protein.
Discussion
Smoking is a major lifestyle factor influencing the health of human beings.35,36 It is well established that cigarette smoke induces inflammation to lungs. During inflammation to any organ including lungs, the circulating enzymes ALT and AST levels in the blood usually rise. In the present study, AST and ALT levels are significantly higher compared to controls, but the levels are within the normal range. Chronic cigarette smoking is a recognized risk factor for cardiovascular diseases. The ratio of LDL to HDL cholesterol was more important than individual levels of HDL cholesterol and LDL cholesterol. LDL/HDL is therefore an accurate measure of heart disease. Low ratio indicates lower risk of heart attack, while high ratio indicates higher risk. In the present study, LDL/HDL and total/HDL cholesterol ratio in smokers was significantly higher suggesting that moderate smokers are vulnerable to cardiovascular risk. Previous reports revealed that moderate and chronic cigarette smoking may contribute to the development of atherosclerosis by increasing the activation of circulating monocytes and platelets as well as their aggregation and adhesion to the endothelium.5,37
Free radicals play an important role in the initiation and progression of various diseases. Superoxide radicals (O2•−), hydroxyl radicals (OH2•−), peroxyl radicals, hydrogen peroxides etc are the most common ROS. The nitrogen-derived free radicals are nitric oxide and peroxy nitrite anions.38,39 A single puff of cigarette smoke contains 1014–15 ROS. 40 Gas phase of smoke contains a steady state level of nitrogen, carbon and oxygen-centered radicals and produces esters and peroxyesters of nitrous and nitric acids. The gas phase also causes pulmonary alveolar macrophage to produce more active oxygen species. The tar semiquinone radical reduces oxygen and produces superoxide. 41 In this study, increase in plasma and red cell membrane lipid peroxidation were observed. Lipid peroxidation has been implicated in a number of deleterious effects such as increased membrane rigidity, osmotic fragility, decreased cellular deformability, reduced erythrocyte survival and lipid fluidity. 10 Free radicals produced by cigarette smoke might be responsible for increased plasma and red cell lipid peroxidation. Lipid peroxides formed by cigarette smoking may further enhance the formation of carbon-centered fatty acyl radicals that react with oxygen to form peroxyl radicals. Superoxide and hydroxyl radicals are known to cause marked injuries to the surrounding tissues and organs. Removing superoxide ion and hydroxyl radical is probably one of the most effective defense mechanisms against a variety of diseases. Superoxide radicals are dismutated by SOD and hydroperoxyl radical are removed by catalase. GSH in association with GPx metabolizes hydrogen peroxide to water, thereby protecting mammalian cells against oxidative damage. 42 Earlier studies reveal that heavy and chronic smoking decreases red cell antioxidant status.43,44 Our results shows that decrease in reduced glutathione (GSH) with an increase in the activities of SOD, CAT and GPx were observed in moderate smokers. A positive correlation has been observed between lipid peroxidation and activities of SOD, CAT and GPx of smokers. Since lipid peroxides are substrates for GPx and hydrogen peroxide is substrate for CAT, activities of these enzymes might have increased to scavenge superoxide, lipid peroxides and hydrogen peroxides formed by cigarette smoking. These results suggested protective role of defense enzymes towards oxidative insult induced by cigarette smoking.
In the present study, erythrocyte of smokers does not result in any significant change in hemolysis. However, an increase in plasma and red cell nitrates and nitrites were observed. The individual concentrations of membrane total phospholipid and cholesterol concentrations are changed, overall phospholipid and cholesterol (C/P) ratio was unaltered, and suggesting cigarette smoking induced hemolysis might be suppressed by the changes of physical properties of the membrane. At the same time, the antioxidant role of nitric oxide (NO) may not be ruled out. NO has multiple physico-chemical properties that make it a potentially more effective lipid antioxidant than α-tocopherol. 45 Nitric oxide readily crosses cell membranes and concentrates in lipophilic milieu by virtue of its uncharged character, low molecular mass and relatively high lipid/water partition coefficient.46,47 It reacts to terminate chain propagation reactions catalyzed by lipid alkoxyl and peroxyl radical species, also inhibits free radical-mediated cholesterol peroxidation by forming nitrosated cholesterol. 48 NO is also known as determinant of RBC mechanical behavior and at some critical concentration, NO appears necessary for preserving the cellular mechanical property. 49 NO is effective in modulating membrane fluidity and plays multifaced roles depending on the condition, level of its production and type of oxidative stress that is induced and its functional role can vary between cell type. 50 In previous studies, NO has been shown to ameliorate hemolysis in bone marrow transplantation patients. 51
In conclusion, increased lipid peroxidation, antioxidant status and NO with no significant change in red cell hemolysis suggested that antioxidant enzymes and NO are quenching free radicals produced by smoking and protecting red cells from hemolysis in moderate cigarette smokers. Our study also revealed that moderate smoking at initial stages seems to influence plasma components mainly lipoproteins and very little influence on red cell and organ damage. The underlying mechanisms causing alterations in cholesterol and phospholipids leading to unaltered C/P ratio needs further in-depth study.
Footnotes
The authors declare that there are no conflicts of interest.
Acknowledgements
Authors thank University Grants Commission (UGC), New Delhi, India for providing JNSRF to Pannuru Padmavathi.
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
