Abstract
This study was designed to investigate the electrophysiological, hemodynamic and biochemical parameters of mercuric chloride and methylmercury exposure on cardiovascular functions and its modulation by melatonin in vivo. Wistar albino rats were divided into six group containing 10 animals each. Mercuric chloride (3.75 µM/L) in drinking water and methylmercury (0.5 mg/kg/day) through gavage, given for 1 month, induced a statistically significant increase (p < 0.001) in left ventricular end diastolic pressure, blood and cardiac tissue mercury content and myocardial lipid peroxides compared to control. Significant attenuation (p < 0.05) of baroreflex sensitivity and depletion of myocardial endogenous antioxidants (p < 0.001) viz. Reduced glutathione (GSH) and superoxide dismutase (SOD) were also found in the mercury-exposed groups as compared to control group. Mercury exposure followed by subacute treatment with melatonin (4 µg/mL/day) in drinking water for 1 month significantly lowered (p < 0.01) left ventricular end diastolic pressure and lipid peroxide levels and increased baroreceptor sensitivity (p < 0.001) and also levels of GSH and SOD (p < 0.001) as compared to mercury-exposed rats. The results of our study provide clear evidence that elevated oxidative stress and altered baroreflex mechanisms caused by mercury intoxication may be the contributing factors responsible for impairment of cardiovascular functions and melatonin may exhibit cardioprotective property against subacute heavy metal intoxication and enhance the antioxidant defense against mercury-induced oxidative myocardial injury in rats.
Introduction
Mercury (Hg), a worldwide pollutant transported by air and water throughout the atmosphere, poses a great health risk to global health. It is recognized as one of the most dangerous environmental contaminants. 1 Mercury induces severe alterations in the tissues of humans as well as experimental animals including neurological abnormalities 2,3 and peripheral neuropathy. 4 This has been evidenced by the tragic epidemics in Japan (Minamata disease) and Iraq. 1 Exposure to mercury either as inorganic (mercuric chloride) or organic (methylmercury) form can cause detrimental effects on cardiovascular system, including reduction of arterial blood pressure, 5,6 systemic and pulmonary vasoconstriction 7,8 and reduction in myocardial contractility. 8,9 Mercury can bind with plasma proteins, enter red blood cells and cross blood-brain and placental barriers. 10
Mercury can react reversibly with any thiol group and hence can bind freely to the protein thiol group, thereby damaging DNA, RNA, membrane structures and protein chains present in the cellular machinery. There is no single specific mechanism of toxicity ascribed to mercury but likely involves multiple coordinated effects on several parallel processes in the cell. An imbalance in the antioxidant-protective mechanism leading to oxidative stress in the cells is being identified as a major effect of mercury exposure. Elimination of free radicals by treatment with antioxidants and or free radical scavengers has shown a great reduction in myocardial dysfunction induced by mercury exposure. 11,12 Several studies have reported associations between subacute exposure to mercury and cardiovascular functions; however, manifestations vary from one study to another. In spite of decades of research, the toxic mechanisms of subacute administration of mercury on cardiovascular system still remains an enigma.
Melatonin (N-acetyl-5-methoxytryptamine), a direct free radical scavenger and extracellular antioxidant, which can detoxify numerous reactive oxygen species (ROS) including hydrogen peroxide (H2O2), hydroxyl radical (.OH), peroxyl radicals (ROO.), and singlet oxygen (1O2), and also reactive nitrogen species (RNS) such as nitric oxide radical (NO.) and peroxinitrite (ONOO-), 13–15 is involved in the regulation of many physiological systems, including cardiovascular system. 16,17 Melatonin influences blood pressure and myocardial contractility in spontaneously hypertensive rats 18 and increases the antioxidant reserve. 19 Decreased melatonin levels were reported in various pathological conditions including hypertension with nondipper pattern, 20 impairment of heart failure, 21 ischemic heart disease, 22 or in patients after acute myocardial infarction. 23 Additionally, melatonin's antioxidant actions are derived from its stimulatory effect on superoxide dismutase, glutathione peroxidase, glutathione reductase, and glucose-6-phosphate dehydrogenase and its inhibitory action on nitric oxide synthase. 24 Melatonin in vivo attenuates sympathetic tone by several mechanisms including direct activation of melatonin receptors, scavenging free radicals or increasing NO availability in the central nervous system. 25
Extensive research has been done on effects of melatonin on ischemia-reperfusion injury, circadian rhythms and sleep-wake cycles, but scientific evidence available regarding the role of melatonin against subacute heavy metal intoxication in vivo is insufficient. Also, hemodynamic variability due to subacute administration of inorganic and organic mercury intoxication has not been reported specifically and information existing about mercury intoxication on autonomic nervous system responsible for impairment of cardiovascular regulatory mechanisms is also very scarce. However, results obtained on scientific models with different analytical levels are partially controversial and the mechanisms behind mercury influence on cardiovascular system are still not completely understood. Therefore, this study was designed to evaluate the in vivo effects of melatonin on hemodynamic and oxidative stress parameters in an open–chest anesthetized rat model of mercury intoxication that probes into the possible mechanisms of neural control of arterial blood pressure regulation and cardioprotection offered by melatonin against subacute administration of inorganic and organic mercury.
Material and methods
Chemicals
Mercuric chloride, methylmercury and melatonin were obtained from Sigma chemicals (St. Louis, Missouri, USA). All other chemicals used were of analytical grade. Double distilled water was used for all biochemical assays.
Animals
Study was performed on Wistar Albino rats of either sex weighing between 200 and 250 g procured from the animal house of University College of Medical Sciences (UCMS) and Vallabhbhai Patel Chest Institute (VPCI), University of Delhi, Delhi, and acclimatized under standard laboratory conditions at 25 ± 2°C, 50% ± 15% relative humidity and normal photoperiod (12 h light: dark cycle) for 7 days. Commercial pellet diet (Amrut Feeds Pvt. Ltd., Delhi, India) and water were provided ad libitum.
The study was conducted in compliance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India, and animals were maintained according to the recommendations approved by National Accreditation Board of Testing and Calibration Laboratories (NABL). Ethical clearance for the experimental protocol was obtained from the Institutional Animal Ethical Committee (IAEC), VPCI and UCMS & GTB Hospital, Delhi, India.
Exposure and treatment of animals
The study was conducted in the six groups of animals, each comprising 10 animals. Group I served as normal control in which animals were kept on normal balanced diet and did not receive any form of mercury exposure or melatonin treatment. Group II was melatonin per se group, where animals were administered melatonin (4 µg/mL) in drinking water for 1 month. Group III served as inorganic mercury group, in which animals were given mercuric chloride (3.75 µM / L) in drinking water for 1 month. Group IV was organic mercury group, in which rats were given methyl mercury (0.5 mg/kg /day) through gavage for 1 month. Group V worked as mercuric chloride + melatonin group, where exposure of mercuric chloride (3.75 µM/ L) in drinking water for 1 month was given to the animals as in group III followed by treatment with melatonin (4 µg/mL) in drinking water for 1 month as in group II. Group VI served as methyl mercury + melatonin group, in which exposure of methyl mercury (0.5 mg/kg/day) through gavage for 1 month was given to rats as in group IV, followed by treatment with melatonin (4 µg/mL) in drinking water for 1 month as in group II.
Surgical procedures for recording hemodynamic parameters
The rats were anesthetized with urethane (1 g/kg) given through intraperitoneal route. Tracheotomy was performed to insert endotracheal tubing. Right femoral artery was cannulated and catheter connected to a pressure transducer (Statham-P23D) via three-way stopcock was inserted for recording arterial blood pressure (ABP). Femoral vein cannulation was done for injecting various drugs. Right carotid artery was cannulated and the catheter was advanced to left ventricle for recording left ventricular pressure (LVP). After opening the chest, small animal ventilator was used to ventilate animals (Columbus Instruments, Columbus, OH, USA) and respiratory rate was synchronized with the rat’s spontaneous rate (60−80 strokes/min). The pericardium was then incised and heart was exteriorized.
Perivascular ultrasonic flow probe was placed around ascending aorta for recording cardiac output using a blood flow meter (Transonic, Ithaca, NY, USA). The animals were allowed to stabilize for 30 minutes. Cardiovascular parameters such as arterial blood pressure (ABP), heart rate (HR), LVP, LVP differential (LV[dP/dt]), maximum LVP differential (LV[dP/dt]max), and cardiac output (CO) were recorded on Power Lab data-acquisition system (4SP, AD Instruments, Australia) with a computerized analysis programme (Chart 5.4.2, AD Instruments, Australia). Animals were exposed to mercuric chloride (group III), methylmercury chloride (group IV), and melatonin (group II) subacutely for 1 month and modifications in the above mentioned hemodynamic parameters were recorded after the exposure of animals to both the mercuric forms followed by treatment with melatonin.
Measurement of baroreflex sensitivity
HR was plotted against arterial systolic blood pressure (SBP) to obtain baroreflex curves indicating blood pressure-HR relationship. Baroreflex sensitivity (BRS) was measured by administering 20 µg/mL/kg phenylephrine (vasoconstrictor) and 20 µg/mL/kg sodium nitroprusside (vasodilator) through venous catheter. The resultant changes in HR at corresponding rise or fall in SBP were measured at different time intervals. The relationship between increase in SBP evoked by phenylephrine and associated bradycardia or decrease in SBP evoked by sodium nitroprusside and associated tachycardia were assessed by regression analysis for individual animal. The regression coefficient (slope of regression line), expressed as beats per minute per mm of mercury (beats/min/mmHg), was taken as an index of BRS.
Biochemical assays
After the completion of the experiment, animals were sacrificed under deep anesthesia and blood samples were immediately collected from heart. Serum was separated by centrifugation at 5000 rpm for 10 minutes and stored at −80°C for biochemical analysis. Serum samples were analyzed for the levels of malondialdehyde (MDA) and reduced glutathione (GSH).
MDA concentration in serum samples was determined spectrophotometrically as thiobarbituric acid reactive substance (TBARS) by the method of Wright et al. 26 In a 0.05 mL serum sample, 0.73 mL phosphate buffer (0.1 M, pH 7.4), 0.2 mL ascorbic acid (100 mM), and 0.02 mL ferric chloride (100 mM) were added. Total volume was 1.0 mL. Reaction mixture was incubated at 37°C in a water-bath for 1 h. Reaction was stopped by adding 1.0 mL 10% trichloroacetic acid. Following addition of 1.0 mL 0.67% thiobarbituric acid, all the tubes were placed in boiling water-bath for 20 minutes and then shifted to crushed ice-bath before being centrifuged at 2500 × g for 10 minutes. The amount of malondialdehyde (MDA) formed in each of the samples was assessed by measuring optical density of the supernatant at 535 nm using spectrophotometer (Spectramax 384 molecular device US) against blank using extinction coefficient of 1.56 × 105 M-1cm-1.
GSH was determined as protein-free sulfhydryl compounds by the method of Jollow et al. 27 A total of 100 µL of sample of serum was precipitated with 100 µL of sulphonic acid (4%). The samples kept at 4°C for 1 h were then centrifuged at 1200 × g for 20 minutes at 4°C. The assay mixture contained 0.1 mL filtered aliquot, 2.5 mL phosphate buffer (0.1 M, pH 7.4), and 0.4 mL DTNB (100 mM) in a total volume of 3.0 mL. The yellow color developed was read immediately at 412 nm on a spectrophotometer (Spectramax 384 molecular device US).
For analysis of activity of superoxide dismutase (SOD), clear hemolysate was prepared from blood samples collected from animals, and activity of SOD in each test sample was calculated according to pyrogallol auto-oxidation method as described by Marklund et al.
28
The reaction mixtures were prepared for various concentrations of standards, tests, and controls. The assay mixture of 3 mL volume consisted of 100 μL each of 1 mmol/L EDTA and 1 mmol/L diethylene triamine pentetic acid (DTPA) in air-equilibrated Tris HCl buffer (50 mmol/L; pH 8.2). To this, 100 μL of standards (with varying concentrations of SOD enzyme) or test sera was added. In controls, neither test sample nor standard was added to assay mixture to obtain uninhibited auto-oxidation of pyrogallol. Finally, 100 μL volumes of 0.2 mmol/L pyrogallol were added in all vials to start a reaction. Changes in absorbance at 420 nm were recorded in spectrophotometer at every 10 seconds interval for a period of 4 minutes. The average change in the absorbance per minute was calculated, and the percentage of inhibition in standards and test samples were calculated using the formula:
Estimation of cardiac biochemical markers
Myocardial TBARS, a marker of lipid peroxidation and myocardial endogenous antioxidants, for example, SOD and glutathione, was estimated. A decrease in myocardial endogenous antioxidants with rise in TBARS was considered oxidative stress.
Estimation of mercury levels in whole blood and left ventricular cardiac tissue by cold vapor atomic absorption spectrophotometery
For estimation of mercury in whole blood, samples were withdrawn by direct puncture of cardiac tissue and stored immediately in heparinized vials for further estimations. To determine mercury level in cardiac tissue, post-mitochondrial supernatant (PMS) was prepared by homogenizing and centrifuging left ventricular tissue in chilled iso-osmotic buffer for 20 minutes at 12,000 rpm. Supernatant was carefully collected and immediately stored at −80°C for further analysis. Both PMS and whole blood were subjected to Cold Vapor Atomic Absorption Spectrophotometery (CV-AAS) for determination of mercury content in control, mercury-exposed, and melatonin-treated groups.
Statistical analysis
Results were presented as mean ± standard error of mean (SEM). Student’s t-test was used for un-paired data. Analysis of variance (ANOVA) followed by post hoc Tukey’s test was used for inter-group comparisons. One-way repeated measures of ANOVA, with bonferroni and multiple comparison tests were used for evaluation among all data sets. Acceptable significance was recorded when p values were less than 0.05. BRS was assessed by calculating the regression coefficient from the slope of regression line. Graph pad prism, sigma (3.01) and microsoft excel (version 2000, SR-I Professional) for windows were used for analysis.
Results
Effect of mercury exposure and melatonin treatment on hemodynamic parameters
Subacute administration of both inorganic as well as organic mercury did not result in any significant changes (p > 0.05) in SBP, diastolic blood pressure (DBP), mean arterial pressure (MAP), pulse pressure (PP), HR, and LVP, whereas a significant increase (p < 0.001) in left ventricular end-diastolic pressure (LVEDP) was observed in mercury-exposed rats as compared to control animals (Figures 1, 2 and 6 , Table 1 ). The ratio of first derivative of maximum differential of LVP to that of left ventricular SBP (LVdP/dtmax/P) was significantly decreased (p < 0.001) in metal-exposed animals when compared to control (Table 1). Per se melatonin administration did not show any significant changes (p > 0.05) in the hemodynamic parameters in contrast to mercury exposure groups (Figure 3 , Table 1) but contributed significantly to restoration of reduced LVEDP induced by mercury (p < 0.001) in the experimental animals (Figures 4 and 5 , Table 1).
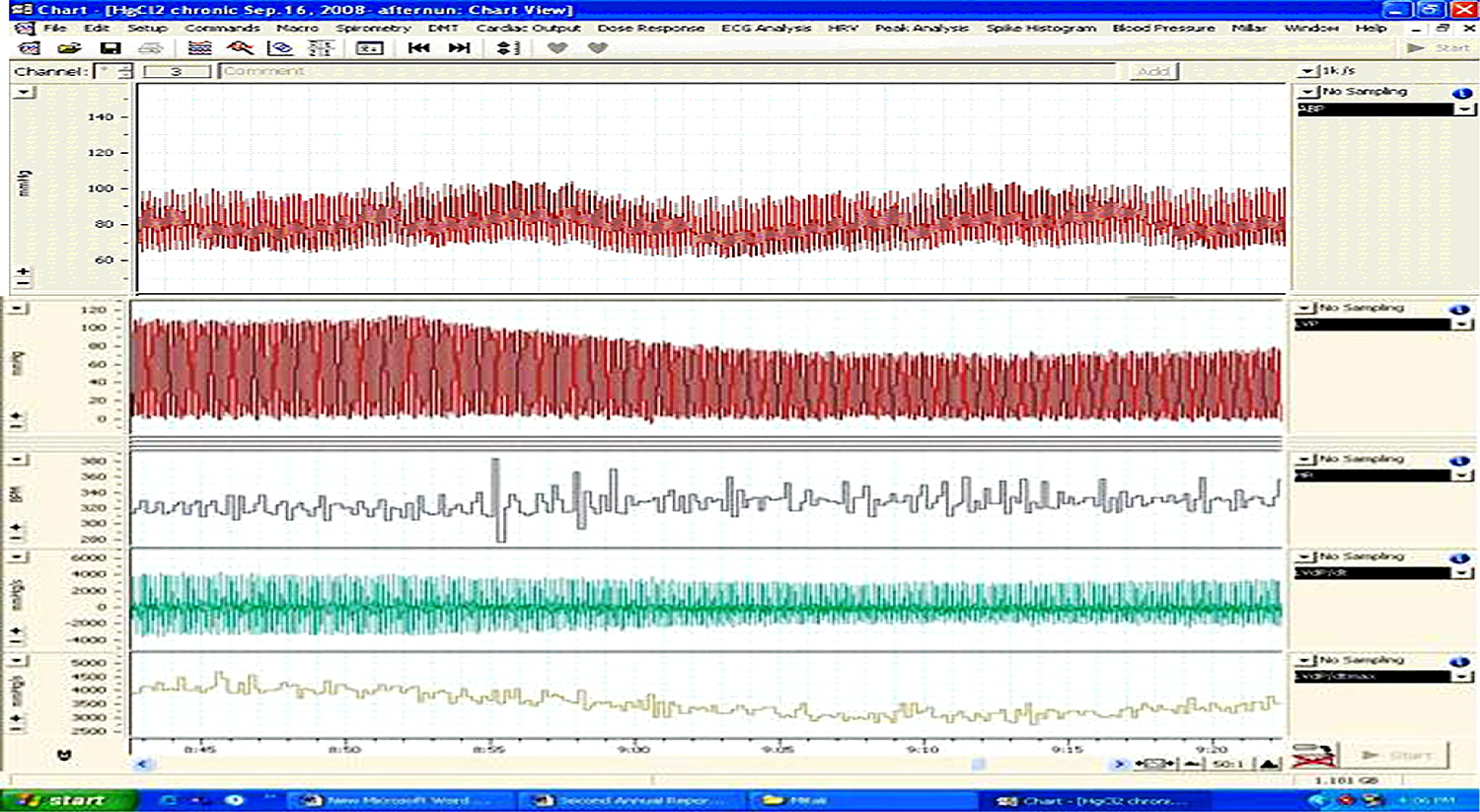
Original tracing of changes in hemodynamic parameters in a rat exposed subacutely to mercuric chloride; ABP: arterial blood pressure, LVP: left ventricular pressure, HR: heart rate, LVdP/dt, LVdP/dtmax.

Original recording of changes in hemodynamic parameters in rat exposed subacutely to methylmercury; ABP: arterial blood pressure, LVP: left ventricular pressure, HR: heart rate, LVdP/dt, LVdP/dtmax
Effect of mercury and melatonin on hemodynamic parameters

Abbreviations: MAP: mean arterial pressure, HR: heart rate, SBP: systolic blood pressure, PP: pulse pressure, LVP: left ventricular pressure, EDP: end-diastolic pressure.
a p < 0.001 vs control.
b p < 0.01 vs HgCl2.
c p < 0.01 vs MeHg.
d p < 0.001 vs HgCl2.
e p < 0.001 vs MeHg.

Original tracing of changes in hemodynamic parameters in a rat exposed subacutely to melatonin; ABP: arterial blood pressure, LVP: left ventricular pressure, HR: heart rate, LVdP/dt, LVdP/dtmax.

Original tracing of changes in arterial blood pressure in a rat exposed subacutely to mercuric chloride followed by subacute treatment with melatonin; ABP: arterial blood pressure, LVP: left ventricular pressure, HR: heart rate, LVdP/dt, LVdP/dtmax

Original tracing of hemodynamic parameters in a rat exposed to subacute methylmercury exposure followed by subacute treatment with melatonin; ABP: arterial blood pressure, LVP: left ventricular pressure, HR: heart rate, LVdP/dt, LVdP/dtmax.
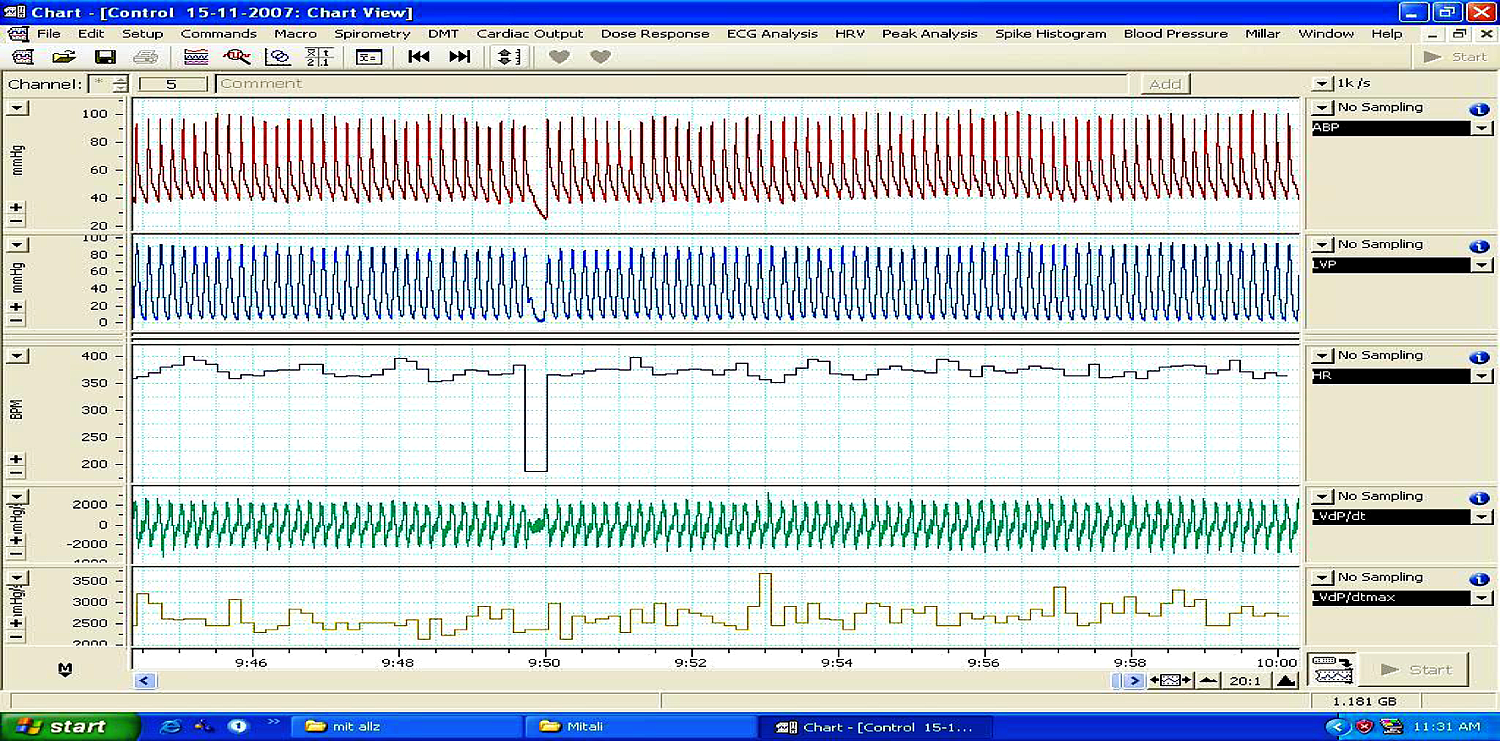
Original representative tracing of hemodynamic parameters in a control animal; ABP: arterial blood pressure, LVP: left ventricular pressure, HR: heart rate, LVdP/dt, LVdP/dtmax.
Effect of mercury exposure and melatonin treatment on vasoconstrictor and vasodilator responses
Significant attenuation of BRS (Table 2 ) was seen after subacute exposure of mercuric chloride (p < 0.05) and methylmercury (p < 0.001) as compared to control group. Mercury exposure in rats decreased the contractile responses along with sensitivity in response to phenylepherine (vasoconstrictor) and sodium nitroprusside (vasodilator). Melatonin per se did not affect (p > 0.05) the BRS but it reversed significantly (p < 0.001) the marked attenuation in BRS caused by mercury exposure in rats (Table 2).
Effect of mercury and melatonin on baroreflex sensitivity (beats/min/mmHg)

a p < 0.05 vs control.
b p < 0.001 vs control.
c p < 0.001 vs HgCl2.
d p < 0.001 vs MeHg.
Effect of mercury exposure and melatonin treatment on oxidative stress parameters
Myocardial LPO was found to be significantly (p < 0.001) higher in the mercury-exposed groups in contrast to control group. Treatment with melatonin significantly decreased the elevated level of LPO (p < 0.001; Table 3 ). Significant decrease (p < 0.001) in levels of myocardial endogenous antioxidants viz. GSH and SOD was seen in the mercury-exposed groups as compared with the control group, and increased significantly (p < 0.001) with melatonin treatment, as compared with the group of animals exposed to mercury either inorganic or organic (Table 3). Melatonin per se group (Group II) did not show any significant (p > 0.05) changes in any of the above-mentioned parameters in as compared with the Group I rats.
Effect of mercury and melatonin on oxidative stress parameters
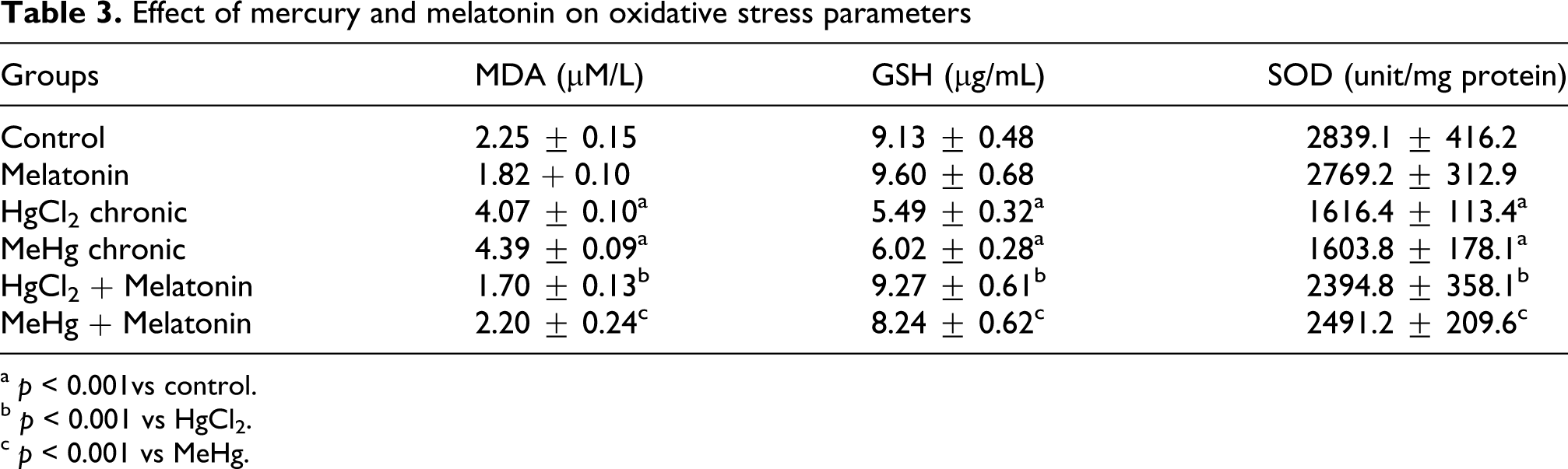
a p < 0.001vs control.
b p < 0.001 vs HgCl2.
c p < 0.001 vs MeHg.
Estimation of mercury levels in whole blood and left ventricular cardiac tissue
Groups I, II, V, and VI did not exhibit mercury content to the detectable limits of CV-AAS, whereas subacute administration of mercury in groups III and IV observed significant elevation of mercury levels in whole blood as well as in the cardiac tissue (Table 4 ).
Accumulation of mercury and melatonin in blood and left ventricular cardiac tissue

Abbreviation: MDL: minimum detection limit.
Discussion
In the present study, long-term administration of both forms of mercury in animals resulted in marked rise in left ventricular end diastolic pressure, indicating ventricular dysfunction. Reduction in first derivative of maximum differential of LVP (LVdP/dtmax) when compared to control indicated impairment of myocardial contractility. This relates to slowing of calcium transients that frequently accompanies cardiac fibrosis 29 and may contribute to the elevation in left ventricular end diastolic pressure. 30 Increased LVEDP indicated an increased preload or incomplete emptying of the left ventricle and impaired left ventricular performance as in myocardial infarction and heart failure. The ratio of first derivative of maximum differential of LVP to that of left ventricular SBP (LVdP/dtmax/P) was significantly decreased in mercury-exposed group indicating functional damage in the left ventricular cardiac tissue. Mercury-exposed groups followed by treatment with melatonin resulted in significant restoration of left ventricular end-diastolic pressure suggesting attenuation of degree of ventricular dysfunction as is supported by other studies. 31,32
The role of baroreflex function is to provide rapid and efficient stabilization of arterial pressure on a beat-to-beat basis by means of strategically located arterial sensors, which are sensitive to high blood pressure. Animals exposed to subacute mercury failed to show corresponding change in the HR to the rise in blood pressure on phenylephrine (PE) slow bolus injection or fall in blood pressure on sodium nitroprusside (SNP) administration suggesting loss of negative feedback mechanism through nucleus tractus solitari. A significant restoration of BRS was seen after treatment with melatonin suggesting repair of impaired arterial blood pressure control. The bradycardia response to rise in blood pressure after phenylepherine administration and the tachycardia response to fall in blood pressure after sodium nitroprusside injection were found normalized in contrast to metal-exposed groups.
The results of CV-AAS suggest that when given subacutely, mercury is taken up by erythrocytes and tissues, has a low clearance rate from the organs, and rate of accumulation of mercury in organs varies directly with the period of administration. Post-mitochondrial supernatant of the left ventricular tissue and blood samples showed the level of accumulated mercury significantly higher than the control group of rats. The reason behind accumulation of mercury in cardiac tissue and blood may be due to its binding to sulfhydryl groups on cysteine, which may compromise the function of enzymes like sarcolemmal Na+ K+-ATPase, and Ca+-ATPase and various ion channels. Mercury has previously been shown to be a potent inhibitor of enzymes, especially that containing sulfhydryl groups. 33 It also impairs the enzymes sulfite oxidase and cysteine dioxygenase interfering with sulfur oxidation, creating a lack of sulfate. It has also been postulated that after absorption into tissues, mercury is slowly oxidized to form divalent inorganic Hg2+ that acts as the activated form and causes accumulation in the target organs and causes toxic effects through binding to sulfhydryl groups in proteins. 34
Rats exposed to mercury followed by treatment with melatonin did not exhibit mercury content under detectable limits indicating heavy metal chelation with the antioxidant leading to a concentration that limits its own detection by CV-AAS.
Free radicals generated by mercury exposure initiate lipid peroxidation of the membrane-bound polyunsaturated fatty acids, leading to impairment of the membrane structural and functional integrity. 10 This concurs with our findings wherein the levels of lipid peroxides were observed significantly increased in animals subjected to mercury exposure.
Although the exact mechanisms of free radical generation are not yet completely understood, it has been presumed that reduced glutathione and SOD depletion triggered the production of ROS that induce lipid, protein, and DNA oxidation. 35 Our results showed that mercury exposure in rats caused a significant depletion in GSH levels and activity of SOD, indicating in vivo generation of free radicals. Our results are in agreement with other studies. 36,37 ROS accumulation induced by subacute concentrations of mercury resulted in cytotoxicity in myocardial and endothelial cell monolayers. 38
Melatonin has been reported to influence the blood pressure in spontaneously hypertensive rats, but no significant effect was observed in normotensive animals. 18 In agreement with these findings, current study exhibited that per se administration of melatonin had insignificant effect on hemodynamic parameters in rats.
Experimental evidence has shown that melatonin, besides its free radical scavenging actions, promotes the activity of GSH-Rd, thereby helping to maintain high levels of reduced GSH. 39 The protective actions of melatonin can also be attributed to the reduction of free-radical-mediated lipid peroxidation and increased antioxidative defense like SOD and catalase. 40 In agreement with these findings, our study showed significant reduction in level of lipid peroxides in animals exposed subacutely to mercury followed by melatonin treatment. GSH levels depleted after mercury exposure were also restored and SOD activity was significantly raised as compared to mercury-exposed groups after melatonin treatment.
Significant restoration of BRS was seen after treatment with melatonin suggesting the repair of neural control of arterial blood pressure regulation, impaired by subacute mercury intoxication as is supported by other studies. 41
Animals following melatonin treatment did not exhibit mercury content under detectable limits despite being exposed to mercury, indicating heavy metal chelation with the antioxidant leading to a concentration that limits its own detection by CV-AAS.
Melatonin’s mechanism of action is unclear; however, the ability of melatonin to decrease free radical generation and to bind with heavy metals may possibly involve the following mechanisms: (a) direct detoxification of free radicals and radical products, (b) stimulation of the activities of antioxidative enzymes, (c) inhibition of the activities of prooxidative enzymes, (d) up-regulation of the GSH-synthesizing enzymes, (e) synergistic actions with endogenous antioxidants, (f) mitochondrial actions of melatonin that reduce oxidative stress, and (g) melatonin as a chelating agent binding heavy metals.
Extent of cardioprotection offered by the melatonin is associated with near normal activity of the diagnostic marker enzymes in serum and significant decrease in levels of lipid peroxides in heart tissues of rats that is indicative of significant cardioprotective effect and myocardial membrane integrity offered by melatonin. In conclusion, present study demonstrated that cardiotoxicity induced by inorganic and organic mercury results due to impairment of hemodynamic parameters, baroreceptor control and oxidative damage in experimental animals. Cardioprotective potential of melatonin is also demonstrated in ameliorating mercury-induced cardiotoxicity through its free radical scavenging cascade, restoration of BRS, thus repairing neural regulatory mechanisms of arterial blood pressure control.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.

