Abstract
The grey seal (Halichoerus grypus) has been part of the Baltic Sea fauna for more than 9000 years and has ever since been subjected to extensive human hunting, particularly during the early phases of its presence in the Baltic Sea, but also in the early 20th century. In order to study their temporal genetic structure and to investigate whether there has been a genetically continuous grey seal population in the Baltic, we generated mitochondrial control region data from skeletal remains from ancient grey seals from the archaeological sites Stora Förvar (Sweden) and Neustadt (Germany) and compared these with modern grey seal data. We found that the majority of the Mesolithic grey seals represent haplotypes that is not found in contemporary grey seals, indicating that the Baltic Sea population went extinct, likely due to human overexploitation and environmental change. We hypothesize that grey seals recolonised the Baltic Sea from the North Sea. during the Bronze Age or Iron Age, and that the contemporary Baltic grey seal population is direct descendants of this recolonisation. Our study highlights the power of biomolecular archaeology to understand the factors that shape contemporary marine diversity.
Introduction
The accelerating loss of biodiversity indicate that we are in the midst of a sixth mass extinction (Barnosky et al., 2011). In previous mass extinctions, loss of wildlife populations and species is often attributed to geological or climatic events, however within the past few centuries, human activities, such as commercial whaling and sealing (Scott Baker and Clapham, 2004), fishery (Dulvy et al., 2003), game hunting (Ripple et al., 2015) and habitat disturbances may be a major contributor to species loss. Overexploitation is far from a recent phenomenon (Keighley et al., 2019; Rodrigues et al., 2016), even in our early prehistory, overexploitation led to the collapse of terrestrial megafauna populations (Sandom et al., 2014), raising the question to what extent prehistoric hunting have affected marine megafauna.
Archaeological evidence demonstrates the wide presence of prehistoric marine hunter-gatherers around the coasts of the Baltic Sea with a life-style dominated by a marine diet, including molluscs, fish and marine mammals. The Baltic Sea is a semi-enclosed brackish water basin that was formed as an icy freshwater lake following the last glacial retreat and has since alternated between being a brackish sea and a freshwater lake (Andrén et al., 2011; Berglund et al., 2005). The different Baltic Sea phases were driven by deglaciation, isostatic rebound and climatic changes, and the Baltic Sea attained its current form some 8000 years ago (Andrén et al., 2011). Five marine mammal species have been recovered from the Baltic Sea zooarchaeological record, grey seal, harbour seal (Phoca vitulina), ringed seal (Pusa hispida), harp seal (Pagophilus groenlandicus) and harbour porpoise (Phocoena phocoena) (Glykou, 2014; Glykou et al., 2021; Olsen et al., 2010, 2016; Schmölcke, 2008; Sommer and Benecke, 2003; Sommer et al., 2008; Storå and Lõugas, 2005). Of these, the harp seal is now extirpated and the Baltic harbour porpoise and harbour seal are threatened species. Similarly, being an abundant species in the 19th century, the grey seal was close to extinction in the 20th century due to the effects of hunting and environmental pollution (Harding et al., 2007). However, with successful conservation actions, the population of grey seals in the Baltic Sea has been restored, currently reaching over 30,000 counted individuals from an estimated 3000 during the 1970s (Harding et al., 2007; HELCOM, 2018). Today, this population size is even considered to be harmful to professional fisheries, exemplified by damage to catch and gear (Olsen et al., 2018). The drastic change in grey seal abundances during the last 100 years, from being a frequently hunted species to almost extirpated, and then abundant again, raises the question on how the presence of grey seal has varied in the Baltic Sea during prehistoric times, and whether such fluctuations were caused by climatic changes, anthropogenic impacts or both.
The grey seal inhabits the Atlantic coasts ranging from western Canada to the Kola Peninsula, Russia (Hall and Russel, 2017). Based on morphological, behavioural and genetic data, two subspecies have been recognised; the Baltic grey seal (Halichoerus grypus grypus) and the Atlantic grey seal (Halichoerus grypus atlantica) (Berta and Churchill, 2012; Fietz et al., 2016; Graves et al., 2009; Klimova et al., 2014; Olsen et al., 2016). Baltic grey sealpups are usually born on ice, although land-breeding populations are known (Hall and Russel, 2017; Hook and Johnels, 1972; Jüssi et al., 2008). The time of breeding for the Baltic population is usually from February to March but the season differs substantially geographically (Coulson, 1981; Hall and Russel, 2017).The pups have a mean lactation period of 18 days followed by a 25 days long post-weaning fast on the ice or on land (Hall and Russel, 2017). In areas where they breed on land, the grey seals have shown to have a high degree of site fidelity often returning to these sites (Pomeroy et al., 1994). Radiocarbon dates of skeletal material from grey seals, retrieved from archaeological sites, show that the grey seal entered the Baltic Sea at least 9000 years ago which place them among the first seal species that immigrated to the Baltic sea (Lindqvist and Possnert, 1997, 1999; Pettersson and Wikell, 2013). As archaeozoological remains show, the species primarily resided in the southern and central Baltic Sea and was uncommon in the northern parts (Lindqvist and Possnert, 1997; Sommer and Benecke, 2003; Ukkonen, 2002).
We shed new light on the history of grey seals in the Baltic Sea using 14C-dating and mitochondrial DNA analysis from two archaeological sites in the Baltic Sea area, Stora Förvar (Sweden) and Neustadt (Germany) and compare them to historic and modern grey seal samples from the same area. Strikingly, while the grey seal is the most common species in the oldest layers at the site of Stora Förvar, they are almost entirely replaced by ringed and harp seals in the zooarchaeological record after a c. 2000 years long hiatus in the cave sequence starting c. 6000 cal BC (see Supplemental Figure S1, available online) (Apel and Storå, 2018, 2020; Apel et al., 2018; Lindqvist and Possnert, 1999; Pira, 1926). The cause of this hiatus and the associated shift in the frequency of grey seal bones is unknown, and could result from both human impacts and environmental change. The complex natural history of the Baltic Sea and the well-known cultural history of the area, provides a perfect natural setup to address questions on the effects of hunting and environment on marine megafauna, and can provide us with important insights on current biodiversity loss in the course of human disturbances and the ongoing global warming.
Materials and methods
Study area and samples
The great extent to which marine mammals were hunted during the Mesolithic period is evident at the Stora Förvar cave on the island of Stora Karlsö, located c. 6.5 km west of Gotland (Figure 1a). Here the cultural layers demonstrate an almost unbroken chronology from the Mesolithic period until today, reflecting changes in both society and environment. The majority of the grey seal bones in the Mesolithic cultural layers (c. 7200–c. 6000 cal BC), at Stora Förvar derives from pups, 0–3 months of age, and sub-adults less than 2–3 years of age, whereas adult specimens are uncommon (Apel and Storå, 2018; Pira, 1926). Young grey seals were thus the primary target, maybe related to the fact that pups do not enter the water during weaning and the post-weaning fast, making them an easily caught prey (Bennett et al., 2007). The high frequency of young specimens has led to the conclusion that Stora Karlsö housed a breeding colony of grey seals during the Mesolithic period (Lindqvist and Possnert, 1999; Pira, 1926).
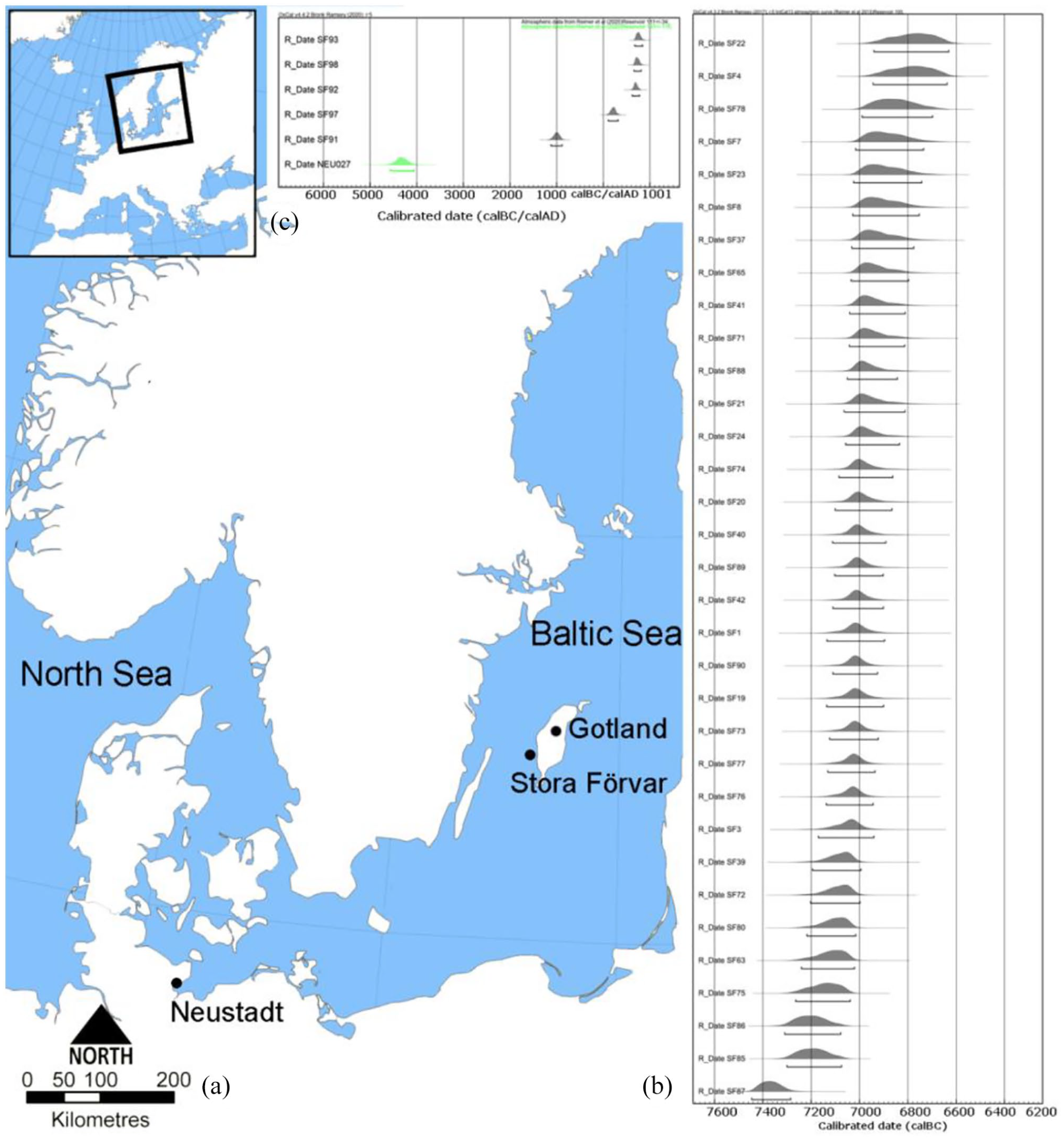
(a) Map of the present Baltic Sea showing the archaeological sites mentioned in the text. (b) Calibrated radiocarbon dates from grey seal bones retrieved from layer F8–F13 in the cave Stora Förvar, Sweden (see Supplemental Table S1, available online) using 100 years as the reservoir effect according to Lindqvist and Possnert (1997 and 1999). (c) Calibrated radiocarbon dates from the younger layers A2, B/F2 and A5 in the cave Stora Förvar, Sweden calibrated with a 111 ± 34 years reservoir effect and the Neustadt (Germany) sample calibrated with a 193 ± 116 years reservoir effect (Glykou et al., 2021). Modified from Wikipedia commons, attribution: Norman Einstein (GFDL (http://www.gnu.org/copyleft/fdl.html) or CC-BY-SA-3.0).
The seal bones from the costal Late Mesolithic-Early Neolithic site Neustadt (Germany) in South-Western Baltic (Figure 1a) date to 4400–3800 cal BC. The zoological finds at the site show a focus on the exploitation of marine resources, mainly seals and fish. The most common seal species found here is the harp seal, followed by the grey seal (Glykou, 2014; Glykou et al., 2021). According to the Minimum Number of Individuals, grey seals are represented by seven individuals. Age determination show the presence of yearlings and adult grey seals (Glykou, 2014).
The selection of bone samples chosen for analysis was based on the temporal distribution of grey seals in the Baltic Sea and the relative chronology of the archaeological contexts included in this study. Sample selection was restricted to the temporal bone because it is one of few skeletal elements in seals that allow an identification to species level, while its high mineral density makes it one of the most frequently found, and best preserved, bone (Hodgetts, 1999; Storå, 2001). In the case of Stora Förvar, the sampled bones (n = 63) represent all Mesolithic archaeological layers evenly. Sample selection from Neustadt (n = 6) was based on the Minimum Number of Individuals (see Supplemental Table S1, available online) (Glykou, 2014).
Radiocarbon dating
Collagen extraction and radiocarbon dating
Collagen extraction was performed on the bones using a modified Longin protocol (Brown et al., 2006), including ultrafiltration, 30 kDa Amicon filters, to remove humic substances and fragmented collagen. A total of 42 collagen samples ranging between 2.1 and 3.5 mg were sent for radiocarbon dating and isotope analysis to the Ångström laboratory at Uppsala University. Radiocarbon dates were calibrated using OxCal v4.4.2 (Ramsey, 2009) using the atmospheric curve, since the Baltic Sea is not a true marine environment (Reimer et al., 2020). All dates are given as cal BC with 2σ.
Reservoir effect
The levels of contemporary 14C in the atmosphere show a global uniformity not seen in marine environments where the levels can vary, depending on for example, environmental factors, usually expressed as the reservoir effect (Ascough et al., 2005; Lougheed et al., 2013). Organisms that incorporate 14C from marine environments can be affected by a reservoir effect making their apparent age look older than they are and this has to be accounted for. The marine reservoir effect in the Baltic Sea varies spatially and temporally since it depends on salinity and regional freshwater run-offs (Lougheed et al., 2013). The reservoir age for the Baltic Proper during the Mesolithic has been estimated to approximately 100 years (Lindqvist and Possnert, 1997, 1999) and this estimate was used for calibration in OxCal v4.4.2. For the Late Mesolithic/Early Neolithic samples from Neustadt, we used a reservoir offset of 193 ± 116 years, and for the Bronze Age/Iron Age samples from Stora Förvar a reservoir offset of 111 ± 34 years was used (Glykou et al., 2021).
δ13C values as indicator of past environment
Finally, the δ13C values were used as an indicator of the environment occupied by past Baltic Sea grey seals, where a less negative value indicates marine conditions and high negative values indicate freshwater conditions (Eriksson and Lidén, 2002; Glykou et al., 2021; Smith et al., 1996).
DNA extraction, library preparation and sequencing
All lab work was performed in the dedicated aDNA facility at the Archaeological Research Laboratory, Stockholm University, using standard methods for aDNA work. The bones were irradiated on two sides with 1 J of UV light in a UV crosslinker and the outer surface of the bone was removed before sampling. Each bone was sampled using an electric drill set at 5000 RPM to remove 110–150 mg bone powder for DNA extraction. DNA extraction was performed in a separate aDNA laboratory using a modified version of protocol C (Yang et al., 1998). For each DNA extraction, a master mix of 2.9 ml EDTA, 30 µl Triton X and 30 µl Proteinase K (10 mg/ml) was added to the bone powder and incubated for 24 h at 38°C. Additional 30 µl Proteinase K was then added to the extracts followed by two additional hours of incubation at 55°C. One extraction blank was added for every five samples. The extract was reduced to 100 µl using Amicon Ultra centrifugal filtres (Merck Millipore, Burlington, USA) and purified using the MinElute PCR Purification Kit using the protocol recommended by the manufacturer (Qiagen, Hilden, Germany).
DNA library preparation and sequencing
Double stranded DNA libraries were prepared using 20 μl of extract, as described in Meyer and Kircher (2010) except for the fragmentation step since aDNA is already fragmented. One library blank and an extraction blank was included for every six extracts. The quantity of DNA in the prepared libraries and blanks was estimated in duplicates using a real time PCR (qPCR). Each reaction-mix of 25 µl consisted of a master-mix of 12.5 µl Maxima SybrGreen, 10.5 µl H2O and 0.5µl each of the primers IS7 and IS8 (10 mM). Two samples, SF10 and SF34, were excluded at this step since the amount of quantifiable DNA was too low. Each library was amplified in five replicates, with one PCR blank added. Each PCR-mix of 25 µl contained 15.25 µl ddH2O, 2.5 µl 10X TaqGold Buffer, 2.5 µl 25 mM MgCl, 0.25 µl dNTPs 0.5 µl IS4, 0.5 µl Index-primer, 0.5 µl AmpliTaq Gold and 3 µl Library DNA. The cycling conditions were 94°C for 12 min followed by 9–16 cycles at 94°C for 30 s, 60°C for 30 s, 72°C for 45 s and a final extension at 72°C for 10 min. Amplified libraries were pooled and purified with AMPure XP beads (Agencourt AMPure XP, Beckman Coulter, Brea, USA). The DNA libraries and PCR blanks were tested on a Qubit using the Qubit® dsDNA HS Assay Kit (ThermoFisher, Waltham, USA). DNA libraries were then quantified on a 2100 Bioanalyzer using the High Sensitivity Kit (Agilent Technologies). A total of 67 libraries were pooled and sequenced on an Illumina HiSeq 2500 platform 2 × 125 base pairs (bp) and an Illumina NovaSeq SPrime 2 × 150 bp at the National Genomics Infrastructure (NGI), Stockholm, Sweden.
Bioinformatic data processing
The raw reads were processed and mapped against the grey seal mitochondrial genome (NCBI accession: NC_001602) using the Paleomix pipeline (v1.2.13.1) (Schubert et al., 2014). The processing of the raw fastq-files included SAM/BAM file manipulation using SamTools (v. 0.1.19) (Li et al., 2009), removal of adapters by AdapterRemoval (v2.2.0) (Schubert et al., 2016) and removal of PCR-duplicates by Picard MarkDuplicates. BAM-files were realigned using GATK Indel realigner, and aligning and mapping using BWA (v0.7.15) (Li and Durbin, 2010). Data authentication was tested by mapDamage (v2.0.6) (Jónsson et al., 2013), which proved that all samples included in the final data analysis have clear signs of deamination, which is typical for ancient DNA (Willerslev and Cooper, 2005). Subsequently, the programme Geneious Prime 2020.1.2 was used to map the realigned bam files to a 635 bp long grey seal reference sequence including the mitochondrial control region, based on which consensus sequences were produced. Samples were hereafter aligned and trimmed down to the 435 bp long control region. This alignment was then trimmed down further to the 350 bp long d-loop, creating a second dataset. This enabled us to explore temporal changes in genetic composition by comparing the ancient Baltic samples to previously published historic (19th century) and modern grey seal mitochondrial control region haplotypes from the Baltic Sea (n = 190) and the North Sea (n = 149) (Fietz et al., 2016). We also compared them with modern d-loop data from grey seals from the North East Atlantic (NEA) (n = 115) and the North West Atlantic (NWA) (n = 19) (Klimova et al., 2014). In the further analysis, we have divided the samples into the following categories: Stora Förvar Mesolithic Period, Stora Förvar Bronze and Iron Age, Neustadt Late Mesolithic/Early Neolithic and finally the Baltic Sea historic and modern samples.
Haplotype networks
The script TempNet in R 3.5.1 (Prost and Anderson, 2011; R Development Core Team, 2018), with the use of the libraries ‘pegas’ (Paradis, 2010) and ‘ape’ (Paradis et al., 2004), was used to produce a control region minimum spanning temporal network with three, time categories. The first phase consists of Mesolithic samples from Stora Förvar, the second phase consists of Mesolithic/Neolithic samples from Neustadt and Bronze Age/Iron Age samples from Stora Förvar and the third phase consists of historic and modern Baltic grey seal samples (Fietz et al., 2016). The TempNet network sort haplotypes based on pairwise differences attributable to mutations, and displays the data in three dimensions so that presence or absence of haplotypes can be followed through time. For comparison with the modern grey seals from the North Sea, the north-west Atlantic Sea and the north-east Atlantic Sea, we created two PopArt minimum spanning networks (Leigh and Bryant, 2015); one in which singletons in the historical and modern samples were identified and, for simplicity one without singletons.
Haplotype diversity and differentiation
The programme DnaSP v. 6.12.03 (Rozas et al., 2003) was used to calculate the number of polymorphic sites (S), number of haplotypes (H), haplotype diversity (Hd) and nucleotide diversity (Pi) for the Baltic data set (ancient and modern). DnaSP was further used to calculate pairwise KST values, in order to estimate the level of genetic differentiation between the grey seal populations at different time periods. The different programmes used (R, PopArt and DnaSP) show some variation in the numbers of haplotypes defined, which is due to differences in the underlying models determining when a sequence is different enough to be regarded as a separate haplotype. Furthermore, the programme DnaSP was unable to read unknown or missing data, hence two such positions were manually excluded beforehand. In the initial 435bp alignment, position 155 was removed due to an R in sample SF83 at this position, and position 176 was removed due to a Y in sample NEU24 at this position (R = either A or G and Y = either C or T). The manual removal of ambiguous positions as well as variation in number of defined haplotypes by the programmes does however not affect the overall results presented in the paper.
Demographic history
A Bayesian Skyline Plot was constructed in BEAST (Bouckaert et al., 2014) for the Baltic Sea samples. This is an estimate of population dynamics based on molecular sequences (Drummond et al., 2005). HKY was used as evolutionary model (Hasegawa et al., 1985) with kappa set at level 50, and an evolutionary clock rate at 0.0001392. The radiocarbon dates were used as tip dates in the analysis. We used the sex identification protocol from Bro-Jørgensen et al. (2021) to investigate if Mesolithic hunters preferentially targeted female or male grey seals.
Results
Radiocarbon dates
The radiocarbon dates of the grey seals from the archaeological deposits confirm the earlier suggestions (Apel et al., 2018; Pira, 1926) of a hiatus in presence of grey seals in the Baltic Sea. The dates for the majority of the Mesolithic grey seals from Stora Förvar fall between 7100 and 6800 cal BC (Figure 1b, see Supplemental Table S1, available online). The oldest Mesolithic specimen (SF87) is dated to 7453–7275 cal BC, whereas the youngest Mesolithic specimen (SF22) is dated to 6942–6625 cal BC. After that grey seals are absent, until they reappear in the younger layers in Stora Förvar that date to the Bronze Age/Iron Age, that is, between 1123–883 cal BC and cal AD 675–844. Only one seal from Neustadt was radiocarbon dated, and the date 4566–4056 cal BC (Figure 1c) is concordant with previous radiocarbon dates from this site (Glykou, 2014) to the Late Mesolithic/Neolithic.
δ13C values
The δ13C values of the Mesolithic Stora Förvar grey seals fall between −14.1‰ and −20.6‰, that is, ranging between marine and freshwater isotope values. However, only two seals yielded marine values, SF80 (−15.7‰) dated to 7230–7011 and SF86 (−14.1‰) 7313–7067 cal BC (Figure 2, see Supplemental Table S1, available online). The majority of the δ13C values (n = 38) fall between −17.9‰ and −20.6‰ indicative of a brackish-freshwater environment (see Supplemental Material, available online for extended discussion on the reservoir effect).
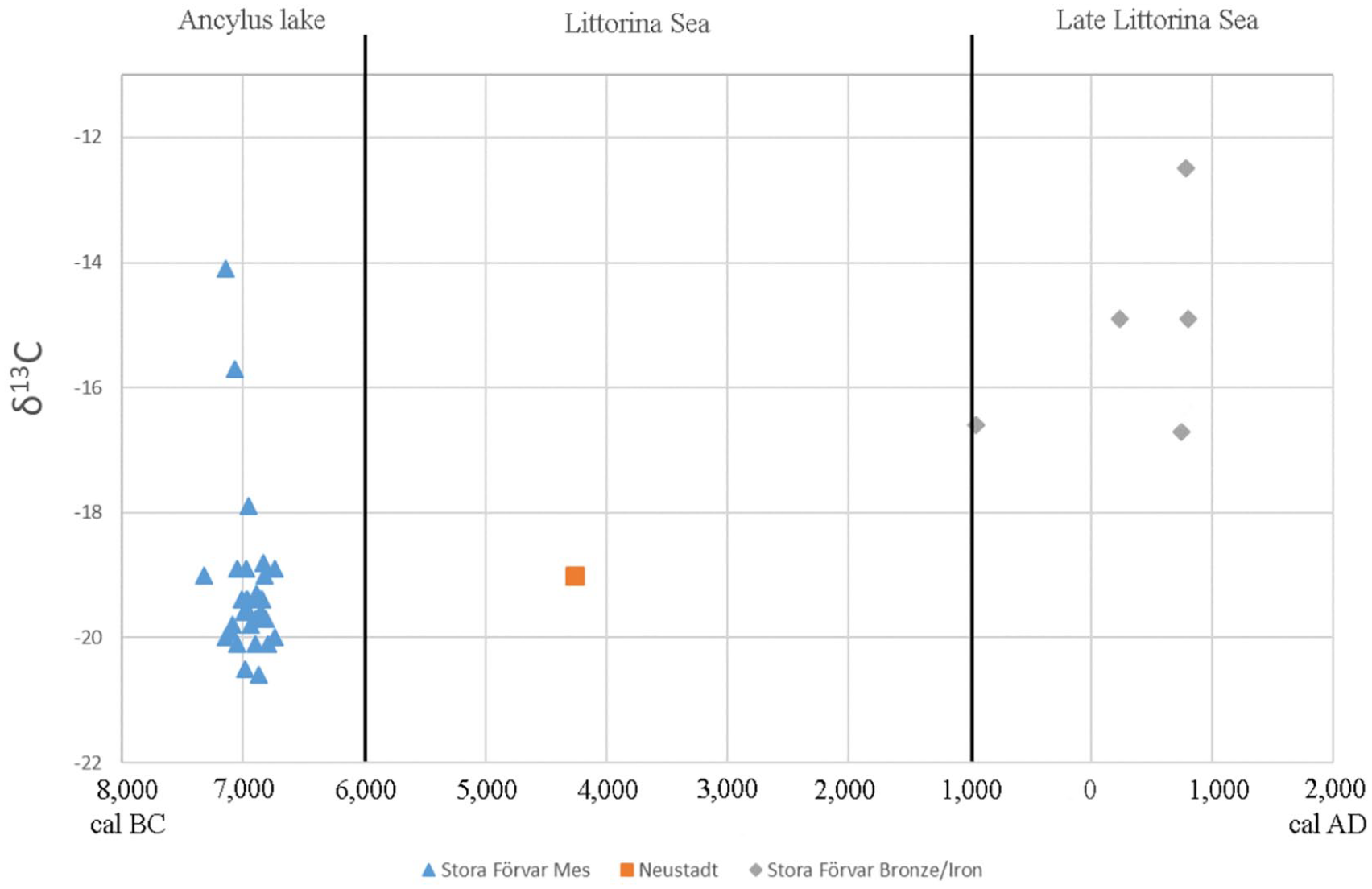
δ13C values for grey seals indicating a shift in the Baltic Sea environment from freshwater-brackish to more marine conditions. The Baltic Sea stages according to Berglund et al. (2005).
Ancient DNA
DNA preservation
The success rate for the DNA-analysis was remarkably high, only four out of the 69 samples were excluded either due to too low amounts of quantifiable DNA or due to a low percentage of mappable reads (see Supplemental Table S1, available online). This is attributable to the exceptional preservation conditions in the Stora Förvar cave. Two additional samples were excluded from further analysis, since they turned out to be other seal species; sample SF35, harp seal and SF94, harbour seal.
Haplotype networks
The temporal haplotype network illustrates that the early Mesolithic grey seals from Stora Förvar comprise 12 haplotypes with 32 of the 51 samples belonging to the same large haplogroup (Figure 3). However, only three of these haplotypes are present in the middle layer, that is, Mesolithic/Early Neolithic samples from Neustadt and Bronze Age/Iron Age samples from Stora Förvar, and only two haplotypes from Mesolithic Stora Förvar are present in the historical and modern Baltic Sea grey seal population, despite our large samples size (190 samples; 55 haplotypes). Strikingly, the main haplotype, represented by 32 grey seals in the Mesolithic Stora Förvar sample, and one from Neustadt, has now been lost. The close connection between the late Bronze Age/Iron Age haplotypes from Stora Förvar and the modern and historical haplotypes in the Baltic Sea populations is apparent from the links between the middle and the top level in the temporal network (Figure 3). On a larger geographic scale, the PopArt network illustrates the genetic relationships between the ancient samples in the Baltic Sea and modern Atlantic seals (Supplemental Figures S2 and S3, available online). This global haplotype network strongly supports that the Mesolithic grey seal haplotypes have been lost The PopArt network also demonstrates that the oldest Baltic Sea haplotypes are more closely related to modern and historic haplotypes from the Baltic Sea than they are to haplotypes from the North-West and North-East Atlantic.
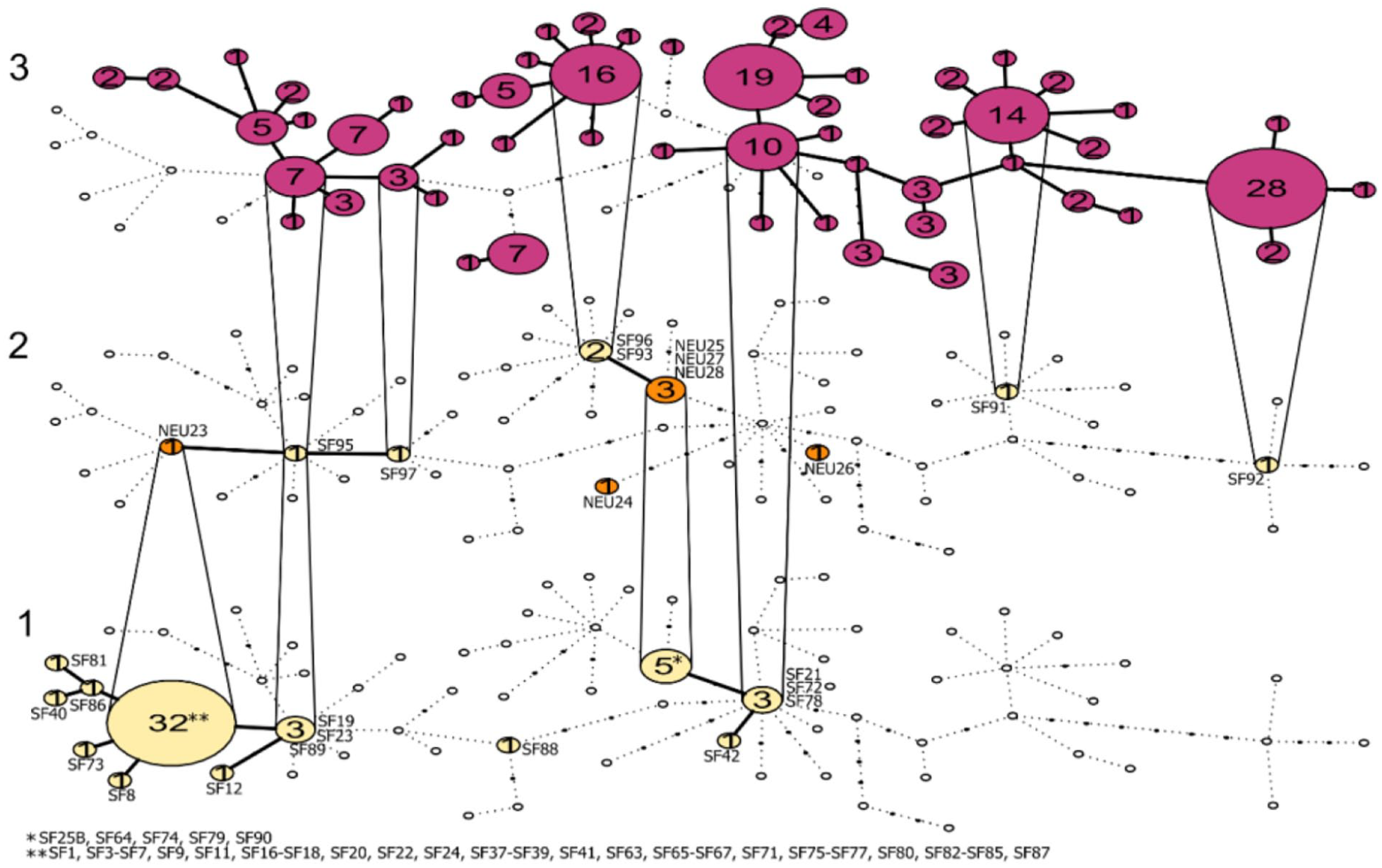
Grey seal haplotypes visualised in a temporal network showing the genetic relationship between the ancient and historic/modern grey seals in the Baltic Sea. (1) Stora Förvar Mesolithic; (2) Neustadt and Stora Förvar Bronze Age/Iron Age; and (3) Historic/modern Baltic grey seals from Fietz et al. (2016). The black dots are missing haplotypes and the white circles are haplotypes that are missing from that particular phase but found in others. Yellow = Stora Förvar, Orange = Neustadt, Purple = Baltic Sea historic/modern.
Genetic diversity and differentiation
The Mesolithic samples from Stora Förvar had a statistically lower haplotype and nucleotide diversity (Hd = 0.598; Pi = 0.00609) compared to all other samples from the Baltic Sea (Figure 4, see Supplemental Table S2, available online). The genetic diversity of the younger samples from Stora Förvar, Neustadt, and the historic and modern samples was overall similar and not significantly different.
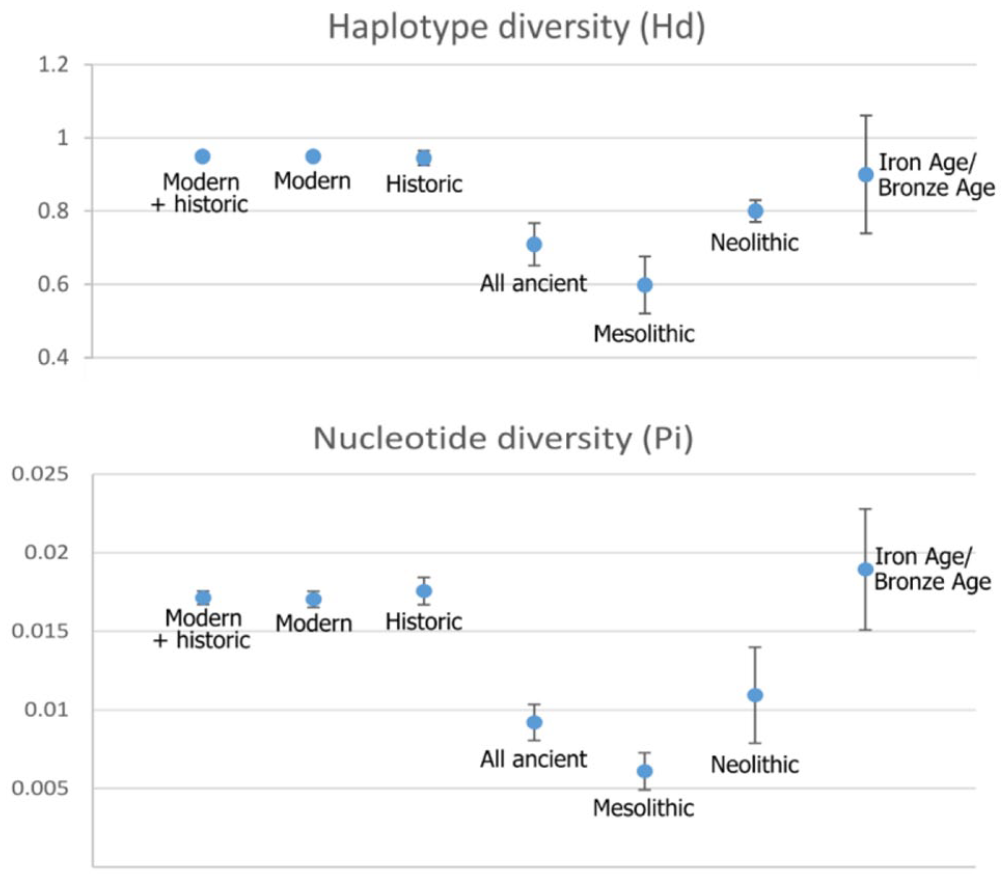
Haplotype diversity (Hd, above) and nucleotide diversity (Pi, below) as calculated in dnaSP from Baltic grey seals. The error bars show the standard deviation (SD) (see Supplemental Table S2, available online).
In support of the haplotype networks, the pairwise estimates of genetic differentiation support the genetic uniqueness of the early Mesolithic samples from Stora Förvar (see Supplemental Table S3, available online). Further, the Late Mesolithic/Early Neolithic samples from Neustadt are not genetically different from the Bronze Age/Iron Age samples from Stora Förvar, but they differ significantly from all historic/modern samples. In contrast, the Bronze Age/Iron Age samples from Stora Förvar are not significantly genetically differentiated from the modern grey seals.
Demographic history
The Bayesian Skyline Plot suggests a low effective population size during the Mesolithic Stora Förvar phase and an increase during the Bronze Age/Iron Age period, which could reflect an immigration of animals to the Baltic Sea (see Supplemental Figure S4, available online). Also visible is the decrease in effective population size during the intensive hunt at the first half of the 20th century, that almost drove the Baltic grey seal to extirpation. We found no difference in the number of males versus females in the archaeological samples, neither between the different phases nor between sites (older stages at Stora Förvar: 29 females, 22 males, younger stages at Stora Förvar: four females, three males, Neustadt: three females, three males) (see Supplemental Table S1, available online). Consequently, hunting does not seem to have been biased towards a certain sex.
Discussion
The radiocarbon dates and ancient DNA analyses shed new light on the fascinating history of the Baltic Sea grey seal. We find that grey seals were present around Gotland as early as 7453–7275 cal BC, which place them among the first immigrants into the young Baltic Sea. However, grey seals gradually disappear in the zoological material from Stora Förvar already c. 6600 cal BC. Strikingly, our comparison of DNA from Mesolithic, Bronze Age/Iron Age, historical and modern Baltic Sea grey seal populations indicate that the disappearance from the zooarchaeological record is associated with a dramatic loss of mtDNA haplotypes, with only three out of 15 ancient haplotypes present in the historical and modern Baltic Sea grey seal population. It is plausible that a small breeding population survived in the Baltic sea, but given the loss of haplotypes we find it more likely that the absence of grey seals reflects a local extinction during the Mesolithic with the reappearance in the Bronze Age/Iron Age resulting from immigration. From this time-point onwards, the Baltic grey seal population show a genetic continuity and uniqueness from grey seals in the North Sea lasting until today (Fietz et al., 2016; Klimova et al., 2014).
The disappearance of a genetic unique grey seal lineage during the Mesolithic coincides with what seems to be the most intensive phase of sealing by hunter-gatherer communities around the island that lasted for 300 years, between 7100 and 6800 cal BC (Figure 1b). This confirms the observations made by Pira already in 1926 (Pira, 1926), who suggested that grey seals in this area during the Mesolithic period, were hunted at a non-sustainable rate that eventually led to local extinctions. Interestingly, humans utilised the cave for yet another 500 years after the grey seals disappeared from the site, as shown by a series of radiocarbon dates published by Apel et al. (2018). The radiocarbon dates in our study also indicate that the grey seal subsequently was rare in the Baltic, that is, between the Neolithic and the Bronze Age (Ukkonen, 2002) (Figure 1c).
In addition to hunting, the grey seals disappear in connection to a number of environmental events during the Mesolithic period. One being the so-called 8.2 kyr BP event, which had a major impact on all life in and around the Baltic Sea. This was a short period lasting for about 150 years (Kobashi et al., 2007) of cold and dry winters in the Northern hemisphere at 6200 cal BC (Alley and Ágústsdóttir, 2005; Alley et al., 1997; Mayewski et al., 2004). However, our data show that the grey seals disappear from Stora Karlsö already 6600 cal BC, some 400 years before the 8.2 kyr BP event. Indeed, although our radiocarbon dates show that the grey seal disappeared from the Gotland area earlier than was previously assumed, we cannot exclude that other environmental events were the final or contributing cause of their disappearance. The fluctuating sea levels, also known as the Littorina transgressions, starting around 6500–6200 cal BC (L1 transgression), with a maximum sea level c. 5800–4200 cal BC, and with the last transgression c. 3200–3000 cal BC caused changes in the salinity in the Baltic basin (Berglund et al., 2005). The change in sea salinity, as the freshwater in the Baltic basin turns to a brackish sea, affected the primary production, which in turn could have caused cascade effects on the grey seal population. It has, for example, been suggested that environmental factors and associated trophic bottom up effects might be more important than hunting in regulation of marine mammal populations (Baylis et al., 2015).This may have been a contributing factor to why the grey seal disappeared.
The δ13C values of the Mesolithic Stora Förvar grey seals range between −14.1‰ and −20.6‰, but the majority of the values ranges between −17.9‰ and −20.6‰ which suggests a brackish or even freshwater environment, implying that the influx of salt water to the Baltic was very low. These δ13C values are similar to previous studies on Baltic grey seals from the same period (Apel et al., 2018; Eriksson and Lidén, 2002). Of certain interest are the two samples with deviating, more marine δ13C values (−14.1‰, −15.7‰), suggesting that these seals either originate from or foraged in a marine environment for example, in the Kattegat or North Sea. Intriguingly, the two seals, SF80 (−15.7‰) and SF86 (−14.1‰), date to 7230–7011 and 7313–7067 cal BC respectively (Figure 2, see Supplemental Table S1, available online). It is however, possible that the reservoir effect for these two specimens are higher than the 100 years applied to other seals which had more depleted δ13C values, considering their more marine isotope values. Therefore, we applied a 400 years reservoir effect on these specimens, as suggested for marine materials from the region of Kattegat, which gave the dates 6897–6653 (SF80) and 6991–6766 cal BC (SF86) that would place these seals among the younger grey seals in the Mesolithic Stora Förvar zooarchaeological material (see Supplemental Figure S5, available online). The samples, one male (SF86) and one female (SF80), do not deviate genetically from the other early Mesolithic seals from Stora Förvar. Considering that grey seals move over large areas, but have a strong site fidelity suggests that their marine isotope signatures result from foraging movements rather than immigration. Given their radiocarbon dates close to the disappearance of grey seals at Stora Förvar, it is tempting to speculate whether such movements were driven by environmental changes in the Baltic Sea marine ecosystem. Another issue that arises here is whether these seals were living in the freshwater Ancylus lake or in the brackish Littorina Sea, as the timing of the influx of marine water to the Baltic basin is strongly debated (Bennike et al., 2021; Berglund et al., 2005).
The low genetic diversity and supposedly genetic isolation of Mesolithic grey seals at Stora Förvar could have made them sensitive to extensive hunting and rapid changes in the environment. Such changes may include collapse of prey populations, alterations of breeding habitat or introduction of competitors, predators or novel pathogens. Human harvest overexploitation of wild populations is a known cause of loss of genetic variation (Allendorf et al., 2008), and it is increasingly clear that even early human exploitation could be on a scale causing substantial impacts of mammalian populations and species for example, Keighley et al. (2019). Hunters prey choice can determine the effect hunting has on a population (Caughley, 1978) and the seal hunters at Stora Förvar have been primarily targeting young grey seals of both sexes (29 females and 22 males). This could have had detrimental effects on the grey seal population since many large mammals are especially sensitive to juvenile mortality (Gaillard et al., 1998). Grey seal females show strong fidelity to their breeding sites (Karlsson et al., 2005), and studies on the seal culling programme in Denmark in the late 19th and early 20th century showed that grey seals in comparison to the sympatric harbour seal is sensitive to intensive hunting pressure, became locally extinct and only very slowly re-established lost breeding colonies (Galatius et al., 2021; Olsen et al., 2018). Thus, if hunting and/or environmental change caused the disappearance of Baltic grey seals during the Mesolithic, it might have taken a substantial immigration of animals to re-establish the Baltic Sea grey seal population.
Conclusions
Our radiocarbon and aDNA analyses of zooarchaeological material suggest that grey seals appeared around Gotland as early as 7400 cal BC, but then disappeared c. 6600 cal BC. The loss of haplotypes and strong genetic differentiation between the Mesolithic samples and samples from subsequent periods, strongly suggest that the majority of the early grey seal colonisers of the Baltic Sea were extirpated. Our genetic data further suggest that their reappearance to Stora Förvar during the Bronze Age/Iron Age is the result of an immigration event of animals either completely recolonising the region or mixing with a small population of animals that survived and bred during the intermediate period. From this immigration, we found genetic continuity in the Baltic grey seal population until modern time. We find it likely that a combination of intensive hunting and environmental events contributed to the disappearance of the grey seal from the Baltic Sea during the early Mesolithic period, potentially providing one of the earliest examples of wildlife population collapse due to human overexploitation.
Supplemental Material
sj-docx-1-hol-10.1177_09596836221080764 – Supplemental material for The Baltic grey seal: A 9000-year history of presence and absence
Supplemental material, sj-docx-1-hol-10.1177_09596836221080764 for The Baltic grey seal: A 9000-year history of presence and absence by Hans Ahlgren, Maiken Hemme Bro-Jørgensen, Aikaterini Glykou, Ulrich Schmölcke, Anders Angerbjörn, Morten Tange Olsen and Kerstin Lidén in The Holocene
Footnotes
Acknowledgements
The skeletal material in this study has been provided by the museum collections at the Swedish History Museum, Stockholm, Sweden and the Archaeological State Museum Schloss-Gottorf, Schleswig-Holstein, Germany. We would like to thank Jan Storå, Osteoarchaeological Research Laboratory, Stockholm University for the osteological analysis of the Stora Förvar material. We would also like to thank SNIC and Uppsala Multidisciplinary Center for Advanced Computational Science (UPPMAX) for resources provided for the projects delivery02388, delivery00942.
Authors’ contributions
HA, MHBJ, AG, AA, MTO, KL participated in the design of the study and drafted the manuscript. HA and MHBJ carried out the molecular lab work. MHBJ carried out sequence alignments and the statistical analyses. US provided zoological material from Neustadt and helped draft the manuscript. All authors discussed results, reviewed the manuscript text and gave final approval for publication.
Data accessibility
The datasets supporting this article are accessible via the electronic supplementary material. The mtDNA haplotypes are uploaded in GenBank under Accession nos. MZ354058-MZ354120.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union’s EU Framework Programme for Research and Innovation Horizon 2020 under Marie Curie Actions Grant Agreement No 676154 as part of ‘ArchSci2020’; and the Swedish Research Council, Sweden [grant number 2015-02151].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
