Abstract
It is a common assumption that – compared with the Mesolithic – the adoption of Neolithic lifeways was accompanied by a higher risk of infection and the development of epidemic diseases. Such a hypothesis seems plausible when considering singular archaeological parameters like increasing population density and palaeopathological indicators of poor health. However, evidence for the far-reaching consequences of epidemics has not yet been examined. Thus, the relevance of infectious diseases as triggers for transformation processes in the Neolithic remains to be identified. By reviewing specific archaeological, genetic and palaeopathological proxies gained from groups of individuals that inhabited the German loess zone from the early to the final Neolithic, we provide a diachronic view of the periods between 5500 and 2500 BCE with regard to postulated indicators of epidemic events. Our analyses of the archaeological proxies suggest major transformations in domestic strategies and mortuary practices, especially in the middle and late Neolithic. Interestingly, mass burials indicative of epidemic events are lacking. Ancient DNA results on pathogens confirm single infections throughout the Neolithic, but there is no clear evidence for diseases of epidemic proportions. The osteological records are not conclusive since the majority of osseous changes are unspecific with regard to the cause and course of infections. We conclude that currently neither biological nor archaeological proxies suggest substantial contributions of epidemics to Neolithic transformations in the German loess zone. This finding contrasts with the general assumption of a higher risk of infection and the development of epidemic diseases during the Neolithic.
Keywords
Introduction
Major epidemiological and demographic transitions in human history have been associated with specific changes in subsistence, social organization, technology and medicine (Barrett et al., 1998; Omran, 1971). When the Neolithic lifestyle arrived in Central Europe about 7500 years ago, a major epidemiological and demographic transition began (Armelagos et al., 1991; Bocquet-Appel and Bar-Yosef, 2008; Bocquet-Appel and Dubouloz, 2004; Gronenborn and Petrasch, 2010). The Neolithic lifestyle introduced more sedentary subsistence strategies involving the use of plant and animal domesticates as a dietary resource and the accelerating creation of anthropogenic landscapes and residential areas (Kristiansen, 1982; Midgley, 1992; Müller, 2001, 2011). Furthermore, this period was characterized by a general increase in population size and agglomeration (Shennan, 2018; Whittle, 2018; Zimmermann, 1996). It has been hypothesized that the Neolithic transition was accompanied by a rise of infectious diseases compared with Mesolithic hunter-gatherers due to substantial changes in the human ecology, influencing, for instance, demography, housing conditions, hygiene and diet (Barrett et al., 1998). This hypothesis has traditionally been supported with the following arguments: (1) Infectious agents require a certain concentration of hosts to thrive. The living conditions in many agricultural communities were crowded compared with Mesolithic times; thus, pathogens were able to spread easily, affecting entire groups and ultimately reaching endemic levels. (2) Cohabitation with animals allowed the transmission of pathogens to humans. (3) Contaminated water sources and close contact with human/animal faeces and waste also facilitated infection. (4) Unbalanced diets and physical stress weakened individuals, and consequently reduced their resistance to infections that could further aggravate nutritional deficiencies. Infants, the elderly, the malnourished and those not previously exposed to pathogens were at a particularly greater risk (Armelagos et al., 1991; Larsen, 2006). Overall, infections are supposed to have brought about an unprecedented disease load, morbidity and mortality to the Neolithic farmers (Barrett et al., 1998; Lambin et al., 2010; Shchelkunov, 2011).
Recent fine chronological dating and demographic modelling indicate drastic fluctuations after 5000 BCE (early Neolithic), showing severe population decline between 3000 and 2500 BCE (final Neolithic) (Hinz et al., 2012; Müller, 2015; Müller and Diachenko, 2019; Shennan and Edinborough, 2007). These collapses were not linked to extreme climatic conditions, as has previously been suggested (Shennan, 2013). They did, however, coincide with sociocultural changes as reflected in the emergence of different archaeological groups. Supportive evidence from ancient DNA (aDNA) studies suggests shifts of genetic patterns in the population at similar time points (Allentoft et al., 2015; Brandt et al., 2013; Haak et al., 2015) and may therefore be connected with the demographic declines mentioned above (Kristiansen and Larsson, 2005). The reasons for these changes still remain unknown, but it is tempting to speculate that they were driven or at least facilitated by infectious diseases of epidemic proportions (Rascovan et al., 2019).
Epidemics are caused by pathogens of bacterial, viral, fungal or parasitic origin that harm their hosts and readily spread from person to person. Whether individuals succumb to a disease depends largely on their health status as well as the size of the infectious load and the virulence of a particular strain. Today, an epidemic is declared when the observed incidence reaches a defined threshold above the known baseline rate (Green et al., 2002). Since such quantitative epidemiological figures do not exist for prehistoric periods, we explore archaeological archives to identify potential consequences of acute infectious outbreaks that affected many people in a relatively short period of time, and to provide insights into the living conditions of the respective populations. Therefore, we use the following archaeological and bioarchaeological methods and parameters: (1) Housing conditions are used to reconstruct changes in socio-environmental interactions that might have facilitated the origin and transmission of infectious diseases; (2) changes in burial practices are considered as a socio-ritual response to epidemic events, including exceptional findings of mass burials or frequent inhumations in rapid succession; (3) by means of aDNA methods, infectious agents are detectable in skeletal remains, and virulence can be assessed by genomic analyses; and (4) palaeopathological examination of human bones can determine the presence and frequency of lesions, some of which are specific for certain infections, and can provide insights into the health status at the individual and population levels.
We concentrate on a geographic transect that ranges from the German Alps to the Harz Mountains and from the Rhine to the Elbe river, covering the major loess areas in Germany (Figure 1). Their fertile arable land favoured the Neolithic lifestyle and led to a high diversity in archaeological entities and their associated markers (Müller, 2009). Here, we consider a time frame between 5500 and 2500 BCE, ranging from the early to the final Neolithic (Table 1).
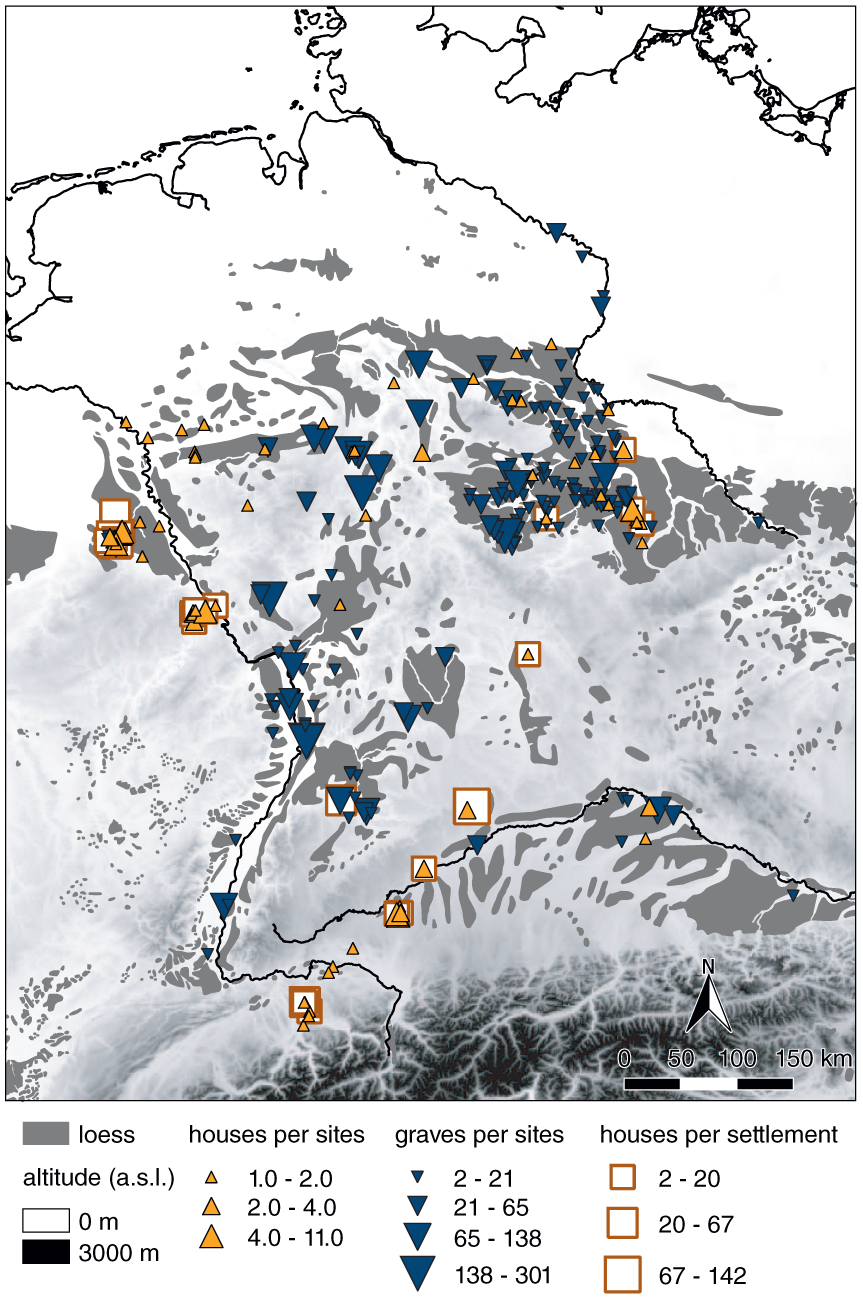
Area of study (based on Haase et al., 2007). Sites with archaeological proxies used in the statistical analyses (results shown in Figure 2).
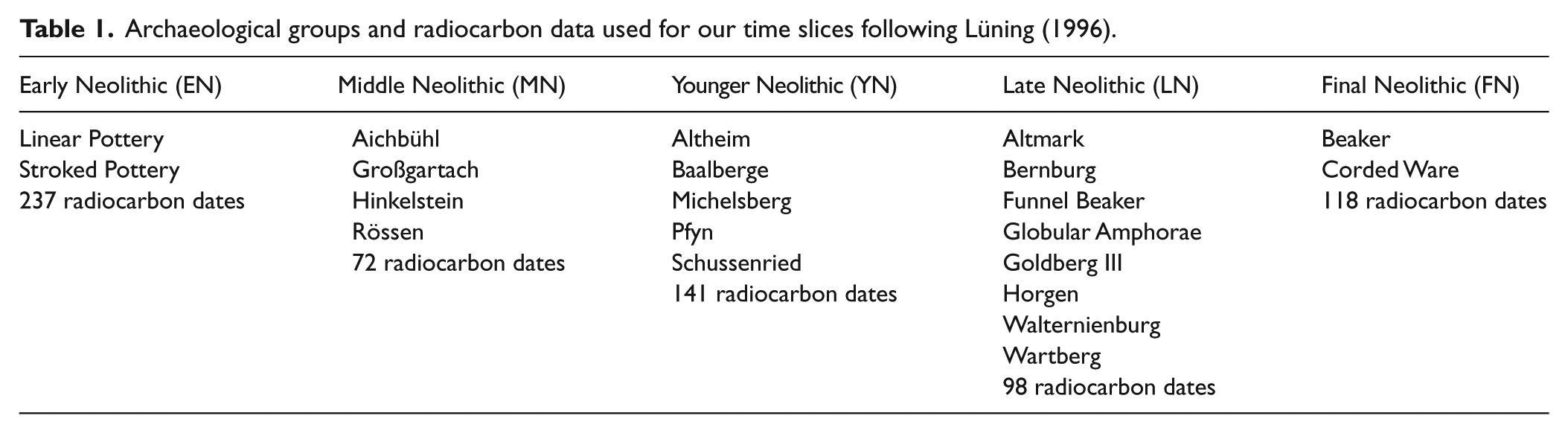
Archaeological groups and radiocarbon data used for our time slices following Lüning (1996).
Archaeological evidence for epidemics
Past living conditions that are relevant for the reconstruction of epidemics such as population density, breeding strategies or cohabitation with animals cannot be assessed directly by primary archaeological data. However, we expect changes in mortuary practices and domestic strategies to reflect major transformations as a potential consequence of epidemics. As proxies, we use available primary data, namely, house size, number of houses per settlement and number of inhumations per cemetery or number of burials, respectively.
Five time slices of approximately equal length were defined based on related archaeological groups (Table 1, Figure 2):
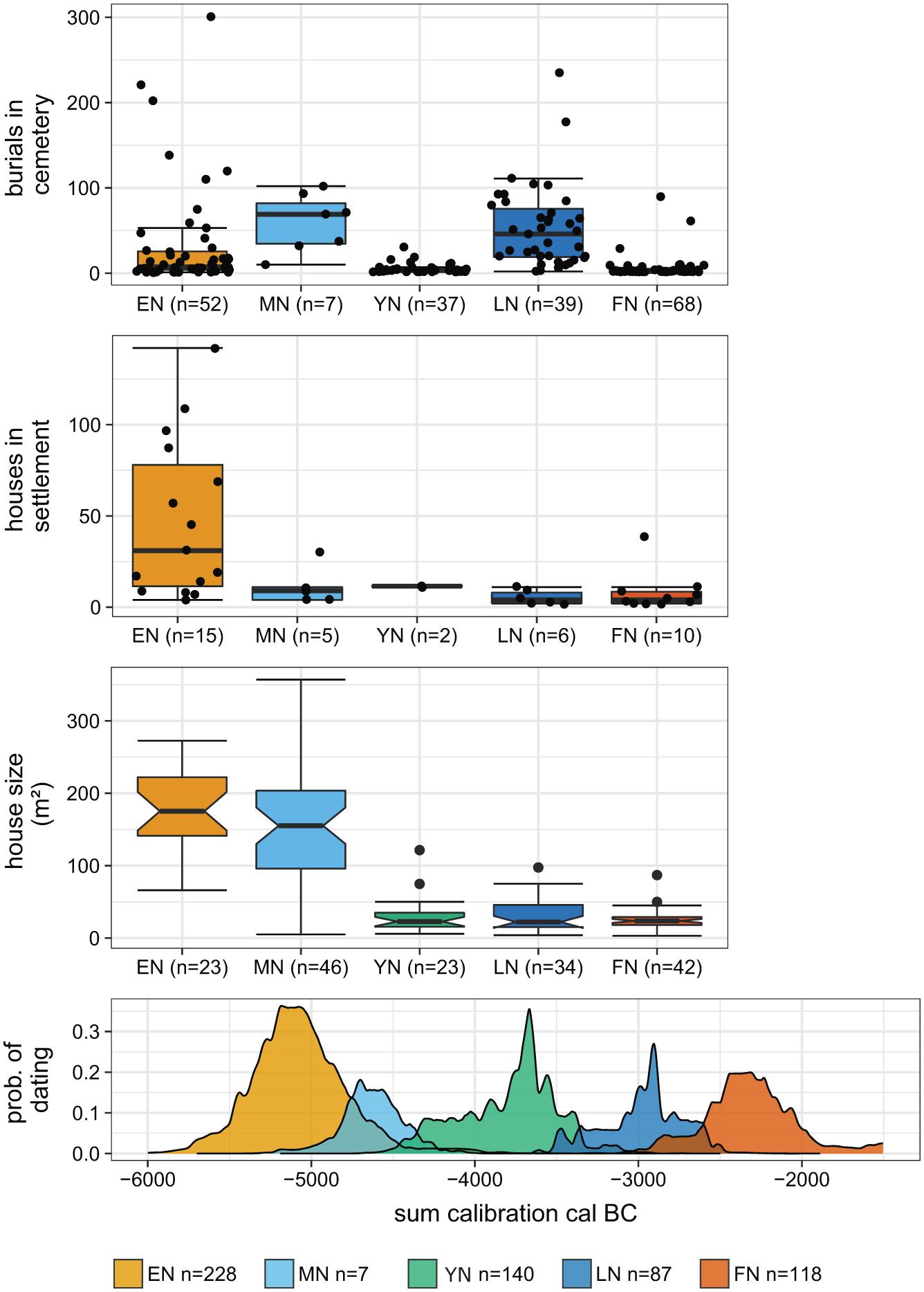
Transformations in settlements and burial communities in the Neolithic German loess zone. Statistical analyses of archaeological proxies from the area of study. The data are presented as box-and-whisker plots across time slices. The plot for house size has additional notches to illustrate a 95% confidence interval for differences in medians (McGill et al., 1978). EN: Early Neolithic; MN: Middle Neolithic; YN: Younger Neolithic; LN: Late Neolithic; FN: Final Neolithic .
The early Neolithic (EN), associated with the Linear Pottery Culture (LBK), expands from the Danube to the Rhine and to the northern part of the area selected here (Jeunesse, 1997; Price, 2000).
The middle Neolithic (MN) features several regional cultures but is dominated by Rössen.
The younger Neolithic (YN) comprises mainly Michelsberg sites and a phase characterized by huge enclosures (Gleser, 2012; Seidel et al., 2016).
The late Neolithic (LN) is described by heterogeneous archaeological groups (see Table 1).
The final Neolithic (FN) is closely linked with Corded Ware communities and the Bell Beaker complex (Furholt, 2014; Müller, 2009).
The absolute dating of each time slice is represented by a smoothed sum calibration (10-year moving average) of all related data from the RADON database (47.48° latitude, 6.09° longitude to 51.95° latitude, 14.32° longitude), excluding those with a standard deviation greater than 300 (Figure 2).
Demographic estimations of associated populations have been carried out in several studies based on proxies indicating settlement activities, such as site density, number of houses or house size (Birch-Chapman et al., 2017; Müller, 2007; Shennan, 2009; Zimmermann et al., 2009; Zimmermann and Wendt, 2003). Another approach uses inhumations and life expectancy, number of inhumations per location, site density (Schiesberg, 2012), a combination of settlement size and inhumations (Balkowski and Hartmann, 2015) or pollen data to estimate land use (Lechterbeck et al., 2014). Since population size estimation based on radiocarbon dates is problematic (Contreras and Meadows, 2014), we use house sizes and number of houses per settlement as indirect proxies for tracing changes in domestic and residential strategies. However, these parameters should not necessarily be regarded as living space as important information on interior structures and house usage is often missing (Birkenhagen, 2003; Lüning and Reisch, 2011; Schlichtherle and Wahlster, 1986; Stäuble and Lüning, 1999; Zürn, 1965). Although variation in living space might also be related to sociocultural preferences (Casselberry, 1974), it is the most abundant and often applied parameter for demographic estimations.
Here, we only focus on the changes in house size and number of houses per settlement. Both parameters are considered to reflect behavioural changes in domestic organization that can be influenced by epidemics and vice versa (Figure 2).
During the early and middle Neolithic, at least 75% of large houses exceeded 100 m². However, the different architectural structures of middle Neolithic (Rössen) houses provided more living space, while the house size per se did not change. In the younger Neolithic, a maximum of 75% of houses reached up to 50 m². This decrease towards smaller house sizes throughout the Neolithic is striking and persistent. This major change of living space indicates transformations in the socio-economic behaviour that may also be due to epidemic events. Clearly, the reduction of house sizes is a complex process that had a sustainable effect on architectural design and knowledge, social organization and domestic activities such as storage, cooking and heating.
Interestingly, the number of houses in each settlement showed a rapid decrease from the early to the middle Neolithic. Missing information on house generations in subsequent periods prevents further calibration of the dataset as, for instance, has been done for early Neolithic settlements (e.g. Mischka, 2004). In the early Neolithic, 75% of the settlements comprised more than 10 houses. In contrast, from the younger Neolithic onwards, the number of houses for each settlement was smaller (with one exception in the final Neolithic).
Despite a generally smaller database during and after the transition from the early to the middle Neolithic, we can observe fundamental changes in the domestic strategies in the middle Neolithic period associated with both house size and number of houses per settlement. This finding may be compatible with human responses to epidemics.
Changes in burial practices and increases in the number of burials in cemeteries may be evaluated in terms of greater mortality, possibly caused by epidemics. The detectable number of individuals per burial is influenced by funeral practice, and thus reflects different behavioural strategies dealing with death, which are, for instance, visible in mass graves after disastrous events (Gowland and Chamberlain, 2005; Margerison and Knüsel, 2002). For some archaeological groups, no common burial practice is known, or a general lack of burials precludes further information.
The number of burials per cemetery differed throughout the Neolithic. A major change appears in the younger Neolithic, characterized by a clear decrease and less variability in the frequency of burials per site. The subsequent late Neolithic contrasted with greater numbers and variability of burials per cemetery. In the final Neolithic, there were more cemeteries, however, with fewer burials. We also observe changes in burial practice. In the early Neolithic, cemeteries were dominated by groups of single flat graves with inhumations in crouched positions (Häusler, 2001; Hofmann and Orschiedt, 2013; Veit, 2008). Irregular burial depositions are known from different contexts, such as in settlements, enclosures and mass graves as well as cremations (Hofmann and Orschiedt, 2013; Veit, 1996, 2008). In the middle Neolithic, the cemeteries were composed of single flat graves with the individuals placed in a stretched position and various orientations (Fischer, 1956; Häusler, 2001; Hofmann and Orschiedt, 2013; Meier-Arendt, 1975; Rötting, 1977; Spatz, 1999; Veit, 2008). For the younger Neolithic groups, diverse burial practices are known (Figure 3), while the primary character of graves from the Michelsberg group is still under discussion (Hofmann and Orschiedt, 2013; Nickel, 1998). The late Neolithic is the only time slice characterized by collective burials (i.e. graves with successive inhumations over several hundred years), while single burials in flat graves were rare (Dirks, 2000; Fischer, 1956; Häusler, 2001; Hofmann and Orschiedt, 2013; Schierhold, 2014). The common burial practice changed again in the final Neolithic, from collective to single and multiple burials in flat graves (Fischer, 1956). These individuals were laid down in a crouched position, in a gender-specific manner (Dresely, 2004; Fuchs, 2013; Furholt, 2014). Some irregular burial depositions with cremations are also recorded for this period (Fischer, 1956).
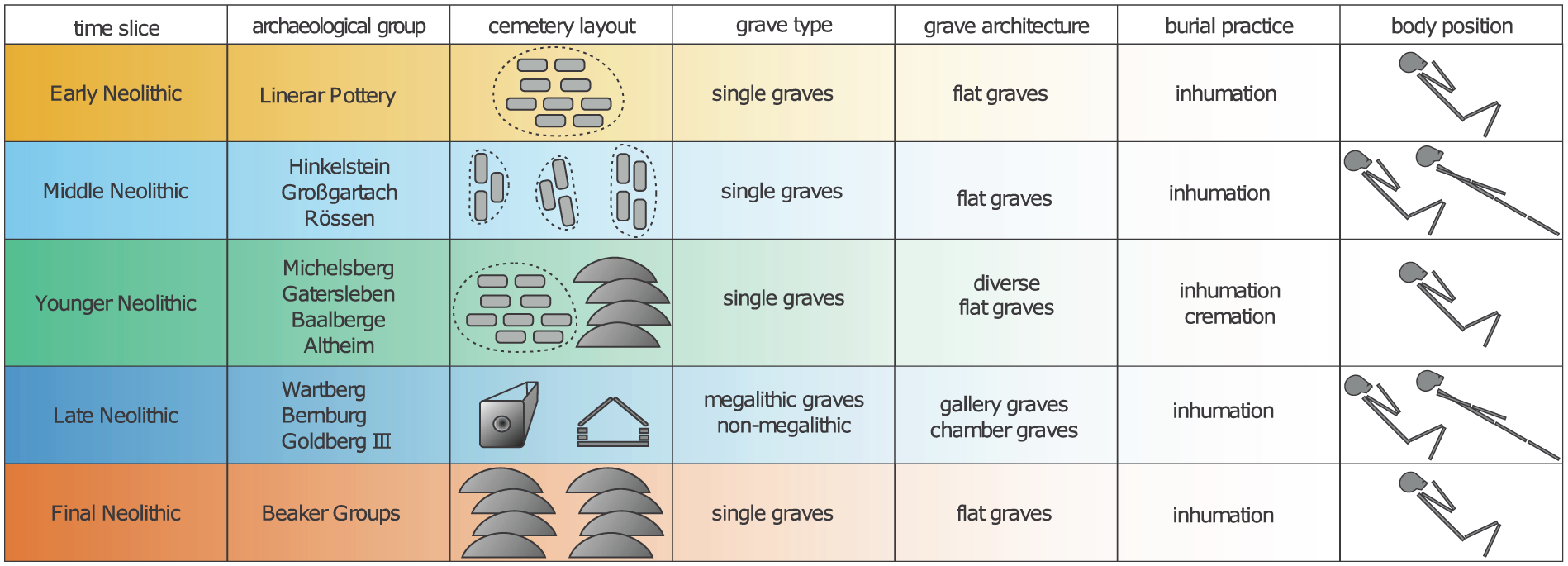
Time slices, archaeological groups, and characteristics of most common mortuary practices in Neolithic Germany (data from Hofmann and Orschiedt, 2015; Furholt, 2014; Schierhold, 2014; Veit, 2008; Häusler, 2001; Dirks, 2000; Preuß, 1998; Nickel, 1998; Veit, 1996; Rötting, 1977; Spatz, 1999; Meier-Arendt, 1975; Fischer, 1956).
In summary, there are no clear indications of phases that reveal an increase in the number of burials throughout the Neolithic periods which could be associated with epidemic events. It is remarkable that the younger Neolithic graves did not show clearly defined burial customs, indicating a loss of widely distributed funeral practices and representing the lowest number of burials in cemeteries. Furthermore, at the end of the Neolithic, multiple burials occurred more often, suggesting the simultaneous death of people, which, in turn, may be related to epidemic events.
aDNA evidence for epidemics
aDNA analysis has drastically expanded the scope of palaeopathology as full genomic investigations are now feasible. The greatest advantages of today’s aDNA studies of infectious diseases are (1) the possibility to reliably identify a pathogen and to reconstruct its genome sequence thanks to optimized protocols which allow the recovery and sequencing of sufficient amounts of DNA (Bos et al., 2011, 2014; Feldman et al., 2016; Krause-Kyora et al., 2018a, 2018b; Spyrou et al., 2016, 2018), (2) the identification of transmission and virulence factors likely to cause extreme symptoms at the individual level (e.g. death) and/or population level (reduction in the number of people; Taubenberger et al., 2005; Tumpey et al., 2005) and (3) tracing genomic changes in a pathogen through time, reconstructing its evolutionary history (Feldman et al., 2016; Krause-Kyora et al., 2018a, 2018b; Maixner et al., 2016; Schünemann et al., 2013; Spyrou et al., 2018).
Although aDNA research has in general made huge progress over the last decade, it is still challenging to find authentic pathogen DNA due to issues of preservation and contamination. In addition, the bioinformatic analysis of the ancient genome sequences requires the comparison with reference data. These reference sequences predominantly originate from pathogens that are clinically relevant and/or known as causative agents of epidemics today. This bias may preclude the detection of ancient strains that went extinct and have no close present-day relatives. Another obstacle is that fast-killing microbes do not usually leave specific lesions on skeletons. In the absence of such external guiding factors, a targeted aDNA analysis is not possible, and the detection of a Neolithic epidemic is virtually a chance finding. To increase the odds of success, the large-scale screening of hundreds or thousands of individuals for pathogen DNA is required – and a tiny bit of luck (Krause-Kyora et al., 2018a, 2018b; Mühlemann et al., 2018a, 2018b).
Keeping these limitations and caveats in mind, we here review the reported aDNA-based pathogen findings from the European Neolithic and slightly later periods, venturing outside the designated German loess zone to also include relevant observations from neighbouring regions (Figures 1 and 4; Table 1).
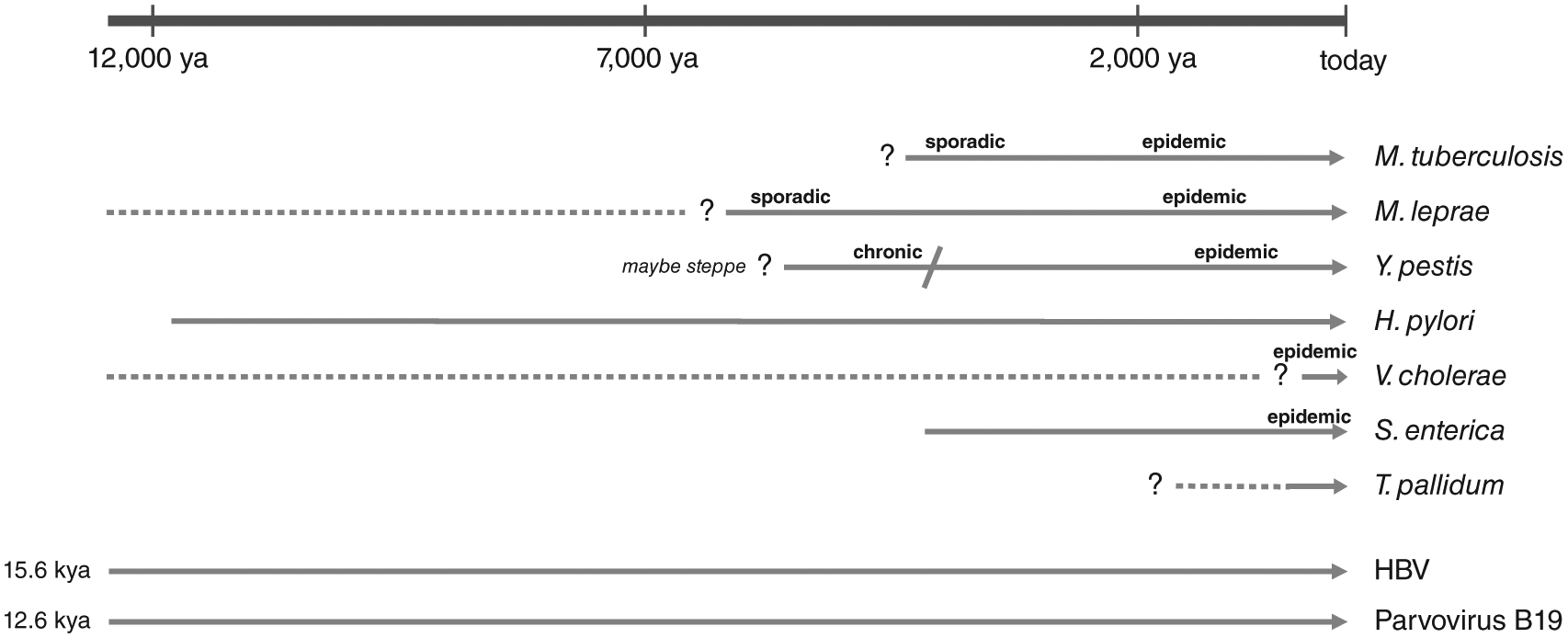
aDNA-based chronology of pathogens causing some infectious diseases known today.
The earliest direct evidence of a human pathogen, the hepatitis B virus (HBV), dates to 7000 years ago (Krause-Kyora et al., 2018b). The virus is highly contagious and is currently one of the most widespread pathogens, diagnosed in over 250 million people worldwide (Krause-Kyora et al., 2018b). In 53 Neolithic skeletons from Central Germany, two full HBV genomes were recovered from individuals associated with the Linear Pottery (early Neolithic) and the Funnel Beaker Cultures (late Neolithic), respectively. The Neolithic forms appear to represent distinct lineages that likely went extinct (Krause-Kyora et al., 2018b). Twelve additional HBV genomes have been recorded from Bronze and Iron Age contexts across Central-Western Eurasia, compatible with the spread of the virus during migrations at that time (Mühlemann et al., 2018a). The virus has been estimated to have infected humans at least for the past 15,000 years (Mühlemann et al., 2018a).
Parvovirus B19 V (B19 V) is a ubiquitous human pathogen that can lead to fifth disease (erythema infectiosum), a common rash-causing childhood disease. B19 V can easily be transmitted via the respiratory or blood-borne route. The infection is usually mild and may even take an asymptomatic course. B19 V DNA is thought to persist lifelong in an infected individual (World Health Organization (WHO), 2017). In a large-scale screening of 1578 ancient humans, viral genomes were detected in 20 skeletons that came from a wide geographic area (Europe, Central Asia, Greenland). The earliest B19 V positive sample dates to the early Neolithic from Russia (6795 years ago), indicating a long-term association with humans which is estimated to go back some 13,000 years (Mühlemann et al., 2018b).
The oldest complete genomes of bacterial pathogens found so far also date to the Neolithic period. Yersinia pestis (Y. pestis) appeared in Eurasia already ~5000 years ago (Rascovan et al., 2019; Rasmussen et al., 2015). This early form may have been able to cause pneumonic plague (Rascovan et al., 2019), though it most likely lacked the efficient flea-based transmission to humans (Rasmussen et al., 2015). It was only much later (3800 years ago) that the bacterium acquired specific mutations which might have increased its virulence to humans (Spyrou et al., 2018). Y. pestis was quite frequent in Bronze Age individuals (Rasmussen et al., 2015). One scenario is that Y. pestis lineages already expanded across Eurasia during the Neolithic, causing a plague pandemic that contributed to the Neolithic decline (Rascovan et al., 2019); another hypothesis is that they were carried into Europe during the migration of Bronze Age populations (e.g. those associated with the Yamnaya culture) from the steppe (Andrades Valtueña et al., 2017). In either case, evidence of massive plague pandemics, as they were, for instance, recorded for the Medieval Black Death, is still missing for the Neolithic and the Bronze Age periods (Fornaciari, 2017).
Helicobacter pylori (H. pylori), a gastro-intestinal pathogen, was identified in the stomach of the Iceman, a 5300-year-old Copper Age mummy from the Italian Alps (Maixner et al., 2016). Although H. pylori is carried by approximately half of the world’s population today, 90% of the infected people do not have any symptoms such as stomach ulcers or gastric carcinoma. In-depth analysis of the ancient H. pylori genome showed that it was a cytotoxic strain that could trigger strong inflammatory immune responses in its host. Yet, whether the Iceman suffered from gastric disease and stomach pain could not be confirmed histologically, owing to the poor preservation of the stomach mucosa (Maixner et al., 2016).
Similar to H. pylori, which is thought to have accompanied humans for the last 100,000 years (Achtman, 2016), it was also suggested that Mycobacterium tuberculosis (M. tuberculosis), the cause of tuberculosis (TB), is an ancient pathogen that left Africa together with its host ~60,000 years ago (Comas et al., 2013). However, two ancient genome studies suggest estimates of fewer than 6000 years for the emergence of the most recent common ancestor of the bacterium (Bos et al., 2014; Kay et al., 2015). Whole M. tuberculosis genomes from Neolithic specimens have not been generated yet. So far, only low-resolution polymerase chain reaction (PCR)-based analyses have been performed on DNA extracted from Neolithic skeletons with TB-specific pathologies. The results indicate that the lesions might have resulted from M. tuberculosis infections (Hershkovitz et al., 2008; Masson et al., 2015; Spigelman et al., 2015). However, one criticism of the PCR-based method is that it is more prone to contamination and does not allow the differentiation between authentic M. tuberculosis and that of closely related Mycobacterium species in the soil which could also end up in the aDNA extract (Müller et al., 2014). How long M. tuberculosis has afflicted humankind still remains an open and controversial question (Achtman, 2016). Whether TB was already present before and during the Neolithic or developed much later can only be resolved by genomic data from Neolithic samples, which will also allow detailed functional, evolutionary and phylogeographic analyses.
Based on this literature review, various infectious agents have been successfully identified in Neolithic contexts by aDNA research. The detection of authentic bacterial and viral DNA is a proof-of-principle and demonstrates the great sensitivity of the applied methods. But as discussed above, there is no guarantee of pathogen detection due to the intrinsic pitfalls of aDNA work. In addition, a positive finding in an individual cannot determine its ultimate cause of death, since an infection may have led to an asymptomatic or latent course of the disease, or may have resulted in symptoms milder than those we see today.
Interestingly, of the microbial organisms detected in Neolithic remains, HBV, P19 V and H. pylori are estimated to have been present already in hunter-gatherer groups. It is also noteworthy that the pathogens were mainly detected in single remains indicating sporadic cases, or a few specimens from different sites and periods that are widely separated from each other through space and time.
Osteological evidence and skeletal pathology
Skeletal remains make up the primary object for investigating physical stress and illness which humans experienced during life, including infectious diseases. Osteological and bioarchaeological studies showed growing evidence that, compared to hunter-gatherers, early agricultural communities were exposed to higher physical stress. This is associated with intensified physical work, nutritional imbalances as well as an increase in acute and chronic diseases. (Armelagos et al., 1991; Ash et al., 2016; Barrett et al., 1998; Bickle, 2013; Cohen, 2008; Krenz-Niedbala, 2014; Macintosh et al., 2016). If the Neolithic transition was also accompanied by a rise in infectious diseases, we would expect the following findings in the palaeopathological records: (1) a significant increase in skeletal symptoms specific and/or unspecific for certain infectious diseases; (2) a high frequency of unspecific skeletal symptoms indicating poor health status that rendered the individuals more susceptible to succumb to a deadly infection; and (3) large numbers of infants and elderly without perimortal injuries in mass burials, as a reaction to sudden and frequent deaths that were caused by rapid and fatal disease courses.
As most infections primarily affect soft tissues and only a few leave traces in the osteological records in a chronic stage, a disease diagnosis often remains difficult. Therefore, it is worth taking into account both skeletal morphology and molecular pathogen evidence. Several skeletal inflammatory responses are associated with infection, but only some disease-causing pathogens are considered to develop specific skeletal pathology, such as Treponema pallidum (among others inducing syphilis) or Mycobacterium leprae (Baker and Armelagos, 1988; Ortner, 2011; Roberts and Manchester, 2013; Waldron, 2009). To date, neither bacterium has been found in Neolithic Europe (Donoghue et al., 2015; Harper et al., 2011). Skeletal morphologies suggestive of leprosy (e.g. rhinomaxillary syndrome facies leprosa and/or deconstruction of hand and foot bones) are reported for the Late Copper Age in Hungary (3780–3650 cal. BCE; Köhler et al., 2017), the Anatolian Bronze Age (2700–2300 BCE; Mellink and Angel, 1969) and early Bronze Age in Scotland (2300–2000 BCE; Roberts, 2007), but not for the Neolithic in Germany. In addition, the diagnostic value of these cases is controversial and their sporadic occurrence does not speak for epidemic extents (Mark, 2019). In line with results from aDNA research, osteological evidence for leprosy is growing for the later periods from the Iron Age onwards (Donoghue et al., 2015; Mariotti et al., 2005), with a peak in morbidity during the Middle Ages (Aufderheide and Rodriguez-Martin, 1998; Roberts and Manchester, 2013).
Up to now, TB is the only infectious disease with characteristic skeletal pathomorphology that has been discussed for the Neolithic (Santos, 2015; Schultz and Schmidt-Schultz, 2015; Tuli, 2016). In osteological examinations, associated lesions are described particularly for the lower vertebral bodies, with ventral collapse due to osteomyelitis resulting in an angular kyphosis (‘Pott’s disease’, Figure 5; Balázs et al., 2015; Dass et al., 2002), or in large joint elements, such as knees and hips (tuberculous arthritis; Nerlich and Lösch, 2009; Roberts, 2015; Tuli, 2016). The underlying pathological processes are tuberculous arthritis that arises from reactivation of bacilli lodged in bone during the original primary mycobacteria infection (Schirmer et al., 2010). Clinically, tuberculous arthritis is assumed to result from the spread of an initial infectious focus in the bone to the joint. Infrequently, tuberculous bacilli travel from the lung to the spine causing Pott’s Syndrome in the end stage of disease (Garg and Somvanshi, 2011).
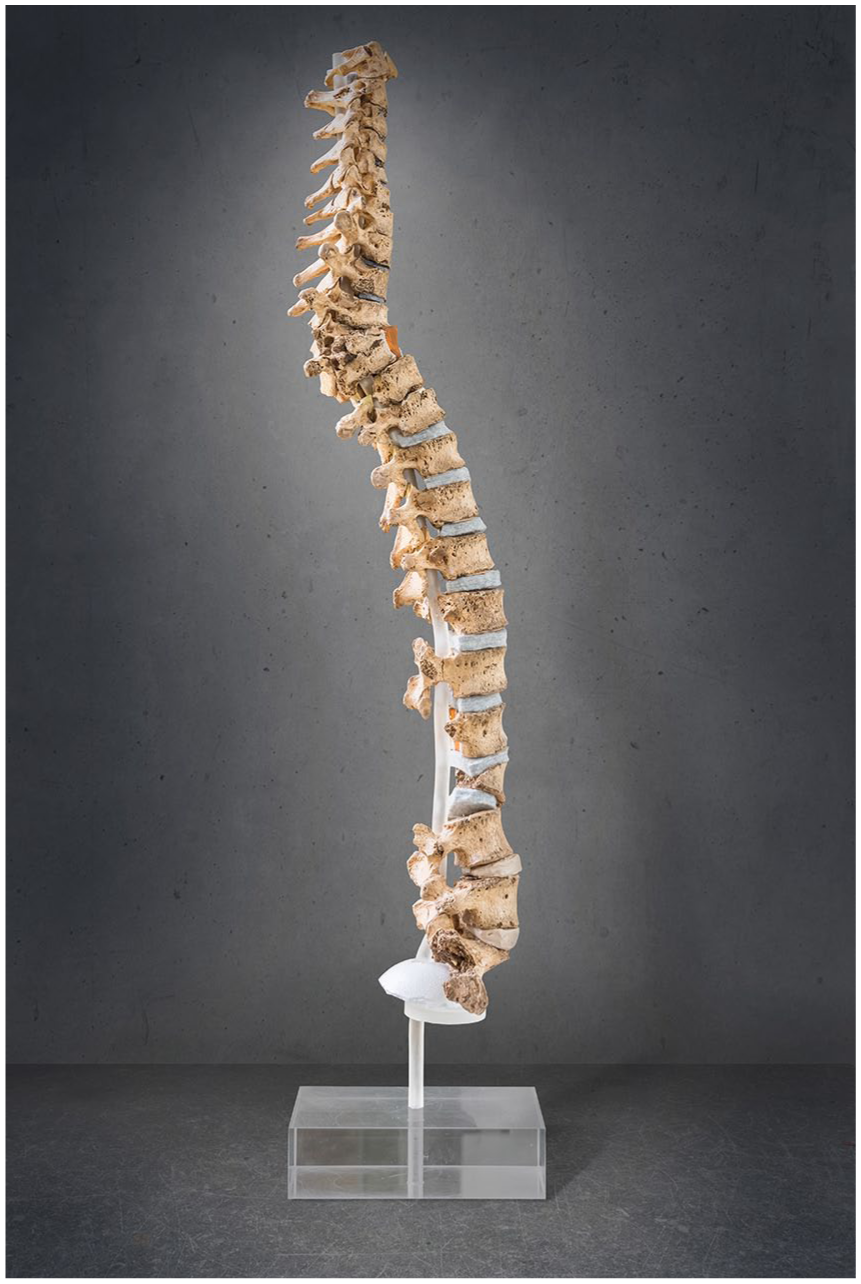
Pott’s Syndrome, diagnostic of TB osseous symptoms in the spine (ventral collapse of the upper thoracic vertebral bodies leading to angular kyphosis), found in an individual from a Rössen (middle Neolithic) context (Bartels, 1907; Ludwig, 2007; picture Heidelberg University Hospital, publication agreement with O. Bock-Hensley/Deutsches Tuberkulose-Archiv Heidelberg).
There is a large number of skeletal symptoms suggestive of TB from prehistoric contexts, such as hypertrophic (pulmonary) osteoarthropathy (especially of the ribs; Masson et al., 2013; Mays and Taylor, 2002; Webb and Thomas, 1986), serpens endocrania symmetrica (SES; Hershkovitz et al., 2002), impressions on the endocrania and pelvic bone due to tubercula involvement (Schultz and Schmidt-Schultz, 2015) and others (Nerlich and Lösch, 2009; Spekker et al., 2012). The diagnosis of TB in Neolithic human remains should be based on several lines of evidence, that is, molecular pathogen detection (e.g. by aDNA, lipids) as well as specific and unspecific skeletal symptoms, ideally in the same individual. To date, these studies have reached their methodological limits (see preceding section; Hershkovitz et al., 2015; Masson et al., 2015; Müller et al., 2016; Nerlich and Lösch, 2009). This also applies to the large-scale examination of 118 LBK (early Neolithic) individuals from Central Germany (Nicklisch et al., 2012). In this population, 22 individuals exhibited suspicious rib lesions and three had diagnostic symptoms of Pott’s disease. Other reported TB cases are sparse for the Neolithic German loess zone, and there are only few in the European context (Canci et al., 1996; Köhler et al., 2014; Masson et al., 2015; Pósa et al., 2015; Spekker et al., 2012). Pott’s Syndrome was found in a skeleton from a Rössen (middle Neolithic) site (Bartels, 1907; Ludwig, 2007). The individuals buried at the LBK cemetery Wandersleben exhibited extremely high rates of inflammatory symptoms on the nasal and paranasal passages (100% affected) as well as pleuritic areas of the rib cage (71%). These lesions indicate chronic diseases of the lower and upper airways, which may be caused by TB or TB-like infections, even if other characteristic symptoms are lacking (Klingner, 2016). Clinical observations further revealed that only few infected individuals develop osseous manifestations (e.g. 2–5%, Roberts, 2015; 3–5%, Rühli et al., 2005). As a consequence, early-stage cases of TB remain undetected in osteological examinations. Furthermore, non-tuberculous mycobacterial infections can also cause similar pathologies in the skeletal system (Park et al., 2014). Osteoarticular affection usually arises secondary to direct infection. Thus, diagnostic osteological TB markers alone cannot be used to estimate the infection rate during the Neolithic.
Other infectious agents found in aDNA studies from Neolithic Germany such as P19 V, H. pylori or HBV leave no traces in human osteological material or manifest in unspecific and/or secondary symptoms of inflammation and physical stress. The latter would not be able to induce sudden death peaks of epidemic proportions on their own.
The pathomorphology of most skeletal infections is apparent by lesions of the inner or outer medullar bone surfaces, such as decreased and/or increased bone formation, alterations of the bone anatomy (e.g. periostitis, osteitis, osteomyelitis; Ortner, 2003) and may be mirrored in secondary symptoms of physical stress, such as cribra orbitalia (Figure 6) and porotic hyperostosis (both associated with anaemia; Brickley, 2018; Rivera and Mirazón Lahr, 2017; Walker et al., 2009; Wapler et al., 2004), and enamel hypoplasia (tooth formation disorders due to physical stress; Hillson, 2008; Scott, 2002; Suckling, 1989; Waldron, 2009). In general, more systematic research with clinical reference is needed to understand the reciprocal effects and aetiology of such physical stress markers that are often used to trace the consequences of the Neolithic lifestyle. For instance, clinical studies associate fifth disease (erythema infectiosum; P19 V) with severe anaemia in patients with haemolytic disorders and rheumatologic manifestations in adults when the disease is persistent (Heegaard and Brown, 2002; Mor et al., 2016; Naciute et al., 2016; Naides et al., 1990). Such processes might also involve the skeletal body (e.g. cribra orbitalia, finger joint lesions; Ortner, 2011; Waldron, 2009). On the other hand, iron deficiency is considered to be protective against TB (Boelaert et al., 2007; Meller and Berthold, 2008; Prados-Rosales et al., 2014).
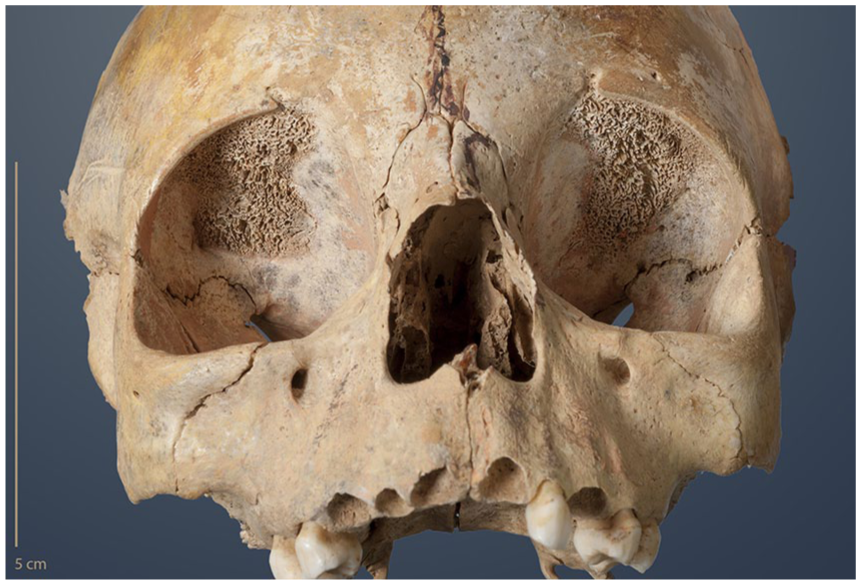
Osseous symptoms of unspecific stress, example of cribra orbitalia as an anaemic reaction. A 3- to 4-year-old infant buried in the collective grave of Niedertiefenbach (Wartberg Group, late Neolithic) showing strong trabecular growth at the orbital roof as a result of increased blood production (picture: S. Jagiolla; Rinne et al., 2016).
In a diachronic study that included 593 individuals from the Mesolithic and early to late Neolithic periods, Wittwer-Backofen and Tomo (2008) investigated unspecific stress markers in order to compare physical burdens before and during the establishment of the Neolithic lifestyle. The authors surmised a decline in health during the early Neolithic in the German loess zone (Carli-Thiele, 1996; Wittwer-Backofen and Tomo, 2008). Other palaeopathological studies on skeletal series (e.g. Jechtingen, Lauda-Königshofen, different Baalberge cemeteries, Wiederstedt or Niedertiefenbach) from various Neolithic periods draw a different, and rather patchy, picture of incidence rates (e.g. Alt et al., 2014; Funke, 2006; Menninger, 2008; Meyer et al., 2004; Rinne et al., 2016; Rinne et al., in press). For a comprehensive interpretation of these heterogeneous results, the respective socio-economic backgrounds, their influence on the population lifestyle and the varying qualities of the primary data have to be considered.
The presumed higher susceptibility of children and elderly to a lethal course of an infection, frequent epidemic events or an overall higher disease burden would be reflected in an over-representation of the respective age classes in skeletal series. The study by Wittwer-Backofen and Tomo (2008) indeed suggests an increase in the proportion of subadults in the early Neolithic, thus a higher risk of death early in life at the onset of Neolithic lifeways. However, current data on life expectancy show a high level of variability on the regional scale (e.g. Dresely, 2004; Fritsch et al., 2008). Furthermore, the potential role of epidemic events has not been thoroughly considered in this regard, and a comprehensive study on mortality data on both regional and supra-regional scales is needed.
In general, there are only few mass graves (more than five individuals buried at the same time; cf. Eggert, 2001) known from Neolithic Germany. The most famous one, Talheim, shows many males with signs of interpersonal violence rather than exceptionally high frequencies of infants and elderly (Wahl et al., 1987). Similar characteristics apply to other graves dated to the early Neolithic (Halberstadt, Dresely and Alt, 2015; Schöneck-Kilianstätten, Lohr, 2013). Therefore, it is unlikely that these burials are connected to epidemic events. The grave from Wiederstedt is exceptional in this regard, as the buried individuals revealed an age distribution (8 subadults out of 10 individuals) typical of an epidemic. There were also no indications of lethal traumatic injuries (Meyer et al., 2004).
The application of the mass grave definition excludes many other burial contexts with multiple individuals that may also reflect the sudden death of several people in a short period of time, due to devastating events such as infectious diseases or famine. Multiple burials are in general more often known from the later Neolithic periods, for example, from Corded Ware (final Neolithic) or Salzmünde contexts (late Neolithic) (Alt et al., 2017; Dresely, 2004). One exception is the multiple grave located in the vicinity of the Michelsberg earthwork Bruchsal-Aue (4250–3650 cal. BCE). The multidisciplinary analysis of the skeletal remains draws a connection between the large number of infants, symptoms of anaemia as well as the presence of potentially deadly parasite infections (Keller et al., 2015). However, multiple burials that show a deviation in the age distribution appear only sporadically. In addition, such deviations are often hard to ascertain due to the small number of buried individuals (for Middle East Germany: Fuchs, 2013; for Southwest Germany: Ortlof, 2014; Trautmann, 2008). Therefore, neither the number of mass/multiple burials nor the demographic distribution among the buried individuals indicates phases of epidemics in Neolithic Germany, although some sites might reflect local disease outbreaks.
In summary, the current information gained from the skeletal material is insufficient for supporting or rejecting the hypothesis of a significant increase in infectious diseases. To exploit the available osteological resources to the maximum, the following recommendations have to be implemented: (1) comprehensive and comparable reporting of physical stress parameters and skeletal lesions; (2) consistent application of diagnostic methods and tools, including an agreement on specific coding systems; and (3) adequate consideration of clinical expertise in palaeopathological diagnosis and interpretation.
Conclusion
In this review, we apply a multidisciplinary approach combining archaeology, genetics and osteology in order to test the hypothesis of epidemic-driven transformations in the Neolithic.
The archaeological record shows fundamental changes in domestic strategies and mortuary practices from the 5th to the 4th millennium BCE (younger to late Neolithic). Although the data are sparse, there is a clear reduction in house size, number of buildings and architectural design. In general, this period is characterized by substantial changes, for instance, in the construction of huge enclosures, long-distance contacts and the Mesolithic–Neolithic transition in Northern Germany and Scandinavia (Andersen, 1991; Bakker, 1991; Bertemes, 1991). For the younger Neolithic (Michelsberg group), these changes cannot be linked with palaeopathological or genetic evidence for infectious diseases due to the poor record of human remains. In the late Neolithic, shifts in burial practices led to heterogeneous cemetery layouts, diverse grave architecture and collective graves. In the final Neolithic, multiple burials suggesting the simultaneous death of several individuals are observed at higher frequency. Interestingly, this observation coincides with smaller settlements and house sizes. Hence, these multiple burials might reflect single local disease outbreaks in smaller communities. There are no convincing numbers of mass burials with individuals showing no signs of interpersonal violence. Accordingly, mass burials indicative of epidemic events seem to be missing.
From the palaeopathological perspective, various symptoms indicate infectious diseases in the Neolithic osteological record. Although one study suggests a higher disease burden during the early Neolithic (Wittwer-Backofen and Tomo, 2008), comparative datasets from later time slices are not available, hampering comprehensive diachronic evaluations. In the future, it is necessary to conduct consistent osteological investigations across additional and larger skeletal series.
The only infectious disease observed so far in the Neolithic that leaves diagnostic changes on human bone is TB or TB-like infections. However, these lesions are rare in the palaeopathological record. Genetic studies suggest that M. tuberculosis emerged rather late, around 5000 years ago (Brynildsrud et al., 2018). Thus, in the late Neolithic, TB may have been present only sporadically and not in epidemic proportions. However, the actual incidence of TB infections could have been higher, hidden in unspecific skeletal pathomorphology.
Modern aDNA techniques allow the detection of a wide range of bacterial and viral pathogens from skeletal remains. Indeed, current genetic results show some infectious diseases in Neolithic populations. Interestingly, of the few pathogens detected, HBV, P19 V and H. pylori are estimated to have been present already before the Neolithic. It is also noteworthy that the pathogens were mainly detected in single remains, indicating sporadic cases, or a few specimens from different sites and periods. However, we are aware of the limitations given by the preservation of the bioarchaeological records, and new aDNA findings may change the picture we have drawn here. Especially, whether the early Y. pestis strains were already able to cause a Eurasian pandemic during the Neolithic and the Bronze Age remains to be clarified; to date, the lack of convincing numbers of mass burials does not support this scenario.
Up to now, the proxies examined here have not provided direct evidence for large-scale infections of high impact in the German loess zone between 5500 and 2500 BCE. Thus, based on this review, there is no convincing support for the hypothesis of epidemic-driven transformations in this area during the Neolithic. However, future work will certainly improve the current data availability and will offer additional and more detailed insights into the role of infectious diseases at this important turning point in human prehistory.
Supplemental Material
Fuchs-et-alii_InfectiousDiseases_information-supplemental-material_revised – Supplemental material for Infectious diseases and Neolithic transformations: Evaluating biological and archaeological proxies in the German loess zone between 5500 and 2500 BCE
Supplemental material, Fuchs-et-alii_InfectiousDiseases_information-supplemental-material_revised for Infectious diseases and Neolithic transformations: Evaluating biological and archaeological proxies in the German loess zone between 5500 and 2500 BCE by Katharina Fuchs, Christoph Rinne, Clara Drummer, Alexander Immel, Ben Krause-Kyora and Almut Nebel in The Holocene
Supplemental Material
Fuchs-et-alii_supplemental_material – Supplemental material for Infectious diseases and Neolithic transformations: Evaluating biological and archaeological proxies in the German loess zone between 5500 and 2500 BCE
Supplemental material, Fuchs-et-alii_supplemental_material for Infectious diseases and Neolithic transformations: Evaluating biological and archaeological proxies in the German loess zone between 5500 and 2500 BCE by Katharina Fuchs, Christoph Rinne, Clara Drummer, Alexander Immel, Ben Krause-Kyora and Almut Nebel in The Holocene
Supplemental Material
HOL857230_References_to_supplement – Supplemental material for Infectious diseases and Neolithic transformations: Evaluating biological and archaeological proxies in the German loess zone between 5500 and 2500 BCE
Supplemental material, HOL857230_References_to_supplement for Infectious diseases and Neolithic transformations: Evaluating biological and archaeological proxies in the German loess zone between 5500 and 2500 BCE by Katharina Fuchs, Christoph Rinne, Clara Drummer, Alexander Immel, Ben Krause-Kyora and Almut Nebel in The Holocene
Footnotes
Acknowledgements
This review is the result of collaboration between the CRC 1266 subprojects D2 ‘Third millennium transformations of social and economic practices in the German Lower Mountain Range’ (C. Rinne, C. Drummer) and F4 ‘Tracing infectious diseases in prehistoric populations’ (B. Krause-Kyora, A. Nebel, A. Immel) as well as palaeopathological expertise from Z1 ‘Zentralised tasks’ (K. Fuchs). We thank Carsten Reckweg for editing figures as well as Sarah Jagiolla, Oswinde Bock-Hensley and the Deutsches Tuberkulose-Archiv Heidelberg for providing photos. We also thank Christy Baker Starrman for English proofreading.
Funding
This study was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation – Projektnummer 2901391021 – SFB 1266).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
