Abstract
In the archaeological record, ash and charred organic material are the only indications of the type of fuel used by ancient societies to feed their fires. This potential source of information may help further understanding of past human behaviour in relation to fuel selection, applied type of fire and function of fires lit in hearths. This study examined ash from reference samples and ash and char samples recovered from an Iron Age peatland site in Vlaardingen, the Netherlands. Local availability and abundance made it possible for the occupants of the site to choose from different fuel resources. Peat and cattle dung were readily available, while wood was less abundant. Reference samples from various locations were collected to investigate the properties of ash. Different types of wood, peat and cattle dung were accordingly ashed and analysed. In total, two techniques were used, that is, chemical analyses (x-ray fluorescence (XRF)) for the determination of the elemental composition and microscopic studies on field images of these ashes, mounted on glass slides, to investigate phytoliths quantitatively (Si and Ca types), siliceous aggregates and spherulites. The properties of the archaeological samples were compared with these results. The archaeological char samples were used for identification and analysed using reflective microscopy to study structure and temperature by reflectance (%Ro). It provided the necessary information to determine the type of fuel used by the Iron Age farmers and obtain more information on the function of the present fire structures. The results are strong indications that Iron Age farmers used all available fuels, that is, peat, cattle dung and wood, to feed their fires. The integrated application of phytolith, geochemical and char analyses has a high potential for the identification of the fuel selected by ancient societies.
Introduction
In the Iron Age (725–25 BC), the landscape of the Western coastal parts of Belgium and the Netherlands is characterized by the presence of mainly peat and clay (Bakels, 2012). Peat was formed in coastal lagoons, which were the result of the relative rise of the sea level in earlier periods. So, much of the area became covered with peat whereby some parts raised to levels (domed) above extreme high water during exceptional storm floods and thus becoming a rather dry land. It was rarely inundated, and it is thought that only during periods of high water due to spring tide and during periods of high discharge of the rivers Rhine, Meuse and Scheldt that the area became submerged. Since a considerable number of house plans were discovered in the area, it is supposed that during the Iron Age, people for the first time occupied the peat areas. The house plans showed a living compartment for people and attached to it a stable compartment for cattle (Eijskoot et al., 2012; Waterbolk, 1975). Local drainage made the cultivation of non-cereal crops and animal farming possible (Koot and Bakels, 2002). The Iron Age inhabitants can be considered as farmers living from agriculture including plant and cattle breeding. Pollen research has shown that trees were rather dearth in the area. Tree(s) pollen were identified as mainly alder (Alnus) and some ash (Fraxinus) (Brinkkemper, 1991; Koot and Bakels, 2002). Within the house features, much char, ash and heated bones were observed concentrated in structures apparently used to light fires within.
It is supposed that in the dwellings, fires were made to generate heat necessary for activities such as preparation of meals, providing warmth and so on. To initiate these fires, an interaction has to take place between a certain fuel, air and heat from an external heat source – the three basic elements of the fire triangle (Emmons and Atreya, 1982). Fuel would have originated from one of the three main resources, namely, (1) local vegetation (including harvest residues), (2) fossil fuel and (3) animal by-products. Fuel is a material composed of inorganic and organic fractions and water (Braadbaart et al., 2012; Canti, 2003), the latter providing the potential energy that can be transformed into the required heat energy. Once the outside heat source has been introduced to the fuel, heat is absorbed, and as a result, the fuel will rise in temperature. This process will drive the chemical reactions converting gradually the organic constituents (i.e. cellulose and starch) of the fuel, through substances with mainly aromatic constituents. From a temperature of 280–300°C, a new solid material and a large amount of flammable volatiles are formed. The solid residue is usually referred to as the char, a highly carbon-enriched material composed of a three-dimensional network with predominantly randomly orientated aromatic units. Char derived from wood is often called charcoal, but in principle, all plant materials can be thermally converted into chars (Braadbaart et al., 2012; Rein, 2009). Only when these temperatures are reached and with enough air, both new products are able to oxidize. The latter process is a highly exothermic process, generating and releasing the required heat. As long as the temperature remains above 300°C, both reactions will release heat. The residue of the char is the inorganic ash. When temperature decreases and not enough air is made available, the fire will extinguish. Under these conditions, the remains of a fuel, that is, ash and char, provide the only evidence of the original type of fuel that was used, and possibly, the original function of the fire structure can be determined. The identification of fuels by their ash is still far from being a routine practice in archaeology, as is shown among others by the investigation of the studied site. This could be mainly due to lack of a standard methodology, although recent studies have paid much attention to this issue (Braadbaart et al., 2012; Canti, 2003; Gur-Arieh et al., 2014; Lancelotti and Madella, 2012; Mentzer, 2014; Peters et al., 2001; Pierce et al., 1998; Portillo and Albert, 2011; Shahack-Gross and Ayalon, 2013, and the references cited therein). The study of ash (and char) should be part of an archaeological investigation in order to obtain a fuller picture of fuel use. Ash from different plant materials used as fuel is characterized by the presence of distinctive inorganic compounds, mainly consisting of silica and calcium. In archaeological research, ash is usually investigated by chemical analyses of the constituent elements and microscopic studies. The latter method uses the mineral inclusions present within the plant tissues referred to as phytoliths. They comprise both opal phytoliths and calcium oxalate crystals (Brochier, 1983; Canti, 2003; Franceschi and Nakata, 2005; Gur-Arieh et al., 2014; Lancelotti and Madella, 2012; Piperno, 2006). In this study, additionally, the presence of spherulites and siliceous aggregates were investigated. The former are calcium carbonate crystals derived from animal dung (Canti, 1998). The latter are a different form of silica-containing material, cemented biologically into aggregates consisting of amorphous Si-, Al-, Fe- and K-rich mineral phases (Albert and Weiner, 2001; Schiegl et al., 1996).
From various fuels, the comparative differences between their remains, that is, ash and char, have been investigated in order to establish the means by which fuel types can be distinguished from each other (Braadbaart et al., 2012; Canti, 2003). In order to use this information in archaeological research, also the variation in the available fuel types from place to place must be taken into account. For this purpose, it will be necessary to use reference samples of fuels that may have been available to the societies under investigation. For peat areas in the Iron Age, in principle, fuel types from three resources were available, that is, local vegetation, peat as a fossil fuel and dung as an animal by-product.
Concerning ash remains, reference samples of wood, peat and cattle dung were ashed and the properties determined (see also, for example, Albert and Weiner, 2001; Braadbaart et al., 2012; Canti, 2003; Gur-Arieh et al., 2013; Lancelotti and Madella, 2012). To discern possible differences between the char remains, the results of an earlier study were used (Braadbaart et al., 2012, Figure 4). Subsequently, the results of the analyses conducted on the reference ashes and chars were assessed and used to investigate the ash and char samples recovered from the archaeological site selected for this study: the so-called Vlaardingen – De Vergulde Hand West (VHW). This Iron Age site, situated in a wetland and a peat area, was extensively investigated and, therefore, provides much contextual information (Eijskoot et al., 2012). The objective of this study is to verify whether the applied analyses on reference material are appropriate to use on archaeological material and are useful to determine which fuel types were used. In addition, it is investigated whether the results for ash and char are comparable. The results may clarify the function of the fires structures lighted by the inhabitants. This study may give further insights into the workings of a particular prehistoric society and the landscape it operated in. It will also provide information pertaining to the relative level of generation, control and application of heat (pyrotechnology) used in past societies.
Material
Reference material used for analysing ashes
Wood
Since Alnus and Fraxinus trees were growing in the area of investigation (Eijskoot et al., 2012), wood samples from these species were obtained from the trunk of full-grown trees growing in a forest in Heemstede, the Netherlands, situated about 5 km from the North Sea coast. The diameter of the samples was about 5–10 cm with the bark left attached.
Peat
In total, three types of peat were investigated, identified as (1) Sphagnum, (2) reed and (3) sedge peat. Sphagnum peat was collected from the Meerstalblok, the central part of the bog reserve at ‘Bargerveen’ (located south-east of Emmen in the north-eastern part of the Netherlands) where peat has been cut and excavated for centuries (Boon et al., 1986). The sample was dated at a 14C age of ca. 3000 BP. This type of peat is composed primarily of Sphagnum, cotton grass (Eriophorum vaginatum) and ericaceous rootlets. Reed peat was donated by the Department of Physical Geography at the University of Utrecht, the Netherlands. The sediment core was obtained from a location near Linschoten, in the middle of the Netherlands. Peat was formed on top of Pleistocene cover sand during the Holocene. The analysed peat sample contained not only reed (Phragmites australis) but also sedge species and dated to ca. 6000 BP. Sedge peat was donated by ADC, Amersfoort, the Netherlands. Sample 1931 from pit 43 was obtained from an excavation along road N57 in the province of Zeeland in the south-western part of the Netherlands. The sample was characterized by undetermined sedge material and cotton grass. Unfortunately, the date of the sample was not determined, but based on samples obtained in the vicinity, the age is comparable to the peat present during the occupation of the VHW site (i.e. Middle Iron Age). The peat samples were dried in ambient air for 1 month prior to use.
Cattle dung
Galloway cattle grazing in a meadow provided dung (CDS), which was collected in the spring of 2008 from the farm at the Medieval Yard Schothorst (which forms part of the Centre for Nature and Environmental Education on the Schothorst Estate, near Amersfoort, the Netherlands). This farm practices traditional Medieval animal husbandry where, in spring and summer, the cattle graze on meadow grass, and mineral supplements are only given when absolutely necessary and then in very small quantities. In addition, cattle dung (CDH) was collected from Highland cattle free roaming in a forest in Heemstede, the Netherlands. Both dung samples were air-dried for 1 month before being used.
Archaeological material
The archaeological site selected for this study is the so-called Vlaardingen – VHW site, which was extensively investigated, and accordingly, much contextual information is available (Eijskoot et al., 2012). In building Vz09-G01, two fire structures can be distinguished, that is, structures 1 and 2 (Figure 1). Structure 1, situated in the living compartment, is described as a hearth consisting of a loam platform wherein pottery sherds have been pressed. The context of this fire structure suggests that it was mainly used to prepare food and as a source of heat to keep the occupants warm. During its use, it was regularly cleaned and raked out. Structure 2, situated inside the cattle compartment, shows a concentration of large quantities of char and ash. Here, the loam platform with pottery sherds is not present. It is assumed that here other activities took place such as drying of peat and dung. The ash samples recovered were used as received. Regarding char, the samples were extracted by the excavators through wet sieving and flotation, and as a result, ash residues and small and friable pieces of char were lost. There is a tendency or bias that only the larger pieces of the recovered char were selected for botanical purposes and identified as mainly Alnus and Fraxinus. Fuel use was apparently not a research question and not further investigated, as, in general, it was assumed that wood would have been used as fuel. Although only a limited number of charcoal samples were still available for this study, it may provide useful information. From fire structure 1, in the living compartment of the building, three ash samples and one char sample were used for further study, that is, ash samples 1039 (28 g), 1154 (8 g) and 1214 (367 g), together with char sample 1606 (4 g; Figure 1). Ash sample 993 (5 g) was obtained just outside this building near the entrance, of which the context shows to be raked out material. From fire structure 2, situated in the cattle compartment, char sample 1555 (4 g) was recovered. Ash sample 5113 (38 g) was obtained just outside building Vz09-G09. The mass of each sample as received from the site and used for the experiments is given in grams.
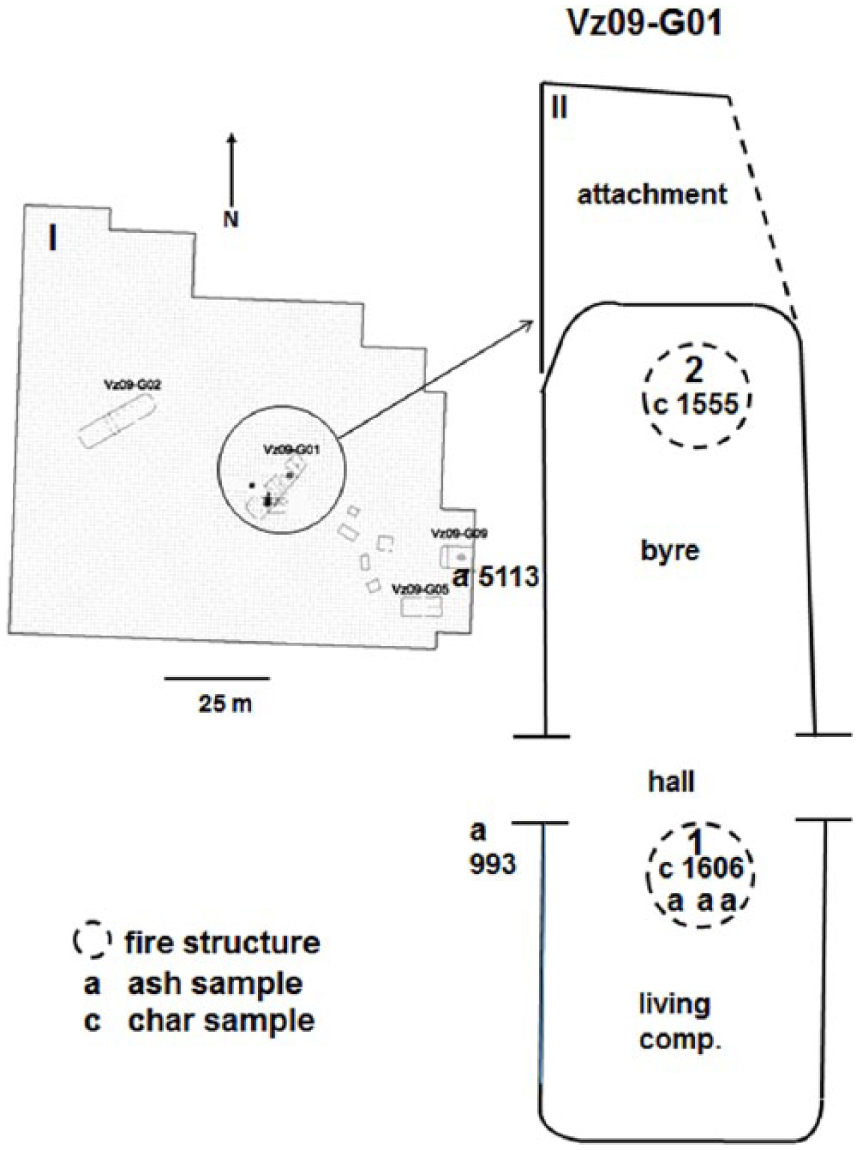
Site of Vlaardingen – De Vergulde Hand West (VHW). (I) General overview of house plans and location of ash sample 5113 and (II) detail of house plan Vz09-G01 showing the location of ash samples 1154, 1039 and 1214 in fire structure 1; ash sample 993 near an entrance of a dwelling; and char samples 1555 and 1606. See also text.
Methods
Ash
Preparation
The reference samples of wood, peat and cattle dung were combusted in an open fire where the temperature was kept below 500°C using a MERA KJ-12 thermometer equipped with a K-type thermocouple. To complete the oxidation of the samples, they were subsequently heated at a temperature of 450°C for 2 hours under air in a muffle furnace. The archaeological ashes were used as received from the site.
Granulometry
The particle size distribution of each ash sample was determined by laser diffraction (Malvern Mastersizer 2000 coupled with a Hydro 2000G inlet). The particles larger than 2000 µm were removed, and the particle size distribution range was measured between 0.1–2000 µm.
Elemental analyses
From each ash residue, five samples were measured, and the mean of the normalized (corrected for organic compounds) results was calculated. The equipment used is the HH x-ray fluorescence (XRF) Thermo Scientific Niton XL3t device with GOLDD detector equipped with a silver anode operating at a maximum of 50 kV and 40 µA. This handheld device was used in the laboratory, and the samples were measured on a stable flat surface. This device is well suited for the measurement of up to 25 elements simultaneously in the analytical range between sulphur (atomic number 16) and uranium (atomic number 92). Light elements (magnesium, aluminium, silica and phosphorous) can also be measured with the same detector using a helium purge. The samples were measured in bulk mode. The device was factory calibrated.
Principal component analyses
The FOMpyroMap multivariate programme and the ChemomeTricks programme for the calculations were used (Klap et al. 1996).
Microscopic preparation for silica and calcium compounds
Opal phytoliths
It is known that opal phytoliths are strongly present in ash of grass in which the concentration can be up to 20 times higher relative to ash of wood (Karkanas et al., 2002). To increase comparability, wood samples were chemically treated to remove non-siliceous components (i.e. highly fragmented char), thus increasing relative opal phytolith concentrations and greatly improving identification. It is noted that only the ash samples selected for opal phytolith analyses were chemically treated. For the calcium oxalate crystals and spherulites analyses, no chemical treatment was applied. Chemical treatment was carried out as described by Albert and Weiner (2001). In short, 50 mg of each ash sample was treated with a mixture of 5 mL 3N HCl and 5 mL of 3N HNO3 for 30 minutes at 100°C. The insoluble fraction was washed three times with deionized water. Next, the sample was placed in a glass beaker, and 10 mL of 30% H2O2 was added. The H2O2 evaporated on a hot plate at 70°C and the residue dried in a stove at 60°C. For the microscopic study, 4 mg of each sample was thoroughly mixed with 2 mL deionized water. From each mixture, 0.05 mL was mounted on a glass slide. In this way, each slide contained an equal amount of ash, that is, 0.1 mg, which made a comparison between the different ashes possible. These slides were examined using trans-illumination under a Leica DM6000 M microscope. From each slide, five field views were selected at random and photographed. The field views are prepared using a magnification of 250× (see also scale bars in relevant pictures). This study uses a quantitative approach in the sense that for the opal phytoliths, a ‘field of view’ system is used whereby the number of opal phytoliths is counted in each of five at random selected fields of view. Next the mean (with standard deviation (SD)) is calculated using the in this manner counted number in the five field views and is referred to as the frequency. To obtain the larger particles (leaf surfaces) present in ashes flattened out for photography, ashes were suspended in propylene oxide, and drops were spread on microscopic slides and evaporated. To make microscopic preparations, the dried ashes were covered with Epon and a cover slip and dried in an oven at 40°C.
Calcium oxalate crystals
As described above, 4 mg of chemically untreated ash from the reference samples was mounted on a glass slide and studied under the Leica microscope.
Spherulites
For this study, chemically untreated ash from the cattle dung samples was mounted on glass slides and studied using a Zeiss Axioskop 40 microscope under cross polarized light (XPL).
Siliceous aggregates
The presence in the glass slides is noted.
Char
Reflectance
The char samples were first examined by incident light microscopy using a Leica KL200 stereomicroscope and a Leitz Ortholux II microscope using standardized settings for hardwood determination (IAWA Committee, 1989). From these observations, char samples were selected at random to be examined by reflective light. Reflectance measurements were performed on five such specimens of char samples 1555 and 1606, which were embedded in resin and polished in accordance with standard methods defined in ISO 7404, part 2 (2009; for details, see Braadbaart and Poole, 2008, Braadbaart et al., 2012). This method allows to study samples smaller than 1 mm. Reflectance was measured under oil immersion at a wavelength of 546 nm using the above described Leitz microscope. For each specimen, a total of 100 randomly performed reflectance readings were taken (to obtain a statistically acceptable population), and the mean reflectance was calculated. Reflectance measurements were performed in accordance with standard methods, which have been extensively described in Braadbaart (2004). The derived temperatures were determined using the calibration curves of Braadbaart and Poole (2008). In addition, the structural morphology of each archaeological char was examined under reflected light and oil immersion using a Leitz motorized DMLA microscope equipped with an xyz stage and a Basler video camera. For this purpose, the same samples (embedded in resin, as described above) were used (Figure 2).

Photographs of char structures under reflected light, scale bar = 60 µm. (a) Sample 1555 showing a typical cell structure of wood, (b) sample 1555 showing a typical structure whereby a large part does not show the typical cell structure of wood but a homogeneous structure and (c) sample 1606 shows a compact structure representing charred peat or dung, with hair-like structures (arrow).
Results
Reference material – ash
Elemental analyses
Wood
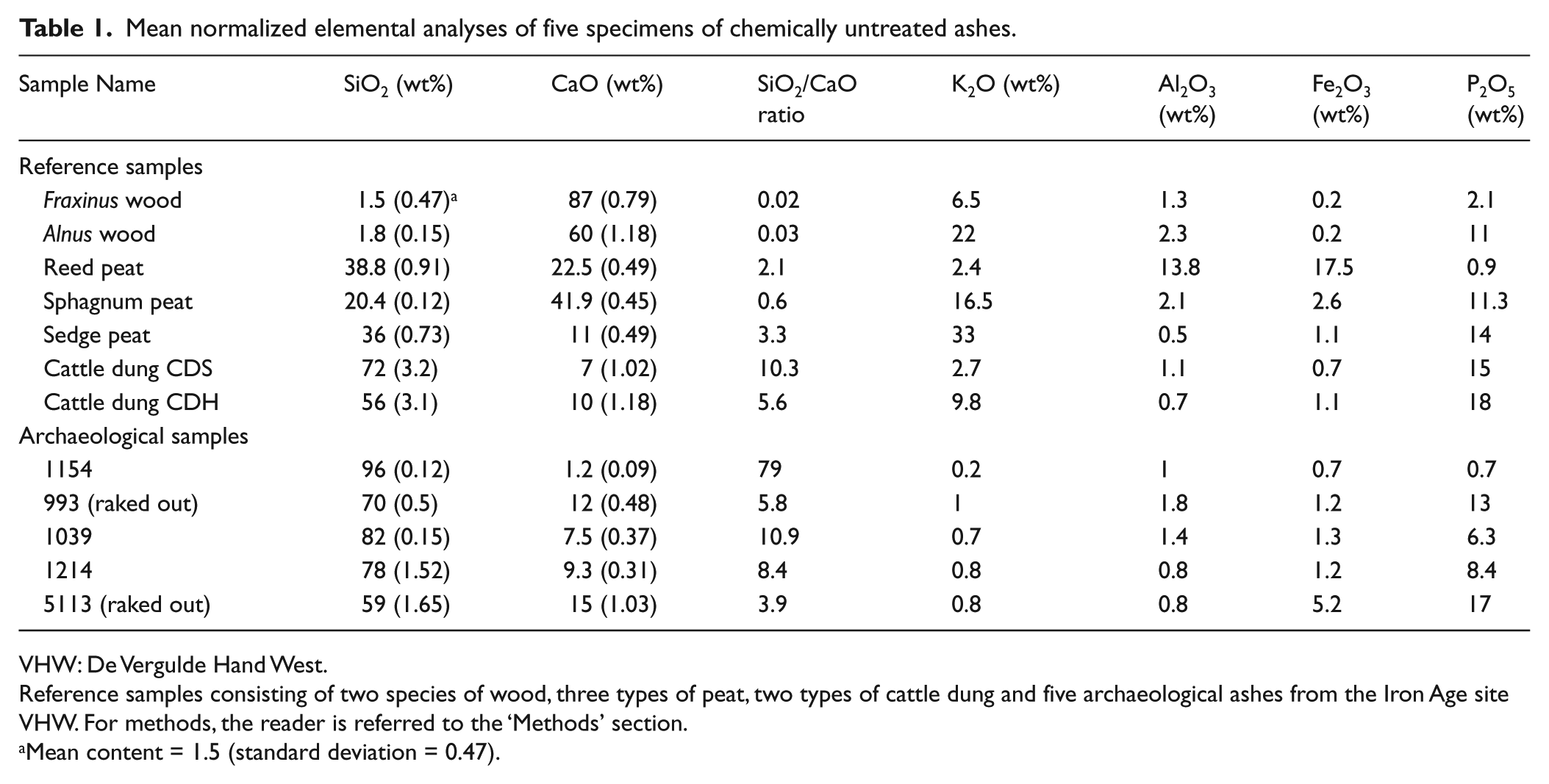
The Si content of the wood ash samples was far lower compared with the Ca content, resulting in a low SiO2/CaO ratio of 0.02 and 0.03 (Table 1).
Mean normalized elemental analyses of five specimens of chemically untreated ashes.
VHW: De Vergulde Hand West.
Reference samples consisting of two species of wood, three types of peat, two types of cattle dung and five archaeological ashes from the Iron Age site VHW. For methods, the reader is referred to the ‘Methods’ section.
Mean content = 1.5 (standard deviation = 0.47).
Peat
The Si content in the reed and sedge peat ash samples was higher than the Ca content. For sphagnum peat, the Ca content was higher than the Si content. This results in a SiO2/CaO ratio ranging from 0.6 to 3.3. The contents of the other measured elements vary considerably in the three peat types. Al and Fe content in reed peat ash was relatively high compared with the other two peat ash samples, while the K and P content was lower (Table 1).
Cattle dung
The Si content in both samples was rather high, while the Ca content was somewhat lower compared to that of peat ash. It means that the SiO2/CaO ratio was high and varies between 5.6 and 10.3 (Table 1).
Microscopic analyses
Wood
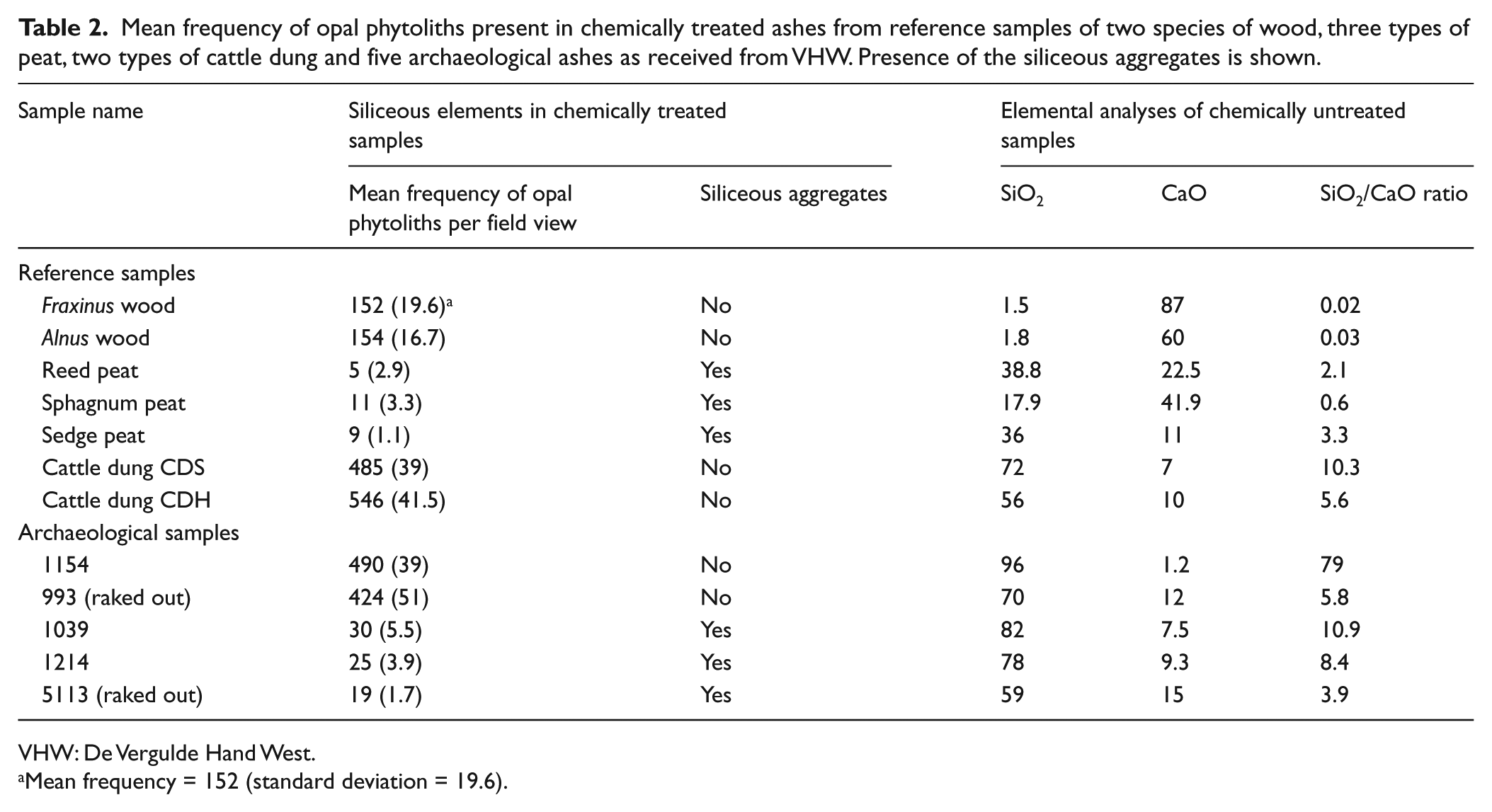
Opal phytoliths. The frequency of the opal phytoliths in the field views in Alnus ash was 154 (SD = 16.7) and for Fraxinus 152 (SD = 19.6; Table 2). The main morphotypes were globulars (arrows in Figure 3). In both wood species, blackish aggregates were present, which appeared under XPL as mainly small (5 µm) grains of calcium carbonate.
Mean frequency of opal phytoliths present in chemically treated ashes from reference samples of two species of wood, three types of peat, two types of cattle dung and five archaeological ashes as received from VHW. Presence of the siliceous aggregates is shown.
VHW: De Vergulde Hand West.
Mean frequency = 152 (standard deviation = 19.6).

Photograph of a field view of chemically treated ash from modern wood (Fraxinus).
Calcium oxalate crystals. The crystals were particular rich in the wood (chemically untreated) ash samples.
Spherulites and siliceous aggregates. Both features were not observed in the wood samples.
Peat
Opal phytoliths. Remarkably, the ashes of peat types originally identified as reed and sedge containing did not harbour any of the corresponding opal phytoliths (field views 5 (SD = 2.9) and 9 (SD = 1.1)). For the sphagnum peat sample, the field view was 11 (SD = 3.3; Table 2). In the three samples, some elongates were observed. The remainder features were isolated, often indistinct, miscellaneous forms and fragments.
Calcium oxalate crystals. An occasional crystal was observed in the three peat samples.
Spherulites. No spherulites were observed.
Siliceous aggregates. The main features visible in the three peat ashes were siliceous aggregates, often opaque from organic material occluded within these aggregates (Figure 4). Under XPL, the aggregates do not show any indications of the presence of calcium carbonate.

Photograph of a field view of chemically treated ash from peat (reed).
Cattle dung
Opal phytoliths. The frequency of the opal phytoliths in the field views was 485 (SD = 39) for Heemstede dung (CDH) and 546 (SD = 41) for Schothorst dung (CDS). The two treated modern cattle dung ashes were characterized by a high abundance of typical grass morphotypes (Figure 5; Twiss, 2001).
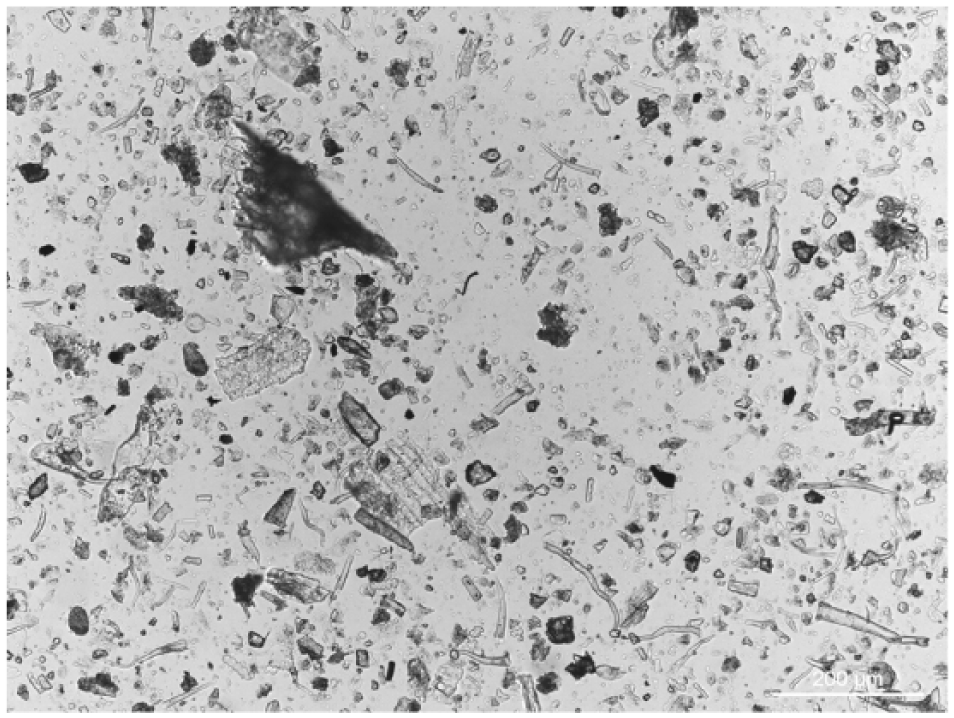
Photograph of a field view of chemically treated ash from modern cattle dung (CDH).
Calcium oxalate crystals. Just an occasional crystal was observed in the two samples.
Spherulites. For this investigation, chemically untreated ash samples have been used. In both dung ash samples, the characteristic shapes and patterns for faecal spherulites commonly seen under XPL (Canti, 2003) were not observed.
Siliceous aggregates. No aggregates were observed.
Archaeological material – ash
Granulometry
The particle size distribution of the samples showed that the size of the particles is between 10 and 100 µm for 80 wt% of the total sample, which is characteristic for ash particles (Braadbaart et al., 2012; Brochier, 1983).
Elemental analyses
Samples 1154, 993, 1039, 1214 and 5114
The results of the elemental analyses performed on the five ash samples are summarized in Table 1. The five samples were all characterized by a SiO2/CaO ratio ranging from 79 to 3.9 (Table 1). Sample 1154 had the highest Si content and the lowest Ca content. The elemental analyses have been carried out on ash samples that were not chemically treated. To verify the influence of the chemical treatment on the samples for opal phytolith analyses, they were analysed after this treatment. The results show that the SiO2 content of all samples increased to around 95 wt%.
Microscopic analyses
Sample 1154
Opal phytoliths. The frequency in the field views is 490 (SD = 39). The field image is characterized by a high abundance of well-known grass-related and sedge-related opal phytoliths (Figure 6a). In addition, rather large pieces of plant tissues were found as parts of vascular tissue and leaf surfaces. Typical grass-type stomata were observed in conserved leaf surfaces (Figure 7).
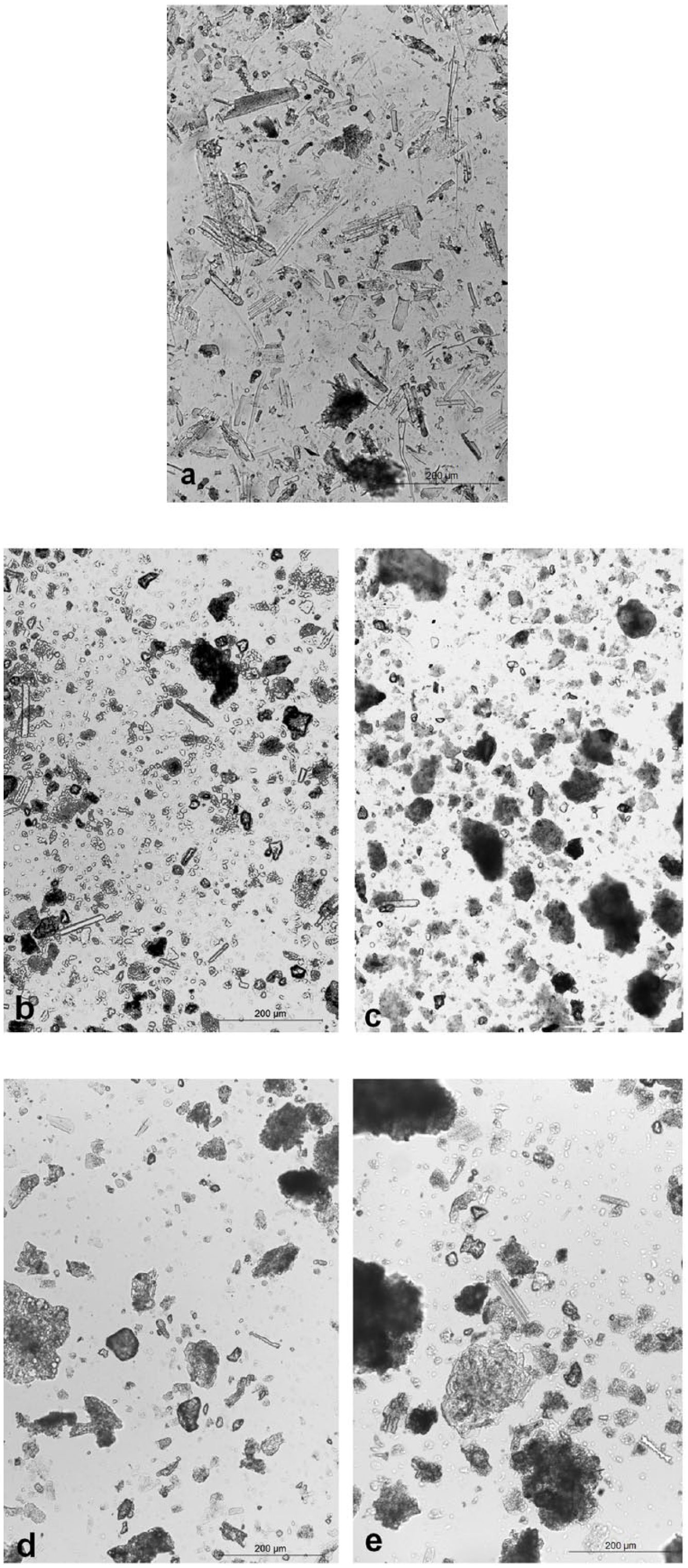
Photographs of field views of chemically treated archaeological ash samples (a) 1154, (b) 993, (c) 1039, (d) 1214 and (e) 5113.
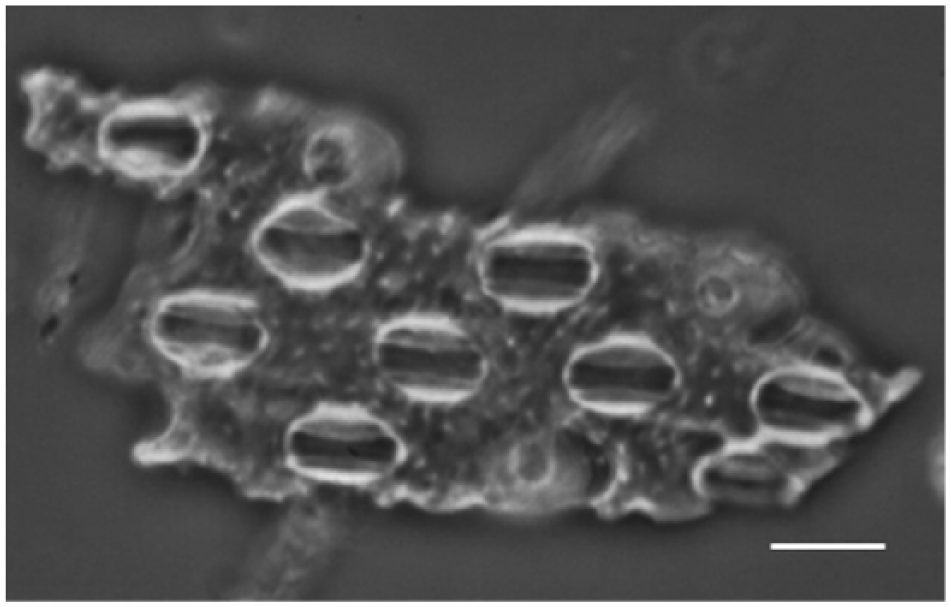
Leaf structure with typical grass stomata from archaeological ash sample 1154. Scale bar = 10 µm.
Calcium oxalate crystals. No crystals were observed.
Spherulites. No characteristic shapes and patterns of spherulites were observed under XPL.
Siliceous aggregates. No aggregates were present.
Sample 993
Opal phytoliths. The frequency in the field views was 424 (SD = 51). Compared with sample 1154, identical morphotypes were present.
Calcium oxalate crystals. No crystals were recognized.
Spherulites. No characteristic shapes and patterns of spherulites were observed under XPL.
Siliceous aggregates. No aggregates were present.
Samples 1039, 1214 and 5113
Opal phytoliths. The frequency of the opal phytoliths in the field views was 30 (SD = 5.5) for sample 1039, 25 (SD = 3.9) for sample 1214 and 19 (SD = 1.7) for sample 5113. Only a very few elongates were observed.
Calcium oxalate crystals. No crystals were recognized.
Spherulites. No spherulites were observed.
Siliceous aggregates. The field images of these samples (Figure 6c–e) are almost identical and show mainly siliceous aggregates sometimes covered with black opaque material.
Archaeological material – char
The two char samples, 1555 and 1606, were composed of multiple specimens of charred material ranging from 2 to 5 mm. They were black and friable. From sample 1555, related to fire structure 2 in the cattle compartment (Figure 1), 173 fragments were examined under incident light microscopy of which 153 (88%) identified as Alnus and Fraxinus. In total, five of these fragments were selected for reflective microscopy. Here, the characteristic cell walls of wood were observed (Figure 2a). It is noted that in some samples, the cell wall material from numerous cells seemed to have been transformed into a homogeneous mass showing no cells and clearly different from the characteristic wood cell structure (Figure 2b). The mean values of the reflectance readings for these fragments ranged from 0.61%Ro to 4.426%Ro (SD = 0.02–0.31%Ro). These values correspond to temperatures in the range of 370–670°C. From sample 1606, recovered from fire structure 1, a total of 107 fragments were examined under incident light microscopy, of which 52 fragments (49%) were identified as Alnus and Fraxinus, while 55 fragments could not be identified. To examine also other charred samples, five of the unidentified fragments were selected for reflective microscopy and showed a compact, white reflective groundmass consisting of charred, partly decomposed plant material (Figure 2c). The characteristic structures that have been previously attributed to charred hairs were observed (Braadbaart et al., 2012). The mean values of the reflectance readings for these fragments range from 1.397 to 3.590 (SD = 0.05 to 0.29), corresponding to temperatures of 370–580°C.
Discussion
Earlier investigations show that the landscape of the investigated Iron Age area has mainly peat or clay soils where trees were dearth and where people and cattle were sheltered under the same roof (Eijskoot et al., 2012). Peat and cattle dung, both excellent and efficient fuels, when dried (Braadbaart et al., 2012), were thus readily available near the farms This means that it is quite plausible that these fuel types were used by Iron Age farmers to make their fires instead of, or at least together with, wood. On the other hand, it is also possible that the cattle dung was used by the farmers to improve the growing conditions of the crops they were growing.
Reference samples
The main diet for cattle was grasses, although cattle foraging in a forest may also eat leaves and twigs. Grasses have a high relative Si content and contain a wide variety and well-documented types of opal phytoliths (Piperno, 2006). The results of this study show that opal phytoliths are still abundant in the cattle dung samples after having passed their intestinal system (Table 1). The SiO2/CaO ratio is high and ranges from 5.6 to 10.3. Wood ash was characterized by a low SiO2/CaO ratio of 0.02 and 0.03 (see also Canti, 2003, and Braadbaart et al., 2012). It is well known that the abundance of opal phytoliths is rather low in wood ash (Karkanas et al., 2002), while their morphotypes also differ from grass opal phytoliths. The strong difference in abundance of opal phytoliths between the cattle dung ash and the wood ash is also clearly observed in their frequency obtained from the field views (see methods). Since the frequency is calculated from the amount of counted phytoliths in these field views it is considered as a quantity. To distinguish between the remaining ashes of both types of fuel, this quantitative method is very useful as shown by the results of this study. Moreover, for archaeologists who study fuels used by ancient societies, it is a practical and relatively easy method.
Therefore, using the investigated properties of the ashes, that is, the SiO2/CaO ratio and the frequency of opal phytoliths in a field view, is a practical method for distinguishing between the use of wood and dung as fuel (Table 2).
The reference samples of both wood and cattle dung are modern materials. In contrast, peat is a fossil fuel that has been affected by diagenetic processes. Moreover, the three types were recovered from different sites in the Netherlands away from the area under study because most of the peat in the VHW area has been removed in the past for different reasons. This may have an influence on the elemental composition and explain the measured differences (Table 1). The resulting SiO2/CaO ratio for peat ranges from 0.6 to 3.3, higher than the ratio of wood and much lower than cattle dung ratios. Regarding the presence of opal phytoliths, it is striking that no typical reed, sedge or grass morphotypes were observed in the field views of the peat types. The major component observed in the field views are silica aggregates, which were characterized by an amorphous Si-rich, Al-rich, Fe-rich and K-rich material (Albert and Weiner, 2001). In total, three obvious explanations exist for the absence of opal phytoliths in peat and accordingly in their ash: (1) there was no silicon available to form the opal phytoliths, (2) they have been removed by the flow of ground water or (3) they disintegrate for several reasons during their stay in the soil. The first reason could be valid in raised peatlands, where only nutrient-poor rain water is present to feed plants, and in turn, no Si is available for the deposition of the opal in their cells. This is at least true for the VHW site investigated here. However, it is known that mosses and some sedge taxa do not seem to contain opal phytoliths, which could be the explanation for the lack of opal phytoliths in some peats (Bozarth, 1993; Ollendorf et al., 1987). The second reason seems rather obvious when the peat is situated in a wetland. Regarding the third reason, it is generally accepted that quartz (SiO2) dissolves only in strongly alkaline environments (pH > 9), but it cannot be ruled out that under neutral and reducing conditions, the opal comprising phytoliths may dissolve or disintegrate (Williams and Crerar, 1985; Williams et al., 1985; Thorn, 2004–2005). In this regard, it is noted that the solubility of opal-A is 17 times higher than that of quartz (Fraysse et al., 2006). Other reasons may be the presence of a considerable amount of Al (Carnelli et al., 2002; Wüst and Bustin, 2003) and the process of silica recycling (Alexandre et al., 1997; Derry et al., 2005). It is obvious that more research is necessary to explain the presence of the siliceous aggregates and the rather low frequency of opal phytoliths in the peat samples.
Spherulites, often present in animal dung (Canti, 1998; Shahack-Gross, 2011), appear to be lacking in the ash of both cattle dung reference samples. This is in accordance to the observations of Brochier (1996, Table 1), which show an absence of spherulites in cattle dung.
In summary, phytolith and chemical analyses from reference ash samples have a high potential for the identification of the origin of archaeological ashes (Table 2).
Archaeological samples
The elemental analyses of the five archaeological ash samples showed high SiO2/CaO ratios (Table 1), with sample 1154 exhibiting a Si content of 96 wt%, the highest of the group. The Ca content was still relatively high in the investigated samples, which shows that the Ca compounds may have been rather stable during their time in the soil. This is in accordance with the findings of Vassilev et al. (2013 and references cited therein), showing that these compounds are not easily dissolvable, even in the acidic environment usually existing in peat. In all five samples, the percentage of the elements K and Al is rather low suggesting that compounds containing these elements have dissolved or otherwise reacted during their stay of about 2000 years in the soil. This is not valid for the elements Fe and P, whereby the percentage of P did not seem to be affected. The Si and Ca contents were still comparable to those derived from the reference dung samples. This indicates that these ashes, based on their high SiO2/CaO ratio, likely originated from dung. However, peat cannot be completely excluded, depending on the type of peat. This is confirmed by the principal component analysis (PCA) performed on the elemental analyses (Figure 8). Here, the two raked out samples (993 and 5113) have a tendency towards peat because of their relative high P content, but this could be also the result of the fact that they were dumped on P-containing soil, raked out from inside the house, outside the dwellings.
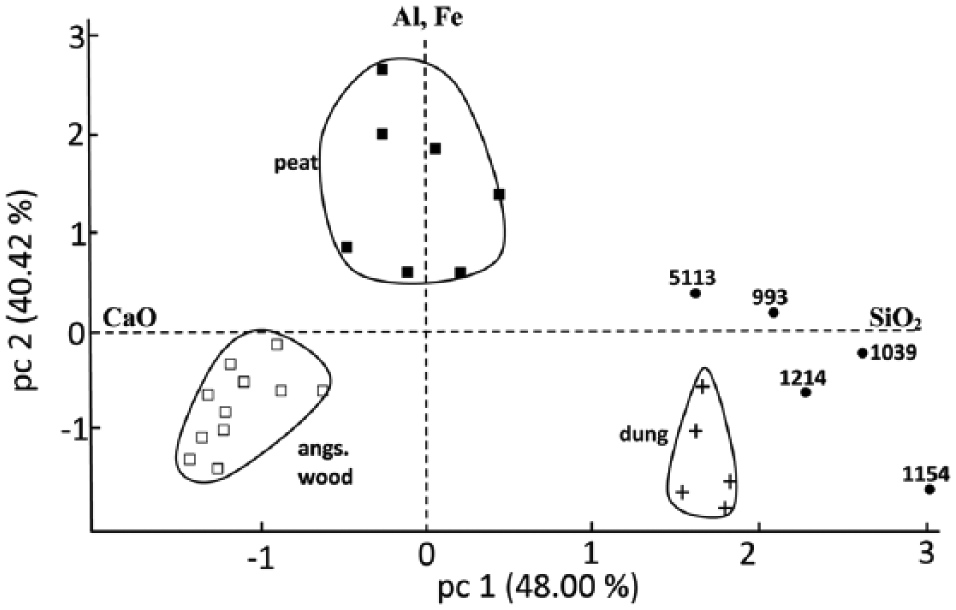
Score plot of principal component analysis of the chemical analyses of the reference and archaeological ashes. The archaeological ashes are the mean of five measurements. The horizontal 0-axis shows the directions where CaO (left) and SiO2 (right) tend to cluster in the discriminant space.
The microscopic studies on ash samples 1154 (Figure 6a) and 993 (Figure 6b) showed high frequencies of grass phytoliths in their field views, and when compared with the field view of the modern dung ashes, they were strikingly similar (Figure 5; Table 2). The field views of samples 1039, 1214 and 5113 (Figure 6c, d and e) showed mainly siliceous aggregates, often black coloured (Table 2). The resemblance to the field views of the reference peat ashes was striking. Since samples 1154 and 993 are most likely derived from cattle dung they were selected to study under XPL for the presence of spherulites. None were observed, as was also the result in the reference samples. In summary, the results of the elemental analyses and the microscopic studies are not completely compatible. The elemental analyses are clear about the use of dung as fuel, while the presence of siliceous aggregates in three samples makes the use of peat also possible. This shows the importance for a study to explain why and how these aggregates are formed.
Char sample 1606 from fire structure 1 in the living compartment consists of 49% of charred wood. Reflective light was used to identify the other half as having been derived from peat or dung. The presence of hair-like features (Figure 2c) makes it most likely that these fragments originate from cattle dung (Braadbaart et al., 2012, Figure 4). The results of the analyses on the ash samples 1039, 1214 and 1154 and the char sample 1606, all retrieved from fire structure 1 in building Vz0G-G01, are apparently in contrast. The ash samples indicate that the fuel used was cattle dung and/or peat, whereby no indications of the use of wood as fuel were found. However, the char sample shows that around 50% of the investigated fragments originate from wood. This disparity could be explained by the amount of ash present in the different fuels. Cattle dung may contain 20–40 wt% of ash; in peat, the amount of ash ranges between 3.5 and 6 wt%, while the ash content of wood is only around 1–2 wt% (Braadbaart et al., 2012, and the references cited therein). This may cause wood ash to be very poorly represented in the total amount of ash recovered from a fire structure where more types of fuel were used, and thus, its characteristic properties can easily be missed in the retrieved ash samples. Regarding the char, the more friable charred peat and dung fragments may have been lost during the various excavation processes, such as flotation and sieving. In addition, there is the tendency by excavators to recover only the larger pieces of char, which originate mainly from wood as explained above, to make botanical identification of the species easier. This creates a bias towards the question asked in this study: which fuel has been used by the farmers?
From fire structure 2 in the cattle compartment, only char is available. So no comparison with ash samples can be made. The fragments of sample 1555 (Figure 2a) showed all the characteristics of charred wood under reflective light, which corresponds to the observation under incident light that almost 90% is charred wood. In Figure 2b of this sample, a feature was observed whereby the cells of wood were not observed, instead a homogeneous mass was present. It is suggested that these areas, often called ‘glassy or vitreous’ matter, are attributed to heating conditions during the production of a material such as charcoal. Although the exact mechanism of how these features are formed is not clear yet, it seems reasonable to assume that one or more of the constituting compounds of the cell walls has become viscous and as a result lost their cell structure (Braadbaart et al., 2016).
The temperature inferred from the reflectance measurements on wood char fragments from sample 1555 varied from 370°C to 670°C. The higher temperature can explain the homogeneous areas, without the cell walls. They could be the result of the preparation of wood, dung or peat by heating into a new and more efficient fuel, often referred to as charring (such as industrial charcoal), with a 50% higher heating capacity. In the latter case, temperatures of 500°C and higher are necessary (Braadbaart et al., 2016). The fuels prepared in this way have excellent heating properties and cause less smoke. It is also obvious that the dung and peat fuels have to be dried and thus heated anyway before they can be used as fuel. This may have been a problem when the drying is performed in open air under the existing climatic conditions in the studied area. When being dried inside, it gives a terrible smoke, so the inhabitants may have decided to perform this in the cattle compartment and probably using wood as an additional fuel. It cannot be excluded that this process may have been done together with other technological activities related to forging iron. Moreover, peat and dung are not only more efficient fuels compared to wood, but their fires are easy to maintain over longer periods of time with a more consistent temperature and thus extremely useful to prepare food (Braadbaart et al., 2012). The temperature in sample 1606, from the living compartment, is between 370°C and 580°C. So it is suggested that the fire structure in the living compartment was used for cooking and heating at a temperature in the order of 400°C using the dried or charred peat or dung as fuel, which were prepared in the cattle compartment (Braadbaart et al., 2016). The temperature of 580°C agrees with the temperature measured in the cattle compartment since the here-prepared chars only showed the higher reflectance values related to around 600°C.
In this case, the results show that even despite the limited number of samples and the fact that the results of the analyses from ash and char remains did not provide completely comparable results, this approach may provide interesting information regarding the living conditions of the inhabitants involved. Char as well as ash are extremely vulnerable to excavation processes (Matthews, 2010). Chars from dung and peat are friable and easily destroyed, while the small ash particles are easily lost. Special care needs to be taken to circumvent these problems. Sieving and flotation should be avoided as much as possible when studying ancient fuel remains. It is suggested that subsampling by hand of collected bulk sediment should accompany the normal excavation activities, which involve flotation on a large scale. Adopting an excavation protocol for hearths and other fire structures avoiding wet sieving and flotation and analysing the remains of the fuels, valuable information can be obtained. Information can be used to shed more light to an important objective of archaeological studies, which is how these ancient societies dealt with fuels and their fires and the relative level of generation, control and application of heat (pyrotechnology).
Conclusion
The applied methods for ash, that is, elemental analyses and a quantitative approach for opal phytoliths and reflective microscopy for char samples smaller than 1 mm, have a high potential for the identification of the type of fuel used in archaeological fire structures. Thus, an integrated application of phytoliths, geochemical and char analyses is recommended. It will provide useful indicators of anthropogenic fire usage and function in regard to the available fuel resources and types and thus palaeoenvironment. In an area where more types of fuel are available, it is recommended to use both char and ash samples. The reason for the absence of opal phytoliths in the peat reference ash samples used in this study is unknown and warrants further examination. Special attention should be given to the excavation processes since char from peat and dung is more friable than char from wood and will be easily lost together with the relatively small ash particles (Braadbaart et al., 2012; Matthews, 2010). Adopting a protocol for the excavation of fire structures avoiding wet sieving and flotation or for this purpose subsampling by hand of collected bulk sediment should accompany the normal excavation activities, which involve flotation on a large scale. Although only a limited number of samples were available, some interesting suggestions are made related to the conditions by which the farmers obtained their necessary heat and explain the function of the fire structures present in both compartments. Wood is a less-efficient fuel type compared with peat and dung, and the latter fuels are easy to maintain over longer periods of time with a more consistent temperature and are extremely useful to prepare food (Braadbaart et al., 2012). So one can imagine that the fire structure in the living compartment was used for cooking and heating using peat or dung as fuel. Before being used, both fuels have to be dried at least, and it seems, therefore, reasonable to assume that the fire structure in the cattle compartment was used to prepare the fuels used in the living compartment. The applied methods are especially valid for circumstances as they exist in the investigated archaeological site situated in a peatland where three fuel types are usually readily available but should also be applied at sites where it is often assumed a priori that wood is the only fuel used to light a fire. It is suggested that apart from botanical identification an important part of an archaeological investigation are the in this study suggested methods to analyse the remains of fuels, which in this study even contained leaf structures with typical grass stomata. This could add much valuable information related to not only how ancient societies selected these fuels for their heat sources but also the relative level of generation, control and application of heat (pyrotechnology).
Footnotes
Acknowledgements
The technical assistance of Eric Mulder (Faculty of Archaeology, Leiden University) and Gerda Lamers (IBL, Leiden University) with the phytolith research and of Erica van Hees (Faculty of Archaeology, Leiden University) with identification of chars is highly acknowledged. We thank Harry Veld of Deltares (Utrecht, the Netherlands) for his technical assistance with the reflectance measurements. The comments of Luc Vrijdaghs (Centre de Recherches en Archéologie et Patrimoine (CReA-Patrimoine), Université Libre de Bruxelles, Belgium) and of three anonymous reviewers, which led to a considerable improvement of this manuscript, are greatly appreciated.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
