Abstract
This study presents the synthesis of a chabazite photocatalyst (Cu/TiO₂/SAPO-34) using an innovative hydrothermal method for the rapid degradation of pharmaceutical and organic pollutants. The material was characterized using scanning electron microscope, energy dispersive spectroscopy, X-ray diffraction, Fourier transform infrared spectroscopy (FTIR), UV-vis diffuse reflectance, and photoluminescence spectroscopy for morphology, structure, molecular bonding, and optical properties. The Cu/TiO₂/SAPO-34 was photoelectrochemically characterized using linear sweep voltammetry (LSV), electrochemical impedance spectroscopy (EIS), Photocurrent (PC) transient curves, and Mott–Schottky (MS) analysis. The pharmaceutical and organic pollutant removal ability of the photocatalyst was investigated through a paracetamol and methylene blue photodegradation test, using ultraviolet radiation. The Cu/TiO₂/SAPO-34 photocatalyst exhibited superior electrochemical, light absorption, structural, and photocatalytic properties compared to TiO₂. It demonstrated reduced charge transfer resistance (2007 vs. 3931 Ω), higher photocurrent density (46 vs. 9 μA/cm2), and improved charge migration and optically generated charge carrier separation, leading to improved photoelectrochemical performance. Structural analysis confirmed the formation of the composite with a larger average crystallite size (17.56 vs. 13.11 nm for TiO2). FTIR and EDX analyses verified the presence of Cu, TiO2, and SAPO-34 in the mix. Optical analysis showed reduced bandgap energy (2.61 eV vs. 3.09 eV for TiO2), enhanced UV and visible emissions, and early infrared absorption. The composite outperformed TiO2 in degrading pharmaceutical and organic contaminants, showcasing its potential for advanced photocatalytic and photoelectrochemical applications. The material can suppress biodegradation, preserve the natural world, enhance solar energy absorption, self-clean medical equipment, and purify wastewater from pharmaceutical contaminants.
Keywords
Introduction
The rapid increase in pharmaceutical and organic contaminants in water resources has become a pressing global concern due to their enduring toxicity and resistance to conventional treatment methods. Human activities, including the discharge of domestic wastewater, industrial processes, agricultural runoff, healthcare practices, and improper waste disposal, are the primary sources of pharmaceutical and organic contaminants in water resources. 1 Pharmaceutical contaminants in water originate from the human excretion of unmetabolized medicines, improper drug disposal, and wastewater from households, healthcare facilities, and pharmaceutical manufacturing plants. Additionally, veterinary medicines from livestock and aquaculture contribute through runoff and discharge. 2 Organic contaminants arise from domestic products such as detergents, industrial discharges from various industries, agricultural inputs like pesticides and fertilizers, landfill leachate, and the natural decomposition of vegetation. 3 The persistence of these pollutants poses significant risks to drinking water quality, human health, and aquatic ecosystems, particularly in regions with inadequate wastewater treatment infrastructure, thus creating a need for sustainable solutions.
Advanced photocatalytic materials have gained attention as sustainable solutions for environmental remediation, offering efficient degradation of emerging pollutants under solar energy. Among these, TiO₂ has been widely studied; however, its wide bandgap and limited visible-light activity constrain its practical performance. To overcome these limitations, composite photocatalysts incorporating transition metals and porous supports are being researched to enhance light absorption, charge separation, and catalytic efficiency. 4 The goal is to remove pollutants such as pharmaceutical waste and organic contaminants from surfaces and contaminated water to safeguard the environment and the ecosystem. Most traditional techniques have disadvantages, including the production of secondary pollutants, partial mineralization, and occasionally high costs. One more sophisticated pollutant degradation technique is photoelectrochemical oxidation, which produces potent oxidants that oxidize organics in wastewater nonselectively by utilizing both light and applied potential. 5 Photocatalysis and photoelectrochemical technologies are recognized as among the most effective methods to facilitate wastewater treatment and to prevent toxins and pollutants from adhering to all surfaces to create a safer and healthier atmosphere, reduce the effects of pollution, and protect the climate.6–8
Photoelectrochemical oxidation effectively separates light-generated charges to prevent carrier recombination by migrating electrons to the cathode through the applied bias potential as photons illuminate the catalyst. This averts the disadvantages of electron-hole recombination and catalyst recovery challenges with conventional photocatalysis. 9 Among the photo-responsive materials being researched, TiO2 is singled out due to its availability, nontoxicity, and affordability. However, the huge band gap of TiO2 prevents it from absorbing enough visible light in the UV spectrum, limiting its photocatalytic efficiency.10,11 This has prompted research to focus on developing other visible light-active catalysts with small band gaps and low electron-hole recombination or doping of TiO2 to narrow its bandgap and boost its photocatalytic and photoelectrochemical characteristics.12,13
While most scholars concentrate on doping TiO2 alongside other metals or metal oxides, Zeolites like SAPO-34 are increasingly explored for photocatalysis because of their large surface area, well-ordered porosity, unique microstructural features, high-level adsorption, and exceptional high-temperature stability. They provide effective support for semiconducting photocatalysts. 14 Pan et al. modified SAPO-34 zeolite with Mo-oxide in a simple wet-impregnation process. They found that considerable stable photocatalytic activity may be achieved by adjusting the acidity strength of catalysts. 15 Making use of clear-cut BiVO4 and an amended SAPO-34 porous framework, Wang and collaborators manufactured a dual-function photocatalyst. The developed photocatalyst demonstrated improved ability to transfer electrons, inhibited recombination of electrons and holes, and demonstrated high activity for the photocatalytic destruction of formaldehyde pollutant. 16 Several scholars have also studied the Cu/SAPO-34 blend and verified that it is an appropriate catalyst but needs hydrothermal functionalization to be stable.17,18 Many reports on doping metal oxides with SAPO-34 zeolite to create enhanced photocatalysts have not thoroughly examined their detailed photocatalytic and electrochemical properties, nor adequately explored their application in degrading pharmaceutical waste and organic pollutants.
This study introduces a Cu/TiO₂/SAPO-34 composite that combines the photo-responsive properties of copper with the structural advantages of silicoaluminophosphate to enhance the light-driven performance of TiO₂ in pollutant degradation. The objective of this study is to synthesize and characterize a chabazite-based Cu/TiO₂/SAPO-34 photocatalyst using a hydrothermal method, and to evaluate its photocatalytic and photoelectrochemical performance, particularly its efficiency in degrading organic and pharmaceutical pollutants compared to conventional TiO₂. Such multifunctional photocatalysts offer significant potential for sustainable water treatment and environmental protection by efficiently removing contaminants and addressing challenges linked to ecosystem degradation and climate change.
The experiment
Materials used
Each of the materials used in this study was obtained from Sigma Aldrich and included the following: 40% Colloidal silica (SiO2) suspension in water, 98% aluminum oxide (Al2O3), 85% aqueous solution of phosphorus pentaoxide (P2O5), 99% dipropylamine, 97% titanium IV isopropoxide (TTIP), 35% tetraethylammonium hydroxide in H2O, 99% ethanol, 99% copper (II) nitrate trihydrate, 70% nitric acid, and sodium hydroxide pellet (40.00 g/mol). All reagents were employed without any further purification.
Material synthesis
The nanoparticles used in this study were developed in three-fold. First, TiO2 was synthesized in a heat-assisted sol-gel process. 19 The TTIP was mixed with an ethanol solution in a 1:5 volume proportion. A stock solution made of a 1:50 combination of nitric acid and water was added drop by drop into the hydrolyzing TTIP and ethanol mixture after 30 minutes of action on a magnetic stirrer. The new mixture was left stirring at 60 °C for 2 hours and then cured at 100 °C for a day in an Ecotherm oven. The obtained TiO2 powder was dried and annealed at 300 °C in a furnace for 2 hours.
In the preparation of the Cu/TiO₂/SAPO-34 photocatalyst, SAPO-34 nanosheets were first produced using an adapted hydrothermal approach previously reported. 20 The SAPO-34 nanosheets concoction was made of 85% aqueous solution (10 P2O5 molar ratio), 10 (Al2O3 98%): 16 dipropylamine: 770 H2O: 3 parts of 40% SiO₂ aqueous suspension: 10(tetraethyl ammonium hydroxide or 35% TEAOH aqueous solution), making up an entire solution weight of 120 g. The TEAOH was administered as a structural directing agent (SDA) to prevent all the Al in SAPO-34 from dispersing in water. A round-bottom borosilicate glassware placed in an oil bath at 220 °C was used to hydrothermally synthesize the SAPO-34 nanosheets instead of the traditional autoclave. The obtained suspension was aged while stirring at 65 °C for 4 days. The resulting solution was heat-treated at 220 °C for a day using a laboratory hot plate, then washed twice with water and ethanol to get the SAPO-34 nanosheets, which were then conserved at −30 °C, prior to 500 °C annealing for another 6 hours to get rid of the SDA. Finally, 1.3 g of TiO2 was mixed with 100 mL of purified water containing 800 mg of the SAPO-34 nanosheets and added to a stirring homogeneous dispersion of copper (II) nitrate trihydrate (2.63 g) to create the Cu/TiO₂/SAPO-34 photocatalyst. To reduce the copper in the solution, 10 g of aqueous NaOH was added dropwise while stirring. The nanostructured photocatalyst concoction was brought to 150 °C while refluxing for 3 hours. After three rounds of washing to get rid of any contaminants, the resulting final product was dried and kept in the desiccator till it was used.
The entire nanoparticle synthesis process regarding the nanostructured Cu/TiO₂/SAPO-34 is represented in Figure 1.
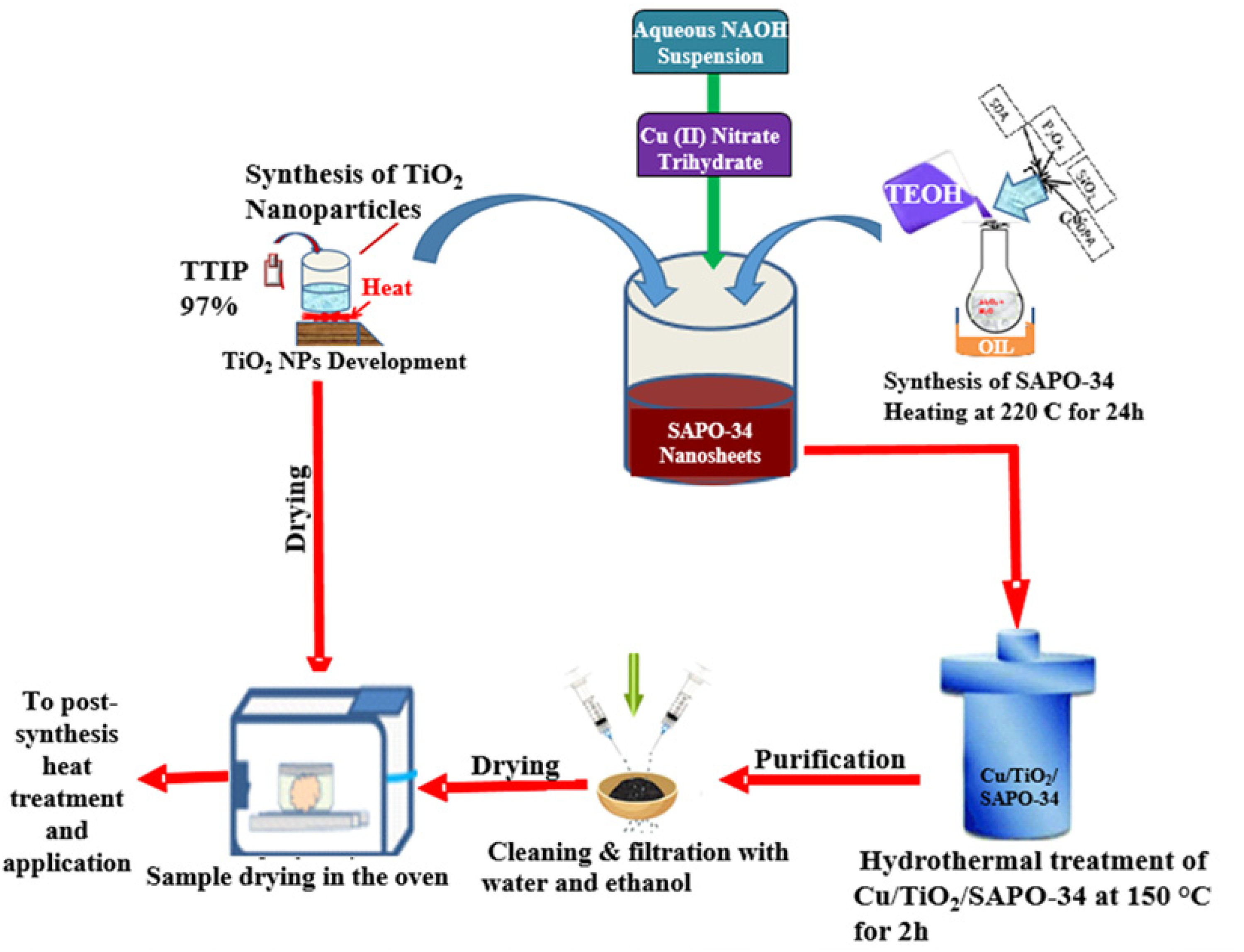
Schematic of the nanoparticles synthesis process.
Characterization
The qualities of the structure of the synthesized powder materials were analyzed by means of an X-ray diffractogram (Philips XPERT-PRO) to identify the peak patterns and Fourier Transform Infrared spectroscopy (SHIMADZU 2450) to determine the molecular bonding bands. The morphologies were characterized using the scanning electron microscope (SEM, X-MAX VEGA3 TESCAN), and an energy dispersive spectroscopy (EDS) Aztec Spectrometer, Oxford Instruments. A Shimadzu RF-6000 photoluminescence (PL) spectrofluorophotometer and a UV-2450 spectrophotometer were used to examine the optical properties. The excitation wavelength was set to 220 nm, and the scan speed was set to 6000 nm/min.
Photoelectrochemical analysis
To perform the photoelectrochemical analysis, 50 mg of every arch specimen was combined with 5 mg of polyvinylidene fluoride (PVDF) synthetic resin and droplets of N-methyl-2-pyrrolidone (NMP) to form a paste, which was then applied with a spatula on the conductive surface of the flourine tin oxide substrate. Prior to characterization, the base containing the sample was heated in an oven set at 60 °C for an hour. An electrochemical workstation ORIEL LCS-100 (AutoLAB) with three electrodes was used to evaluate the prepared samples. The generated photoelectrodes with the specimens were utilized as working electrodes, and the counter electrode was composed of Pt foil, whereas the reference electrode was Ag/AgCl. An aqueous 0.1 M Na₂SO₄ solution was employed as the electrolyte, while the photoanode was irradiated using a 50 W LED lamp delivering a light intensity of 0.13 W/cm2. Electrochemical analyses, including electrochemical impedance spectroscopy (EIS), linear sweep voltammetry (LSV), and Mott–Schottky (MS) measurements, were conducted under dark conditions. Transient photocurrent responses and EIS spectra were obtained at an applied bias of 1 V. The MS plots were recorded at 1000 Hz, whereas LSV was carried out in the potential range of 0 to 1.2 V with a scan rate of 0.01 V/s. Photoelectrochemical characterization was utilized to examine the capability of the synthesized material to drive oxidation–reduction reactions and generate the required electrochemical potential via photon absorption. This methodology enabled the evaluation of light-harvesting efficiency, charge transfer behavior, electron–hole separation, and overall photocatalytic activity. Photoelectrode characterization was performed to determine the maximum photocurrent, the onset potential, and the effectiveness of the nanocomposite in improving the light-harvesting capability of TiO₂.
Photocatalytic property analysis
Treatment of wastewater
The prepared TiO₂ and Cu/TiO₂/SAPO-34 composites were used to treat a real wastewater sample of 100 ppm concentration with a photocatalyst loading of 100 mg for up to 120 minutes under UV light exposure. To evaluate each photocatalyst's ability to remove organic pollutants from real wastewater, the wastewater sample was first examined under a PerkinElmer NexION 300 quadrupole inductively coupled plasma mass spectroscopy (ICP-MS) to spot out all the organic elements polluting the water. Table 1 shows the initial concentrations of the polluting elements identified in micrograms per liter (μg/L).
ICP-MS identified elements and their concentrations in the wastewater.
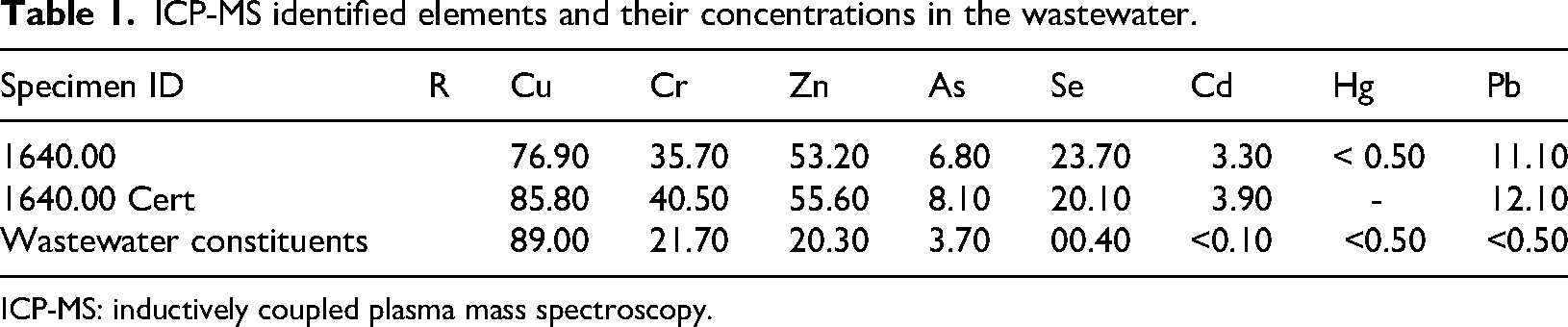
ICP-MS: inductively coupled plasma mass spectroscopy.
Each of the photocatalysts was then employed to treat the wastewater separately in a photodegradation reaction. For 100 mL of wastewater, 100 mg of each photocatalyst was used. The pH of both photocatalysts and wastewater was maintained at 6.5 and 9.5, respectively. The solution for each reaction was positioned 5 cm below a T5 BLB UV lamp of 40 W power and 40 cm in length. Samples were extracted every 30 minutes and labeled, with the first T0 taken after the initial 30 minutes, which is the dark reaction. After 120 minutes, all recovered samples were subjected to ICP analysis again, and the yield percentages of every element eliminated from wastewater are presented in Figure 6.
Typical contaminant concentrations in water resources vary by source. The wastewater treatment influent could contain pharmaceuticals at concentrations ranging from 0.1 to 50 µg/L, antibiotics at 0.1 to 30 µg/L, analgesics and anti-inflammatory agents at 1 to 100 µg/L, while agricultural runoff could contain hormones at levels of 1 to 50 ng/L. Organic contaminants in domestic wastewater include surfactants at higher concentrations of ≥50 µg/L, similar to persistent organic pollutants, which typically have concentrations in the range of 1 to 100 ng/L. 3 The concentrations of the organic elements present in the wastewater used in this study were identified in Table 1.
Photocatalytic remediation of paracetamol
Aligned with the aims of this study, the developed nanostructured Cu/TiO₂/SAPO-34's ability to photodegrade pharmaceutical contaminants was evaluated by letting it photocatalytically degrade paracetamol. Here, the material was used to degrade paracetamol under UV irradiation, and the result was compared with that of TiO2. In the experiment, a 10ppm solution of paracetamol was prepared, and then 100 mg of each sample was diluted and set in a photodegradation reaction chamber under UV light at 500 rpm. The first 30 minutes of the reaction were in the dark to allow for proper adsorption and desorption equilibrium. After which, the UV light was switched on, and the first sample, C0, was taken, followed by subsequent sampling at 30-minute intervals with a sterile syringe and filtered into sample holders through PVDF-L filters of 0.22 μm pore size. All samples were later analyzed through a UV-2450 spectrophotometer (SHIMADZU). The paracetamol clusters were analyzed through variations in the absorbance maxima. Figure 7 presents the characterization results for both samples.
Results and discussion
Morphological properties
The SEM micrographs and the EDS analysis for the fabricated Cu/TiO₂/SAPO-34 photocatalyst, with comparison to that of undoped TiO₂ powder, are displayed in Figure 2. The Cu/TiO₂/SAPO-34 revealed different particle shapes with some obvious clumping (Figure 2(b)). Whereas the TiO2 SEM image shows particles of different shapes clustered together (Figure 2(a)), the Cu/TiO₂/SAPO-34 displays an aggregate of some powder-like particles spread over the trapezoidal and round-shaped SAPO-34 nanosheets. The uniformly distributed particles are attributed to TiO2 and copper oxide formed over the zeolite's surface. 21 The elemental composition graphs for both the TiO2 (Figure 2(c)) as well as the nanostructured Cu/TiO₂/SAPO-34 (Figure 2(d)) show that both compounds were formed without impurities. In the case of Cu/TiO₂/SAPO-34, the EDS examination established the presence of Al, Cu, Na, C, O, Si, and Ti elements in appropriate proportions, as seen in Figure 2(d), which are all elements found in SAPO-34, TiO₂, or copper oxide. The most pronounced elements in the Cu/TiO₂/SAPO-34 structure were found to be O (29.8%), followed by Ti (27.0%) and C (21.5%). Figure 2(c) shows that the TiO2 nanoparticles were formed without any impurities.
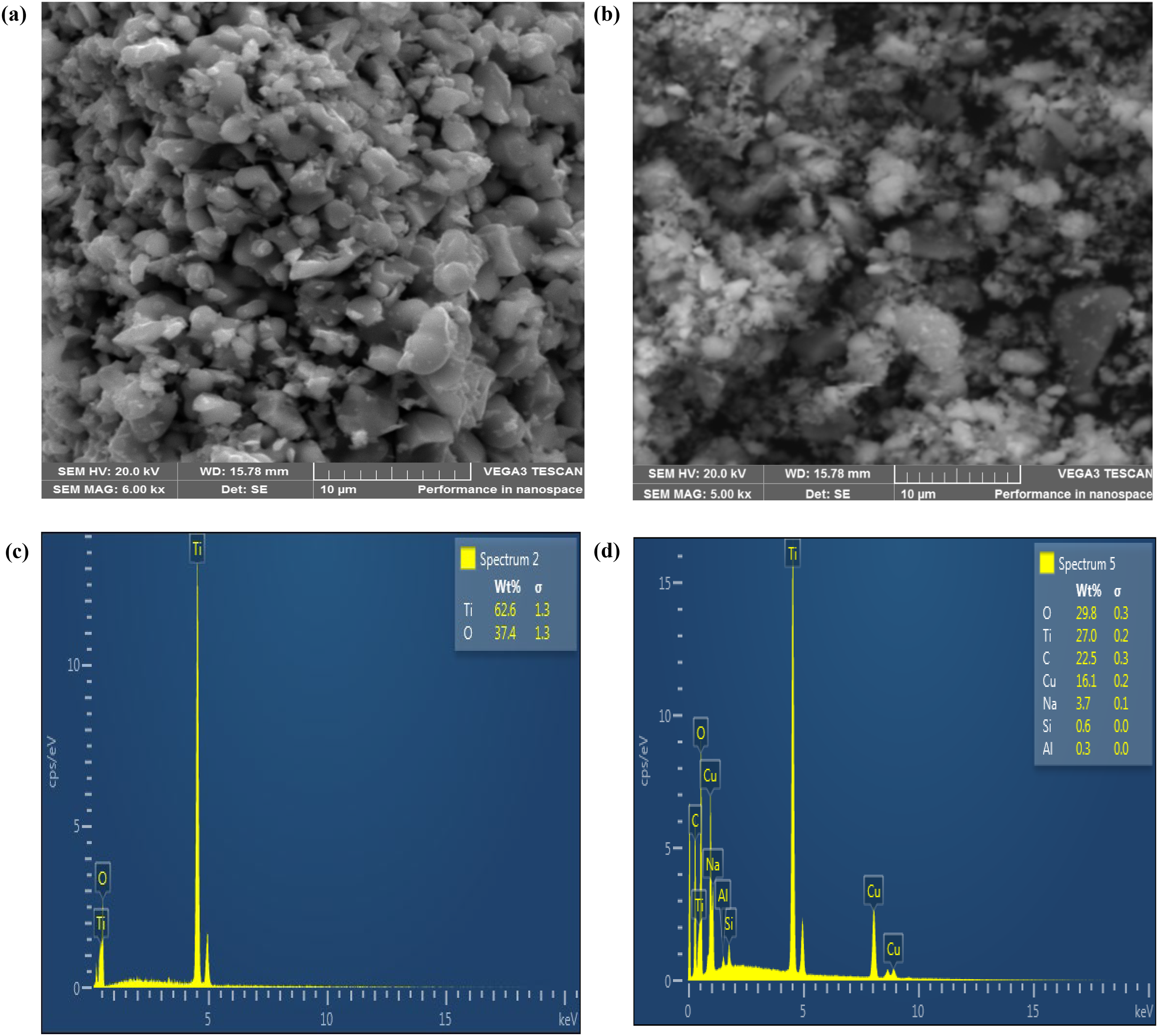
Surface morphology, (a) pure TiO2, (b) Cu/TiO₂/SAPO-34, SEM micrograph, respectively, and EDS analysis of (c) pure TiO2, (d) Cu/TiO₂/SAPO-34, respectively. EDS: energy dispersive spectroscopy; SEM: scanning electron microscope.
Structural properties analysis
Figure 3 shows the structural analysis of Cu/TiO₂/SAPO-34 in comparison with that of ordinary TiO₂. In Figure 3(a), the X-Ray diffraction (XRD) analysis revealed new peak patterns and higher peak intensities for Cu/TiO₂/SAPO-34. The nanostructured Cu/TiO₂/SAPO-34 had diffraction peaks linked to SAPO-34, CuO, and TiO₂. High-intensity peaks associated with CuO are revealed at 2θ equal to 35.33° and 38.60°, matching the hkl indices of (002) and (111), respectively, according to ICSD ref. no 259711. The SAPO-34 formation in the Cu/TiO₂/SAPO-34 mix is revealed by the 2θ peaks at 13.95° and 18.13° in accordant to ICSD 00-47-0429 and matches hkl planes (110) and (021), respectively. 22 The CuO-SAPO-34 species are present in the mix and appear at the diffractions of 65.88°, 68.30°, and 82.77°, corresponding to hkl planes (022), (220), and (222) according to ICCD ref card 04-007-1375 21 Also formed species of SAPO-34/TiO2 appeared at 2θ equal 25.56° and those attributed to anatase TiO2 at 54.05° and 61.51° corresponding to hkl (101) (ICDD 04-002-2750), (105) and (213) (ICDD 01-086-1156) respectively. XRD analysis showed that the nanostructured Cu/TiO₂/SAPO-34 exhibited sharper and more intense diffraction peaks compared to pure TiO₂, indicating an improved crystalline structure. The original TiO₂ peaks were modified in the presence of SAPO-34, confirming the structural enhancement. 20
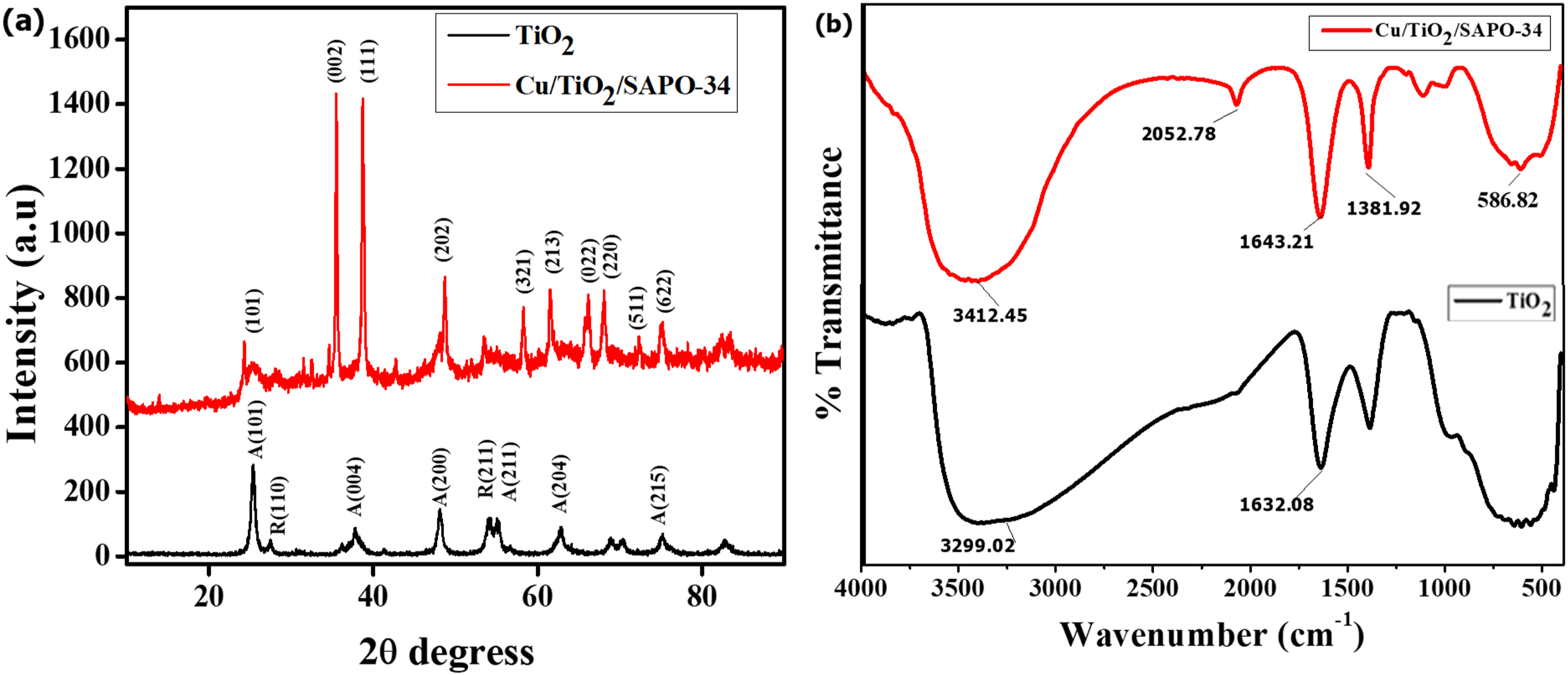
Structural analysis of nanostructured Cu/TiO₂/SAPO-34 and TiO₂ (a) XRD diffraction patterns and (b) FTIR or molecular bonding analysis.
Figure 3(b) displays the Fourier transform infrared spectroscopy (FTIR) spectra of Cu/TiO₂/SAPO-34, where a broad absorption band appears in the range of 450 to 750 cm−1, characteristic of Metal–O–Metal or Metal–O vibrations. 23 The Metal–O modes are associated with Cu–O and Ti–O bonds, while the distinct absorption peak near 673 cm−1 is attributed to Ti–O–Ti and Ti–O stretching vibrations. 24 Those appearing at lower wavenumbers (500–533 cm−1) are attributed to the vibrational modes of Cu–O bonds. These findings confirm the presence of Cu and TiO₂ within the structure. The broad band centered at 3337.70 cm−1 corresponds to the O–H stretching mode, arising from adsorbed water on the nanosheet surface. Sharp peaks near 2103.05 cm−1 are attributed to C–H stretching vibrations. The reduced peak intensities of the Cu/TiO₂/SAPO-34 vibrational bands may result from the influence of TiO₂. Overall, the FTIR results are consistent with the XRD analysis, confirming the successful formation of nanostructured Cu/TiO₂/SAPO-34.
The Debye–Scherrer relation, shown in equation (1), was employed to estimate the average crystallite size of the nanoparticles
With λ being the X-ray wavelength, k = 0.94 is a constant that depends on the shape of the particles, θ stands for the diffraction angle, and β the full breadth of the diffraction patterns at full-width half-maximum intensity. The results showed that the nanostructured Cu/TiO₂/SAPO-34 had an average particle crystallite size of 17.56 nm, whereas for TiO₂, it was 13.11 nm. This indicates that the produced nanostructured Cu/TiO₂/SAPO-34 had larger crystallites, which aligns with the pronounced XRD peaks and the SEM analysis, affirming an enhancement in the structural properties.
Optical analysis
The optical characteristics of the synthesized nanostructured Cu/TiO₂/SAPO-34 and the TiO₂ nanoparticle were established with the aid of diffuse reflectance spectroscopy (DRS) and PL spectroscopy. The outcomes of the optical analysis are shown in Figure 4. The optical transmittance patterns for the nanostructured Cu/TiO₂/SAPO-34 and that of TiO₂ are shown in Figure 4(a). The transmittance spectra in Figure 4(a) indicate that TiO₂ exhibits approximately 80% transmittance, covering the ultraviolet and visible wavelength ranges. In contrast, the nanostructured Cu/TiO₂/SAPO-34 shows merely 20% high transmittance across the UV range, approximately 30% transmittance spanning the visible spectrum, and around 20% transmittance within the near-infrared wavelength range. The absorption spectra in Figure 4(b) show the nanostructured Cu/TiO₂/SAPO-34 exhibiting early absorption in the near-infrared region at approximately a wavelength of 862 nm prior to further absorptions within the UV and light spectrums, whereas TiO₂ exhibits pronounced absorption in the visible and UV light regions. The absorption in the early infrared region affirms that the material can continue its photoelectrochemical degradation action even in the absence of visible light. The bandgap obtained from extending a straight line from the straight portion of the curves to intersect the h(eV) axis of the Tauc plot, as in Figure 4(c), shows that the formation of the nanocomposite dropped the bandgap to 2.61 eV as compared to 3.09 eV for pristine TiO2. The lower bandgap enhanced photoactivity by promoting rapid solar absorption, making the material a better photocatalyst.
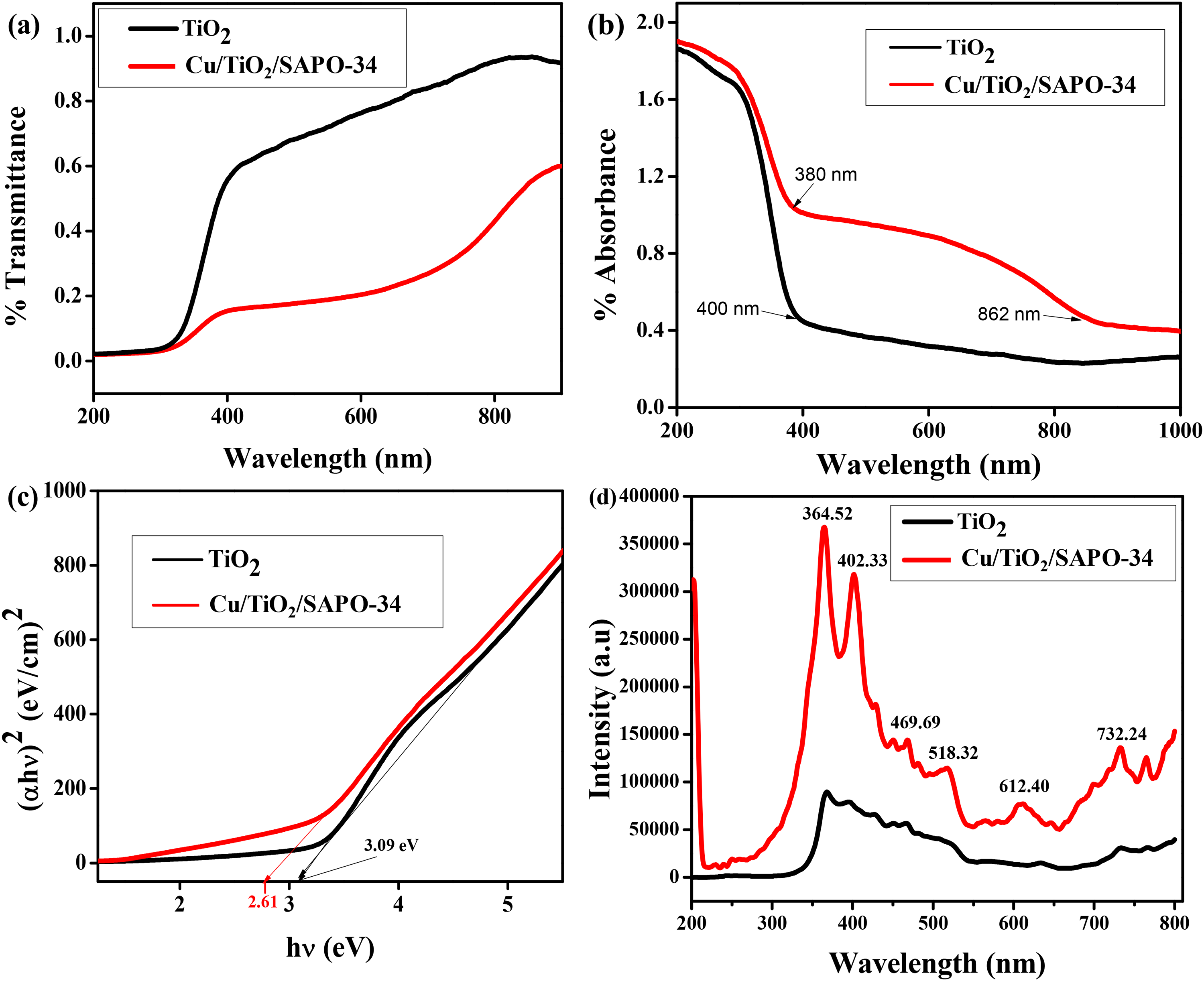
Optical properties (a) transmittance spectra; (b) patterns for the absorbance, (c) bandgap graph for TiO2 and Cu/TiO₂/SAPO-34 from the Tauc relation and (d) PL (photoluminescence) spectral profiles of TiO₂ and Cu/TiO₂/SAPO-34.
Figure 4(d) compares photon-induced excitation and light-harvesting capacity of the nanostructured Cu/TiO₂/SAPO-34 to those of pristine TiO₂ as observed under PL spectroscopy. The PL spectrum of nanostructured Cu/TiO₂/SAPO-34 exhibited emission bands in both the UV and visible regions, with distinct UV peaks at 374 and 397 nm. In the visible range, weak violet emissions appeared at 427 and 443 nm, accompanied by a small blue hump at 457.6 nm, a green peak at 527 nm, and a broad orange emission centered at 600 nm. The presence of multiple visible emission peaks is consistent with the DRS results, confirming that the nanocomposite is photoactive across both UV and visible regions. The green emission at 527 nm and the orange PL peak at 601 nm arise from the ionization of oxygen vacancies, whereas the blue luminescence at 457 nm is attributed to transitions involving oxygen vacancies and interstitial oxygen. 25 As shown in Figure 4(d), the UV peaks at 374 and 397 nm are affected by the metal oxide species and the pronounced presence of SAPO-34 nanosheets. The general impression is that the nanostructured Cu/TiO₂/SAPO-34 has very good optical properties, making it suitable for photocatalytic and photoelectrochemical applications compared to ordinary anatase TiO₂.
Photoelectrochemical properties
The photoelectrochemical features of Cu/TiO₂/SAPO-34 were weighed up to TiO₂ to assess its potential for improved energy efficiency, power density, and cycling performance, contributing to advancements in next-generation energy conversion materials and photocatalysts in the quest to address energy and environmental challenges. Four photoelectrochemical properties were studied, including the LSV, Mott–Schottky (MS) analysis, the EIS, and the photocurrent (PC) response measurements, as shown in Figure 5.
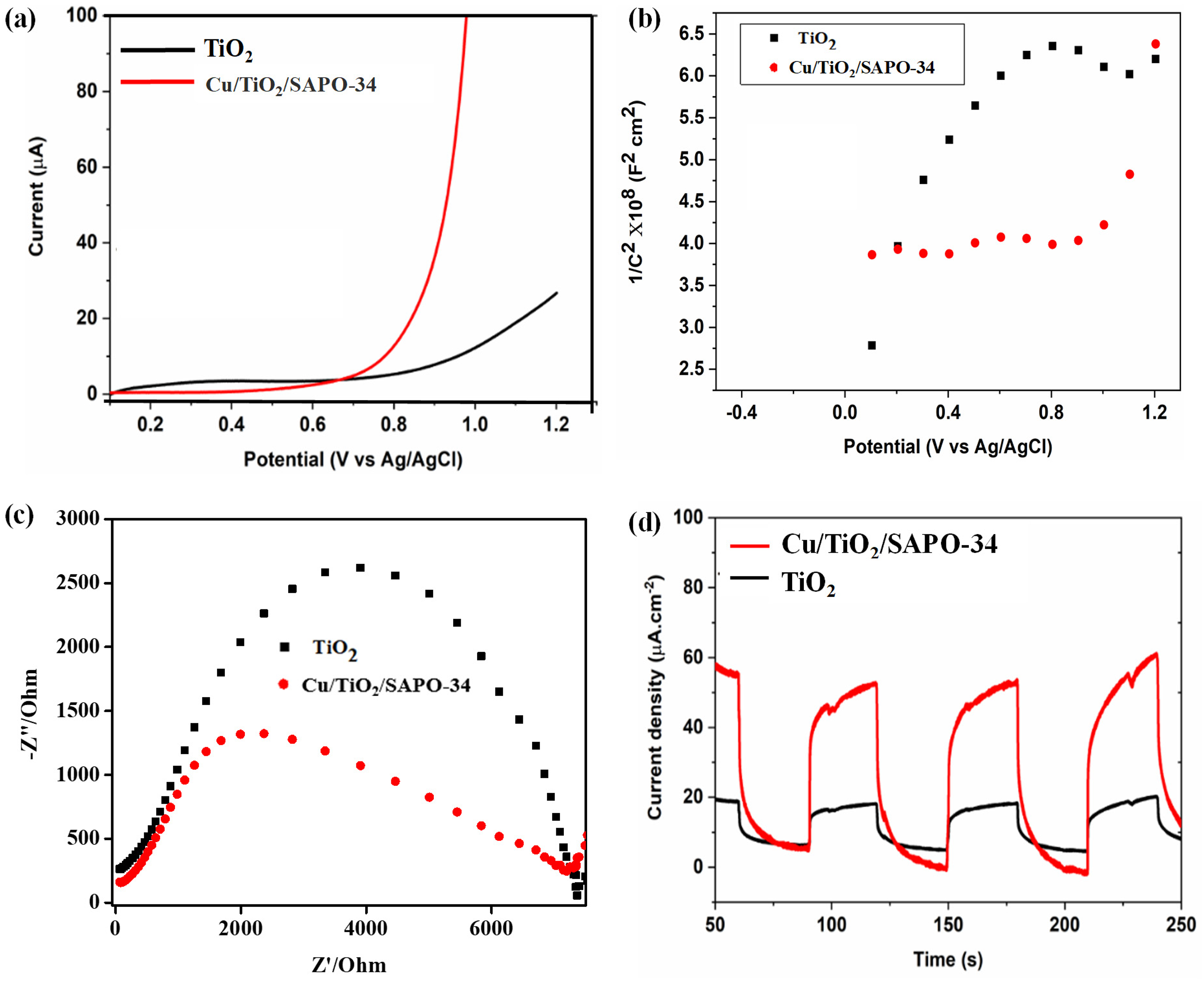
Electrochemical properties of Cu/TiO₂/SAPO-34 and TiO₂; (a) linear sweep voltammetry, (b) Mott–Schottky analysis, (c) EIS (electrochemical impedance spectroscopy) Nyquist plots, and (d) photocurrent transient response curves.
Figure 5(a) shows the LSV analysis of the nanostructured Cu/TiO₂/SAPO-34 versus regular anatase TiO₂. The LSV is employed to determine how a material reacts to the current measured against the applied potential over time to assess the photoelectrode's potential stability. The LSV response curves of TiO₂ and Cu/TiO₂/SAPO-34 photoanodes were recorded in 0.1 M Na₂SO₄ electrolyte, using Pt foil and Ag/AgCl as counter and reference electrodes, respectively, at a scan rate of 0.01 V/s. As shown in Figure 5(a), at an onset potential of 0.9 V, the photocurrent of TiO₂ increased only from 0 to 8.3 μA, whereas the nanostructured Cu/TiO₂/SAPO-34 reached nearly four times this value at 36 μA, continuing to rise with increasing potential across the entire range. The Cu/TiO₂/SAPO-34 curves indicate a change in charge-carrier flux, with no current overshoot, confirming that charge recombination is effectively suppressed. These results indicate that Cu/TiO₂/SAPO-34 exhibits higher current mobility than pure TiO₂, owing to its larger surface area, which facilitates charge-carrier separation. 26
The MS analysis was employed to determine the semiconducting nature of the photocatalyst. The positive MS curves shown in Figure 5(b) for the synthesized anatase TiO2 affirm that it is an n-type semiconductor, while the negative slope for the nanostructured Cu/TiO₂/SAPO-34 material reveals that it is mainly a P-type semiconductor. This result shows a complete transformation of the TiO2 anatomy and is consistent with the structural analysis that, indeed, a new material was truly formed.
The EIS investigation, which was utilized to determine the material's charge transfer resistance, is displayed in Figure 5(c).
A material with elevated charge pass-on aversion will normally possess a huge charge carrier pair amalgamation rate and abject charge detachment efficiency. Thereby rendering it undesired for photoelectrochemical and photocatalytic utilization. The EIS of TiO₂ and Cu/TiO₂/SAPO-34 was compared using Nyquist plots, which display real versus imaginary impedance. A smaller arc radius corresponds to lower charge transfer resistance (Rct) and faster charge-carrier transport. 27 The Cu/TiO₂/SAPO-34 exhibited a much lower Rct (2007 Ω) than TiO₂ (3931 Ω), demonstrating superior conductivity, enhanced charge separation, and reduced electron-hole reunification at its interface.
Figure 5(d) illustrates that the photocurrent density of the photoanode responded consistently to the on/off cycling under LED light illumination. The PC transient response curves evaluate the separation and transport of photo-generated charges. The nanostructured chabazite material exhibited a photocurrent density of 46 μA/cm2, roughly five times higher than that of TiO₂, which measured 9 μA/cm2. This indicates more efficient charge separation and passage from the anode to the cathode in the Cu/TiO₂/SAPO-34 photoanode compared to TiO₂. The enhanced morphology of Cu/TiO₂/SAPO-34 inhibits recombination of photogenerated electron–hole pairs, leading to improved charge separation. Consequently, the Cu/TiO₂/SAPO-34 photoanode demonstrates superior photoelectrochemical performance. The increased photocurrent response is attributed to heterojunctions formed between TiO₂ and Cu-SAPO-34, which improve interfacial charge transfer and reduce reintegration of photo-generated charge carriers. 4 The photoelectrochemical analysis has shown that the nanostructured Cu/TiO₂/SAPO-34 has excellent properties and is a more suitable photocatalyst than TiO₂.
Photocatalytic properties
The material's photocatalytic properties were studied for their application in the remediation of pharmaceutical and organic pollutants from wastewater. Its effectiveness was evaluated by assessing its ability to degrade organic pollutants from wastewater and remediate paracetamol through the decomposition of methylene blue dye.
Photocatalytic degradation of organic contaminants in wastewater
The comparative performance of TiO₂ and Cu/TiO₂/SAPO-34 in eliminating pollutants from real wastewater is presented in Figure 6. TiO2 performance is shown in Figure 6(a), while that of Cu/TiO2/SAPO-34 is shown in Figure 6(b). It was observed that for TiO₂, most elements degraded under UV irradiation, whereas for Cu/TiO₂/SAPO-34, the majority were removed during the dark reaction, with approximately 98% of cadmium (Cd) eliminated in this process. Figure 6(c) clearly compares the percentage removal of each element by the respective photocatalysts. It is seen that for cadmium and selenium, both Cu/TiO₂/SAPO-34 photocatalysts displayed good mortification efficiency (99% and 95%) for Cd and 99% and 94% for selenium (Se), by Cu/TiO₂/SAPO-34 and TiO₂, respectively. In all, the nanostructured Cu/TiO2/SAPO-34 exhibited higher photocatalytic degradation ability for all the elements in the wastewater than TiO2. For instance, while the plain TiO2 only exhibited 58%, 76%, and 83% absorption of zinc, copper, and lead, respectively, Cu/TiO2/SAPO-34 removed 91%, 89%, and 97% of the same elements under the 120-minute reaction time. The superior functioning of the nanostructured Cu/TiO2/SAPO-34 can be credited to the increase in the effective surface area observed with the PL analysis and also the early ultraviolet absorption capability of the material. 28 In essence, Cu/TiO2/SAPO-34 can effectively be used in wastewater treatment and can contribute well to contaminant remediation and environmental sustainability.
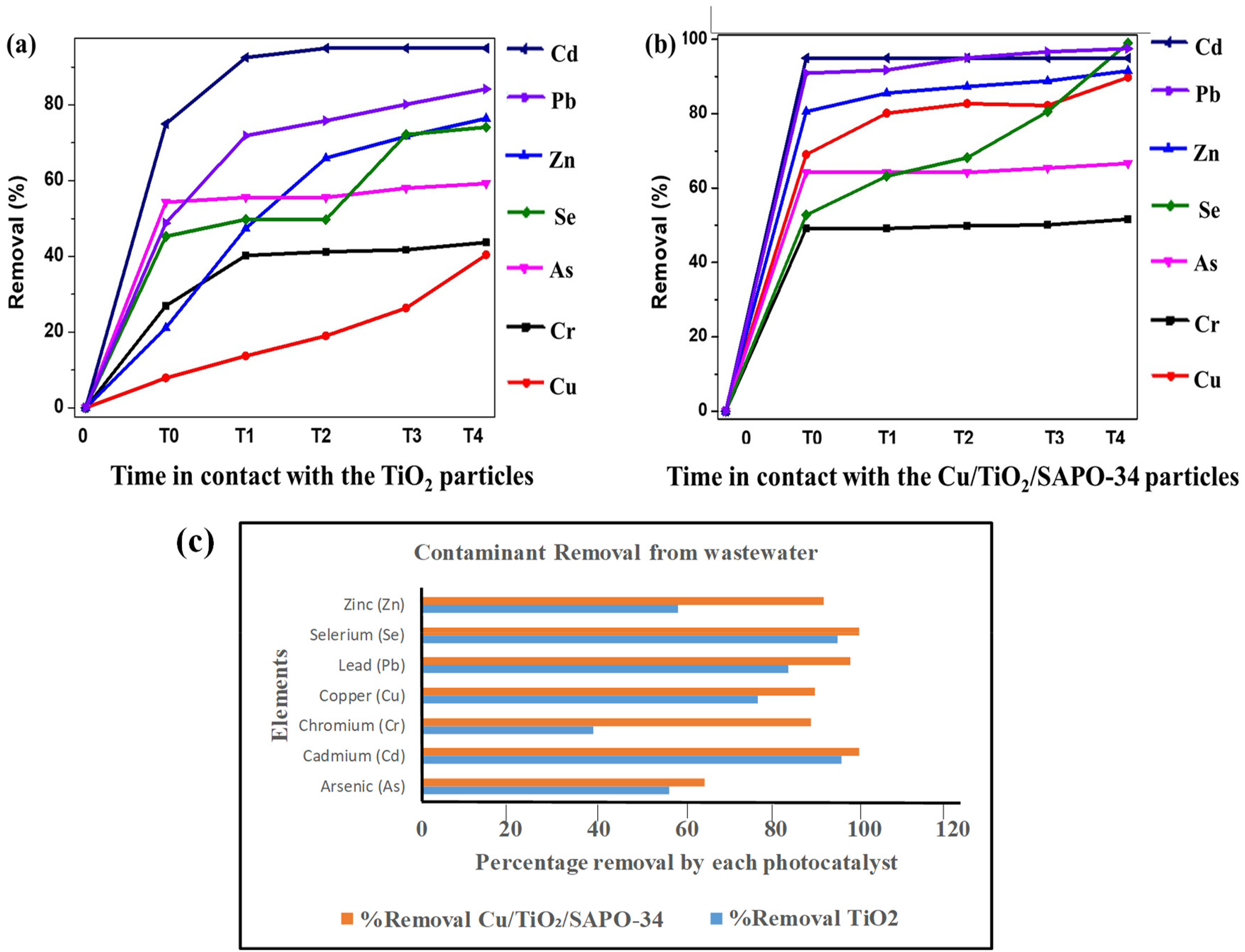
Photocatalytic degradation of contaminants from wastewater by (a) pure TiO2 and (b) Cu/TiO₂/SAPO-34, and (c) comparison of percentage removal of each contaminating element by the different photocatalysts.
Photocatalytic degradation of paracetamol
Here, the nanostructured Cu/TiO₂/SAPO-34 material was used to degrade paracetamol under UV irradiation, and the result was compared with that of TiO₂. Before initiating the photocatalytic degradation reaction, the pH of the 10-ppm paracetamol solution was adjusted to 9 to minimize water solubility and maximize proper adsorption of the catalysts. 29 Figure 7 shows the results of the photocatalytic degradation analysis. For both catalysts, the maxima of absorbance spectra for the different reaction times were identified as around 245 nm, and the peaks gradually diminished in the photocatalytic process. Figure 7(a) shows the degradation evolution curves for TiO2 from 0 to 120 minutes of reaction time. The insert of percentage degradation against time [Ct/C0 vs Time (mins)] reveals that at the end of the 120 minutes of reaction time, only about 39% of the 10 ppm paracetamol have been degraded, of which about 7% took place during the initial 30 minutes of the dark reaction. In contrast, with the nanostructured Cu/TiO₂/SAPO-34, about 30% of the paracetamol was degraded in the dark and around 40% under UV illumination, totaling about 70% of the dye degradation at the end of the 120 minute reaction time as shown in Figure 7(b), in the inset. This shows that the nanostructured Cu/TiO₂/SAPO-34 is a stronger photocatalyst than TiO₂ and will be more suitable for the remediation of paracetamol and other pharmaceutical pollutants from wastewater. The results concur with the optical examination, which established that this nanostructured material is in the early infrared region as well as in the UV spectrum. The doping of the TiO2 with copper and the silicoaluminophosphate has lowered its bandgap and extended its absorption capability beyond 5% in the UV region. 30 This improvement can be attributed to the greater active sites offered by the SAPO-34, high crystallinity, and the well-dispersed TiO2 on its surface, resulting in the low density of recombination centers. 29
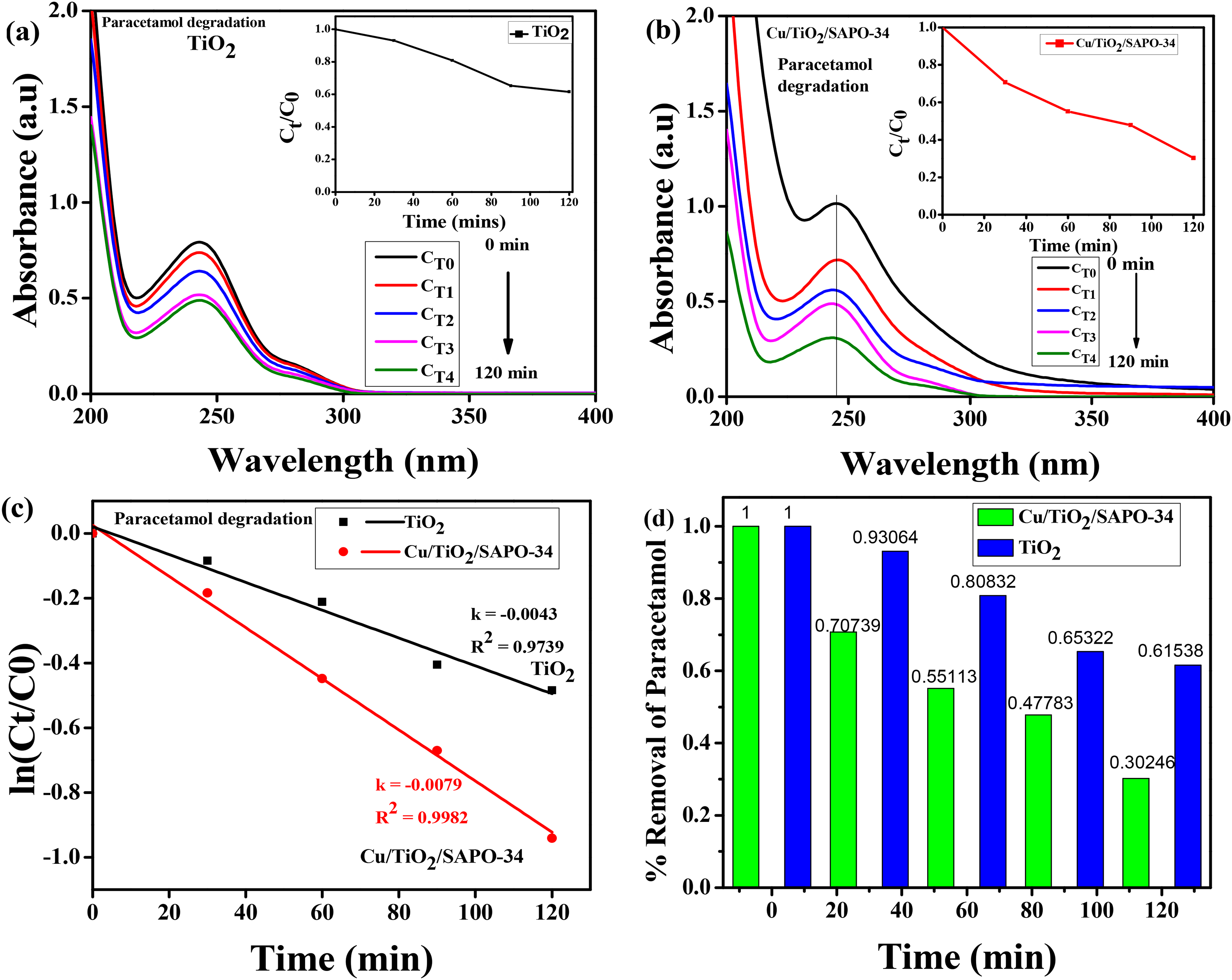
Photocatalytic degradation of paracetamol by TiO2 and Cu/TiO₂/SAPO-34; (a) degradation evolution curves for TiO2 with an inset showing Ct/C0 from 0 to 120 minutes of reaction time, (b) degradation evolution curves for Cu/TiO₂/SAPO-34 with an inset showing Ct/C0 from 0 to 120 minutes of reaction time, (c) plot of ln (Ct/C0) versus time showing determined rate constant value for Cu/TiO₂/SAPO-34 and TiO2, and (d) comparison chart of paracetamol removal (%) over time by TiO₂ and Cu/TiO₂/SAPO-34.
The photocatalytic degradation of paracetamol proceeds according to first-order kinetics. The first-order reaction rate constant (k) for the photodegradation was established from a plot of 
Photocatalyst structure
Based on the proportion of each constituent material in the nanocomposite, the proposed chabazite structure of Cu/TiO2/SAPO-34 is schematically represented in Figure 8(b). This was done by expanding on the SAPO-34 structure and the additives. A 2020 Biovia Material Studio was employed to reconstruct SAPO-34 chabazite anatomy into a 2 × 2 × 2 supercell from a single cell. The crystallographic unit cell of chabazite was acquired from the Materials Studio structural database. All the Si atoms were substituted in turns with P and Al atoms, with each P atom replacing a Si. 32 The structure clearly differs from that of TiO2 (Figure 8(a)) and reflects the outcome of the MS analysis and the morphological results.
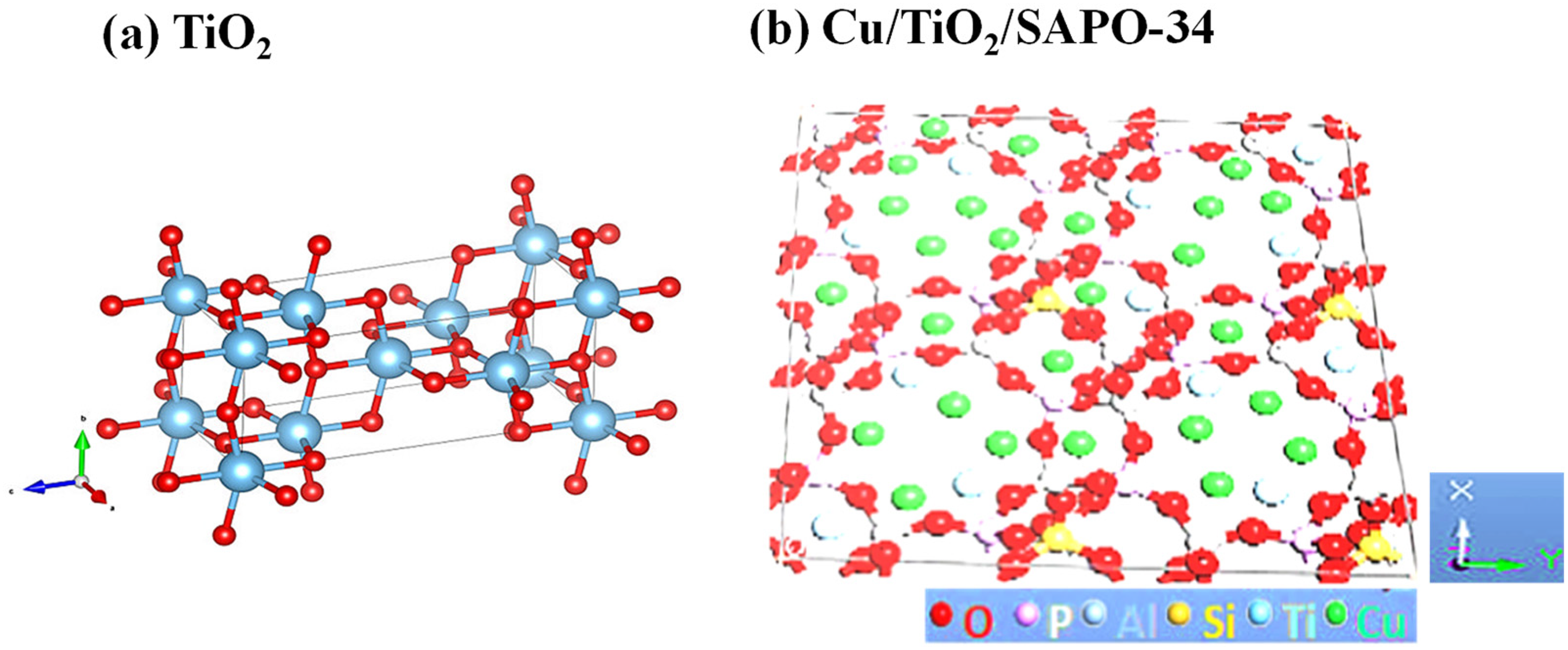
(a) Structure of TiO2 compared to (b) the proposed structure of the nanostructured Cu/TiO₂/SAPO-34 developed in the Material Studio 2.0.
Optical characterization confirmed that the Cu/TiO₂/SAPO-34 photocatalyst possesses a narrowed bandgap, which enhances its photocatalytic efficiency for paracetamol degradation. Under UV irradiation, electrons in the valence band are excited to the conduction band, resulting in the formation of photoactive electron–hole pairs. These charge carriers subsequently react with surface-adsorbed species to form highly reactive hydroxyl radicals (-OH), which initiate the oxidative degradation of paracetamol. The primary pathway involves hydroxylation to form 3-hydroxyparacetamol, followed by dehydration to p-aminophenol, subsequent oxidation to p-benzoquinone-type intermediates, and eventually aromatic ring cleavage leading to complete mineralization. 33 The proposed pathway is illustrated in Figure 9, along with the corresponding mineralization.
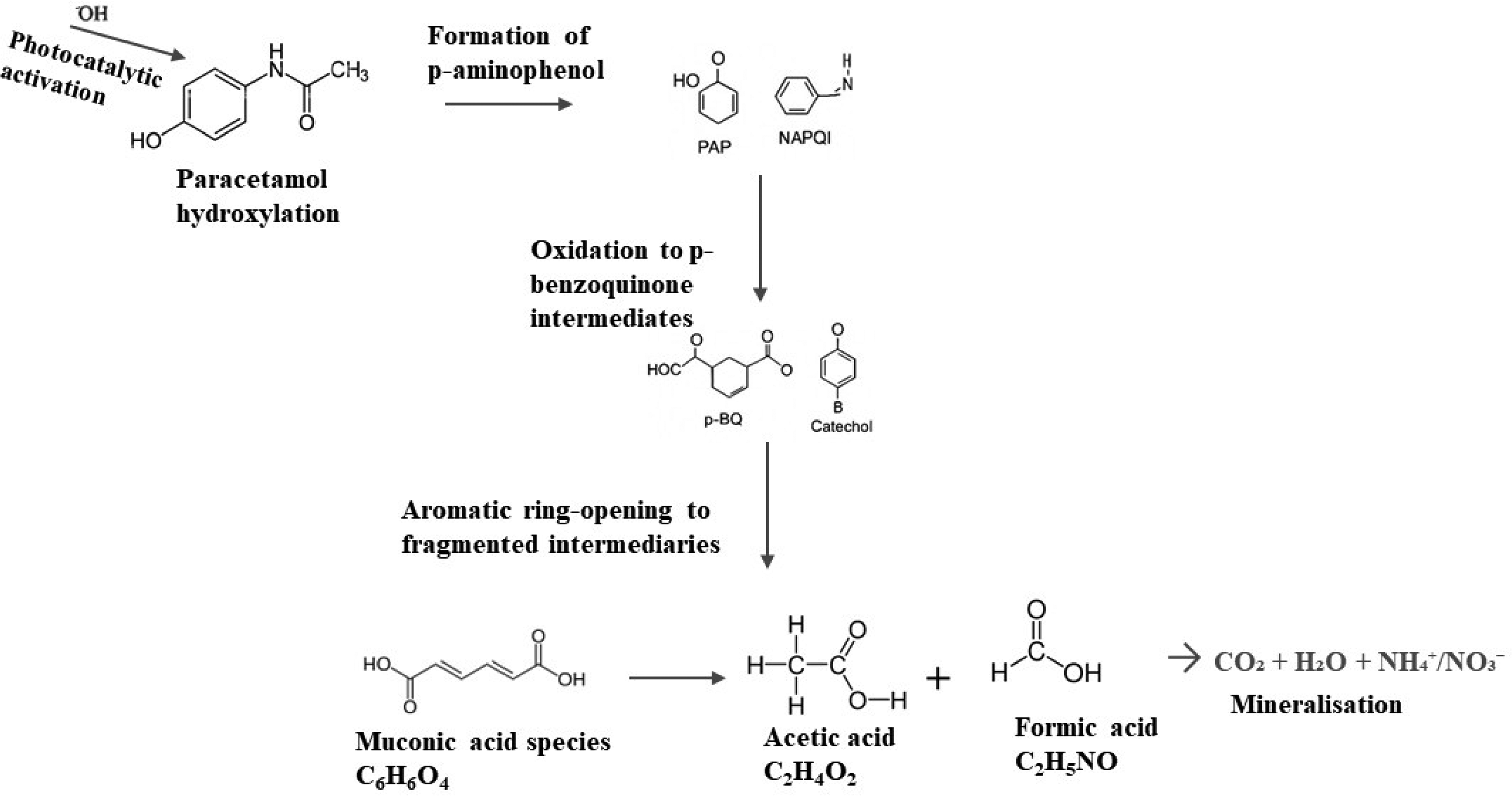
Proposed photocatalytic degradation pathway of paracetamol under UV excitation of Cu/TiO2/SAPO-34 photocatalyst.
The Cu/TiO2/SAPO-34 photocatalyst demonstrates enhanced mineralization of paracetamol due to its visible light responsiveness. The addition of metal dopants significantly improves photocatalytic performance, as these dopants create additional electronic states that narrow the bandgap to approximately 2.61 eV. This reduction facilitates the swift excitation of valence-band electrons under visible light, leading to increased exciton availability and improved degradation efficiency. 34 Figure 10 shows the proposed photodegradation mechanism of the Cu/TiO₂/SAPO-34 photocatalyst, once ignited by UV light.
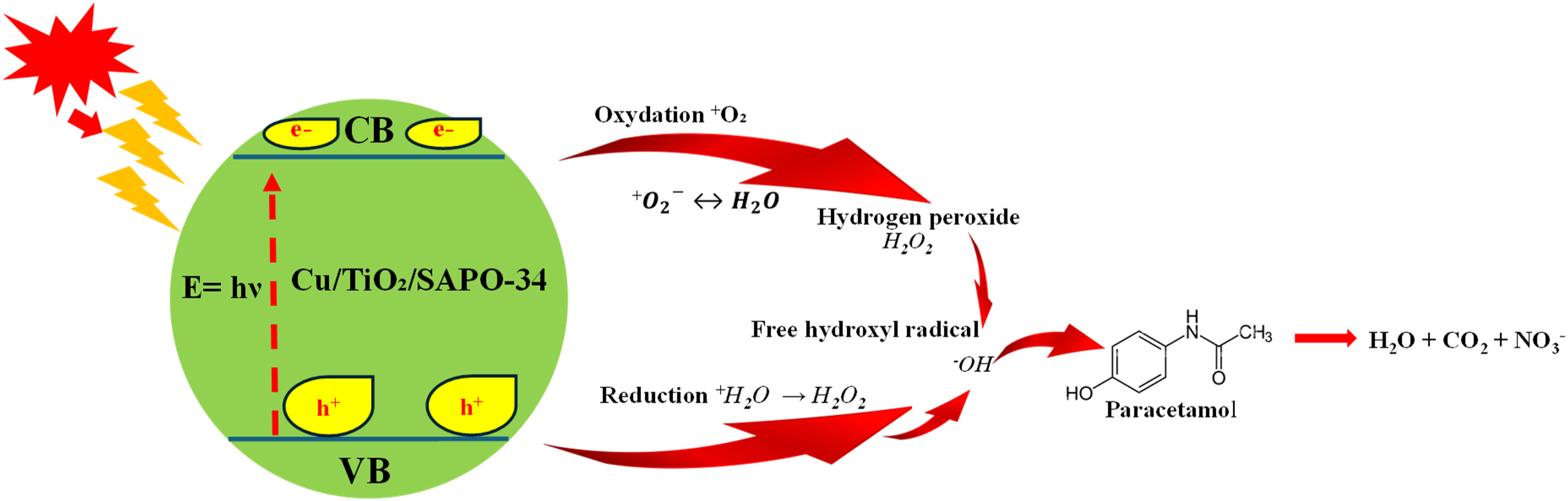
Proposed photodegradation mechanism of the Cu/TiO₂/SAPO-34 photocatalyst on paracetamol under UV light.
Performance analysis
The trend in using titania photocatalysts and composites derived from doping titania nanoparticles with other metals and metal oxides continues to grow. It has been shown that subjecting paracetamol to UV irradiation in the absence of a photocatalyst like TiO2 will yield no significant degradation due to no photolysis. 35 In the presence of a TiO2 photocatalyst, the decrease in paracetamol concentration is possible but negligible due to low absorption. 36 Thus, paracetamol degradation is only possible through photoactivity. Since heterogeneous photocatalysis is a surface phenomenon, the performance of the catalytic activity is related to the specific surface area of the photocatalysts. 37 Therefore, any developed TiO2 composite that improves the surface area in the new structure will improve its photocatalytic performance, as observed in this study. Considering that zeolites such as SAPO-34 provide a high surface area, well-defined pore architecture, unique adsorption properties, and good thermal stability, supplementing them with bimetallics like Cu and TiO₂, with known enhanced electronic, optical, and photocatalytic characteristics, will yield superior results, as demonstrated by the Cu/TiO₂/SAPO-34 in this study.
Conclusion
The electrochemical and photocatalytic properties of a hydrothermally synthesized nanostructured Cu/TiO₂/SAPO-34 photocatalyst were investigated and benchmarked against TiO₂. Both materials were subjected to morphological, structural, and optical characterization, followed by evaluation of their photocatalytic degradation efficiency toward paracetamol and their capacity to remove organic pollutants from wastewater. Morphological analysis confirmed that both photocatalysts were free of impurities. The Cu/TiO₂/SAPO-34 nanocomposite exhibited uniformly distributed powder-like particles with trapezoidal and spherical morphologies. XRD analysis revealed new diffraction features and higher peak intensities for Cu/TiO₂/SAPO-34, whereas TiO₂ displayed predominantly anatase phase. The average crystallite sizes were calculated as 17.56 nm for Cu/TiO₂/SAPO-34 and 13.11 nm for pristine TiO₂. Photoelectrochemical measurements demonstrated that the Cu/TiO₂/SAPO-34 nanocomposite possesses superior charge-transfer and light-harvesting properties, making it a more effective photocatalyst than TiO₂. UV-vis absorption spectra showed that Cu/TiO₂/SAPO-34 exhibits an earlier absorption onset at approximately 861 nm in the near-infrared region, while TiO₂ shows strong absorption primarily in the UV range. Bandgap analysis further indicated a significant reduction in bandgap energy to 2.61 eV for Cu/TiO₂/SAPO-34 compared to 3.09 eV for TiO₂.
Photocatalytic degradation experiments revealed that Cu/TiO₂/SAPO-34 removed more than 70% of paracetamol within 120 minutes, whereas TiO₂ achieved less than 40% removal under identical conditions. Across all reaction stages, including the dark adsorption phase, Cu/TiO₂/SAPO-34 consistently removed a higher proportion of organic contaminants than TiO₂. These findings suggest that the Cu/TiO₂/SAPO-34 nanocomposite is a promising photocatalyst for wastewater treatment, pharmaceutical effluent remediation, surface self-cleaning, and environmental protection.
Footnotes
Acknowledgement
The authors are grateful for the support of the Duke Corporate Education Scholarship, the Global Excellence Stature Award, the University Research Council of the University of Johannesburg, South Africa, and the National Research Foundation of South Africa.
Ethical approval
There are no human participants in this article and informed consent is not required.
Author contributions
Dr Valantine Takwa Lukong performed conceptualization, data collection, and first draft. Dr Christopher N. Chukwuati designed the experiment and performed preliminary review, writing, and editing. Dr Oluchi Vivian Nkwachukwu, material characterization, validation, and manuscript editing. Tien-Chien Jen carried out the final editing, funding acquisition, and supervision. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available on request.
