Abstract
Nickel ferrite nanoparticles were synthesized via the chemical co-precipitation technique and were employed in supercapacitor applications. The particles were characterized using Fourier-transform infrared spectroscopy, X-ray diffraction, and scanning electron microscopy to understand functionality, crystallinity, and surface morphologies, respectively. Electrochemical properties were assessed by cyclic voltammetry, electrochemical impedance spectroscopy, and galvanostatic charge–discharge techniques. An electrode prepared using NiFe2O4 displayed the highest specific capacitance of 98 F g−1 at 50 mA g−1 with an energy density of 2.1 Wh kg−1 at the power density of 80 W kg−1 and 1.6 Wh kg−1 at the power density of 160 W kg−1. After 2000 cycles, the electrode material retained 91% of its capacitance. The results of this study indicate further applications of ferrite nanoparticles in supercapacitor applications.
This is a visual representation of the abstract.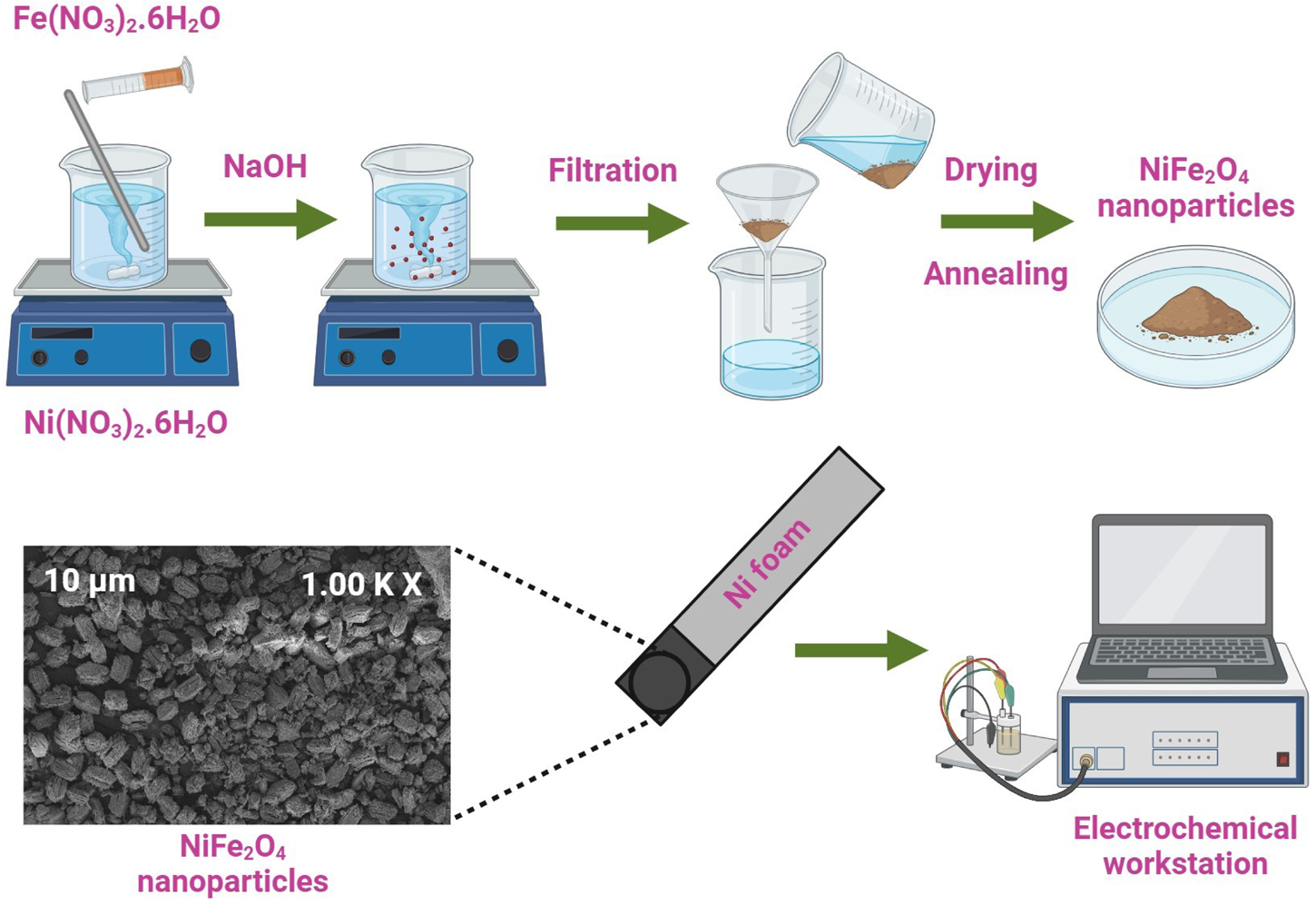
Get full access to this article
View all access options for this article.
