Abstract
The illusion of self-motion provoked by dynamic visual stimulation is known as vection and is a common phenomenon when using visual displays such as virtual reality, video games, or movie theatres. Vection has been historically linked to visually induced motion sickness (VIMS), a phenomenon similar to traditional motion sickness characterized by various symptoms including nausea, fatigue, or eyestrain. Many factors are associated with an individual’s susceptibility to VIMS and vection, but their impact is not well understood. Here, we investigated how field dependence, biological sex, and age are linked to the occurrence of VIMS and vection. To achieve this, we combined the datasets from four independent experimental studies with a pooled sample size of N = 336, including 237 younger and 99 older adults. Our results demonstrated that younger adults experienced significantly more VIMS compared to older adults and that women reported more VIMS than men (although this effect was rather weak). Additionally, field dependence was positively correlated with vection in younger adults, but no relationship between field dependence and VIMS was found. Overall, the findings from this study suggest that field dependence is not a relevant factor related to VIMS. Interestingly, older adults seem to be at lower risk of experiencing VIMS, which is encouraging considering that many novel applications are tailored towards an ageing population for rehabilitation or training purposes.
Introduction
Technologies based on visual displays are an essential part of our daily routine: smartphones, tablets, simulators, or Virtual Reality (VR) applications are highly popular tools for a variety of domains such as entertainment, training, physiotherapy, and rehabilitation.1–3 However, the use of visual devices can cause visually induced motion sickness (VIMS), a subtype of motion sickness caused by dynamic visual content rather than physical motion.4,5 Common symptoms of VIMS include nausea, headache, eyestrain, fatigue, and/or dizziness. Although the exact mechanisms underlying VIMS are not fully understood, several theories have been proposed to explain the cause of VIMS. The most widely cited theory is the sensory conflict theory6–8 and modifications thereof (e.g. subjective vertical conflict theory9,10), proposing that VIMS is rooted in conflicting information delivered by the visual, vestibular, and proprioceptive senses. That is, the visual system may suggest self-motion in the absence of actual physical motion11–13 while the vestibular system accurately suggests stasis. This visual-vestibular conflict, if unexpected or novel to the user, may result in VIMS-related symptoms.
The occurrence of VIMS has been linked to the sensation of vection in numerous studies, but the exact nature of this relationship remains vague (see Ref .14 for an overview). Some studies suggest that vection is the main factor contributing to VIMS,15–17 whereas other studies could not find positive correlations between the two phenomena.18,19 Several factors impact the individual likelihood to experience VIMS and vection, including individual-difference factors such as age, biological sex, or cognitive traits like field dependence. Although these factors have been investigated in the past, there is no consensus on how they may interact with each other and how they may impact VIMS and vection. Thus, in the present paper, we combine data from four separate experimental studies conducted in our laboratory over the past 3 years to investigate how individual difference factors (i.e. age, biological sex, and field dependence) may be associated with VIMS and vection. This approach allows us to draw more robust conclusions from a larger sample size that may offer relevant insights into the importance of individual differences with regards to these two perceptual phenomena.
VIMS, vection, and individual-difference factors
Field dependence
Considered a cognitive style, field dependency describes an individual’s tendency to rely on internal or external contextual information with regards to spatial orientation. 20 The vestibular and proprioceptive systems detect the orientation of the head and the body relative to gravity, providing internal cues that guide the sense of spatial orientation. In contrast, vision provides external cues through reference frames, visual horizon, visual polarity, or object motion. Those who rely more strongly on their internal cues are considered less field dependent compared to individuals who rely more strongly on external cues. Field dependence has been linked to the occurrence of vection and motion sickness in the past, as it may influence the interpretation of conflicting sensory information underlying vection and VIMS. In the context of vection, our previous work 21 found positive correlations between field dependence, vection intensity, and vection duration, suggesting that those who are more field dependent and rely more on visual cues tend to experience more intense and prolonged vection compared to less field dependent individuals (see also22,23). With regards to motion sickness and VIMS, previous studies examining their relationship with field dependence revealed inconclusive findings, with some studies suggesting that individuals who are more field dependent (i.e. who are more sensitive to external or visual cues) may experience more severe VIMS,24–27 whereas other studies could did not find supporting evidence for this assumption. 28 One goal of the present study is to deliver further insights into the link between field dependence, vection, and VIMS. 22
Age
Sensory function and accuracy typically declines as a function of natural ageing, 29 which may have important implications for VIMS and vection in light of the sensory conflict theory. In the context of VIMS, the role of age is not well understood. Only minimal VIMS was found in preadolescent children during a VR study, 30 whereas increased VIMS severity has been reported for older adults (60+ years) in several studies.31–33 However, the robustness of this potential peak in VIMS susceptibility in older adults has been questioned, with recent studies suggesting an opposite pattern with older adults actually reporting less VIMS compared to younger adults.34,35 That is, a review of 8 published papers by Mittelstaedt et al. 36 suggested that half of the included studies found an increase in VIMS in older adults, whereas the other half could not identify age-related differences. Thus, it remains unclear to what extent age impacts the risk of experiencing VIMS.
In the context of vection, the role of age is even less well studied compared to VIMS. A study by Paige (N = 73) 37 suggested that older adults might be more prone to experiencing vection induced by optokinetic stimulation, whereas Haibach et al (N = 45) 38 reported more intense vection in younger compared to older adults. In our own research program, we found mixed evidence as well, with one study reporting increased vection in older adults (N = 38) 39 and another study finding no age-related differences in vection perception (N = 85). 40 Taken together, these studies offer inconsistent results regarding age differences in vection, highlighting the need for further investigation.
Biological sex
Biological sex is one of the most often cited individual-difference factors in the context of VIMS and has been discussed controversially (see Refs. 41,42). Often times, it has been suggested that women might be more susceptible to VIMS than men,43,44 but evidence for this claim is mixed as several studies did not find a difference between the two sexes with regards to VIMS.45,46 Reasons for potential sex-related differences in VIMS remain unclear but it has been speculated that differences in the size of the visual field-of-view, 47 hormonal differences, 48 susceptibility to migraine, 49 and reporting bias 50 may contribute to potential differences in VIMS between men and women. With regards to vection, the studies that investigated potential sex-related differences found inconclusive results. Some studies found no differences in vection measures between men and women,51,52 whereas others have suggested that vection intensity is stronger and vection latency is shorter in women compared to men.53–55 Here, we will deliver further insights that may help to better understand how biological sex may impact the perception of vection.
The present study
The primary goal of the present study was to further investigate how field dependence, age, and biological sex impact VIMS and vection. To achieve this, we combined the data sets of four independent research studies that have been conducted in our laboratories across 3 years from 2022 to 2024. This combined dataset ensures a strong test power given the large sample size (N = 336) and allows us to detect even smaller effects more accurately.
Methods
Included studies
Summary of the experimental settings for the four combined studies including a screenshot of the stimulus.

Participants
Sample size and age characteristics per study split by age group.
Design, stimuli, and apparatus
In the following, we will provide brief summaries of the study design, stimuli and apparatus, and study procedures for each study separately that are relevant to the present paper (see also Table 1).
Makani et al 56 : In this study, the relationship between presence and VIMS was investigated while participants were passively moved through a VR scene showing an outer space voyage for 15 min. Half of the participants were provided with an astronaut self-avatar to increase the feeling of presence whereas no avatar was presented for the other half. A HP Omnicept Reverb R2 was used to present the VR stimulus with a horizontal field-of-view of 114°.
Chung et al 57 : The main goal of this study was to investigate whether providing a thermal wristband that provides cooling or heating sensations to the left wrist may alter VIMS severity. Participants were assigned to one of four experimental conditions and were exposed to a 14-min long video recorded from a camera mounted on the handlebar of a bicycle presented on large projection screen with a field-of-view of 71° horizontally and 60° vertically.
Murovec et al 40 : In this study, the role of stimulus characteristics on vection and VIMS were explored. Participants were seated in front of an array of three adjacent screens covering a total field-of-view of 228° horizontally and 48° vertically. Younger and older participants were presented with a 360° photograph that was presented either intact or scrambled into many segments and rotated continuously to induce the sensation of circular vection about the yaw axis. A total of 24 trials (45 s duration) were presented in randomized order.
Pöhlmann et al 58 : The goal of this study was to investigate whether vibrotactile cues presented to the torso of participants could reduce VIMS during a VR task. Participants were passively moved through a VR scene for 20 min showing a sea voyage while they were asked to use their VR controllers to shoot water from water guns targeted at balloons placed throughout the VR scene. Younger and older adults were randomly assigned to one of four experimental conditions. A HP Omnicept Reverb R2 was used to present the VR stimulus, with a horizontal field-of-view of 114°.
Field dependence, VIMS, and vection measures
In all four studies, the same measures were recorded. To measure field dependence, all participants completed the Computerized Rod and Frame (CRAF) 59 prior to stimulus presentation. During this task, participants were seated in a dark room wearing glasses that limited their visual field to a large projection screen in front of them. The CRAF consists of a squared frame with a vertical ‘rod’ in its centre. Participants were asked to adjust the rod to be aligned with Earth’s vertical, while the surrounding frame was either tilted clockwise (18°), counter-clockwise (−18°), remained stable, or was not present. Deviations from the true vertical were recorded as errors in the alignment of the rod (measured in degrees). That is, larger alignment errors were considered to indicate a higher level of field dependence. The CRAF was presented on a large projection screen (300 cm × 196 cm) with participants wearing a pair of goggles (no lens strength) with customized covering that limited the visual field to the projection screen. Each of the four frame conditions was repeated four times. An averaged CRAF score, representing deviation from true vertical in degrees, was calculated using the following approach: average absolute value of [(Frame −18°) – (Frame 0°)] and [(Frame 18°) – (Frame 0°)]).
VIMS severity was captured using two measures. First, the Simulator Sickness Questionnaire (SSQ) 60 was assessed immediately after stimulus exposure in all studies. The SSQ is comprised of 16 items representing symptoms of VIMS. Participants are asked to rate each symptom on a 4-point scale (none, slight, moderate, severe) to indicate their current state of well-being. Three subscales (nausea, oculomotor, disorientation) as well as a total-score were calculated using a specific weighting procedure suggested by the authors of the SSQ. Second, during stimulus presentation, VIMS was recorded using the Fast Motion Sickness Scale (FMS). 61 The FMS is a verbal rating scale ranging from 0 (no sickness at all) to 20 (severe sickness) that captures the nausea-related aspect of VIMS. Participants were asked to verbally indicate their level of VIMS by reporting a single FMS score every minute (Makani et al., 56 Chung et al., 57 Pöhlmann et al. 58 ) or after each trial (Murovec et al. 40 ). FMS scores were analyzed by calculating the peak score (i.e. the highest FMS score reported throughout stimulus presentation).
Vection intensity and vection duration were measured after stimulus presentation (Makani et al., 56 Chung et al., 57 Pöhlmann et al. 58 ) or after single trials (Murovec et al. 40 ) using standard self-reports. 62 Vection intensity was measured on a scale from 0 (no vection at all) to 10 (very intense vection) and vection duration was measured in percent ranging from 0 (never experienced vection) to 100 (always experienced vection). For the study by Murovec et al., mean scores for vection intensity and duration were calculated and used for data analysis in the present paper.
Data analysis
All data were analyzed using the statistical softwares R (Version 4.2.2) 63 and Jamovi. 64 The significance level was a priori set to alpha = 0.05 for all analyses. The significance level was Benjamini-Hochberg corrected to adjust for multiple comparisons. This method was chosen given that it provides a balance between statistically significant findings and limits false positive occurrences.
Results
VIMS, biological sex, and age 1
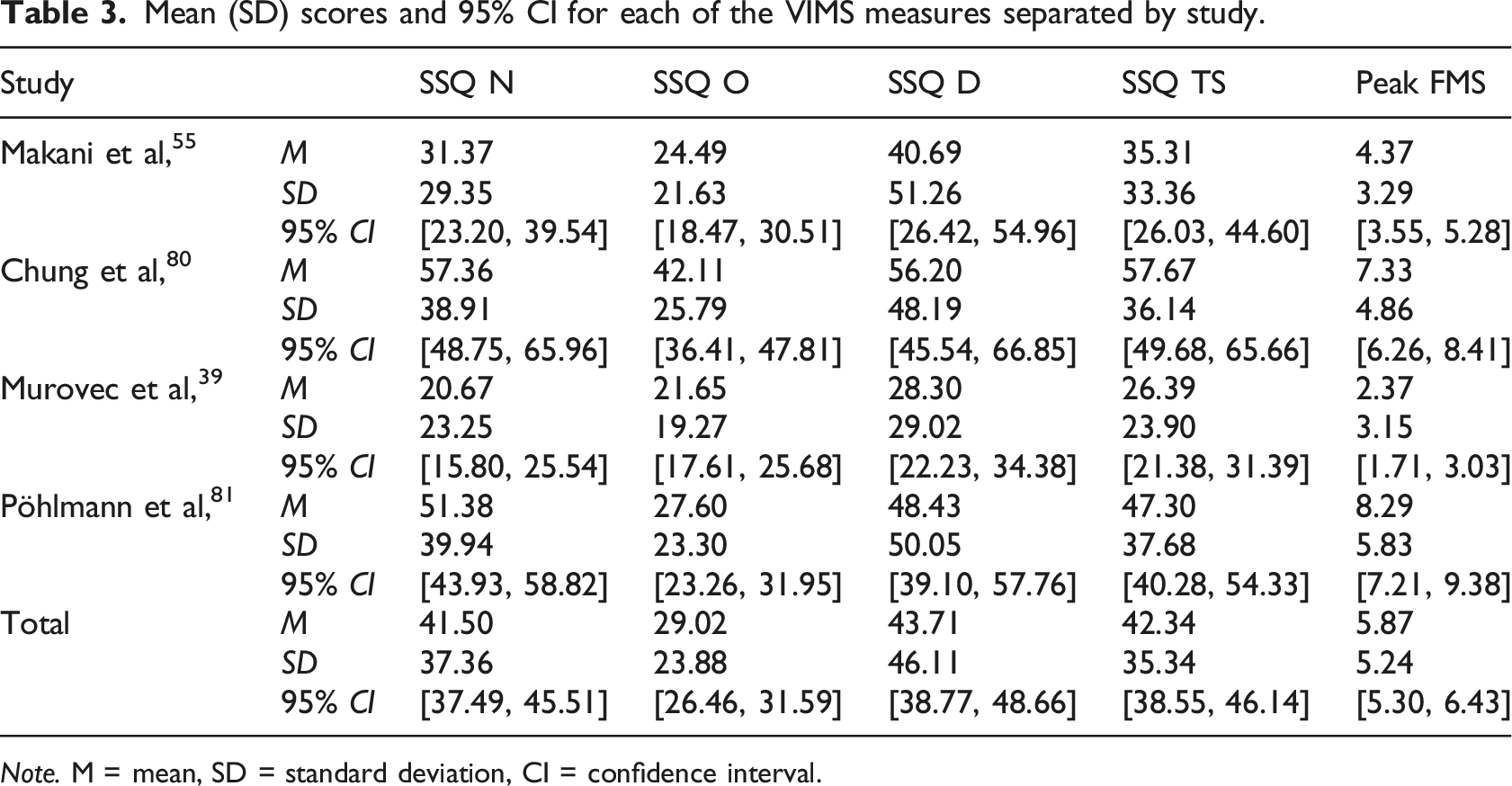
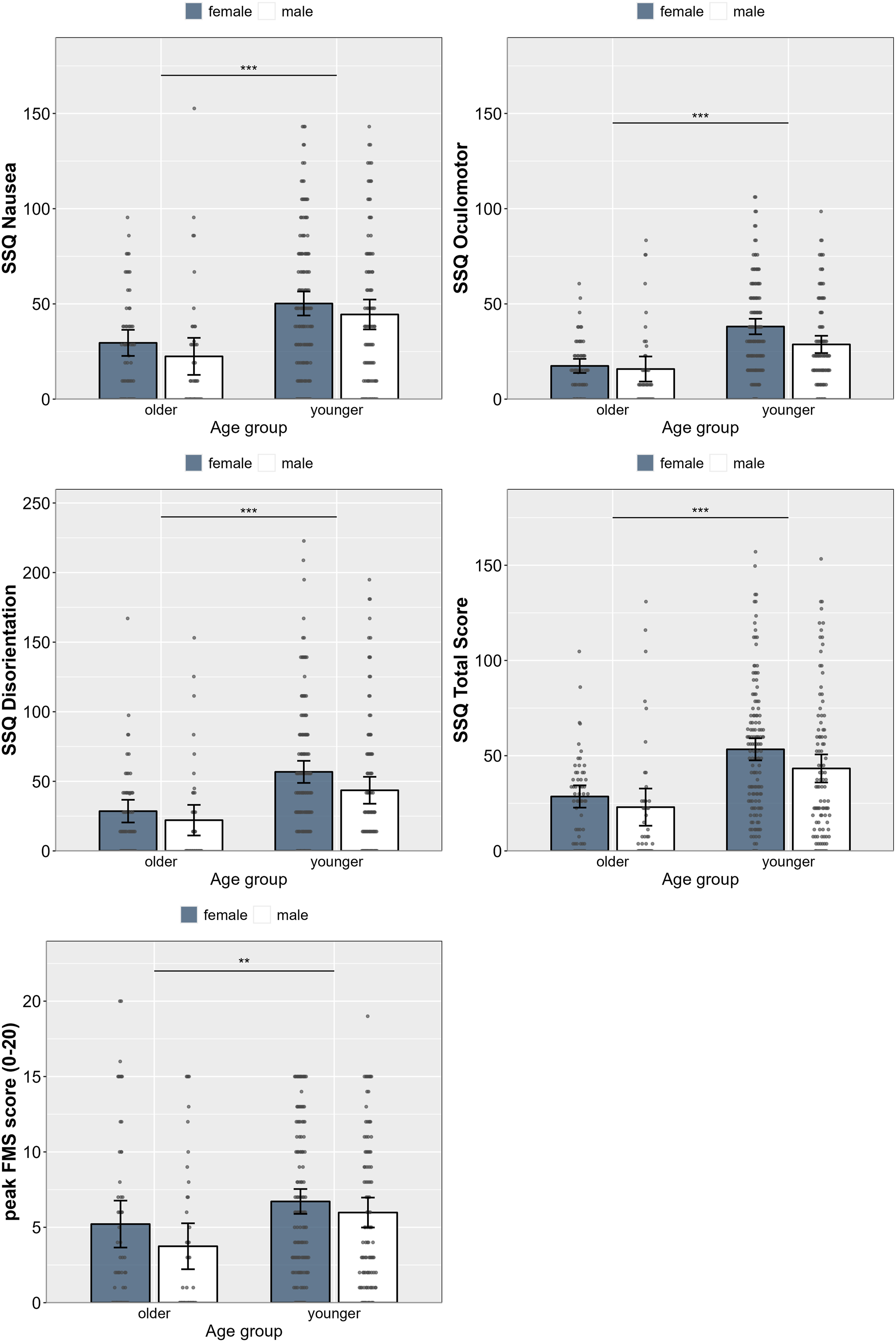
Figure 1 illustrates the distribution of VIMS measures across all studies, whereas detailed results for all VIMS measures separated by study are summarized in Table 3. Mean SSQ and peak FMS scores, combined across all four studies and separated by sex and age group, are depicted in Figure 2. Univariate 2 × 2 ANOVAs including the between-subjects factors age group (younger, older) and biological sex (female, male) were conducted for each of the VIMS variables. A main effect of sex showed for the SSQ subscales oculomotor, F (1, 332) = 8.34, p = 0.004, η2 = 0.02, disorientation, F (1, 332) = 5.24, p = 0.023, η2 = 0.02, and the total score, F (1, 332) = 5.52, p = 0.019, ηp2 = 0.02, but not for the subscales nausea, F (1, 332) = 2.38, p = 0.124, ηp2 = 0.01, and the peak FMS score, F (1, 332) = 2.79, p = 0.096, η2 = 0.01. A main effect of age group showed across all VIMS measures, including the SSQ subscales nausea, F (1, 332) = 24.36, p < 0.001, η2 = 0.07, oculomotor, F (1, 332) = 42.77, p < 0.001, η2 = 0.11, disorientation, F (1, 332) = 22.91, p < 0.001, η2 = 0.06, and the total score, F (1, 332) = 32.59, p < 0.001, ηp2 = 0.09, as well as the peak FMS score, F (1, 332) = 8.75, p = 0.003, η2 = 0.03. No interaction between age group or sex were found. These results suggest that VIMS severity was more intense in younger compared to older adults overall, whereas oculomotor discomfort and disorientation were more pronounced in females compared to males. Violin plots showing the distribution of the VIMS measures across studies and all participants, illustrating that most participants reported VIMS-related symptoms after visual stimulation. Note. SSQ = Simulator Sickness Questionnaire. Note. Grey dots represent individual data points. Mean (SD) scores and 95% CI for each of the VIMS measures separated by study. Note. M = mean, SD = standard deviation, CI = confidence interval. Mean scores for all VIMS measures across all four studies separated by age group and sex, showing significantly more severe VIMS in younger adults across each subscale and overall compared to older adults regardless of sex. Note. Error bars represent 95% CI. Grey dots represent individual data points.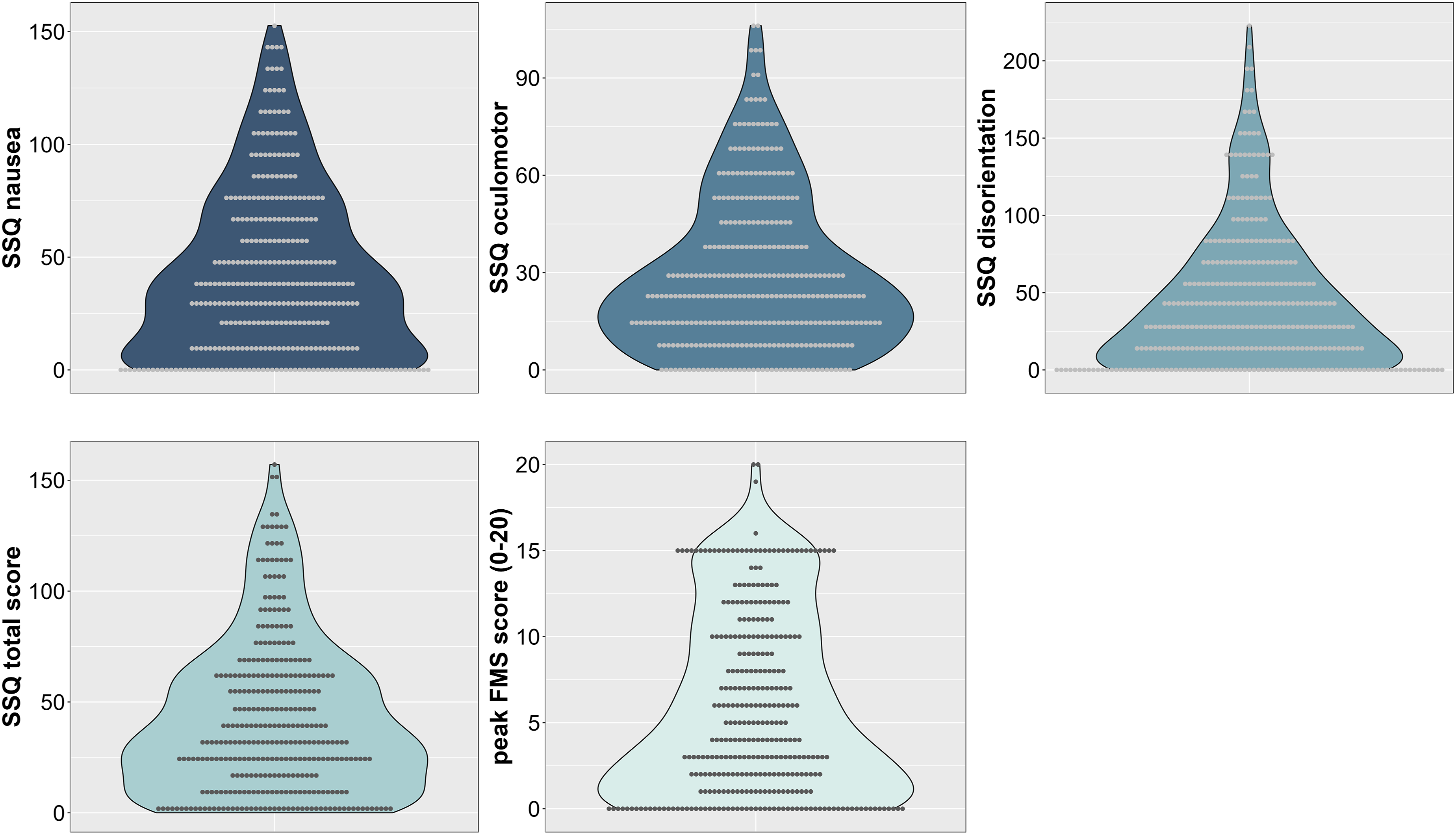
Vection, field dependence, biological sex, and age
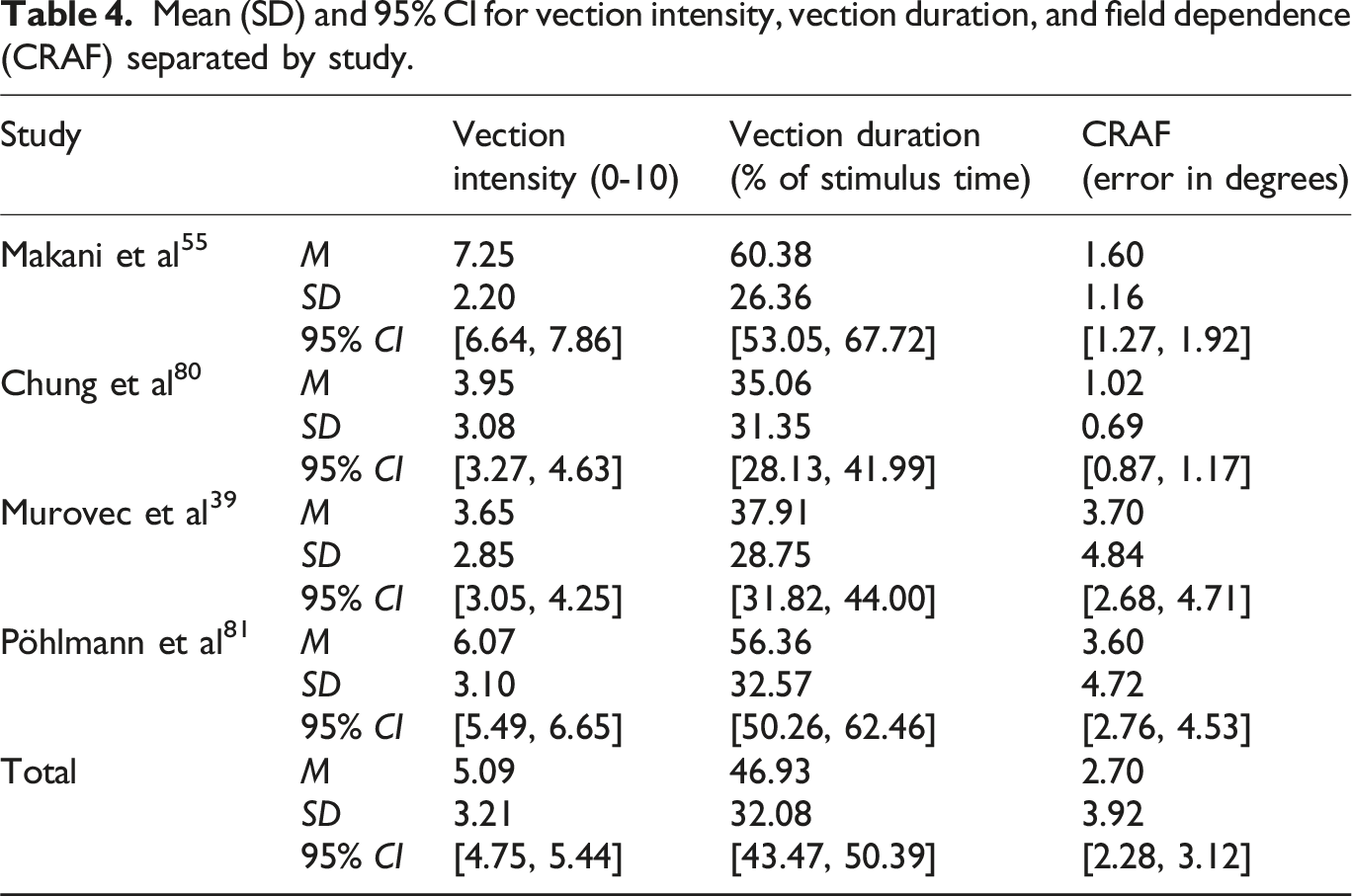
Mean (SD) and 95% CI for vection intensity, vection duration, and field dependence (CRAF) separated by study.
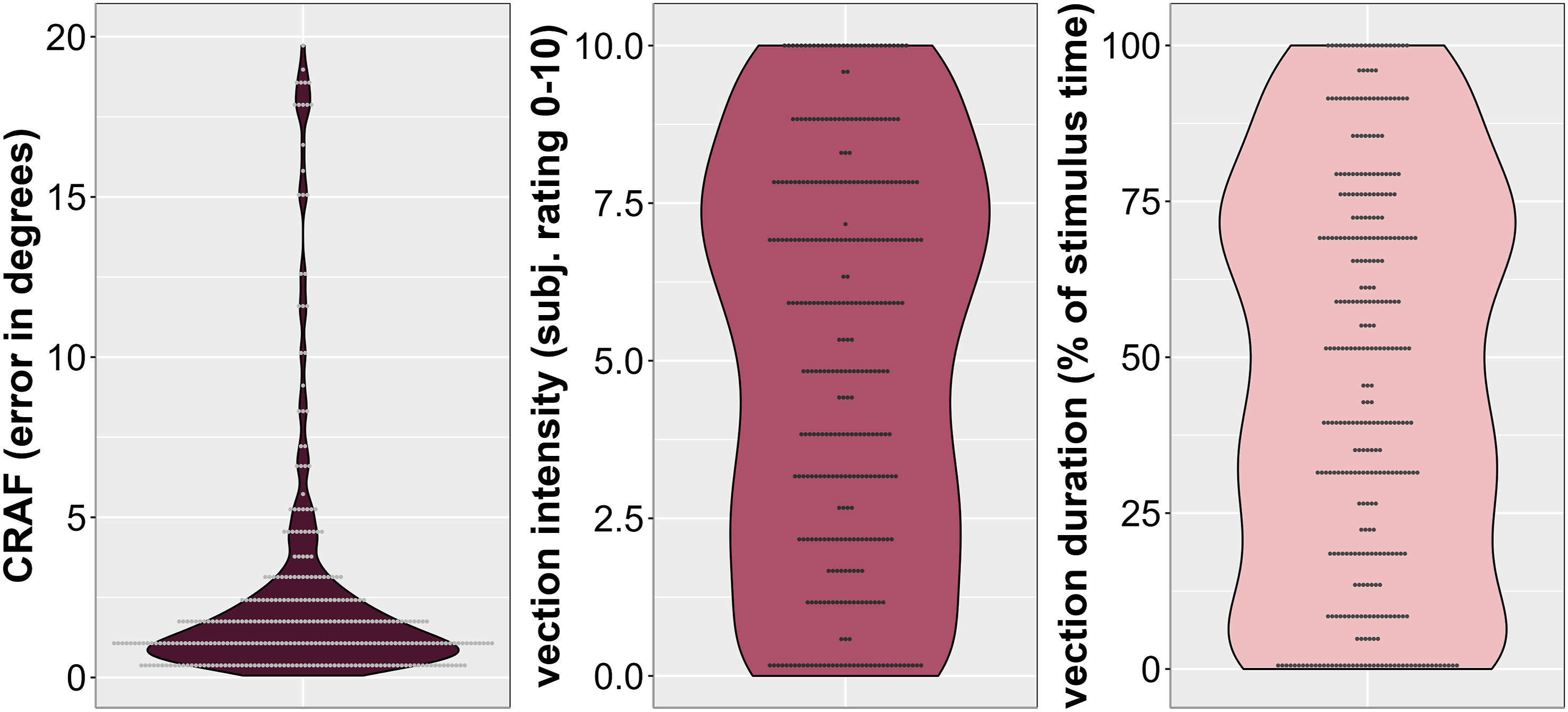
Violin plots showing the distribution for field dependence and vection measures across studies and all participants. Note. Grey dots represent individual data points.
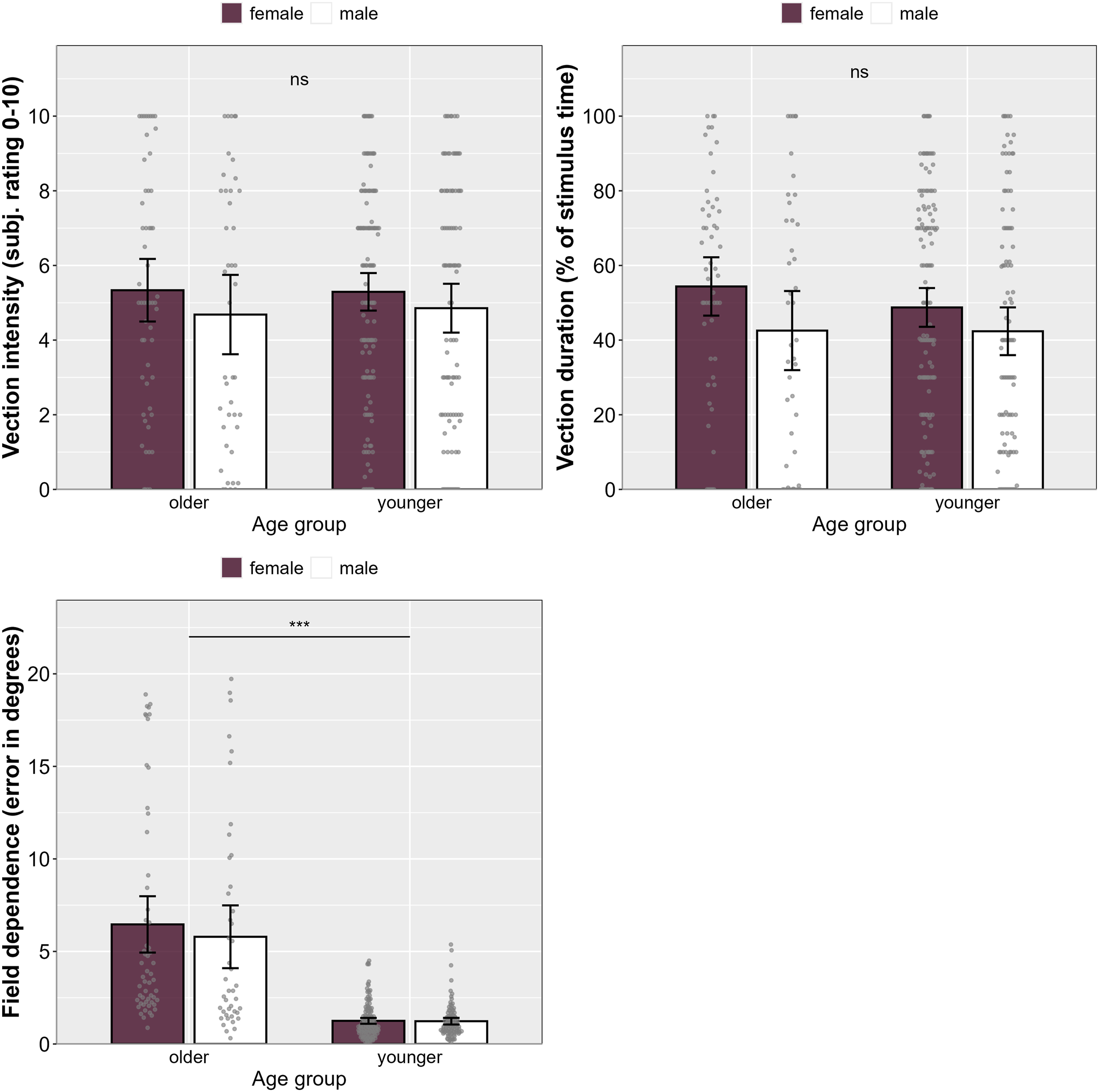
Mean scores for vection intensity, vection duration, and field dependence (CRAF) across all four studies separated by age group and sex, suggesting that age-related differences in field dependence did not translate to any differences in vection intensity or duration. Note. Error bars represent 95% CI. Grey dots represent individual data points.
Correlations between field dependence, VIMS, and vection
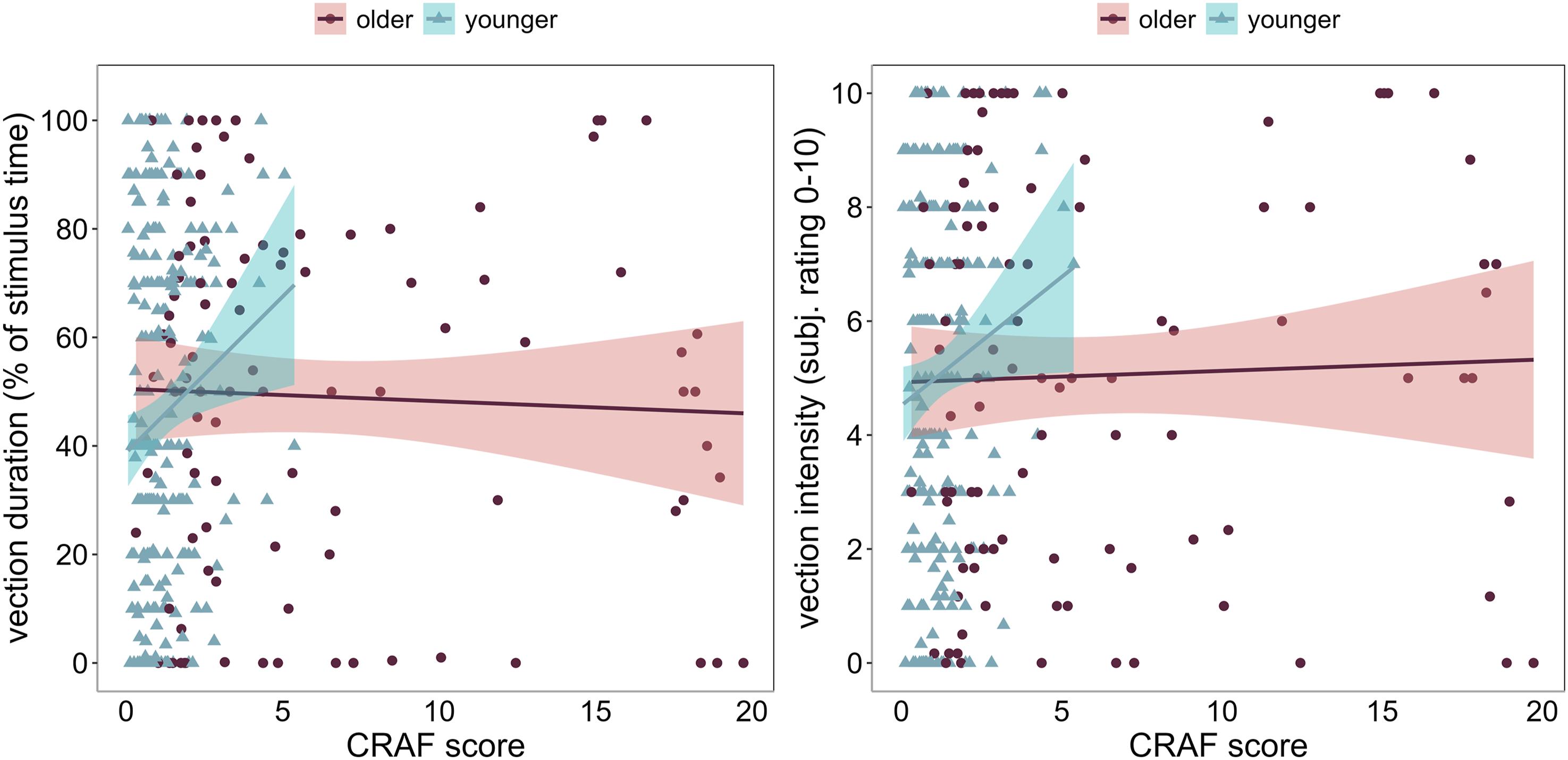
Pearson correlations were calculated for the CRAF scores with all VIMS and vection measures across all four studies combined and separated by age group (Figure 5). Most notably, for both younger and older adults, no significant correlations were found between the CRAF and any of the VIMS measures. In contrast, weak positive correlations were found between CRAF and both vection measures for the younger age group only, suggesting that for younger adults higher CRAF scores were associated with more intense (r = 0.133) and longer lasting (r = 0.168) vection (see Figure 6). For both age groups, weak-to-moderate positive correlations were found between both vection measures and all VIMS measures, with slightly higher correlations found in older (r’s from 0.220 to 0.380) than in younger (r’s from 0.152 to 0.350) adults. These correlations suggest that higher vection intensity and longer lasting vection were associated with higher VIMS scores. Correlation heat map illustrating the relationship between field dependence, VIMS, and vection measures for younger (left panel) and older adults (right panel) across all four studies. Note. FD = Field dependence as measured by the CRAF, N = nausea, D = disorientation, O = oculomotor, TS = total score, int = intensity (*p < 0.05, **p < 0.01, ***p < 0.001). Scatterplot showing the relationship between field dependence (CRAF) and vection duration (left), and field dependence (CRAF) and vection intensity (right), for younger (red) and older (blue) adults. The blue gradient line shows weak but statistically significant correlation between vection duration/intensity and field dependence in younger adults only.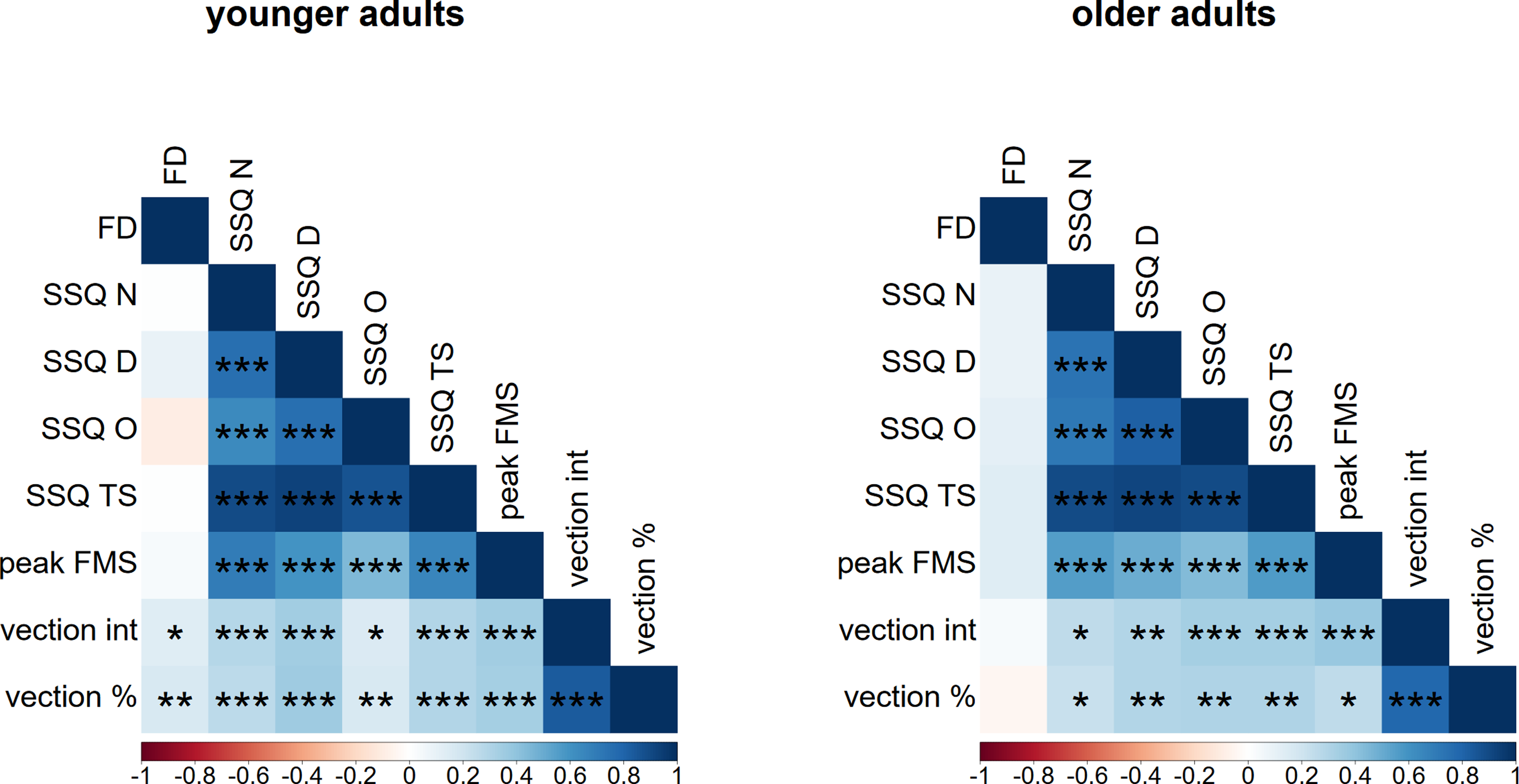
Discussion
The goal of the present study was to investigate how the individual-difference factors field dependence, age, and sex are related to the severity of VIMS, as well as vection intensity and vection duration. To achieve this, we combined data from four independent experimental studies into one large dataset. Overall, older adults reported significantly less VIMS and higher field dependence scores compared to younger adults. Additionally, women reported more VIMS and experienced longer lasting vection compared to men. Field dependence was only significantly correlated with vection intensity and duration in younger adults but not in older adults, and this correlation can be considered rather weak. Instead, vection was positively correlated with all VIMS measures in both age groups with moderately strong effects (up to r = 0.38).
VIMS and individual-difference factors
The relationship between field dependence and VIMS and vection is not well understood. Previous studies suggested that higher field dependence might be positively related to an elevated risk of experiencing VIMS, 27 which, from a theoretical point of view, seems plausible as a stronger dependence on visual cues may increase a potential visual-vestibular conflict (i.e. visual cues indicating self-motion, vestibular cues indicating stasis), which may result in more severe VIMS. This is particularly relevant in context of the subjective vertical mismatch theory, which proposes that a conflict in expected and perceived verticality with respect to gravity is the main contributor to VIMS.9,65 Thus, individuals who are less field dependent could be less susceptible to VIMS, as their perception of verticality remains less challenged during visual stimulation compared to those who are highly field dependent. However, the current study did not reveal any meaningful correlation between field dependence and VIMS, suggesting that field dependence plays a limited role in the development of VIMS at least when the head is not tilted.
Interestingly, the present study found higher VIMS scores in younger compared to older adults. Previously, it has been mostly assumed that older adults are at elevated risk of experiencing VIMS, for instance in the context of driving simulators.31–33 However, more recent studies using VR applications have found the opposite pattern, with older adults being less affected by VIMS.36,66 This trend of decreased VIMS reports among older adults is in accordance with studies on traditional motion sickness, suggesting a decline in susceptibility to motion sickness induced by physical motion with age. 67 The reduced risk in VIMS severity in older adults could be explained by natural habituation.68,69 According to the sensory conflict theory, VIMS occurs in situations with contradicting sensory information that are unexpected or novel to the individual. Across the lifespan, individuals build and consolidate robust neural representations of sensory patterns associated with different types of true or apparent motion that make them less susceptible to experiencing traditional motion sickness and, potentially, also VIMS. Given the heterogenous results regarding age-related differences in previous studies, a more thorough analysis of age-related differences is recommended to better understand the complex relationship between age and VIMS, such as applying study designs that include equally distributed age groups (e.g. 20–30 years and 30–40 years) with a sufficient sample size or by conducting longitudinal long-term studies to investigate how VIMS susceptibility may change over the life span.
Biological sex has long been discussed in controversy as a potential contributor to VIMS susceptibility.42,70 Here, we found partial support for the assumption that women experience more severe VIMS than men. Although this difference was highly significant, the observed effect sizes (η2 = 0.02) suggest that this difference is rather small and it only showed in some of the VIMS measures. A potential sex-related difference might be due to many factors, including hormonal difference48,71 or the openness to report incidences of VIMS. Our study suggests that sex-related differences do exist in VIMS, but that these differences are not very strong or robust, which may explain the heterogenous results found in the literature.
Vection and individual-difference factors
With regards to vection, previous work pointed at a potential positive relationship with field dependence.23,53 Here, we found partial support for this assumption, as positive, significant correlations were found in younger but not older adults, suggesting that participants who were more field dependent experienced more intense and longer lasting vection. Again, this result seems plausible, as those who rely more on visual cues with regards to spatial orientation (i.e. more field dependent individuals) should be more prone to experiencing vection, emphasizing the role of the visual cues in the multisensory perception of motion. The fact that this correlation was not found in older adults might be due to the smaller sample size in older adults compared to younger adults or due to the fact that field dependence scores for older adults showed a much larger variability.
An age-related difference in field dependence between older and younger adults was observed in this study, with older adults being more field dependent than younger adults overall, possibly to compensate for decline of the vestibular and somatosensory systems that occurs with age. During ageing, sensory accuracy tends to naturally decline, resulting in less reliable information from visual and vestibular systems.29,72 Previous studies have suggested that older adults might overweight visual cues, even if they provide unreliable information with respect to spatial orientation, 73 which may explain the current findings. However, field dependence cannot be the only determinant of vection as older adults who are more field dependent do not have more vection than younger adults.
Previous studies found mixed results with regards to age-related differences in vection37,38 and other factors than field dependence have been linked to vection in older adults. For instance, Murovec et al. 39 found increased vection intensity and duration in older than in younger adults using a multisensory stimulus including visual, auditory, and tactile cues. In contrast, when inducing vection without other sensory stimulation, no differences in subjective vection rating 40 or in neurocortical activity 74 related to vection were found between younger and older adults. In the present sample of participants, vection remained rather stable with increasing age. This finding suggests that, regardless of a potential decline in sensory accuracy associated with natural ageing, this phenomenon may remain rather stable over the lifespan.
Sex-related differences in vection have been sparsely investigated in the past, and the few studies that have been published revealed mixed findings. For instance, Darlington 54 reported shorter vection onset times for women compared to men, whereas other studies failed to find any difference between the two sexes with regards to vection.43,75 Here, we did find a significant difference in vection duration, with women reporting longer lasting vection compared to men. Although statistically significant, the weak effect size (η2 = 0.016) suggests that this difference is rather small. No sex-related difference was found with regards to vection intensity, supporting previous studies with similar results. 21
Lastly, we found supporting evidence for the notion that vection and VIMS are positively linked to each other. Although vection is certainly not sufficient to cause motion sickness (i.e. many studies inducing vection do not elicit VIMS), a positive relationship between the two phenomena has been found many times in the literature, with studies suggesting that vection might either be a main contributor to VIMS16,76 or a prerequisite for VIMS. 14 Here, we found supporting evidence that vection and VIMS are indeed positively linked to each other, although the precise nature of this relationship remains vague. For instance, it is possible that stronger vection may cause more severe VIMS due to a perceived stronger visual-vestibular conflict and/or increased postural instability. At the same time, vection and VIMS are often recorded in close temporal proximity, which may lead to a response bias or carry-over effects (e.g. participants associating an increased level of VIMS with increased vection). More recently, Teixeira and colleagues introduced the unexpected vection theory of VIMS, suggesting that variations in the perception of vection during a visual stimulation (e.g. experiencing vection when no vection is expected) may be a key contributor to VIMS.77,78 Although this theory is very recent and more supporting evidence is desirable, the idea of this theory is intriguing, as it resonates well with the current sensory conflict models with vection in the centre of attention. Future work may investigate this theoretical concept and may add to our understanding of vection as a contributor to VIMS.
It is noteworthy that we did not record balance and posture during the present studies which may also impact the level of VIMS17,79 and might be associated with vection. 80 This is particularly interesting with regards to potential age-related differences, as postural stability naturally decreases as a result of ageing. 81 Although we recruited participants who were healthy based on self-reports with no known history of vestibular disorders or balance issues, we cannot dismiss the idea that decreased postural stability in the older population may have impacted our findings. Future studies could combine self-reports with postural sway measures while also considering objective physiological responses associated with VIMS such as alterations in heart rate, respiration rate, or cortical activity. This holistic approach will provide deeper insights into the complex nature of VIMS which can then be utilized for the assessment of VIMS in clinical practice and the identification of at-risk individuals.
Conclusion
The current study investigated the relationship between VIMS, vection, and several individual-difference factors including biological sex, age, and field dependence. Our results indicated that field dependence was, overall, not associated with VIMS. Interestingly, older adults reported significantly lower VIMS severity compared to younger adults, whereas female participants reported greater VIMS ratings and longer vection than male participants (although these effects were rather weak, reflective of the mixed results that have previously been reported). However, vection was significantly correlated with all VIMS measures across both age groups, emphasizing the potential link between these two phenomena. The findings from this study highlight the relevance of selected individual-difference factors in the context of VIMS and vection, while suggesting that field dependence plays a minimal role.
Footnotes
Acknowledgements
This paper contributed to the North American Treaty Organization Research Task Group 346 entitled Assessment of Factors Impacting Cybersickness. We thank Aalim Makani, Dr William Chung, Dr Brandy Murovec, and Dr Katharina Pöhlmann for conducting the experiments included in this manuscript.
Ethical approval
All studies included in this manuscript were conducted in accordance with the principles specified in the Declaration of Helsinki and were approved by the research ethics boards of the University Health Network and Toronto Metropolitan University. Written consent was obtained prior to the beginning of the experiment from all participants.
Consent to participate
All participants provided written consent to participate in the research studies.
Consent for publication
All participants provided written consent that de-identifiable results of the research studies will be published in scientific, peer-reviewed journals.
Author contributions
The authors confirm contribution to the paper as follows:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies included in this manuscript have been supported by grants from the Natural Sciences and Engineering Council of Canada (RGPIN-2017-04387) and Mitacs (IT28850, IT27426) awarded to Behrang Keshavarz.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data of this research study will be made available per individual request pending approval from the ethics boards of the University Health Network and the Toronto Metropolitan University.
