Abstract
Purpose
To estimate the prevalence of endolymphatic sac hypoplasia (EShp)—a proposed specific finding in Ménière’s disease (MD) that defines an endophenotype characterized by bilateral involvement, male predominance, temporal bone abnormalities, and familial clustering—in individuals without MD, to assess its specificity for the condition.
Methods
We analyzed 956 temporal bone CT scans from individuals without MD to assess the prevalence of EShp using the Angular Trajectory of the Vestibular Aqueduct (ATVA) marker. ATVA distribution, reproducibility, and associations with clinical variables were also evaluated.
Results
EShp was identified in 6 ears from 4 individuals, yielding a prevalence of 0.6% per patient and 0.8% per ear. ATVA values had a median of 95.0° (IQR = 12.5°, range 65.9°–159.4°). Interobserver agreement was good (ICC = 0.75), with a mean bias of 6.2° ± 5.4° and 5.5% of ears outside the 95% limits of agreement. No significant associations were found between ATVA and sex, age, or clinical diagnosis.
Conclusions
EShp is rare in individuals without MD, supporting its specificity and potential role in MD pathophysiology. The ATVA marker is reliable and reproducible and may serve as a CT biomarker for the hypoplastic MD endophenotype.
Introduction
Meniere’s Disease (MD) is an inner ear disorder characterized by recurrent episodes of spontaneous vertigo, accompanied by fluctuating sensorineural hearing loss (HL) and tinnitus,1,2 with considerable variability in onset, progression, and severity across patients. Its etiology remains unclear but seems to arise from complex interplay of genetic and environmental triggering factors. Current diagnostic guidelines are based primarily on clinical symptoms, 3 making it challenging due to the diverse presentation between patients and presence of comorbidities such as allergies, autoimmune or autoinflammatory disorders, and migraine.2,4 To address this heterogeneity, current strategies have focused on stratifying MD patients into subgroups using clinical,5,6 genetic,7–10 molecular,11–19 or histopathological markers.4,20–25
The endolymphatic sac (ES) is a non-sensory membranous organ in the inner ear involved in regulating endolymph homeostasis, mediating immune responses, and clearing metabolic waste.26–31 Its intraosseous portion originates in the vestibule and travels through the vestibular aqueduct (VA) to the operculum, where it extends extraosseously to rest on the dura mater.32,33 Developmental abnormalities of the ES and VA have been frequently observed in MD, suggesting a key role in its pathogenesis.21,25,34 Supporting this, Eckhard et al. consistently identified ES pathology in temporal bone specimens of MD patients, differentiating two histopathological subtypes based on abnormalities in its extraosseous region: defined as (i) degenerative or (ii) hypoplastic. 35 The ES in the degenerative subtype displayed epithelial apoptosis and fibrosis, while it terminated abruptly at the operculum in the hypoplastic cases. Bachinger et al. later established a surrogate radiological marker to differentiate these subtypes based on VA trajectory using MRI or CT imaging. 36 In a larger European cohort, the identification of the two subtypes was associated with different clinical profiles. Patients with degenerative endophenotype experienced more frequent vertigo attacks, late onset, reduced vestibular function, and unilateral involvement, suggesting disruptive etiology in adulthood. In contrast, hypoplastic patients exhibited early onset, bilateral involvement, male predominance, and a family history of hearing loss or vertigo, suggesting a genetic basis for early ES developmental collapse. The authors propose these endophenotypes as novel clinical variants of MD, distinguishable by the diagnostic marker. 37
According to these studies, the hypoplastic endophenotype is found in approximately 30% of MD patients. 37 However, fewer than a hundred temporal bone CT scans from control individuals were analyzed,35,36 an insufficient sample size to conclusively determine the absence of this finding in the general population. Therefore, the objective of this study is to confirm the absence of hypoplasia of the endolymphatic sac (EShp) in individuals without MD and, if present, to provide an accurate estimate of its prevalence.
Material and methods
Recruitment of non-MD cohort
A retrospective search of CT was performed in the radio-diagnosis databases of two tertiary hospitals in southern Spain between January 2021 and January 2024. Patients with non-contrast ear CT scans with a thickness of 5 mm in the axial plane were included. Exclusion criteria included (i) a definitive diagnosis of “Meniere’s Disease”, according to the diagnostic guidelines by the Classification Committee of the Bárány Society in 2015 3 ; and (ii) age ≤ 40 years at the time of the CT scan, according to the late onset of MD. That study population was defined as non-MD cohort. Clinical data including age, sex, self-reported country of origin and clinical diagnosis was collected for all participants. Axial CT planes were obtained for both ears to visualize: (i) the vestibule and horizontal semicircular canal and (ii) the vestibular aqueduct’s exit at the operculum. The study was reviewed and approved by the local Ethics Review Board for Clinical Research (No. SICEIA-2024-003488).
Endolymphatic sac hypoplasia (EShp) evaluation
The presence of EShp was interrogated by analyzing the Angular Trajectory of the Vestibular Aqueduct (ATVA) marker on the CT images based on Bächinger et al. methodology, 36 defining EShp when ATVA ≥ 140°. M.M.-M and P.R.-B. measured ATVA independently in each ear in a double-blind manner. Temporal bones in which it was impossible to trace the VA trajectory were deeply examined in the entire CT. Images with poor quality were discarded from the study.
ATVA data analysis
For clinical and ATVA data, categorical variables are presented as absolute counts and frequencies, while continuous variables are expressed as mean ± standard deviation or median with interquartile range (IQR), depending on whether they did follow a normal distribution or did not, respectively. Type of distribution was assessed by the Shapiro-Wilcoxon test.
The prevalence of EShp among non-MD patients was determined based on the number of patients and ears. In addition to hypoplastic ears (ATVA ≥ 140), we also examined ears with intermediate ATVA values between 120° and 140°. ATVA values ≤ 120° are considered normal in healthy individuals or consistent with degenerative ES patology in MD patients. 36 Clinical variables from patients with both hypoplastic and intermediate ATVA were reviewed to identify potential undiagnosed MD or less pronounced but symptomatic forms of ES pathology.
Reliability between measurements was inspected by calculation of Intraclass Correlation Coefficient (ICC), following the interpretation scores established by Koo et al. 38 (ICC ≤ 0.20: poor; 0.2 < ICC ≤ 0.4: fair; 0.4 < ICC ≤ 0.6: moderate; 0.6 < ICC ≤ 0.8: good; 0.8 < ICC ≤ 1.0: excellent). Agreement was evaluated using the Bland–Altman plot to visualize bias between observers.
The association between ATVA and clinical variables were performed using ATVA values averaged between observers. The relationship of ATVA with sex was evaluated with the Wilcoxon rank sum test, while the relation of ATVA and clinical diagnosis was evaluated employing the Kruskall–Wallis test, due to the non-normal distribution of ATVA within groups in these categorical variables. Pearson correlation and linear regression were used to assess the relationship between ATVA and age. A
Results
Characterization of the non-MD cohort
Summary of demographic characteristics in the non-MD cohort.
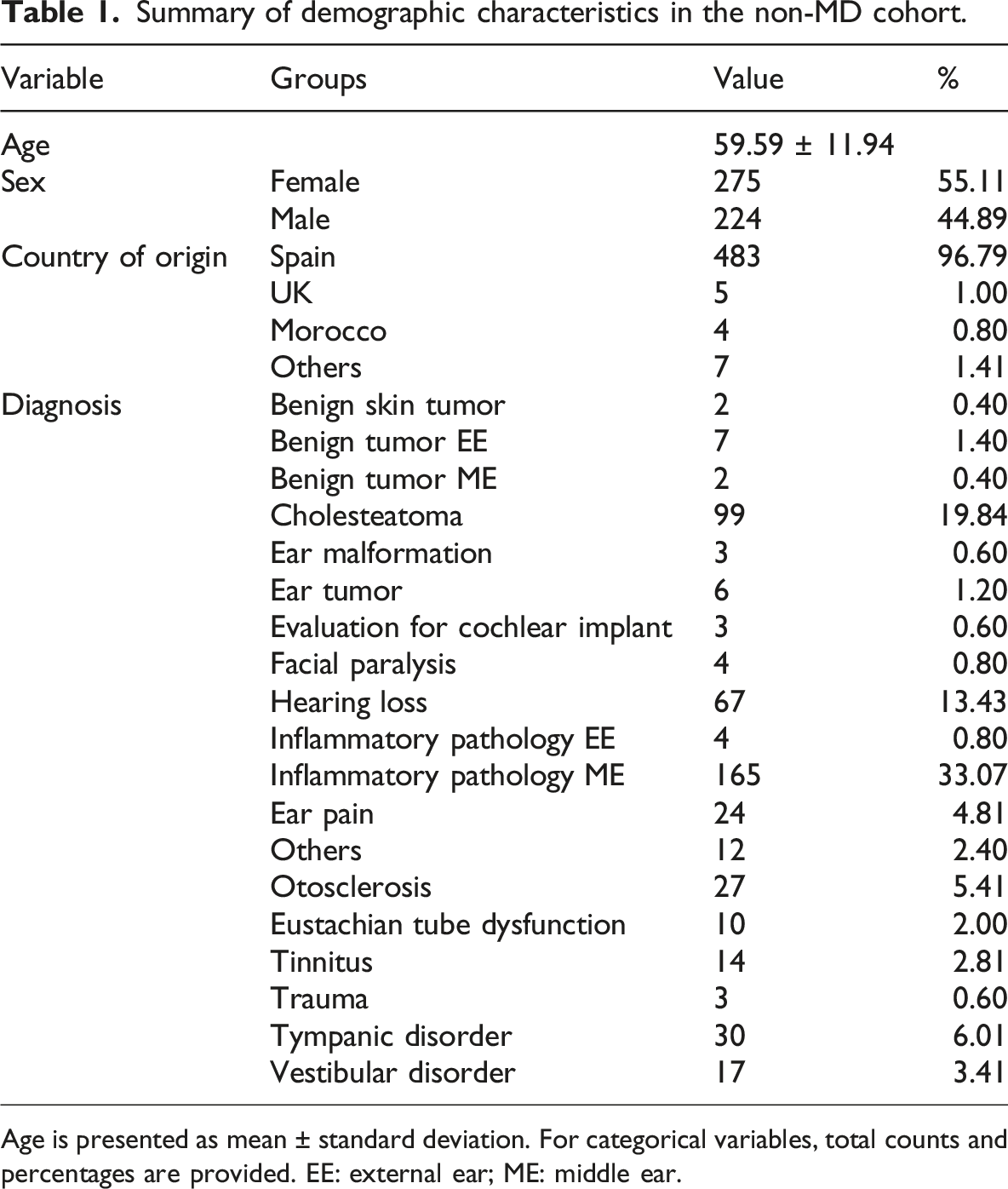
Age is presented as mean ± standard deviation. For categorical variables, total counts and percentages are provided. EE: external ear; ME: middle ear.
Summary of categories and subcategories for diagnostic or reason for CT in patients of non-MD cohort.
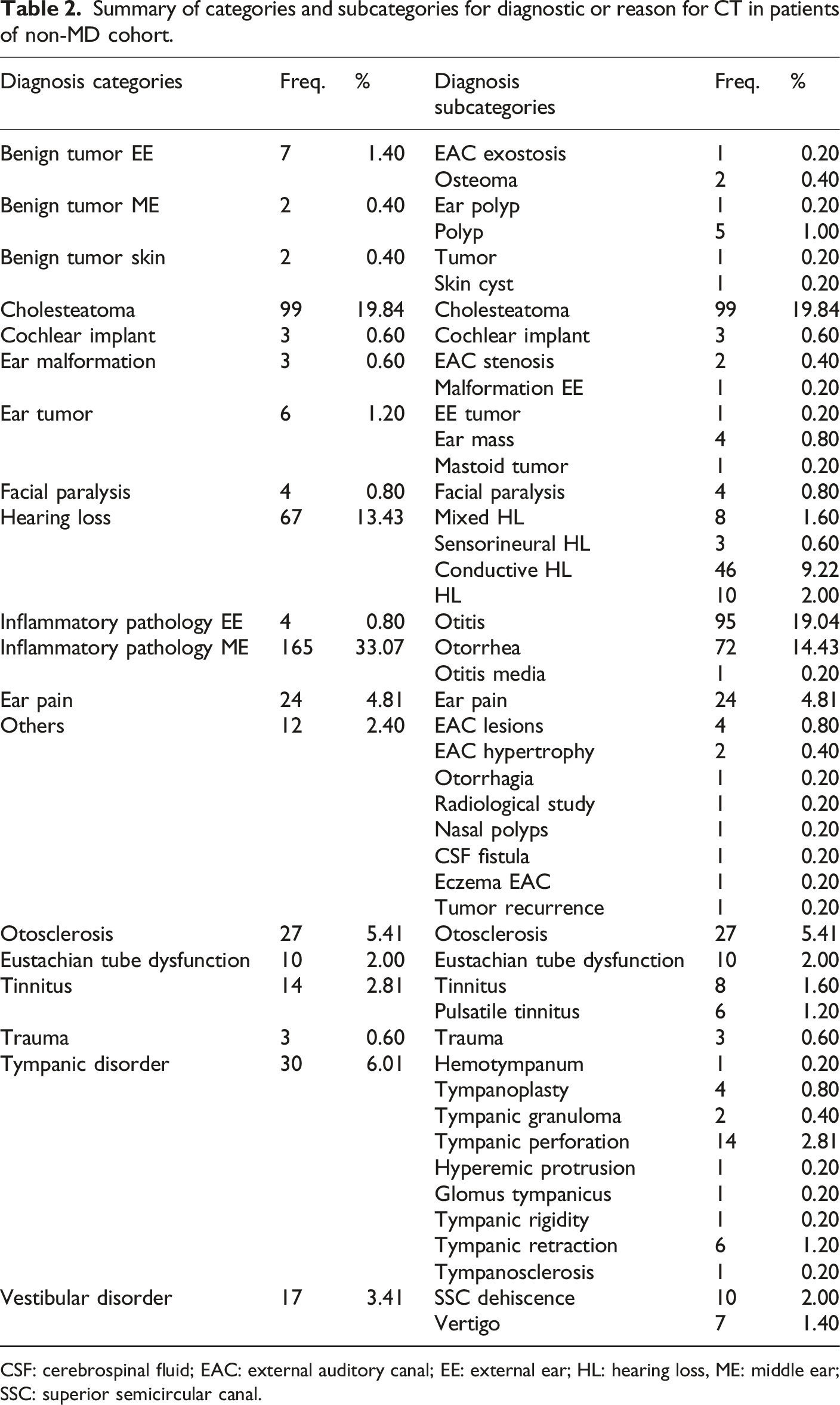
CSF: cerebrospinal fluid; EAC: external auditory canal; EE: external ear; HL: hearing loss, ME: middle ear; SSC: superior semicircular canal.
ATVA distribution and EShp prevalence
ATVA values and clinical variables of patients with at least one hypoplastic or one intermediate value ear.
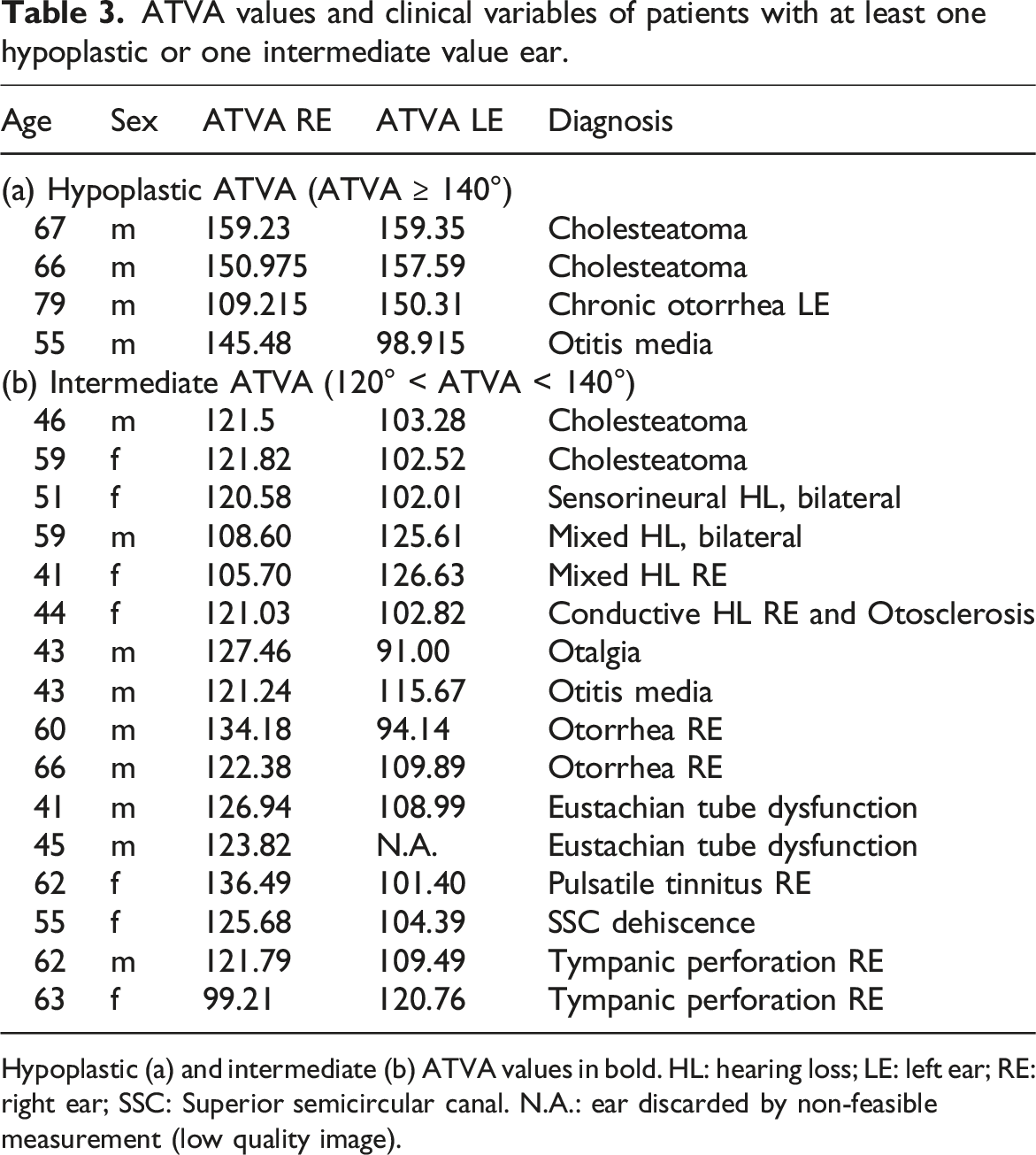
Hypoplastic (a) and intermediate (b) ATVA values in bold. HL: hearing loss; LE: left ear; RE: right ear; SSC: Superior semicircular canal. N.A.: ear discarded by non-feasible measurement (low quality image).
Less pronounced hypoplasia was assessed within the ATVA range of 120°–140° (Table 3, b). Sixteen ears (1.7%), each from a different patient, were identified within this range and associated with various diagnoses, including cholesteatoma (
The five patients with MD-like symptoms were examined in greater detail. Among them, two patients were diagnosed with mixed hearing loss: one female with a left ATVA of 126.6°, affected in the contralateral ear, and one male with a left ATVA of 125.6°, a hearing aid user with severe mixed hearing loss in the right ear and moderate-to-severe mixed hearing loss in the left ear, along with a notable family history of hearing impairment. The third patient was a female with a right ATVA of 121.03°, presenting with otosclerosis and conductive hearing loss ipsilaterally. The fourth case involved a female with bilateral severe-to-profound sensorineural HL, undergoing CT evaluation for cochlear implantation, and a right ATVA of 120.6°. The fifth patient was a female with a right ATVA value of 136.49, with pulsatile tinnitus.
Representative cases from normal, intermediate, and hypoplastic ATVA values are included in Figure 1. Comparison of ATVA measurements between normal (A–A′), intermediate (B–B′), and hypoplastic (C–C′) cases. Two axial CT planes per case were selected to position the anatomical landmarks used for ATVA measurements: (i) the magenta shape and red line marking the bony boundaries of the vestibule and horizontal semicircular canal (A, B, C); and (ii) the trajectory of the vestibular aqueduct as it exits into the posterior cranial fossa, marked by the green line (A′, B′, C′).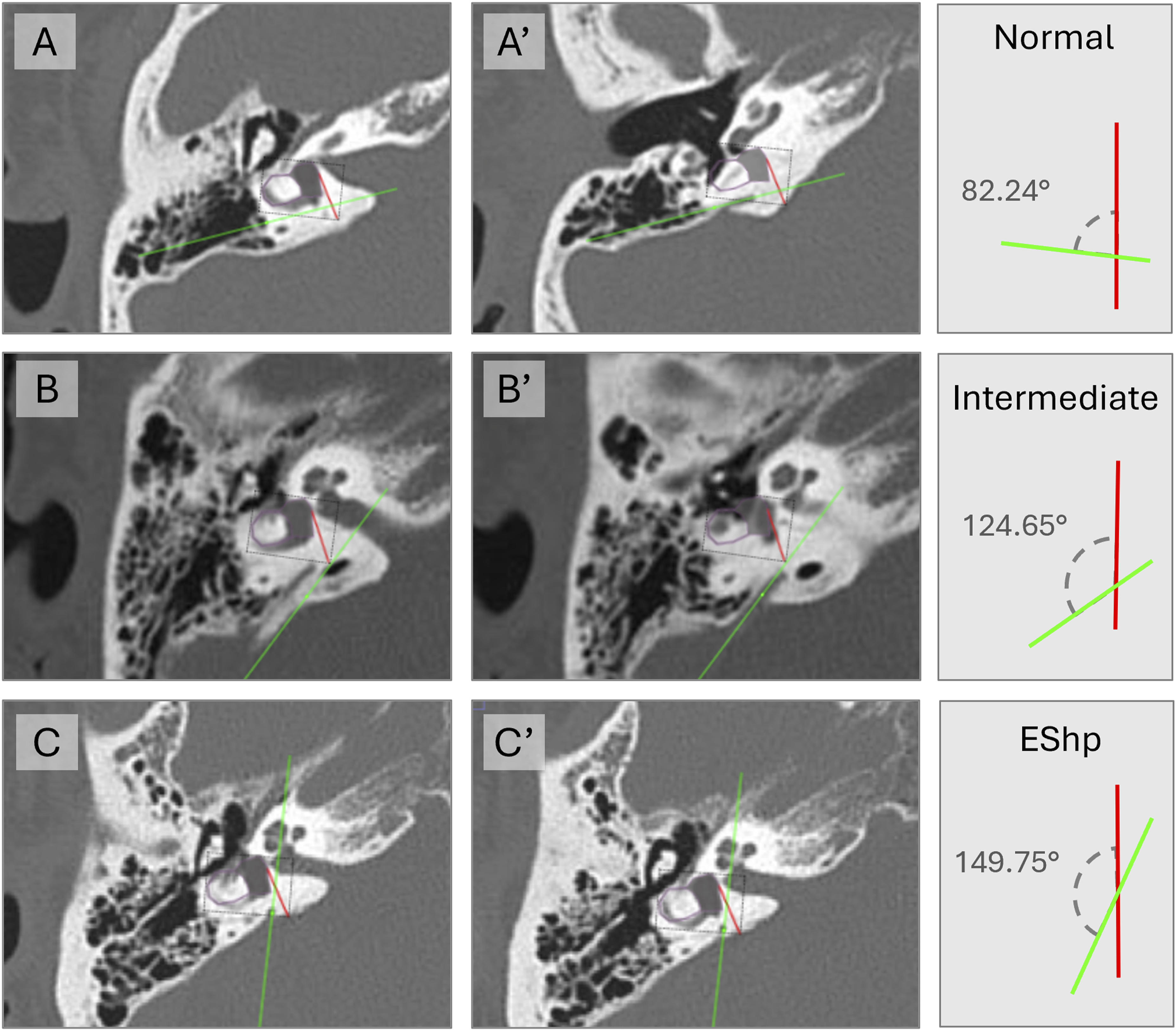
ATVA marker and method reproducibility
ATVA values per each observer, as well as the mean between observers showed a non-normal distribution according to the results of the Shapiro–Wilcoxon test (Figure 2(a)). ATVA median was 95.0°, with IQR of 12.5, minimum value of 65.9° and maximum of 159.4°. The Bland–Altman visualization demonstrated poor interobserver agreement in only 53 (5.5%) (Figure 2(b)). Median and standard deviation for absolute bias was 6.2° ± 5.4°, with a maximum value of 33.9°. Calculated ICC was 0.75 ( ATVA distribution and reproducibility. (a) Non-normal distribution of ATVA for: (i) observer one (blue), W = 0.96, 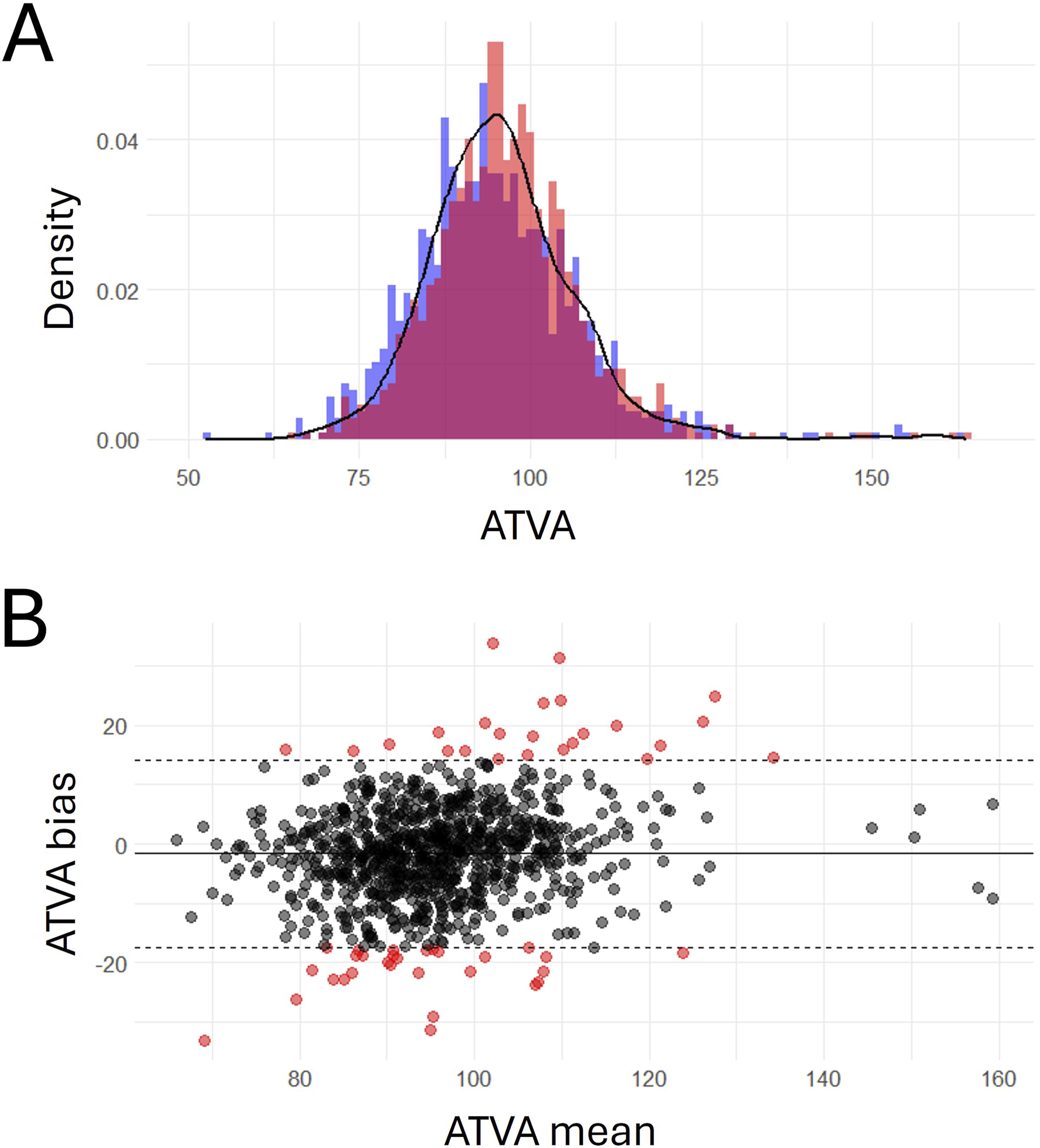
ATVA versus age, sex or diagnosis
ATVA values did not follow a normal distribution in the comparable groups conforming the sex and clinical diagnosis variables. No statistically significant differences were found between the median ATVA angles of men and women (W = 104791; Association between clinical variables and ATVA. No association was found between ATVA and (a) sex (Mann–Whitney W = 104,791; 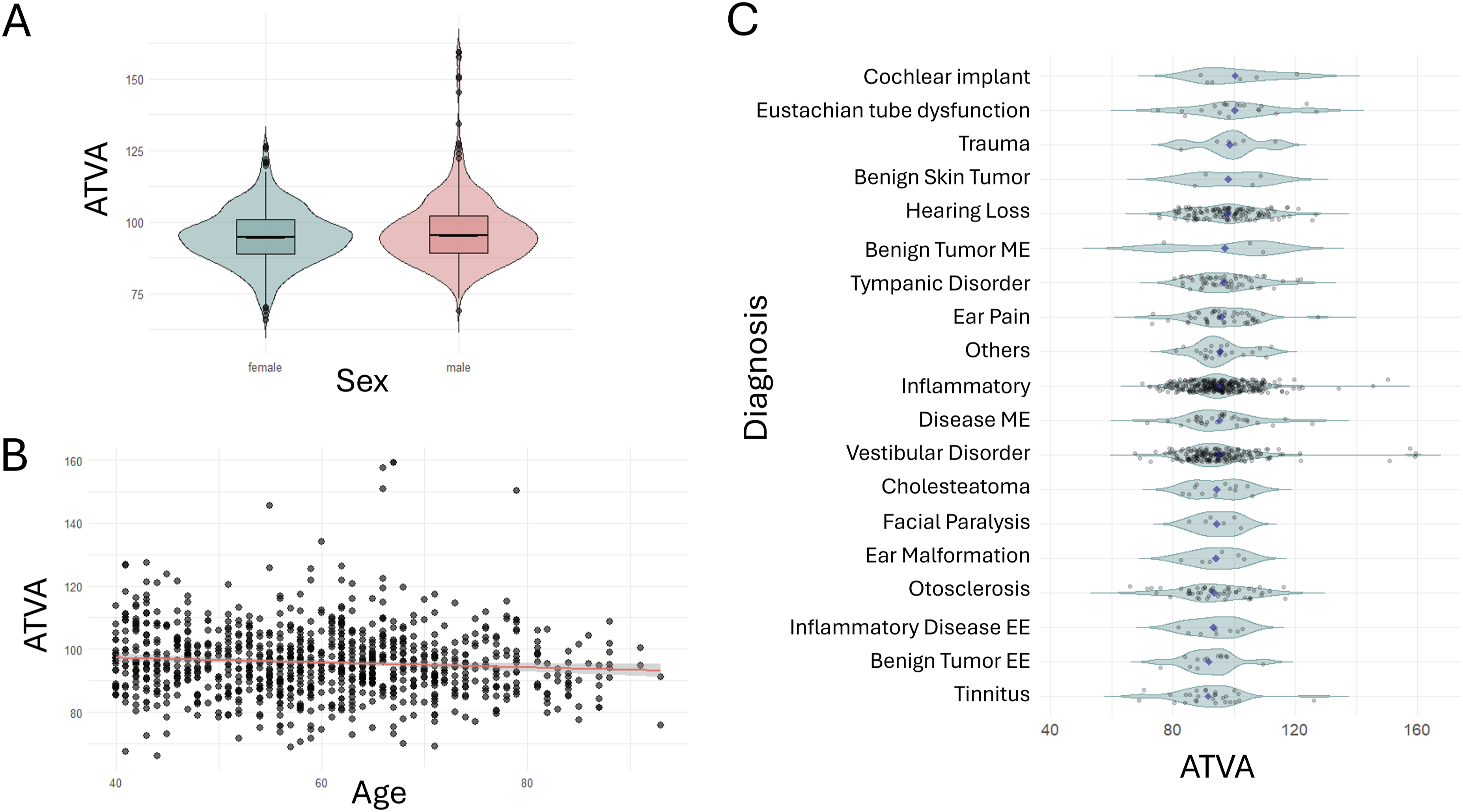
Discussion
The radiological marker ATVA is a surrogate marker for ES pathology, developed to differentiate the two endophenotypes consistently observed in MD.35,37 One of these, the hypoplastic endophenotype, is a rare finding present in approximately 30% of patients, predominantly males, who exhibit early disease onset, bilateral involvement, and a family history of vertigo, sensorineural hearing loss, or MD.35,37 Histopathological analysis of hypoplastic ES reveals a shortening of the endolymphatic sac from the operculum, the region where it normally undergoes its greatest extension and functional epithelial surface growth.35,39,40 Radiological assessment of ATVA measurements on axial CT scans demonstrates that the vestibular aqueduct describes a steep trajectory within the temporal bone, abruptly descending toward the cranial fossa, resembling the morphology observed in fetuses.36,39 These observations support an early developmental arrest as the underlying cause of this endophenotype, suggesting a potential genetic basis.35,37 However, given the rarity of this finding, it is critical to verify that it is truly restricted to MD patients. The limited number of controls in the discovery studies—20 ears from 10 histological cases 35 and 126 ears from 87 histological/radiological cases 37 —highlights the need for evaluation of a larger control population, in order to conclusively establish the specificity of this marker for MD.
Here, we conducted the largest study to date analyzing the presence of hypoplasia of the endolymphatic sac (EShp) in a non-MD control cohort in 494 individuals. Our results support that this finding is rare and likely specific to the disease and reinforce the use of ATVA as diagnostic marker for MD subgroup classification in the clinical practice.
Comparison of Non-MD cohorts evaluated by ATVA.
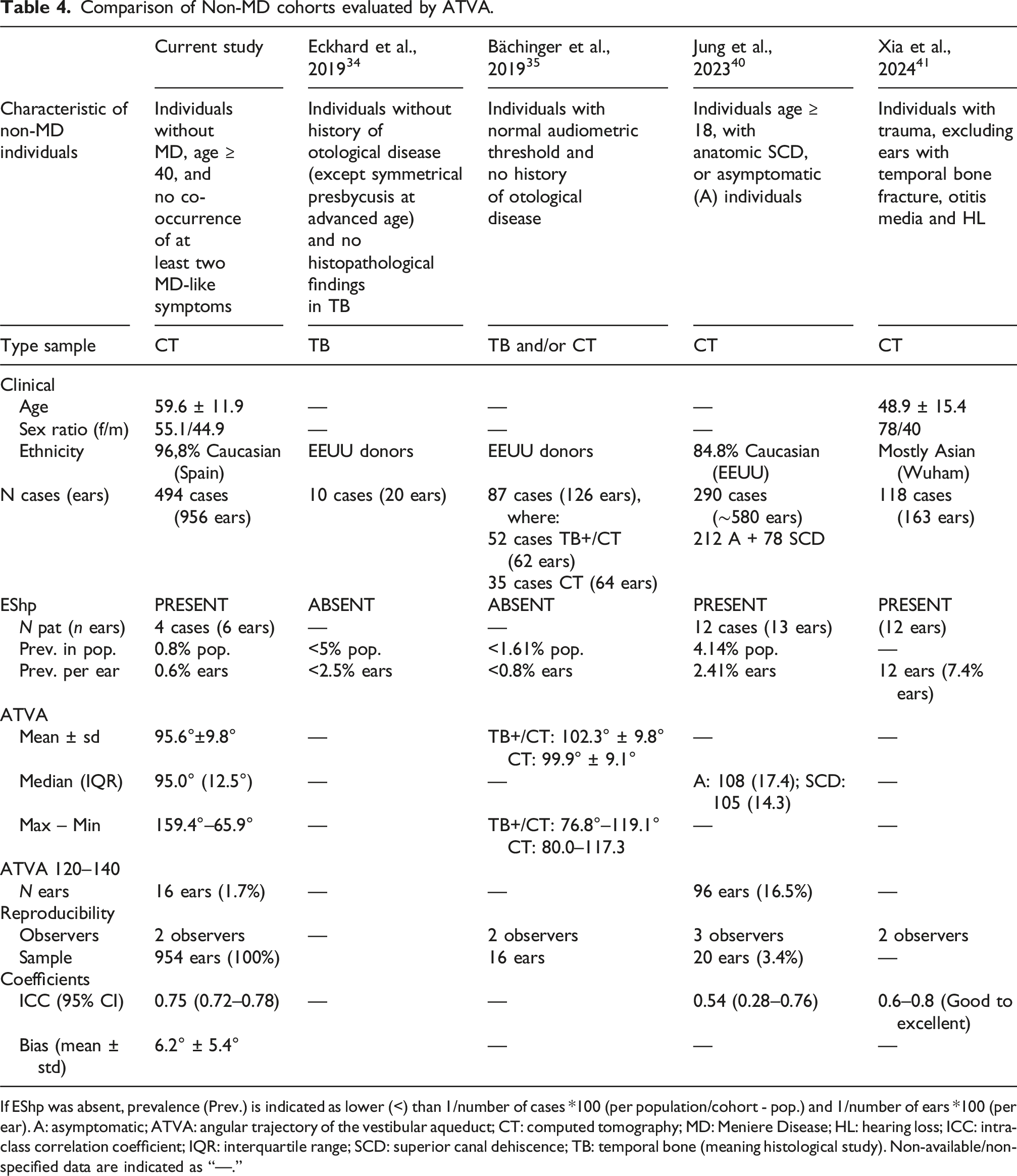
If EShp was absent, prevalence (Prev.) is indicated as lower (<) than 1/number of cases *100 (per population/cohort - pop.) and 1/number of ears *100 (per ear). A: asymptomatic; ATVA: angular trajectory of the vestibular aqueduct; CT: computed tomography; MD: Meniere Disease; HL: hearing loss; ICC: intra-class correlation coefficient; IQR: interquartile range; SCD: superior canal dehiscence; TB: temporal bone (meaning histological study). Non-available/non-specified data are indicated as “—.”
The median ATVA was 95.0° (IQR: 12.5°), reflecting low variability and aligning with previous findings,36,41 though our population showed more extreme maximum and minimum values (Table 4). As the marker angle transitions gradually from degenerated/normal to the hypoplasia, we examined patients with ATVA ear values between 120º and 140° for subtle hypoplasia. This subgroup included 16 ears (1.7%) with diverse diagnoses (Table 3); however, no consistent association was found between intermediate ATVA values and MD-like symptoms such as tinnitus or hearing loss (mixed, sensorineural, or conductive). Jung et al. (2022) reported a higher proportion (16, 5%) of ears in this range, none of which showed with MD-like features. 41 These intermediate angles therefore do not appear to be linked to any specific clinical profile but rather reflect that ATVA varies along a continuous spectrum. Considering this, values between 120° and 140° may represent normal anatomical variation in both control individuals and MD patients with degenerative endophenotype. Although the ≥140° threshold remains a conservative and specific cut-off for hypoplasia, intermediate (120°–140°) and borderline 140° values should be interpreted with caution and ideally assessed in conjunction with other imaging43,44 and clinical markers to determine whether they represent true hypoplasia in MD.
The hypoplastic ES endophenotype has been more frequently described in men with early-onset presentation and is often associated with temporal bone abnormalities such as semicircular canal dehiscence (SCD). 37 This is consistent with our four hypoplastic cases, all male, and with findings by Jung et al. (2022), who identified SCD in 3 of 14 hypoplastic ears within their control cohort. 41 This highlights the value of imaging to detect temporal bone anomalies—such as vestibular canal defects—typically linked to congenital origin and symptoms like sound-induced vertigo, pulsatile tinnitus, autophony, or hearing loss. 45 In fact, other temporal bone features, including retrolabyrinthine bone thickness, posterior contour tortuosity, and pneumatized volume, have also been shown to distinguish between MD endophenotypes.43,44 No correlation was found between ATVA and age, sex, or diagnosis in our entire cohort; and Xia et al. (2024) reported no association between ATVA and peri-vestibular aqueduct pneumatization, vestibular aqueduct morphology, or distance between the posterior semicircular canal and the posterior cranial fossa in control ears. 42
A limitation in these studies has been the selection of an appropriate control cohort, as high-resolution CT scans are obtained from ENT patients with suspected ear-related conditions. To minimize the inclusion of possible undiagnosed MD cases, individuals with confirmed MD and those under 40 years old were excluded, given MD’s delayed onset around age 60. 6 Xia et al. reported a similar mean age of 50, though the minimum age was unspecified, 42 while Jung et al. included individuals over 18 years. 41 In addition to age, the control population in the other investigations excluded individuals with MD-like symptoms, otological disorders, or temporal bone abnormalities (Table 4).
As more investigations utilize the ATVA marker, it is crucial to assess its reproducibility. In this study, both observers measured ATVA on the 956 CT scans. The Bland–Altman plot revealed a lack of agreement in 5.5% of ears, suggesting potential unreliability in a small percentage of measurements. Significant variability was observed in bias, with differences between observers reaching up to 33.9°, which could impact classification as “normal” or “hypoplastic.” The intraclass correlation coefficient (ICC) of 0.75 indicated “good agreement.” “Moderate” and “Good to Excellent” classification has been described by other authors.41,42 However, we observed that ATVA measurement can be particularly challenging in hypoplastic cases, where the vestibular aqueduct is extremely narrow and difficult to trace, sometimes traversing pneumatized regions of the temporal bone. Accurate assessment in such cases requires detailed anatomical knowledge and experience. The method would greatly benefit from standardized protocols and accessible training materials, ideally including CT examples and dynamic axial walkthroughs of challenging or ambiguous cases. These tools would support more consistent evaluations, especially if ATVA is to be adopted clinically for endophenotype identification and guide patient-specific management strategies.
The finding of a higher prevalence of hypoplastic ES in MD patients, as well as its absence or very low presence in control populations, has been consistently replicated across different studies and various population ancestries. The discovery cohort was based in specimens from the temporal bone collection at Mass Eye and Ear, Boston, mainly representing North American donors, 35 and subsequent retrospective and prospective CT studies was performed in Americans, 41 Asians, 42 and Europeans, including Swiss population from Bachinger et al.,36,37 and the present study primarily focusing on Spanish individuals. Collectively, all these studies support that EShp is a rare finding in the general population, but a common finding in MD.
Conclusions
The EShp is a rare radiological finding, with a very low prevalence in patients with otological disorders other than MD. This supports the potential of ATVA as a diagnostic marker for distinguishing between clinical variants and endophenotypes within MD. ATVA’s strong reproducibility and its easy application to CT imaging further supports its implementation in routine clinical practice, enhancing diagnostic accuracy and informing therapeutic decisions. Additionally, ATVA could help identify more homogeneous subgroups for future studies to get a better understanding of the MD pathophysiology through molecular and genetic approaches.
Footnotes
ORCID iDs
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the local Ethics Review Board for Clinical Research (No. SICEIA-2024-003488) with the need for written informed consent waived.
Author contributions
J.A.L-E, J.L.M-R, and P.R-B.: study conception and design; R.M-M, I.B-D, and J.L.M-R: CT acquisition and clinical data collection; M.M-M and P.R-B.: ATVA measurement and database normalization; P.R-B: data analysis and interpretation; P.R-B and J.A.L-E: drafted the manuscript; M-M.M, R.M-M, I.B-D, and J.L.M-R: critically reviewed and revised the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Jose A Lopez-Escamez has received funds to support MD research from The University of Sydney (K7013_B3413 Grant).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are not publicly available due to ethical and privacy considerations. However, de-identified data may be made available from the corresponding author upon reasonable request and pending institutional approval.
