Abstract
Avoiding activities posing bodily threat is adaptive. However, spreading of avoidance to safe activities may cause functional disability in people with chronic pain. We investigated whether costly pain-related avoidance would generalize from one activity to another on the basis of real-life categorical knowledge in 40 pain-free people (30 female; mean age = 25 years; university students and public of Maastricht, The Netherlands). In a computer task, participants moved a joystick to complete activities from two categories (gardening and cleaning). During activities from the avoidance category, pain could be avoided at the cost of task efficiency by deviating from a short, pain-associated joystick movement. Activities from the safe category were never painful. Subsequently, we tested generalization of avoidance to novel pain-free activities from both categories. Participants generalized avoidance to novel activities from the avoidance category despite the novel activities not being paired with pain and despite avoidance costs, suggesting that costly (pain-related) avoidance generalizes from one activity to another on the basis of category knowledge and can thus be wide reaching, creating detrimental consequences.
Keywords
Chronic pain conditions are the leading cause of disability and disease burden worldwide (GBD 2016 Disease and Injury Incidence and Prevalence Collaborators, 2017). Chronic pain affects approximately 20% of adults (Dahlhamer et al., 2018), is the main reason people seek medical care (Institute of Medicine, 2011), and is characterized by significant daily suffering (Becker et al., 1997; Sprangers et al., 2000). Although no existing treatment can eliminate chronic pain, evidence-based psychological treatments reduce the associated disability and distress (Driscoll et al., 2021) and in some cases even reduce pain (de Jong et al., 2008).
Fear-avoidance models posit that catastrophic interpretations of pain increase fear of pain, injury, and/or reinjury, which motivates avoidance of movements and activities believed to worsen the pain or injury (Vlaeyen & Linton, 2012). Excessive avoidance can lead to activity disengagement and physical disuse (Volders et al., 2015). Critically, safety (e.g., the absence of pain or injury) may be misattributed to avoidance, resulting in a self-sustaining cycle of pain-related fear and avoidance, culminating in disability (Meulders, 2019).
One pathway to excessive fear and avoidance is generalization (Guttman & Kalish, 1956; Lissek et al., 2008). That is, the spreading of fear or avoidance from one painful movement to similar movements never paired with pain (i.e., perceptual generalization; Glogan et al., 2021). Higher order reasoning, such as knowledge about concepts and categories, can also contribute to this process (category-based generalization; Dunsmoor & Murphy, 2015). Dunsmoor and colleagues (2012) found that participants conditioned to associate an aversive electrical stimulus with one category (e.g., tools), but not another (e.g., animals), selectively generalized fear to novel, unreinforced members of the previously reinforced category.
Where fear on its own does not interfere with daily life, avoidance can come at significant personal costs, such as reduced physical health and daily functioning (Meulders, 2019). Avoidance is acquired via instrumental conditioning, whereby learning the consequences of one’s actions shapes future behavior (Skinner, 1953). For example, an avoidance response (e.g., moving with reduced spinal motion; Thomas & France, 2007), which results in or is expected to result in the omission of a negative outcome (e.g., a hernia), will be strengthened (i.e., negative reinforcement; Skinner, 1953).
In a recent study by Kloos and colleagues (2022), participants learned to avoid a low-cost pain-associated arm movement by performing a costly, pain-free arm movement in scenes from one category (e.g., outdoor scenes). Subsequently, participants also more often performed the learned avoidance response when presented with novel, unreinforced outdoor scenes, compared with unreinforced scenes from another category (e.g., indoor scenes). Thus, pain-related avoidance learned in one context generalized to other, safe contexts on the basis of participants’ semantic knowledge about environments (Kloos et al., 2022). In musculoskeletal pain conditions, however, fear of movement and activity is the most important predictor of disability (Roelofs et al., 2004). For example, experiencing a shooting back pain when crouching to weed the garden may result in avoidance of this activity that can subsequently generalize to all activities categorized as “gardening,” despite this being a valued pastime.
Therefore, it may be more useful to investigate avoidance generalization in the pain domain as generalization between activities (i.e., responses; response generalization; Skinner, 1953). Although research exists on avoidance generalization between perceptually similar responses (movements; Glogan et al., 2020, 2021, 2022) and category-based avoidance generalization between contexts (Kloos et al., 2022), whether avoidance of a pain-associated activity generalizes to other, categorically related activities has not been explored. Yet category-based generalization increases the scope and complexity of how we learn to avoid harmless activities.
Therefore, we investigated whether pain-related avoidance generalizes from one activity to another on the basis of categorical knowledge about real-life activities. In a computer environment, participants moved a joystick to complete activities from two categories (gardening and cleaning). That is, visual representations of activities were combined with specific joystick movements. During activities from the avoidance category (e.g., gardening), pain could be avoided by deviating from a short, pain-associated joystick movement at the cost of reduced task efficiency. Activities from the safe category (e.g., cleaning) were never paired with pain. Subsequently, we tested generalization of avoidance to novel unreinforced activities from both categories. Participants could always freely choose whether to avoid, giving rise to natural, as opposed to instructed, avoidance (Krypotos et al., 2018). Furthermore, by making avoidance costly, we modeled the real-life costs associated with avoidance in chronic pain (e.g., disability). We expected participants to continue avoiding pain during novel activities from the avoidance category despite avoidance costs.
Statement of Relevance
Excessive avoidance is a key cause and maintaining factor of chronic pain disability. One way in which people may begin to avoid pain-free activities is through generalization, whereby novel activities are steered clear of by virtue of their similarity to a pain-evoking activity. In this research, we investigated whether healthy, pain-free participants generalize pain avoidance on the basis of their existing category knowledge. As expected, participants continued to avoid pain-free activities that were part of the same category (gardening or cleaning) as activities that had been previously paired with pain. This implies that pain avoidance can generalize from one activity to another on the basis of a person’s individual, potentially wide-reaching category knowledge. Chronic pain is often treated pharmacologically, contributing to the current opioid crisis, yet patients also benefit from psychological (cognitive-behavioral) treatments. The current findings underline the multifaceted nature of chronic pain and the need for more multidisciplinary pain therapies, which incorporate medical, psychological, and physical treatments.
Open Practices Statement
The preregistration for the current study is publicly available on OSF (https://osf.io/bghy7). The data and analytical code from the current study can be found in Sections S1 and S2, respectively, in the Supplemental Material available online.
Method
We report (a) all dependent variables and measures that were analyzed for our target research question; (b) all levels of all independent variables and all predictors or manipulations, whether successful or failed; and (c) 3 pilot participants were tested before the official data collection started. Once data collection started, no participant exclusions were made. Thus, 43 people participated in total, with 40 participating in the final version of the experiment.
Participants
A convenience sample of 40 healthy, pain-free volunteers participated in this study (30 female; age: M = 25 years, SD = 5, range = 19–44). The sample size was determined by an a priori power calculation for a dependent-samples t test (based on the difference in mean avoidance behavior between conditions during the test of generalization; G*Power, Faul et al., 2009; α = .05, power = .80, d = 0.50), which yielded a minimum sample size of 34. Because of the novelty of the paradigm and research question, we chose a moderate effect size for determining the power of the study. Furthermore, we collected data from 40 participants to account for lower expected effect sizes during the generalization phase (compared with the acquisition phase). Participants were recruited through the research participation system of Maastricht University (Sona Systems, Nijmegen, The Netherlands), advertisements distributed around the university campus, and word of mouth and social media. Exclusion criteria included being under 18 or over 65 years of age; pregnancy; chronic pain; acute pain in dominant shoulder, arm, elbow, wrist, or hand; current or history of cardiovascular disease and/or psychiatric disorders (e.g., depression, anxiety); electronic implants (e.g., pacemaker); uncorrected hearing and/or vision problems; diagnosed dyslexia or analphabetism; other serious medical conditions; and being advised to avoid stressful situations by one’s general practitioner. All participants provided informed consent and received either 1.5 course credits or €12.50 in gift vouchers as compensation. We terminated data collection once the sample size of 40 was reached. The Ethics Review Committee Psychology and Neuroscience of Maastricht University approved the study protocol (Registration No. 185_09_11_2017_S11).
Apparatus
The experiment was programmed in C# using the cross-platform game engine Unity 2019 (Unity Technologies, San Francisco, CA) and 3D graphics software Blender 2.8 (Blender Foundation, Amsterdam, The Netherlands). The experimental script was run on a Windows 10 Enterprise 64-bit Intel Core desktop computer with 8 GB RAM and an Intel Core i7-6700 CPU processor at 3.40 GHz. An NVIDIA NVS 315 graphics card (Nvidia Corporation, Santa Clara, CA) was used with 1,024 MB of video RAM. The task was presented on a 24-in. ProLite computer screen (B2483HS-B1; Iiyama Corporation, Hoofddorp, The Netherlands). Participants used a Logitech Attack 3 joystick (Logitech International, Lausanne, Switzerland) to perform the task.
Stimulus materials
Pain stimulus
The pain stimulus was a 2-ms square-wave electrocutaneous stimulus delivered by a constant current stimulator (DS7; Digitimer, Welwyn Garden City, United Kingdom) through two reusable 8-mm silver chloride electrodes (Coulbourn V91-01; Coulbourn Instruments, Allentown, PA) filled with SignaGel Electrode Gel (Parker Laboratories, Fairfield, NJ) to the wrist of the dominant hand.
The intensity of the pain stimulus was individually determined using a standard calibration procedure (e.g., Meulders et al., 2011). Participants received a series of electrical stimuli gradually increasing in intensity in 1-, 2-, 3-, or 4-mA increments. The intensity started at 1 mA, and the highest possible intensity was 99 mA (maximum of the constant current stimulator). Participants rated each stimulus on a scale ranging from 0 to 10 (0 = I feel nothing; 1 = I feel something, but this is not painful, it is merely a sensation; 2 = This sensation is not yet painful, but is beginning to be unpleasant; 3 = The stimulus is beginning to be painful; 10 = This is the worst pain I can imagine). Once the participant reached an intensity they would describe as “significantly painful and demanding some effort to tolerate” (roughly corresponding to an 8 on the rating scale), the calibration procedure was terminated, and that intensity was used as the pain stimulus throughout the experiment (pain intensity: M = 20.75 mA, SD = 7.98, range = 2–76 mA).
Activity categories
We used 12 unique activities from two categories of everyday activities: gardening and cleaning. During the acquisition phase, two activities from both categories were presented, and during the generalization phase, four novel activities were presented from both categories. This amounted to six activities from each category. The activities were presented as combinations of a tool and an item matching that tool. For example, a rake was paired with a pile of leaves, and a mop was paired with a puddle of water (see Fig. 1 for an example, Table 1 for all the activities, and Section S3 in the Supplemental Material for visualizations of all activities).
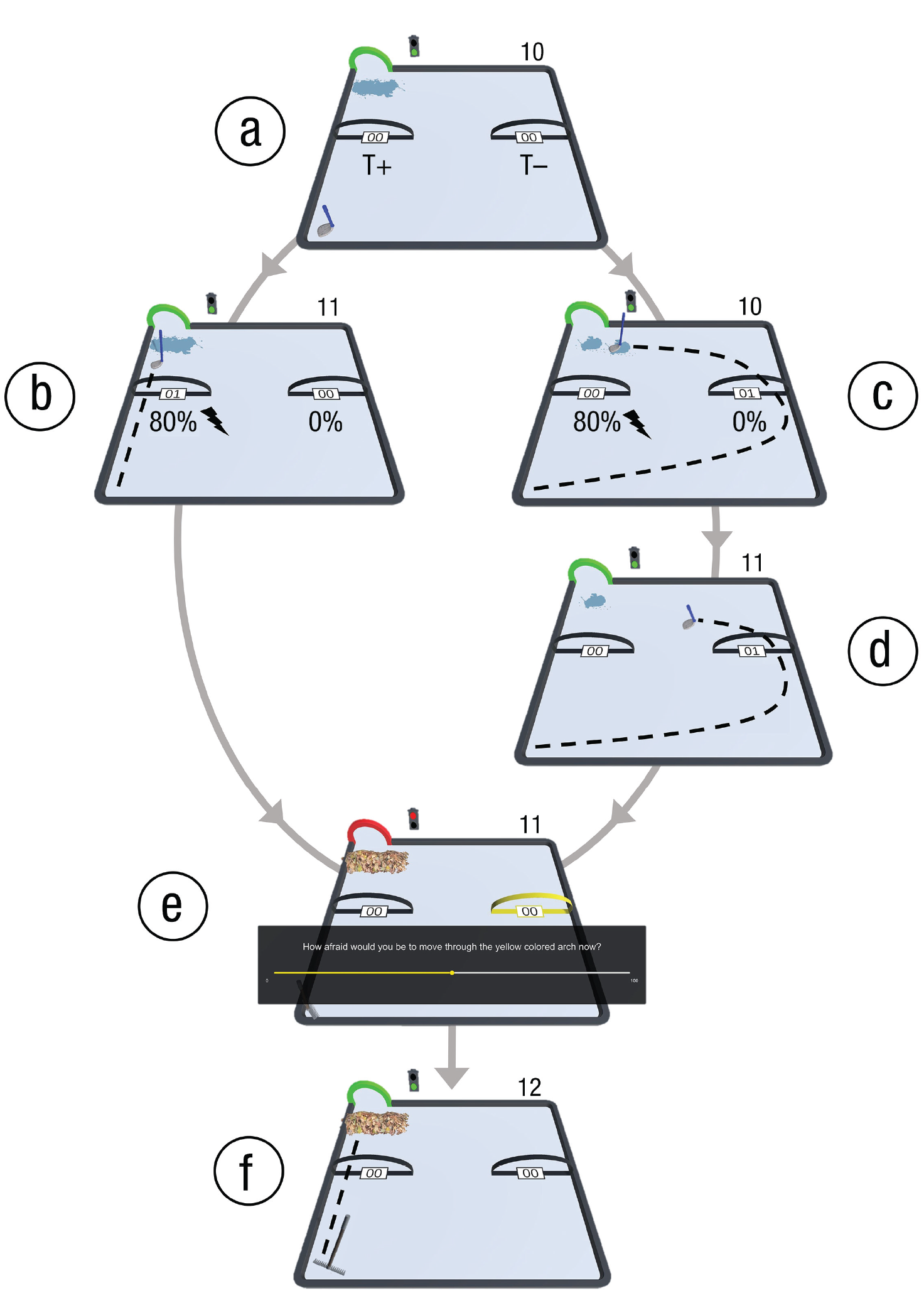
An example trial of the experimental task. (a) The target arch and traffic light turned green as the auditory start tone played. The tool (in this case, a mop) was situated at the lower left corner of the movement plane, and the item (in this case, a puddle of water) was situated in front of the target arch. The movement trajectory arches for T+ and T– were located midway through the movement plane at the left and right sides, respectively. Participants are free to choose between T+ and T–. The trial counter was located above the upper right corner of the movement plane. (b) When T+ was chosen in the avoidance condition, there was an 80% chance that the pain stimulus was presented. The entire item was moved into the target, and the trial counter increased by 1 unit. (c) When T– was chosen, there was no chance that a pain stimulus would be presented. However, there was an 80% chance that only half of the item would move into the target. Thus, the trial was not completed, and the trial counter did not increase. (d) After the first T– movement was performed, the tool returned to the starting position, and the T– movement had to be performed again. The remaining half of the item would be moved into the target. The trial counter increased by 1 unit. (e) Once the item had been moved into the target arch, the target arch and traffic light turned red, and the auditory stop tone played. If the trial included self-report questions, they would appear at the beginning of the new trial, after the new activity had been presented (in this case, raking). (f) After self-reports were provided, the next movement could be performed.
Tool/Item Combinations Used as Activities From the Categories “Gardening” and “Cleaning” in the Current Experiment

Experimental paradigm
Practice phase
The current paradigm was based on the robotic arm-reaching paradigm, a validated operant avoidance-learning paradigm described elsewhere (Meulders et al., 2016). Participants in the current study used a joystick instead of a robotic arm, and they were required to move the joystick to complete everyday activities in a computer environment. On each trial, participants operated the joystick with their dominant hand to move a gardening or cleaning tool from a starting point at the lower left corner of the two-dimensional movement plane to a target, visualized as a green arch at the upper left corner of the movement plane (see Fig. 1). The beginning of a trial was indicated by visual and auditory start signals (a virtual traffic light and the target arch turning green, accompanied by a start tone; for a video of the task, see Section S4 in the Supplemental Material).
Participants could reach the target via two separate movement trajectories, visualized as two black arches midway through the movement plane. Participants could freely choose between the two movement trajectories on each trial. One of the movement trajectories (T+) was the direct pathway from the starting point to the target, and the other (T–) was the longest possible pathway to the target. Furthermore, on their way to the target, participants had to pick up the matching item, which was located in front of the target arch. The item had to be moved into the target.
The trial was completed only after the entire item had been moved into the target. Moving through T+ always resulted in the entire item being moved into the target, whereas moving through T– resulted in only half the item being moved into the target 80% of the time. That is, on 80% of trials in which T– was chosen, only half of the matching item would move into the target. For example, if a participant was presented with the rake and they chose to perform T–, there was an 80% probability that only half of the pile of leaves would move into the target with the rake and a 20% probability that the entire item would move in to the target. Therefore, most of the time the T– movement was chosen, participants needed to perform it twice to complete the activity. Once T– had been chosen, the T+ trajectory arch was blocked until the end of the trial (i.e., the tool would stop before the arch if the participant tried to move through it). Thus, participants were forced to perform T– again to complete the trial. Choosing T– thus came with a cost in terms of distance to the target and task progress. Once the matching item was fully moved through the target arch, a counter positioned above the upper right corner of the movement plane increased by 1 unit (most of the time only after two T– movements). This was to provide participants with information about their task progress. To provide participants with feedback about their choices per trial, we included a separate counter for each trajectory arch, which increased by 1 unit when the tool was moved through the corresponding arch (T+ or T–). Thus, the trajectory arch counter of T– increased by 1 unit whenever T– was moved through, but the overall trial counter increased by 1 unit, only after two T- movements had been performed (80% of the time). Trial completion was indicated by visual and auditory stop signals (the traffic light and the target arch turning red, along with a stop tone).
Eight trials were completed in the practice phase, during which four activities involving different tool/item combinations were presented in pseudorandomized order (gardening category: rake/pile of leaves, wheelbarrow/pile of grass; cleaning category: mop/puddle of water, vacuum cleaner/dust), with the rule that the same category could not be presented on Trials 2 and 7 of this phase. This rule allowed participants to practice providing self-reports of pain-related fear and pain expectancies in both categories. Specifically, at the beginning of Trials 2 and 7, the activity of that trial was presented (e.g., raking or mopping), after which self-reports were collected for both T+ and T– separately (see Self-Reported Pain Expectancy and Pain-Related Fear below). There were no pain stimuli in this phase.
Acquisition phase
This phase was identical to the practice phase, except that the two activities from one category (e.g., gardening; counterbalanced across participants) were now paired with the pain stimulus with an 80% likelihood when moved through T+ (i.e., the avoidance category). Participants learned these contingencies themselves through trial and error. The pain stimulus was delivered when two thirds of the movement had been completed. The two activities from the other category (e.g., cleaning) were never paired with the pain stimulus (i.e., the safe category). Furthermore, moving through T– in any activity was never paired with a pain stimulus. Therefore, participants could avoid the pain stimulus but at the cost of distance to the target and task progress (see Practice Phase above). The acquisition phase comprised four blocks of 12 trials each, during which the four activities were presented in pseudorandomized order. The first trial of the acquisition phase was always in the safe category (e.g., cleaning), and a maximum of two consecutive trials could be in the same category. Furthermore, each activity was presented once during the first four trials of the first acquisition block and the last four trials of the three subsequent acquisition blocks. During these trials, self-reports were collected for both movement trajectories during each activity presented during this phase.
Generalization phase
This phase was similar to the practice and acquisition phases, except that eight novel activities were introduced, four from each category (see Table 1 for a summary). The four original activities were also presented along with the acquisition contingencies (e.g., rake/pile of leaves, wheelbarrow/pile of grass = 80% chance of pain stimulus when moving through T+; mop/puddle of water, vacuum cleaner/dust = 0%). The eight novel activities were never followed by the pain stimulus. However, the avoidance costs remained the same as during the earlier phases, that is, T– also needed to be performed twice during the novel activities. The generalization phase included 12 trials (six from each category) during which all activities were presented once in a randomized order. Self-reports were collected on each trial of this phase. After the generalization phase, an exploratory phase followed in which the combinations of tools and items were intermixed (mixed generalization). The purpose of this phase was to explore (a) whether healthy participants would be conservative and continue to exhibit avoidance behavior even when the combinations did not match and (b) which part of the tool/item combination would drive avoidance (i.e., the tool or the item).
Outcome measures
Behavioral avoidance
Mean maximal deviation
Avoidance behavior was operationalized as the maximal deviation (lateral distance) from the shortest trajectory (a straight line from the start location to the target location) on each trial of the task. Only the first T– movement counted toward this value, given that the second T– movement was forced and thus cannot be interpreted as genuine avoidance behavior. Aligning with the robotic arm-reaching paradigm, the virtual space in which the task was performed was programmed to have a length of 36 cm and a width of 40 cm (Glogan et al., 2020). This information was extracted using the coordinates of each performed movement.
Trajectory choice
Frequencies of choices of T+ and T– were calculated for both categories separately. The program automatically logged this information.
Self-reported pain expectancy and pain-related fear
Self-reports were collected for both movement trajectories. To signal the trajectory to which the question pertained, we highlighted its arch in yellow. Thus, the questions, “To what extent would you expect an electrical stimulus if you would move through the yellow-colored arch now?” (pain expectancy) and “How afraid would you be to move through the yellow-colored arch now?” (pain-related fear) were asked. Questions were presented on screen using a visual analogue scale ranging from 0 to 100 (0 = not at all, 100 = very much). Questions were answered using three buttons on the joystick: one to scroll to the left (toward 0), another to scroll to the right (toward 100), and the third (the trigger button) to confirm the answer.
Data-analysis overview
We preregistered the hypotheses and analysis plan of this study on OSF prior to the start of data collection. We expected larger mean maximal deviations as well as more choices of T– than T+ in the avoidance category, compared with the safe category, during the acquisition phase (category-based avoidance acquisition). We also expected mean self-reports to be higher for T+ compared with T– during the avoidance category and for these to not differ during the safe category (category-based fear acquisition). During generalization, we expected similar patterns of responding but in response to the novel activities (category-based fear and avoidance generalization). We did not have specific hypotheses for the mixed-generalization phase. However, there was a bug in the experimental script, and electrical stimuli were unintendedly delivered during this phase, which we noticed only after data collection. Therefore, this phase was excluded from data analyses.
To test for acquisition and generalization of pain-related fear, pain expectancies (self-reports), and avoidance (maximal deviation), we performed a series of repeated measures analyses of variance (ANOVAs) and a paired-samples t test (avoidance generalization). Repeated measures ANOVAs were followed up with planned contrasts to examine our a priori hypotheses. To investigate the odds of choosing T– rather than T+ in the avoidance category compared with the safe category, we estimated logistic regression models with trajectory choice as the dependent variable and category as the predictor.
The α level was set at .05. For repeated measures ANOVAs, Greenhouse-Geisser corrections were applied to correct for sphericity violations. Holm-Bonferroni-corrected p values are reported for multiple comparisons. The indication of effect size η p 2 is reported for significant ANOVA effects, and Cohen’s d is reported for planned contrasts. The precision of results obtained from our planned contrasts and logistic regressions were estimated using 95% confidence intervals (CIs). Results were deemed statistically significant when CIs did not cross 0. All analyses were performed in RStudio (Version 2021.09.2+382; RStudio Team, 2021) using R (Version 4.1.2; R Core Team, 2021). Repeated measures ANOVAs and planned contrasts were performed with the afex package (Version 1.0-1; Singmann et al., 2021), and logistic regressions were performed using the R package aod (Version 1.3.2; Lesnoff & Lancelot, 2012). All analyses were performed on the complete data set only.
Results
Acquisition
Avoidance
A 2 × 4 repeated measures ANOVA (category: avoidance, safe) × (acquisition block: 1, 2, 3, 4) on maximal deviation data during the acquisition phase yielded the hypothesized two-way interaction, F(2.27, 88.45) = 7.91, p < .001, η p 2 = .17, indicating that the differences in avoidance behavior between categories changed throughout the acquisition phase. As expected, maximal deviations were significantly larger in the avoidance category compared with the safe category, t(39) = 5.66, p < .0001, d = 1.14, 95% CI = [0.66, 1.62]. This was supported by the logistic regression with trajectory choice (T+, T–) as the binary dependent variable and category as the predictor, which showed that participants were 5.57 times more likely to choose T– than T+ in the avoidance category than in the safe category during Acquisition Block 4, exp(1.72), 95% CI = [3.78, 8.29]. Thus, participants learned to avoid more in the avoidance category than in the safe category (see Fig. 2).
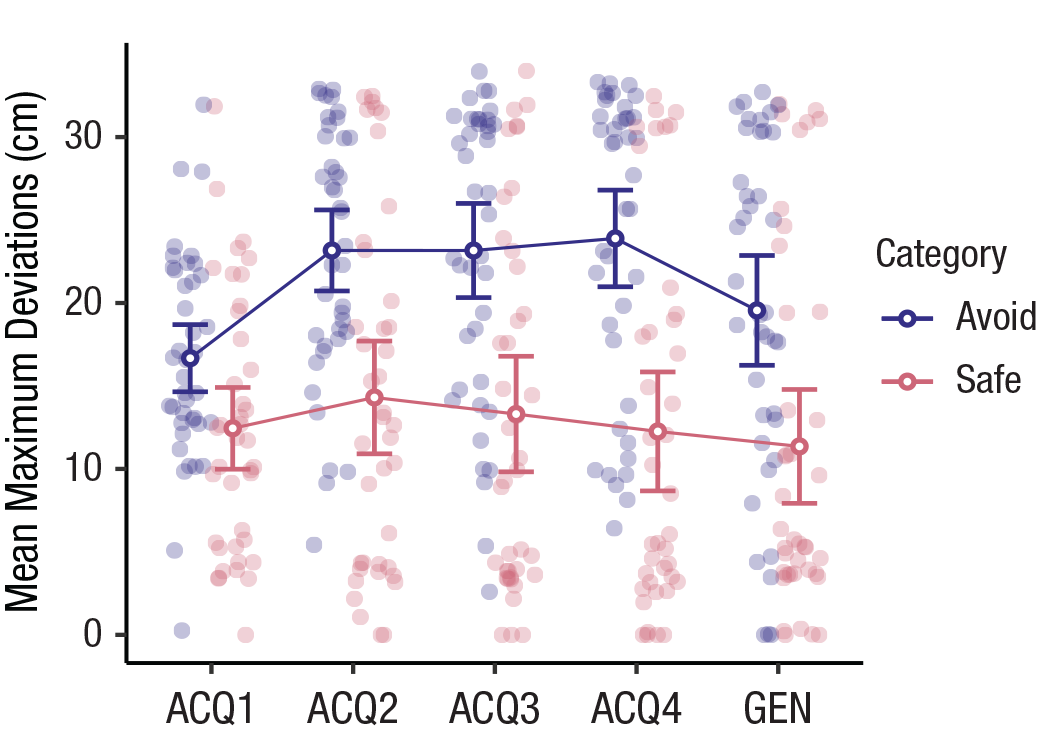
Maximum deviations from the shortest possible movement during the four acquisition (ACQ) blocks and the generalization (GEN) phase, separately for the avoidance and safe categories. Open circles show means, and error bars represent 95% confidence intervals. Filled circles indicate individual participant data.
Self-reported pain expectancies and pain-related fear
A 2 × 2 × 4 repeated measures ANOVA (category: avoidance, safe) × (trajectory: T+, T–) × (acquisition block: 1, 2, 3, 4) on self-reported pain expectancies during the acquisition phase yielded a significant three-way interaction, F(1.26, 49.16) = 24.53, p < .001, η p 2 = .39, suggesting that pain expectancies toward the two trajectories differed between categories and that these differences emerged over time. As expected, planned contrasts confirmed that participants showed higher pain expectancies toward T+ than T– in the avoidance category at the end of the acquisition phase (Acquisition Block 4), t(39) = 14.23, p < .0001, d = 3.45, 95% CI = [2.18, 4.72]. Unexpectedly, these differences also occurred in the safe category, t(39) = 2.46, p = .02, d = 0.63, 95% CI = [0.07, 1.18], albeit to a lesser degree (see Fig. 3).
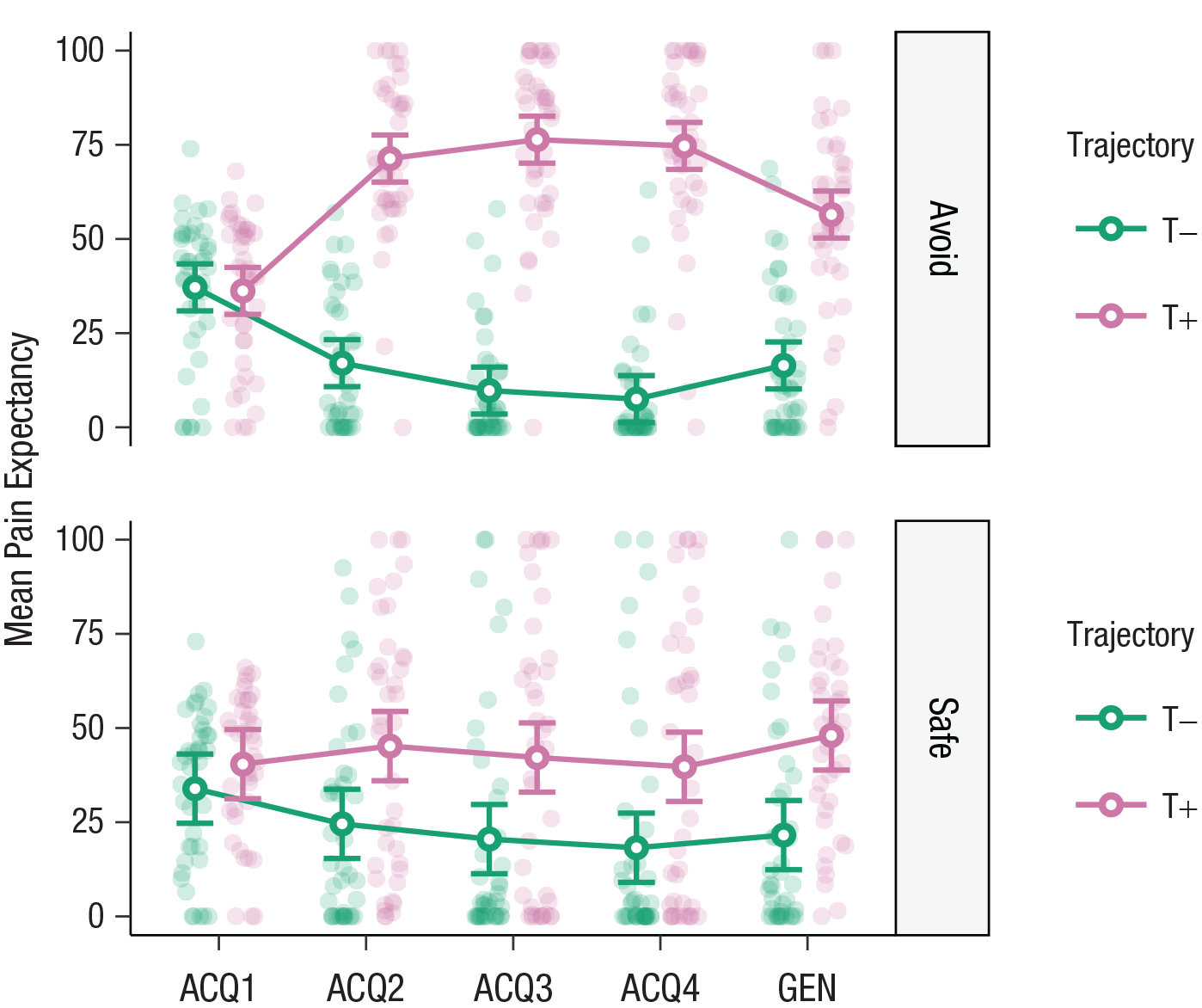
Pain expectancy toward the different movement trajectories (T+, T–) in the avoidance (top) and safe (bottom) categories, separately for the four acquisition (ACQ) blocks and the generalization (GEN) phase. Open circles show means, and error bars represent 95% confidence intervals. Filled circles indicate individual participant data.
A similar repeated measures ANOVA on self-reported pain-related fear revealed comparable effects. There was a significant three-way interaction, F(1.35, 52.58) = 17.41, p < .001, η p 2 = .31, and planned contrasts showed that T+ was feared more than T– during Acquisition Block 4 in the avoidance category, t(39) = 12.30, p < .0001, d = 3.07, 95% CI = [1.88, 4.26]. Again, contrary to our expectations, this difference was also significant in the safe category, t(39) = 2.77, p = .009, d = 0.71, 95% CI = [0.13, 1.28], again to a lesser degree. Thus, participants learned the acquisition contingencies, that is, that T+ was paired with pain and T– was not, in the avoidance category (see Fig. 4). This was reflected in fear learning as well. However, although smaller, these effects transferred to the safe category, suggesting that participants were slightly uncertain about whether both trajectories in the safe category were completely safe.
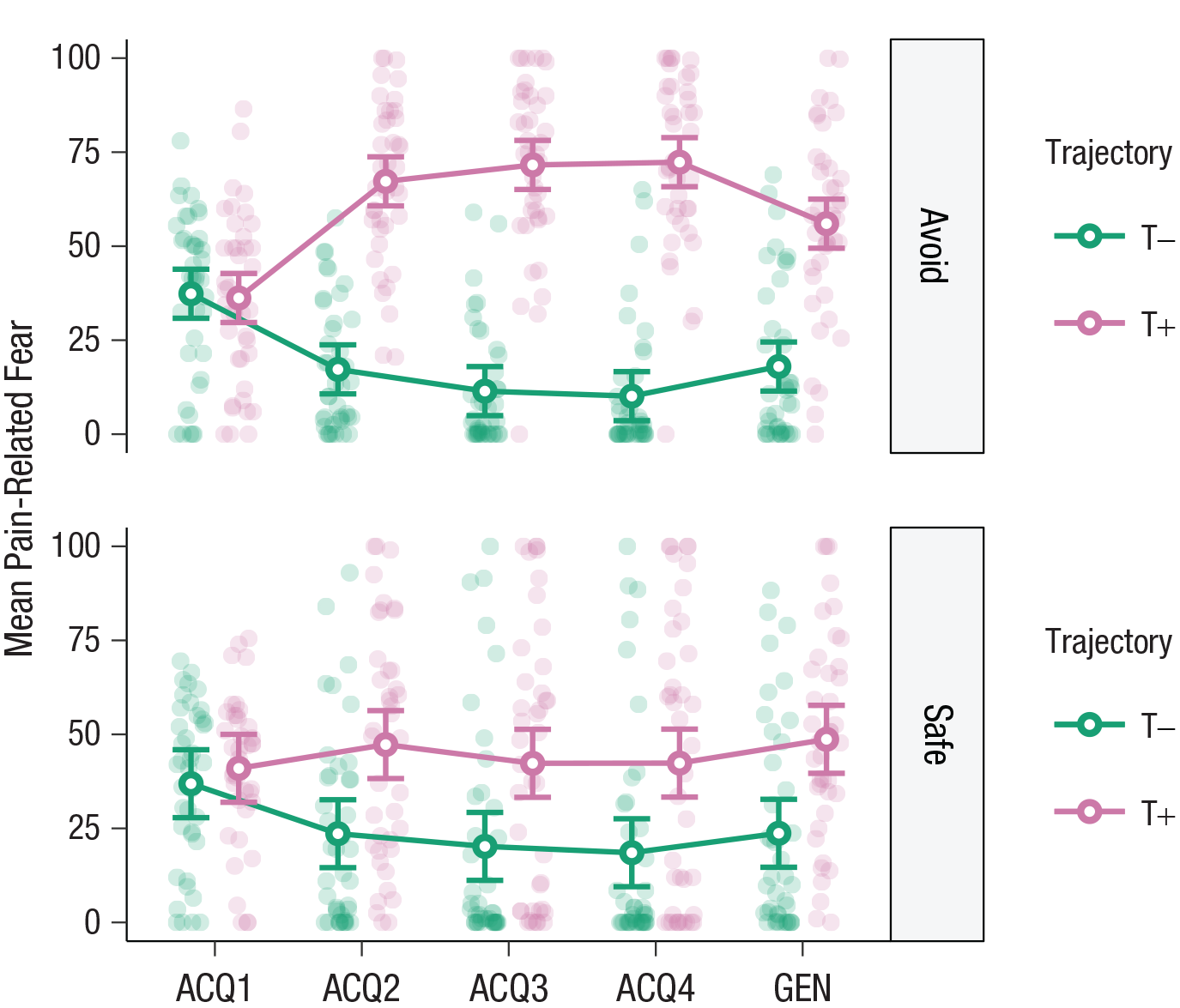
Pain-related fear toward the different movement trajectories (T+, T–) in the avoidance (top) and safe (bottom) categories, separately for the four acquisition (ACQ) blocks and the generalization (GEN) phase. Open circles show means, and error bars represent 95% confidence intervals. Filled circles indicate individual participant data.
Generalization
Avoidance
As expected, a paired-samples t test on mean maximal deviation during the generalization phase, showed that participants deviated more from the shortest movement during novel activities from the avoidance category compared with novel activities from the safe one, t(39) = 5.40, p < .0001, d = 0.78, 95% CI = [0.32, 1.24] (see Fig. 2). However, exploratory analyses showed that maximal deviations were generally lower in the generalization phase compared with the end of the acquisition phase. Interestingly, maximal deviations during the generalization phase were slightly larger for novel (generalization) activities compared with trained (acquisition) ones in the avoidance category, whereas in the safe category, deviations were larger for the trained activities than the novel ones (for the descriptions and visualizations of these analyses and results, see Section S5 and Figs. S5.1 and S5.2 in the Supplemental Material). It should be noted, however, that although these effects reached significance at the p < .05 level, the 95% CIs suggested they were not statistically significant.
These findings were supported by the results of our logistic regression with trajectory choice (T+, T–) as the binary dependent variable and category (avoidance, safe) as the predictor. Participants were 1.80 times more likely to choose T– than T+ during novel activities from the avoidance category, compared with those from the safe category, exp(0.59), 95% CI = [1.14, 2.85]. Taken together, these results indicate that participants generalized avoidance using real-life categorical knowledge. However, participants generally avoided less during the generalization phase compared with the acquisition phase.
Self-reported pain expectancies and pain-related fear
As expected, a 2 × 2 repeated measures ANOVA (category: avoidance, safe) × (trajectory: T+, T–) on pain expectancies during the generalization test revealed a significant two-way interaction, F(1, 39) = 5.11, p = .029, η p 2 = .12, indicating that the differing patterns of pain expectancies in the two activity categories transferred to the novel activities from these categories. In line with results from the acquisition phase, planned contrasts showed that participants expected pain more in response to T+ than T– in the avoidance category, t(39) = 7.65, p < .0001, d = 1.64, 95% CI = [1.29, 1.99]. Although significantly smaller, the same effect also emerged in the safe category, t(39) = 3.93, p = .0003, d = 0.92, 95% CI = [0.63, 1.21] (see Fig. 3). Similarly, exploratory analyses showed that participants expected pain more for T+ than T– during both trained and novel activities. Furthermore, participants expected pain more during trained compared with novel activities in the avoidance category, whereas in the safe category, pain was expected more during novel activities than trained ones, but only for T+ and not T– (see Section S5, Fig. S5.3, and Table S5.1 in the Supplemental Material).
A similar repeated measures ANOVA on self-reported pain-related fear revealed similar effects. There was a significant two-way interaction, F(1, 39) = 5.10, p = .030, η p 2 = .12, and planned contrasts showed that T+ was feared more than T–, t(39) = 7.37, p < .0001, d = 1.49, 95% CI = [1.17, 1.80], during novel activities from the avoidance category. Again, contrary to our expectations, this difference also occurred in the safe category, t(39) = 3.72, p = .0006, d = 0.85, 95% CI = [0.57, 1.12], although it was significantly smaller than in the avoidance category (see Fig. 4). Exploratory analyses showed that participants feared T+ more than T– during both trained and novel activities, similar to pain expectancies. Finally, participants also feared T+, but not T–, more during trained than novel activities in the avoidance category, whereas the opposite was true in the safe category: novel activities were feared more than trained ones (see Section S5, Fig. S5.4, and Table S5.2 in the Supplemental Material). Taken together, these results suggest that participants generalized expectancies and fear of pain toward the novel activities from both categories, although the effect was larger in the avoidance category than the safe category. In general, T+ evoked more pain expectancy and fear than T–.
Discussion
In an innovative paradigm, we combined visual representations of activities with movement to investigate whether pain-related avoidance generalizes from one activity to another on the basis of real-life categorical knowledge. Our hypotheses were largely supported: Despite costs, participants learned to avoid a pain stimulus by deviating more from a pain-associated movement during activities from an avoidance category compared with a safe category. Participants also feared and expected pain more during a low-cost, pain-associated movement (T+) than a high-cost, pain-free movement (T–) in the avoidance category. Critically, these effects also generalized: Participants deviated more from T+ during novel activities from the avoidance category (e.g., gardening) compared with novel activities from the safe category (e.g., cleaning). Participants also continued to fear and expect pain more during T+ than T– when faced with novel activities. Given that we did not train participants on these categories, we conclude that costly pain-related avoidance generalized from one activity to another on the basis of real-life categorical knowledge.
The current findings align with previous research showing that pain-related fear generalizes from one action category to another (Glogan et al., 2019; Meulders, Vandael, & Vlaeyen, 2017) and extends those findings by showing that not only pain-related fear but also instrumental avoidance can generalize from one action to other, categorically similar ones. The current findings also align with a study from Kloos et al. (2022), in which participants generalized costly avoidance to novel scenes from a previously pain-associated category (indoor or outdoor scenes). The current study shows that avoidance can also generalize from one activity to other, categorically associated activities. This is crucial because fear of movement and activity is the most important predictor of disability in musculoskeletal pain conditions (Roelofs et al., 2004).
Surprisingly, participants in the current study reported higher fear and pain expectancies for the pain-associated movement (T+) during activities not only from the avoidance category but also from the safe category, albeit less than in the avoidance category. In line with this, participants also demonstrated some avoidance in the safe category (i.e., deviations were not 0), suggesting that avoidance in the current study may not have been costly enough (Wong & Pittig, 2022). Avoidance is an instrumental response, and instrumental-conditioning procedures involve three crucial elements. The operant response is a behavior influenced by a certain outcome, and the discriminative stimulus indicates whether a specific response will be followed by the outcome (Skinner, 1953). For example, touching (response) a ceramic stove will result in pain (outcome) only when it is red (discriminative stimulus). Thus, although it seems that the activity categories in the current experiment became discriminative stimuli, modulating the operant response–outcome (movement–pain) contingencies (and avoidance), it also seems that T+ still elicited some fear in its own right. This implies that despite learning that one category was safer than the other, participants generally did not trust T+, possibly because there was some avoidance of this movement even in the safe category. That is, participants may have been inferring danger on the basis of their own avoidance behavior (i.e., “I am avoiding; therefore, there must be a threat”; Engelhard et al., 2015; van Dis et al., 2022; van Vliet et al., 2021).
Interestingly, post hoc analyses suggested that participants avoided novel activities more than trained ones in the avoidance category during generalization. This was unexpected because the trained activities were paired with pain, whereas the novel activities were not. The uncertainty associated with novelty may have motivated participants to avoid more in an effort to stay on the safe side (Leng & Vervliet, 2022; Van den Bergh et al., 2021). These findings were not based on a priori hypotheses, however, thus warranting replication and cautious interpretation. Nevertheless, together with the differential self-reports toward T+ and T– in the safe category, these findings draw attention to the myriad unexpected but nontrivial ways in which human participants may feel about or interpret experimental stimuli and manipulations.
Although participants avoided more in the avoidance than in the safe category throughout the experiment, they generally did so less during the generalization phase than the acquisition phase. This aligns with previous research suggesting that avoidance costs motivate healthy participants to explore novel actions (Glogan et al., 2021, 2022). Given that the novel activities were never paired with pain, such exploration may have led to some extinction of avoidance. Our sample was healthy, and previous research has shown that people with chronic pain do not extinguish generalized fear as efficiently as healthy control subjects (Meulders, Meulders, et al., 2017). Whether this is also the case for operant avoidance remains to be tested.
The current results have numerous clinical implications. First, they imply that avoidance can generalize from one activity to another on the basis of categorical relationships between activities, suggesting that the scope of avoidance-inducing behaviors may be increasingly vast and idiosyncratic, based on a person’s unique verbal and semantic networks (Dunsmoor & Murphy, 2015). During exposure therapy for chronic pain, avoidance is typically prevented because it precludes the opportunity to disconfirm threat (Gatzounis et al., 2021; Mineka, 1979). Clinicians may need to take into account that entire categories of behaviors should be targeted and that such networks need to be probed and mapped during treatment. Such an approach would also fit with general recommendations for more person-specific treatment approaches in biomedicine and psychology alike (Insel, 2009; Vicente et al., 2020).
Some limitations deserve mentioning. First, there were only two available joystick movements, and these were the same for all activities. To counter this issue, future studies could use two different avoidance movements (e.g., on both sides of T+) and alternate their availability in order to tease apart the proprioceptive and categorical similarity of the operant responses. Second, although using real-life categories as experimental stimuli extends the generalizability of results beyond the lab, it also reduces experimental control. Compared with experiments using de novo stimuli and categories (e.g., in match-to-sample tasks; Bennett et al., 2015), such lack of control may result in unexpected findings. In the current study, one participant divided the activities into “electronic” and “manual” after the experiment, which also reveals an imbalance in our stimulus selection; there were no electrical tools in the gardening category. Relatedly, not all our activities were entirely realistic; it would have made more sense for a laundry pile to be moved toward a laundry machine rather than the other way around. Future studies should be more cognizant of such details and imbalances. Furthermore, we used arm movements and real-life activities as operant responses to increase immersion in the task. However, because of the joystick’s restricted degrees of freedom, these efforts were ultimately limited. Future research could increase ecological validity by implementing the study in virtual reality. Finally, the current study was conducted in a sample of healthy, pain-free participants, and thus we cannot automatically generalize these findings to people with chronic pain. Nevertheless, it is important to map the basic processes underlying the phenomena we investigate in order to compare clinical samples with healthy people and identify anomalies. Thus, the current study offers a starting point for future research to investigate the differences in category-based avoidance generalization between healthy people and people with chronic pain.
To conclude, using an ecologically valid paradigm in which participants “performed” real-life activities, the current study demonstrated that pain-related avoidance behavior can generalize from one activity to other safe yet categorically associated activities. This form of generalization is highly problematic, given that category-based relations can be extremely wide reaching and idiosyncratic. Thus, a multitude of personalized activities may need to be targeted during psychological therapies for chronic pain to make them as effective as possible in treating these disabling conditions.
Supplemental Material
sj-docx-2-pss-10.1177_09567976231170878 – Supplemental material for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge
Supplemental material, sj-docx-2-pss-10.1177_09567976231170878 for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge by Eveliina Glogan, Peixin Liu and Ann Meulders in Psychological Science
Supplemental Material
sj-pdf-3-pss-10.1177_09567976231170878 – Supplemental material for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge
Supplemental material, sj-pdf-3-pss-10.1177_09567976231170878 for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge by Eveliina Glogan, Peixin Liu and Ann Meulders in Psychological Science
Supplemental Material
sj-pdf-4-pss-10.1177_09567976231170878 – Supplemental material for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge
Supplemental material, sj-pdf-4-pss-10.1177_09567976231170878 for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge by Eveliina Glogan, Peixin Liu and Ann Meulders in Psychological Science
Supplemental Material
sj-xlsx-1-pss-10.1177_09567976231170878 – Supplemental material for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge
Supplemental material, sj-xlsx-1-pss-10.1177_09567976231170878 for Generalization of Costly Pain-Related Avoidance Based on Real-Life Categorical Knowledge by Eveliina Glogan, Peixin Liu and Ann Meulders in Psychological Science
Footnotes
Acknowledgements
This research was presented at the Pain Science in Motion IV Congress, which took place May 19 and 20, 2022, in Maastricht, The Netherlands. We thank Jacco Ronner and Erik Bongaerts for programming, and Richard Benning for creating the graphics of, this experiment. This research was supported by a Vidi grant from the Netherlands Organization for Scientific Research (NWO; grant ID 452-17-002) granted to Ann Meulders. Eveliina Glogan is also supported by a postdoctoral mandate (PDM) from the KU Leuven Research Council Internal Funds (grant ID PDMT2/21/021).
Transparency
Action Editor: Daniela Schiller
Editor: Patricia J. Bauer
Author Contributions
Correction (September 2023):
Article updated online to correct paragraph 2 on pg. 810 due to a production error where text was inserted into the wrong location in the paragraph.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
