Abstract
The diet of pregnant women exposes fetuses to a variety of flavors consisting of compound sensations involving smell, taste, and chemesthesis. The effects of such prenatal flavor exposure on chemosensory development have so far been measured only postnatally in human infants. Here, we report the first direct evidence of human fetal responsiveness to flavors transferred via maternal consumption of a single-dose capsule by measuring frame-by-frame fetal facial movements. Pregnant women and their fetuses based in the northeast of England were involved in this study from 32 to 36 weeks’ gestation. Fetuses exposed to carrot flavor (
Amniotic fluid is the first place where fetuses start to sense their environment, specifically their chemical environment (Brumley & Robinson, 2010). This experience provides continuous sensory information, such as taste and smell, from fetal to neonatal life (Mellor, 2019; Schaal, 2005). The continuity, based on early familiarization, allows newborns to adapt to the postnatal environment (Mellor, 2019).
Among a huge variety of other compounds linked to maternal-fetal genotype and metabolism as well as the maternal environment, the prenatal environment is permeated with aroma compounds conveyed through the mother’s diet (Schaal, 2016). This is the case for both human (Hauser et al., 1985; Mennella et al., 1995, 2001; Schaal et al., 2000) and nonhuman (Figueroa et al., 2013; Hepper & Wells, 2006; Lévy et al., 2020; Smotherman & Robinson, 1987) fetuses who encode and memorize incoming chemical stimuli. In human fetuses, taste buds develop anatomically at 8 weeks’ gestation and can detect tastants from 14 weeks’ gestation (Witt & Reutter, 1998). Additionally, fetal nasal orifices are open to allow amniotic fluid to access olfactory sensory neurons, which can sense odor-active molecules from 24 weeks’ gestation (Witt, 2020). Hence, although they continue to develop anatomically and functionally after birth, fetal chemosensors are sufficiently mature to detect flavors including tastants and odorants in the amniotic fluid in the last trimester of pregnancy (Forestell & Mennella, 2015; Schaal, 2016). Throughout this article, we refer to flavor exposure or experience, acknowledging that taste, olfaction, and trigeminal chemesthesis cannot be dissociated in the womb and that their effects on the fetus may involve one or several of these chemosensory inputs.
To date, the impact of fetal flavor exposure has been investigated using several indirect strategies. First, before ultrasound visualization was possible, fetal chemosensation and swallowing activity of amniotic fluid were inferred from changes in the pregnant mother’s waist size, especially in cases of polyhydramnios, amniotic fluid being in excess because of low fetal swallowing (de Snoo, 1937). Second, preterm infants were studied on the assumption that their chemosensory reactivity could be extrapolated to same-age fetuses for both olfaction (Schaal et al., 2004) and taste (Maone et al., 1990; Tatzer et al., 1985).
A third strategy consisted of testing term-born neonates in the hours or days following birth with chemostimuli that they were assumed to be exposed to prenatally. For example, Hepper (1995) found that neonates 15 to 28 hr old did not show aversive reaction to garlic odor (relative to a control odor) if their mothers had eaten at least four meals containing garlic per week during the last month of pregnancy. Supporting this finding, Schaal et al. (2000) showed that neonates up to 4 days of age, whose mothers consumed anise-flavored sweets in the last 2 weeks of pregnancy, compared with a nonexposed group, preferred the anise odorant. Additionally, Mennella et al. (2001) tested the effects of maternal carrot juice consumption on 5- to 6-month-old infants’ response to carrot flavor and found that those exposed to carrot flavor during the last trimester of pregnancy displayed fewer negative facial expressions toward carrot-flavored cereal than plain cereal. These human findings, added to those of studies on fetal rats, lambs, piglets, and dogs (Figueroa et al., 2013; Hepper & Wells, 2006; Lévy et al., 2020; Smotherman & Robinson, 1987), support the claim that there is evidence of stable, long-term retention of fetal experiences of flavor that can affect food acceptance postnatally.
A fourth strategy consisted of visualizing the spontaneous, immediate responses of surgically prepared fetuses to flavors in utero. This approach has been used in various nonhuman models (Schaal et al., 1991; Smotherman et al., 1991). However, human fetal studies in this field have yet to be undertaken. The aim of the present study was to fill this gap in testing human fetal reactions via ultrasound scanning immediately after exposing them to target flavors ingested by the pregnant mother.
A human fetus can display facial movements (FMs) ascertained by the mobilization of individual facial muscles (Reissland et al., 2016). These FMs can be classified with an objective coding scheme, in which each FM refers to a specific collection of muscular movements. For example, FM11 refers to the appearance of a nasolabial furrow that is formed by the action of the zygomaticus minor muscle, and the lower lip being pulled down toward the chin by the depressor labii inferioris muscle generates FM16 (Ekman & Friesen, 1978).
Statement of Relevance
Through their developing sensory abilities, fetuses are alert to aspects of their environment in the womb. For example, by swallowing and inhaling the amniotic fluid, a fetus can sense the flavors of food eaten by its mother. However, the current literature on human studies has exclusively focused on postnatal outcomes of prenatal flavor exposure. Instead, by analyzing their facial reactions, we present direct, novel evidence that fetuses can discriminate different flavors in amniotic fluid. We found that when fetuses were exposed to carrot flavor, they were more likely to show “laughter-face” reactions, and when they were exposed to kale flavor, they were more likely to show “cry-face” reactions. We also found that facial responses to flavors became more complex as fetuses matured. This study sheds new light not only on fetal sensory abilities but also on the specificity of facial responses to different flavors relating to their discriminative abilities.
Multiple muscular actions induce complex facial expressions, called “gestalts.” The gestalts are the combination of FMs occurring simultaneously or within 1 s of one another, which may consist of double, triple, quadruple, or more fetal FMs (Reissland et al., 2011, 2013). The more FMs contributing to the gestalt, the more complex it is. With fetal age, the complexity of the gestalt increases as evidenced by the increased number of co-occurring movements (Reissland et al., 2011, 2013). These gestalts resemble facial expressions associated postnatally with positive or negative hedonic responses, visible from the second trimester of pregnancy and increasing with gestation (Reissland et al., 2011, 2013). However, it is not yet possible to ascertain hedonic co-occurrence in fetuses. For instance, the “cry-face gestalt” can be described as a series of coordinated FMs encompassing lower-lip depressor (FM16) and nasolabial furrow (FM11), which would be perceived as a cry face by an adult observer but might not be associated with a “sad” hedonic response (for examples of different facial gestalts, see Figs. 1 and 2).
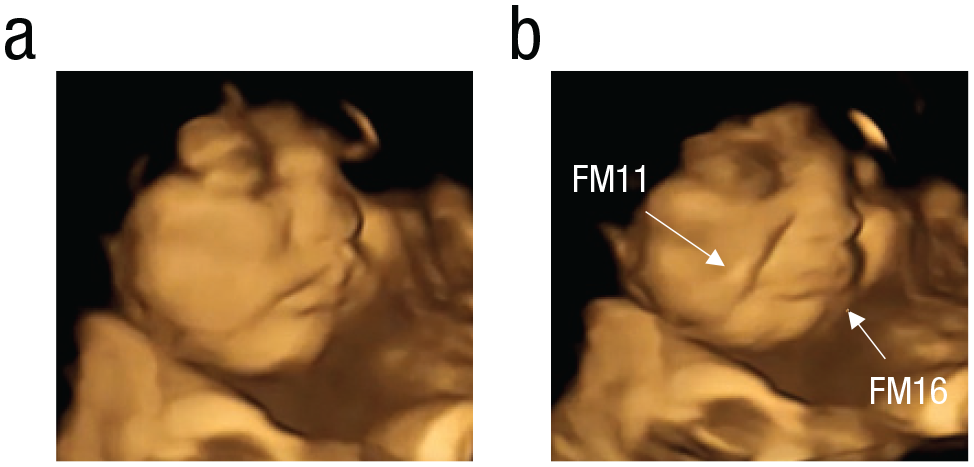
Example of cry-face gestalt of a kale-exposed fetus: (a) baseline, (b) cry-face gestalt (apex). FM11 = nasolabial furrow; FM16 = lower-lip depressor.
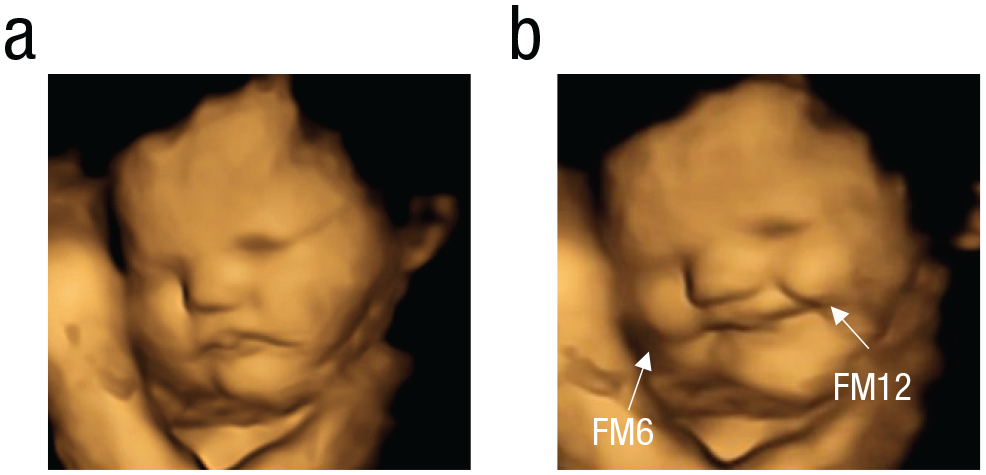
Example of laughter-face gestalt of a carrot-exposed fetus: (a) baseline, (b) laughter-face gestalt (apex). FM6 = cheek raiser; FM12 = lip-corner puller.
The present study coded 4D ultrasound scans with the validated Fetal Observable Movement System (Reissland et al., 2016) to examine how fetuses from 32 to 36 weeks of gestational age react to different flavors transferred to the fetal environment by maternal ingestion. We compared the FMs obtained from scans from three groups of fetuses exposed to either carrot flavor, kale flavor, or no flavor. Although there is no chemical evidence on the stability of compounds of kale or carrot until they reach the amniotic fluid, multiple data exist on the mother-to-fetus transfer of vegetable aromas (for a review, see Spahn et al., 2019). For example, carrot flavor transfer is inferred by a change in infant behavior measured a posteriori (Mennella et al., 2001). Similarly, neonates whose mothers consumed more green vegetables in pregnancy showed higher liking scores for target odors (Wagner et al., 2019). Regarding the taste of vegetables, the complex carrot flavor can be described by adult judges as “sweet” because of its sugar content but also sometimes as having “fruity,” “woody,” or even “petrol” flavors, likely a result of terpenoids present in carrot (e.g., β-carotene; Alasalvar et al., 2001). Kale was chosen because it conveys more bitterness to infants than other green vegetables such as spinach, broccoli, or asparagus (Johnson et al., 2021).
Because these two flavors do not share the same flavor profile, we expected to observe different FM profiles in kale-exposed fetuses and carrot-exposed fetuses. Newborns exposed to carrot juice prenatally had less nose wrinkling, brow lowering, upper lip raising, gaping, and head turning than nonexposed newborns (Mennella et al., 2001). In contrast, newborns drop the corners of their mouths and raise their upper lips when exposed to a bitter solution (Steiner, 1979).
On the basis of this current evidence, we hypothesized first that there would be a significant difference in facial reactions as expressed in facial muscle movements and gestalts between the three groups of fetuses. We predicted a higher rate of “laughter-face” reactions (fetal movements and gestalts) in the carrot group and a higher rate of cry-face reactions (fetal movements and gestalts) in the kale group in comparison with each other and the control group. Second, because of the developmental progression in chemosensory perception and maturation of facial neuromuscular structures, irrespective of what flavor the fetuses were exposed to, we expected to observe developing complexity in these facial gestalts from 32 to 36 weeks.
Method
Ethics
This study was conducted in accordance with the Declaration of Helsinki, and ethical permission for the research reported in this article was granted by Durham University (PSYCH-2019-03-12T15_59_32-wvgf27). All mothers gave written informed consent.
Participants
One hundred mothers between the ages of 18 and 40 years (
Distribution of Mothers Over Flavor Stimuli and Number of Scans Coded
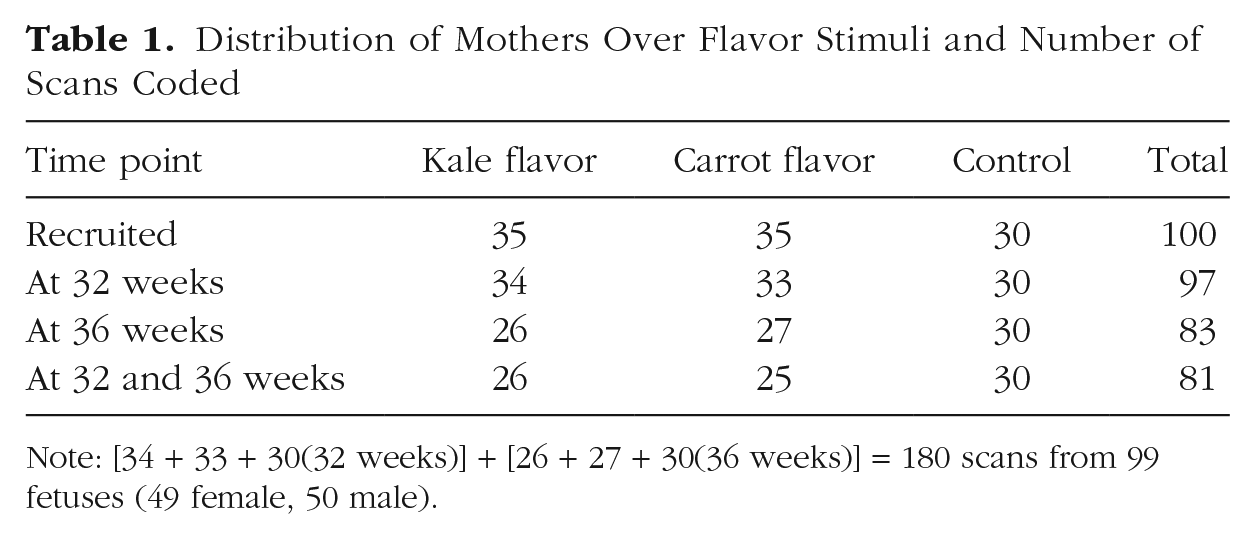
Note: [34 + 33 + 30(32 weeks)] + [26 + 27 + 30(36 weeks)] = 180 scans from 99 fetuses (49 female, 50 male).
All participants were White British, living in the northeast of England. Experimental group participants (carrot:
Flavor stimuli
Participants in the carrot and kale groups received one organic carrot or kale capsule, respectively. The reason for using a capsule was to limit as much as possible the degradation of the flavors before the absorptive digestive process, because the capsules are formulated to reach the small intestine by resisting the gastric passage (Marzorati et al., 2015). In addition, we were able to control maternal likes or dislikes by giving capsules with almost no smell and taste compared with the direct consumption of carrot and kale. One capsule contained approximately 400 mg of carrot/kale powder. This amount corresponds to approximately 50 g of raw vegetables (e.g., one medium carrot or 100 g of chopped kale). The shell of the capsules consisted of hydroxypropyl methylcellulose, which is suitable for vegans and vegetarians (Marzorati et al., 2015).
Procedure
For this study, at 32 and 36 weeks’ gestation, all women underwent nonmedical 4D ultrasound scans in accordance with the British Medical Ultrasound Society guidelines (Society of Radiographers & British Medical Ultrasound Society, 2021). All participants were asked not to ingest anything in the hour prior to their appointment to optimize the effect of the flavor stimuli.
In the experimental groups, participants had abstained from consuming any food and/or drink involving carrot and kale on the day of the scan. Experimental group mothers swallowed one vegetable capsule (carrot/kale) with a mouthful of water approximately 20 min (
To control for the effects of maternal vegetable consumption on fetal exposure to the stimulus, we assessed the frequency of their effective intake of nonbitter (carrot and other nonbitter vegetables including potatoes, peas, beans, and sweet corn) and bitter (kale and other bitter vegetables including lettuce, leeks, and Brassica vegetables such as broccoli and cauliflower) vegetables in the week prior to each scan (see Table 2; for the questionnaire, see the Supplemental Material available online).
Mean Frequencies of Vegetable Consumption in the Week Prior to Scans
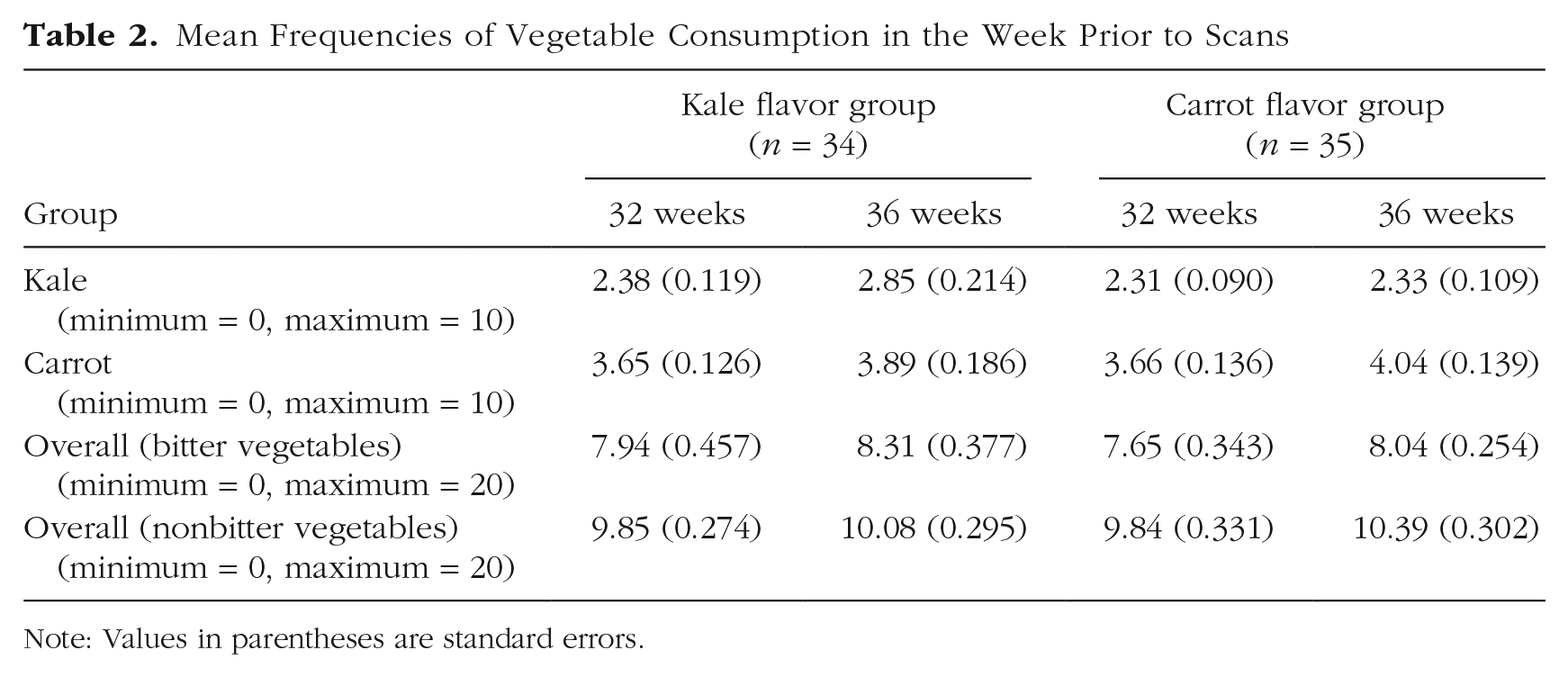
Note: Values in parentheses are standard errors.
Following the completion of waiting time, mothers underwent 4D ultrasound scans for around 25 min. To minimize possible effects attributable to different levels of fetal movements, we started each scan at approximately the same time of day (~3 p.m.). Throughout the scans, by three expert sonographers who were blinded to the study objectives and the group allocation, all mothers laid on their back or on their side in a darkened, quiet room, as they did in their 12- and 20-week scans. They were able to see their unborn child on a screen. At the start of each scan, the sonographer measured femur length, head circumference, and heartbeat to ensure that fetuses were healthy and in the expected range of development. Real-time 4D ultrasound recordings and traditional 2D images were visualized side by side to observe the face and upper torso of the fetus. The scans were recorded (at 24 fps) for off-line analysis using a GE Voluson E8/E10 Expert Ultrasound System with a RAB6-RS transducer (General Electric, Boston, MA). All mothers received a copy of their scans.
Behavioral coding scheme and method of coding
By following procedures validated in previous research, we selected 17 independent FMs to analyze fetal FMs in response to different flavors (see Table 3). Fetuses in the last trimester of pregnancy are able to display these muscle movements (Reissland et al., 2016). The movements, apart from FM11, FM16, and FM24, were selected on the basis of previous research reporting postnatal flavor-elicited reactions (e.g., Mennella et al., 2001; Rosenstein & Oster, 1988; Schaal et al., 2000). Preliminary analysis of the current sample before we decided on the final coding scheme revealed that fetuses were able to express nasolabial furrow (FM11), lower-lip depressor (FM16), and lip pressor (FM24), along with other movements defined in the literature, when exposed to flavors.
Fetal Facial Movements (FMs) Coded and the Muscle Configurations of Facial Gestalts
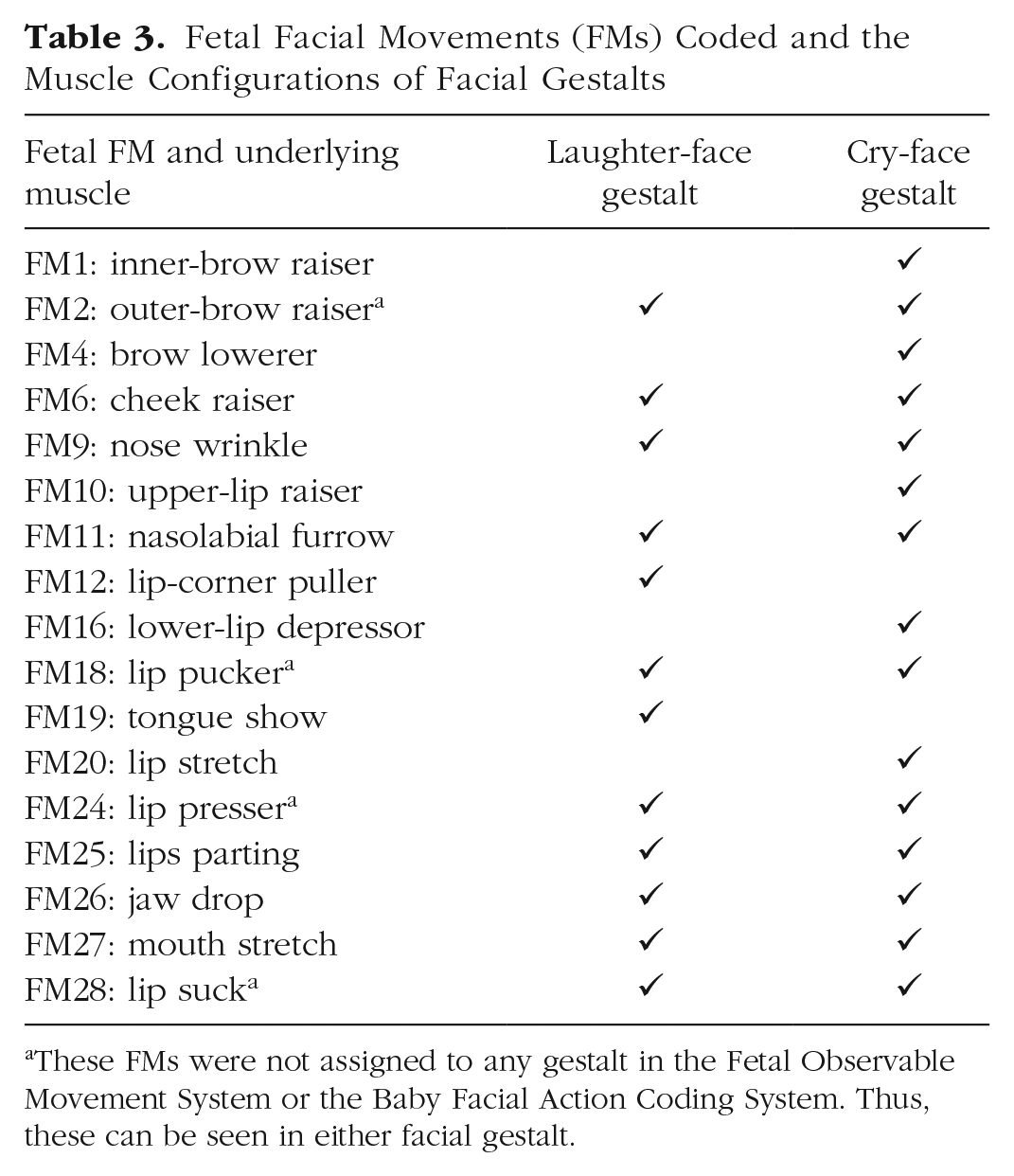
These FMs were not assigned to any gestalt in the Fetal Observable Movement System or the Baby Facial Action Coding System. Thus, these can be seen in either facial gestalt.
In terms of classification of movements, 11 movements were attributed to the gestalts following the Fetal Observable Movement System coding scheme (Reissland et al., 2016). Two movements (FM20 and FM26) were linked to the gestalts following the cry-face matrix in the Baby Facial Action Coding System (Oster, 2006). The rest of the FMs were not specified in the literature; thus, they were exploratory. In the case of the FMs that were assigned to both gestalts, such as cheek raiser (FM6), the final interpretation in corresponding gestalts of these actions depends on reactivity in the other part of the face.
All coding was completed by trained, blinded coders (one main Facial Action Coding System certified coder, two reliability coders) using the Observer (15XT; Noldus, the Netherlands). A lab assistant, who was not involved in coding and analyzing, fully anonymized the data so that the coders were blinded to both experimental and control groups. The coders first observed the scan and marked with “start-stop” codes those sections of the scans where the fetal face was visible enough to code. Although the codable scan duration varied between fetuses because of their position in the womb, the mean of codable length in each group was similar (see Table S1 in the Supplemental Material). Therefore, the relative frequency of fetal FMs per minute was calculated by dividing the number of the given FMs by the codable time and multiplying the result by 60. Second, the total codable footage of each scan was coded frame by frame to identify discrete FMs displayed by the fetus. Third, facial gestalts were identified by summing the number of movements in the gestalt, and the relative frequency of gestalts per minute was measured. Fourth, to measure the complexity of gestalts, we classified the gestalts by the number of concurrent FMs they comprised—single, double, triple, quadruple, or higher. Interobserver reliability analyses were applied by independent coders on 20% of the data set, and the overall agreement was 0.90, with a range of 0.81 to 0.98 using Cohen’s kappa.
Statistical analyses
This study involved longitudinal sampling and mixed design on three different groups (between-subjects design)—two experimental (carrot exposure, kale exposure) and one control (no flavor exposure). All dependent variables—namely, the frequency of discrete fetal FMs, facial gestalts, and the level of complexity—were log-transformed to normalize the data set. The log-transformed scores were transformed back for data presentation.
First, a one-way analysis of variance (ANOVA) was computed to assess whether maternal (age and BMI) and fetal (head circumference at 20 weeks, gestational age at birth, and birth weight) variables differed between groups. Second, Pearson chi-square test was used to test whether there were any significant differences between the groups in terms of fetal sex. Third, using Pearson correlations, we tested whether dependent variables were correlated with maternal mental health variables (stress, anxiety, and depression) and level of education. For missing data in the maternal and fetal independent variables, series’ mean estimations were used. Additionally, an independent-samples
Results
In this repeated measures study, fetal 4D ultrasounds at 32 weeks (
Covariates
Predictors of fetal movements, including fetal sex, maternal age, maternal prepregnancy BMI, gestational age at birth, and birth weight, were initially examined but removed from further analyses because these measures did not significantly differ between groups (
Average time elapsed from ingestion to appearance of the first fetal FMs
In the carrot group, the mean of first FM was observed after 29.20 min (
Analyses of discrete fetal FMs
32 weeks’ gestation
On the basis of 97 ultrasound scans, we conducted analyses of discrete fetal FMs and found that the relative frequency of upper-lip raiser (FM10),
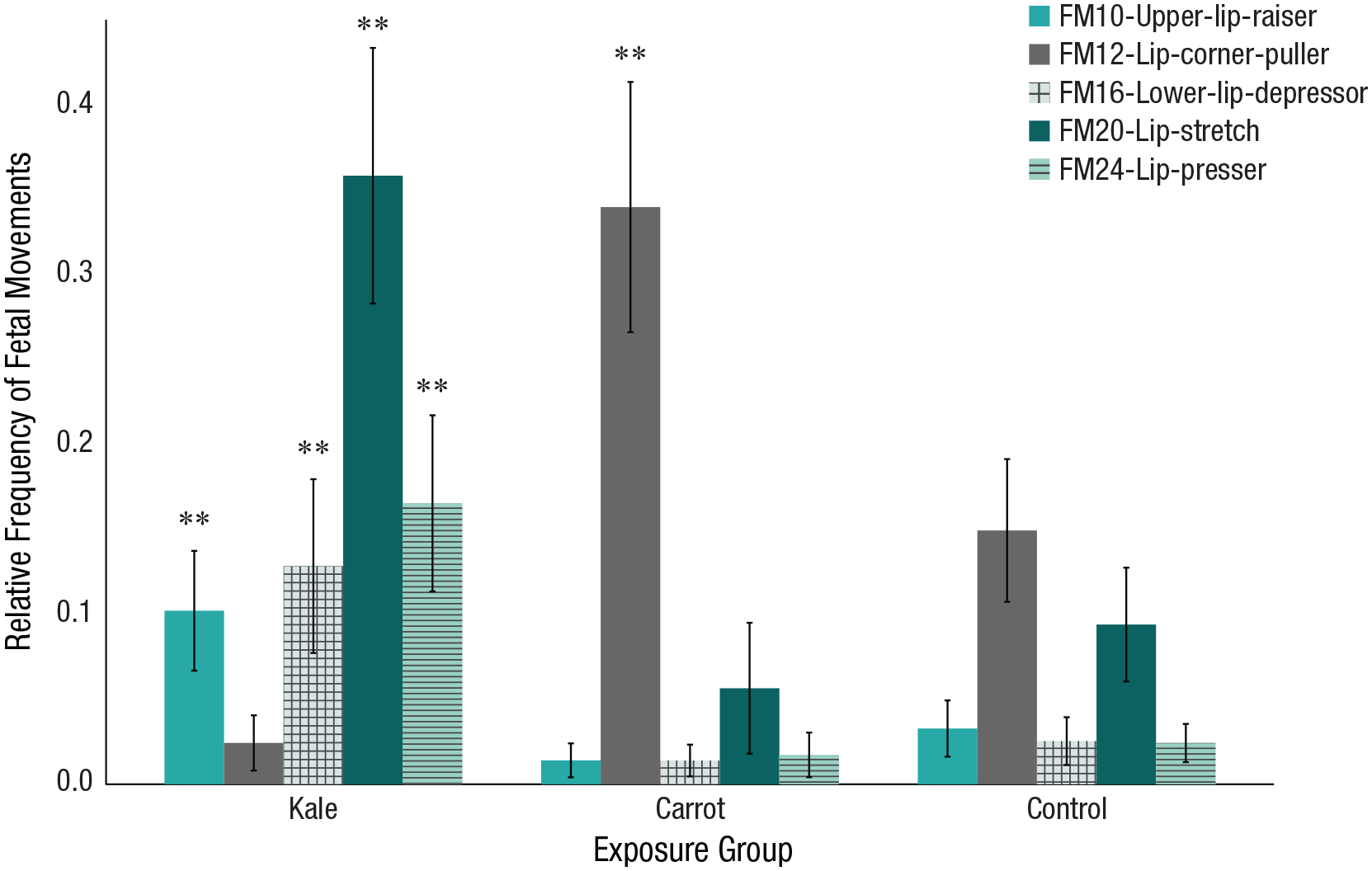
Relative frequency of fetal facial movements at 32 weeks split by exposure group. Error bars represent 95% confidence intervals. Asterisks indicate significant differences (**
36 weeks’ gestation
On the basis of 83 ultrasound scans, we found that the relative frequency of upper-lip raiser (FM10),
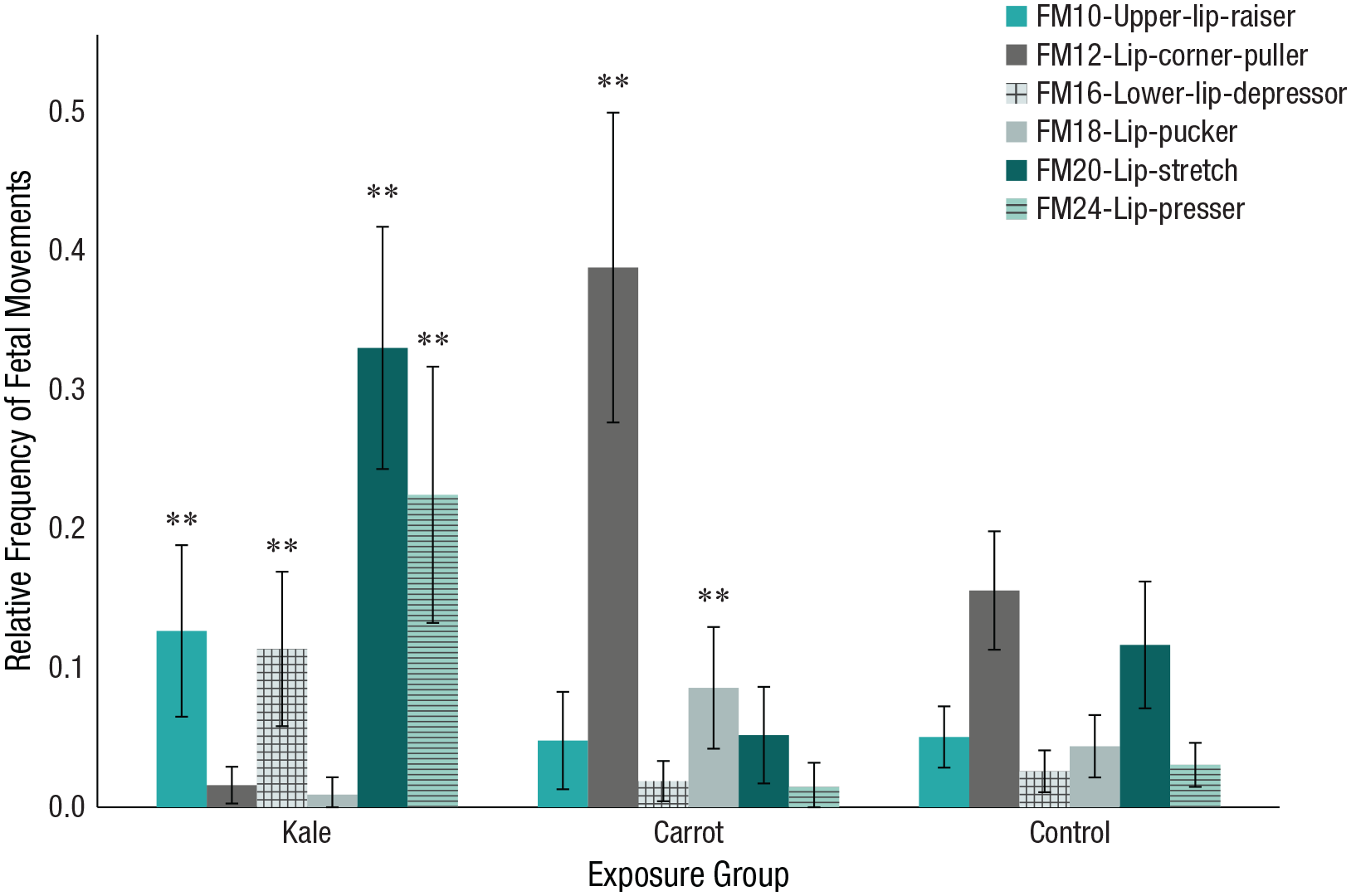
Relative frequency of fetal facial movements at 36 weeks split by exposure group. Error bars represent 95% confidence intervals. Asterisks indicate significant differences (**
Laughter-face gestalt versus cry-face gestalt
Using Roy’s largest root in MANCOVA results, we found a significant effect of flavor exposure on the facial gestalts at 32 weeks,
At 36 weeks, Roy’s largest root revealed a significant difference in the facial gestalts depending on flavor exposure,
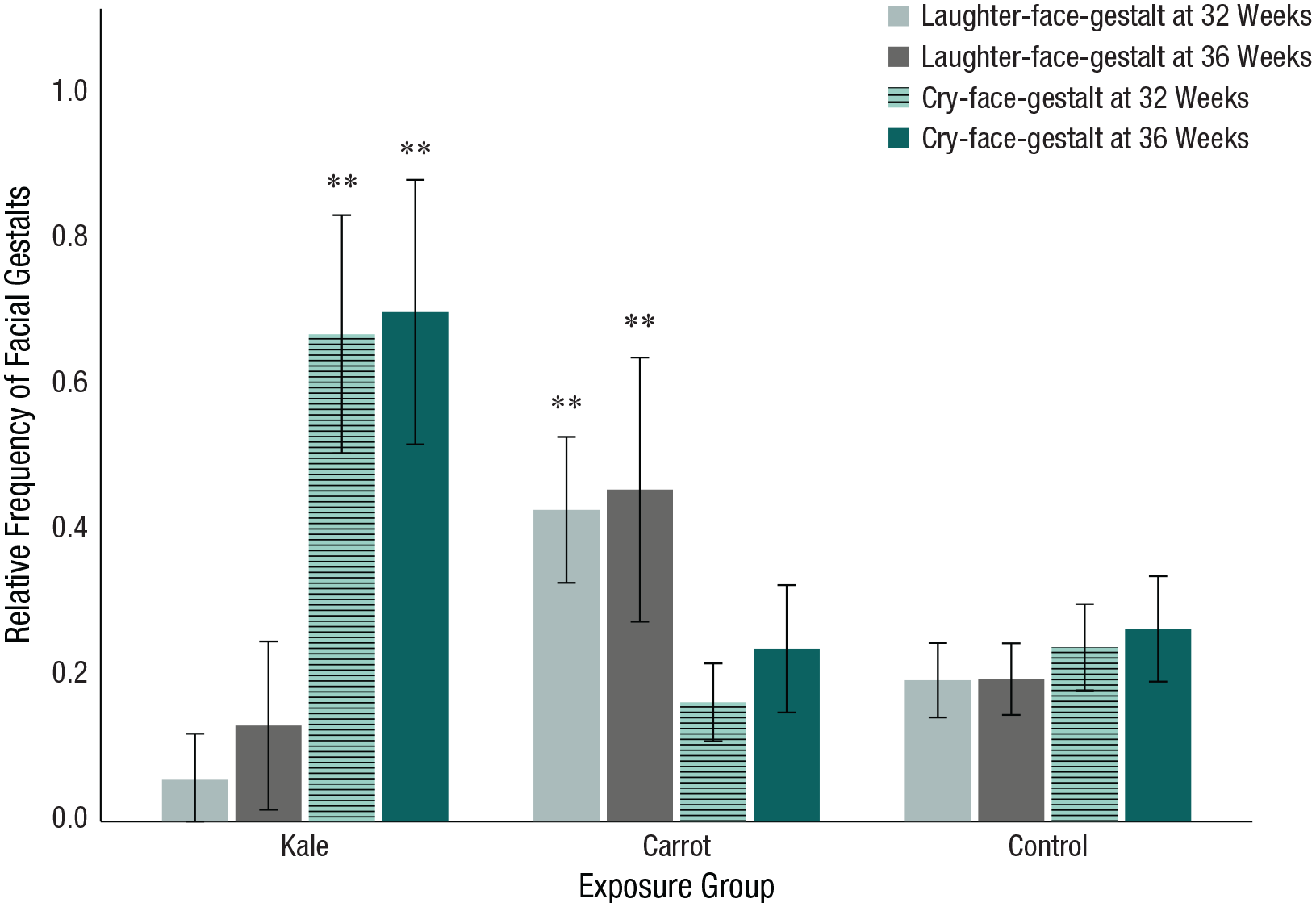
Relative frequency of facial gestalts at 32 and 36 weeks split by exposure group. Error bars represent 95% confidence intervals. Asterisks indicate significant differences (**
Combinations of fetal FMs into complex face gestalts
Whereas facial gestalts consisting of double and triple movements did not show a significant increase in frequency, the overall facial gestalt made of quadruple or more movements increased from 32 to 36 weeks,
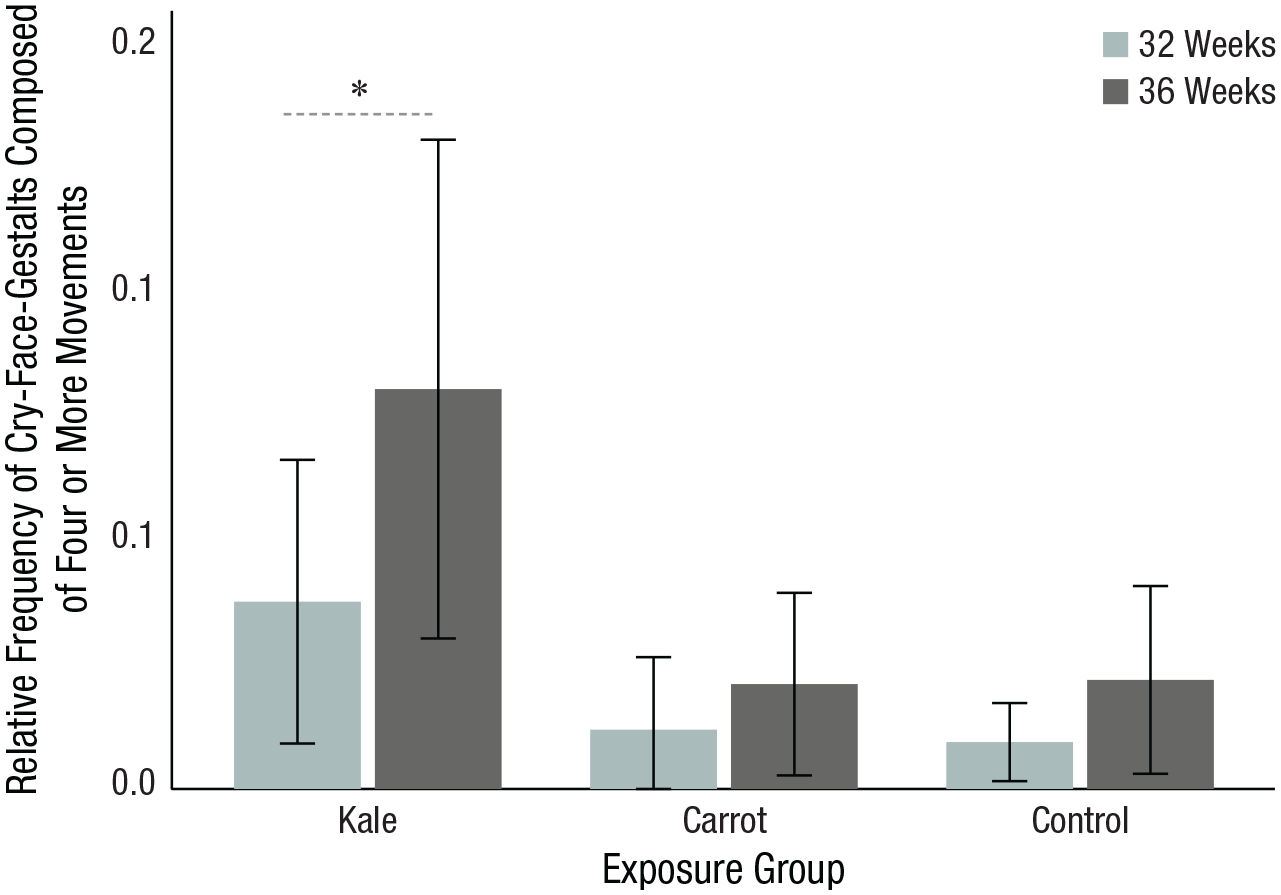
Relative frequency of cry-face gestalts composed of four or more facial movements at 32 and 36 weeks split by exposure group. Error bars represent 95% confidence intervals. The asterisk indicates a significant difference (*
Discussion
This study assessed whether human fetuses ages 32 to 36 weeks react to flavors taken in by their mothers. The main results show that the intake of a single dose of flavorant by a pregnant woman activates fetal FMs as well as a combination of movements that together form a laughter-face or a cry-face gestalt. Moreover, fetuses express different frequencies of facial gestalts in relation to the type of flavor that they are exposed to, namely, more laughter-face gestalts when exposed to a carrot flavor and more cry-face gestalts when experiencing a kale flavor. Finally, as fetuses develop from 32 to 36 weeks, they react to kale flavor, but not to carrot flavor, with an increasing number of different FMs coalescing in more complex facial gestalts. These findings are discussed in more detail below.
The fetus reacts to dietary flavors ingested by the mother
Previous studies have sensorily monitored changes in flavor quality and intensity of the amniotic fluid after the ingestion of odorants or tastants by mammalian females (Hauser et al., 1985; Mennella et al., 1995). The transplacental transfer of flavor molecules has rarely been assessed in tracing them using instrumental chemistry (Schaal, 2005), but numerous physiological and pharmacokinetic studies of drugs have shown that most molecules can cross the placenta, some by passive diffusion and some by facilitated diffusion (Syme et al., 2004). Human studies of fetal chemoreception so far have not included instrumental chemical verification of effective transplacental flavorant transfer into the amniotic fluid but relied only on differential postnatal behavioral outcomes to the flavorants ingested by pregnant women. The present study examined fetal chemosensory reactivity and was able to demonstrate that pregnant women’s flavor intake affects the fetal reactions in utero directly and quasi-immediately.
Fetal reactions observed here in both flavor groups compared with the control group provide evidence that the ingestion of only 400 mg of powdered carrot or kale in a capsule was sufficient to reach fetal chemoreceptors. This effect occurs within a relatively short time: Around 30 min after maternal ingestion of the flavor capsules, we detected observable facial reactions in the fetuses. Thus, in this short time, the flavor content of the capsules undergoes digestion, absorption into the mothers’ bloodstream, metabolization and circulation through the placenta and fetus, collection in the amniotic fluid, and fetal chemoreceptors. So far, we have little knowledge about the biotransformation and transplacental kinetics of flavorants ingested by mothers, but this relatively short transfer time is compatible with the pharmacokinetic data on drugs in the human materno-fetal system (Brown et al., 1990).
Fetal behavior in utero is specific to flavor input
The morphological specificity of fetal reactions observed in the present study aligns with expectations based on neonatal studies on flavor (Maone et al., 1990; Rosenstein & Oster, 1988; Steiner, 1979; Tatzer et al., 1985).
In the kale-exposed group, at 32 and 36 weeks, the relative frequency of upper-lip raiser (FM10), lower-lip depressor (FM16), lip stretch (FM20), and lip presser (FM24), as well as their combinations leading to the cry-face gestalt, was higher in comparison with carrot-exposed and control fetuses. In the case of exposure to carrot flavor, at 32 and 36 weeks, lip-corner puller (FM12) alone and facial configurations involving movements making up the laughter-face gestalt were relatively more frequent in the carrot-exposed group compared with both the control and kale-exposed groups. However, lip pucker (FM18) appeared at 36 weeks but not at 32 weeks. It is known that fetuses can pucker their lips around 32 to 34 weeks (Piontelli, 2015). Individual differences in lip-pucker (FM18) performance may explain the nonappearance of this movement at 32 weeks.
Although lip presser (FM24) and lip pucker (FM18) are not assigned to any of the gestalts in the literature, our findings suggest that FM24 is a reaction to kale flavor and FM18 to carrot flavor. The evidence that fetuses react with differential FMs depending on the flavor of maternal intake indicates that they discriminate these two flavors by at least 32 weeks’ gestation.
Development of flavor-elicited facial expressivity
The present study demonstrates that it is possible to visualize the short-term development of facial expressive reactivity to flavors during late pregnancy. Specifically, the combination of four or more FMs contributing to the cry-face gestalt in the kale-group fetuses increased from 32 to 36 weeks. This finding supports the concept that fetuses exhibit complex motor behaviors associated with cry-face and such behaviors increase with gestational age (Reissland et al., 2011). In contrast, in our sample, there was no evidence that the complexity of the gestalt expression related to the perception of carrot flavor increases with fetal maturation. This contrasting result can be explained by the anatomical substrate engaged in generating laughter-face and cry-face gestalts. The laughter-face gestalt, which occurs in the carrot flavor group, is anatomically more straightforward to generate because the activation of a sole FM12 suffices for its appearance. It thus develops earlier (Kurjak et al., 2003) compared with the complex cry-face gestalt, which occurs more in the kale flavor group. The cry-face gestalt needs the activation of multiple FMs to end with a readable gestalt. Therefore, we might not observe a developmental trend that late in gestation.
Limitations
For this well-powered study, adding a control group from archived data, because of limited resources, may constitute a limitation of the research. Although the archived data include the same stress, anxiety, and depression measures as the original study, we could not collect information in the control group on the frequency of vegetable consumption before ultrasound scans. Given that infants’ chemosensory responses may be influenced by habitual vegetable consumption by pregnant mothers (Wagner et al., 2019), this might also affect fetal reactivity. However, we assume that the data not collected are unlikely to affect the results because the frequency of vegetable consumption was not associated with the FMs of fetuses in both experimental groups.
Another potential confounding factor in the study is the impact of genetic variations in bitter taste perception due to the expression of the TAS2R38 gene (Feeney, 2011). Given that genetically mediated sensitivity to bitter tastants has been associated with food acceptance in infants (Cont et al., 2019), we need to acknowledge that fetuses in our study were likely to comprise a mixture of super-tasters, medium tasters, and nontasters on the basis of single-nucleotide polymorphisms of the TAS2R38 gene. Although we used capsules to bypass mothers’ taste sensitivity, variance in the fetal gene encoding TAS2R38, which dictates individual gustatory differences, may have an impact on fetal FM profile. Additionally, mothers in the experimental groups might have seen the shades of color of the powder inside the translucent capsule, but they could neither taste nor smell the content. Nonetheless, further work is needed to determine whether taster status moderates fetal FM profiles in response to flavor experiences in utero.
Conclusion
The present article is the first longitudinal study to indicate that fetuses are capable of prenatally detecting chemosensory information conveyed by flavor compounds originating in maternal diet. This was shown by fetal facial reactions after either carrot or kale flavor exposure. Fetuses from 32 to 36 weeks react to kale flavor with a cry-face gestalt increasing in complexity as they mature. Because the laughter-face gestalt is less complex than the cry-face gestalt, we were unable to observe an increase in complexity in this case.
Results of this study have important implications for our understanding of the development of human oral and nasal chemoreception, including the nature and timing of behavioral reactions to prenatal flavor exposure, fetal engagement of memory for flavors, and the potential role played by prenatal to postnatal continuity in perception and reactivity to the chemical environment.
Given that experimental evidence indicates that prenatal flavor experience is embedded and is accessed postnatally in humans (Mellor, 2019; Schaal, 2005), it could be argued that repeated prenatal flavor exposures may lead to preferences for certain flavor profiles that are consistent with flavor experienced postnatally in very different contexts. Future studies need to follow up with postnatal behavioral analyses to assess how prenatal flavor exposure can exert influences on postnatal food preferences in the short and long term.
Supplemental Material
sj-docx-2-pss-10.1177_09567976221105460 – Supplemental material for Flavor Sensing in Utero and Emerging Discriminative Behaviors in the Human Fetus
Supplemental material, sj-docx-2-pss-10.1177_09567976221105460 for Flavor Sensing in Utero and Emerging Discriminative Behaviors in the Human Fetus by Beyza Ustun, Nadja Reissland, Judith Covey, Benoist Schaal and Jacqueline Blissett in Psychological Science
Supplemental Material
sj-docx-3-pss-10.1177_09567976221105460 – Supplemental material for Flavor Sensing in Utero and Emerging Discriminative Behaviors in the Human Fetus
Supplemental material, sj-docx-3-pss-10.1177_09567976221105460 for Flavor Sensing in Utero and Emerging Discriminative Behaviors in the Human Fetus by Beyza Ustun, Nadja Reissland, Judith Covey, Benoist Schaal and Jacqueline Blissett in Psychological Science
Supplemental Material
sj-pdf-1-pss-10.1177_09567976221105460 – Supplemental material for Flavor Sensing in Utero and Emerging Discriminative Behaviors in the Human Fetus
Supplemental material, sj-pdf-1-pss-10.1177_09567976221105460 for Flavor Sensing in Utero and Emerging Discriminative Behaviors in the Human Fetus by Beyza Ustun, Nadja Reissland, Judith Covey, Benoist Schaal and Jacqueline Blissett in Psychological Science
Footnotes
Acknowledgements
We thank all sonographers and especially Teay Wei Lit and Marian Ab. Malik for their help in recruiting participants and performing ultrasound scans, and we thank the independent coders for reliability analysis of the data. We are grateful to the mothers who took part in this study.
Transparency
B. Ustun designed the experiment, provided funding, collected data, coded ultrasound scans, analyzed the data, and wrote the manuscript. N. Reissland, J. Covey, B. Schaal, and J. Blissett designed the experiment, provided guidance for data analysis, and reviewed and edited the manuscript. All the authors approved the final manuscript for submission.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
